Upper Gastrointestinal Bleeding
Upper Gastrointestinal Bleeding
Authors: Thomas B. Perera, MD, Assistant Professor, Department of Emergency Medicine, Jacobi/Montefiori Medical Center, Bronx, NY; and Jesse Carron, MD, Department of Emergency Medicine, Jacobi/Montefiori Medical Center, Bronx, NY.
Peer Reviewers: David Overton, MD, MBA, FACEP, FACP, Emergency Medicine Program Director, Michigan State University Kalamazoo Center for Medical Studies, Professor and Chairman of Emergency Medicine, Michigan State University College of Human Medicine; and Keith Grams, MD, Associate Medical Director, Assistant Professor of Clinical Emergency Medicine, Department of Emergency Medicine, Strong Memorial Hospital, Rochester, NY.
I distinctly remember my first experience as a medical student in a public hospital helping the intern handle a patient with vigorous hematemesis. What a mess; blood everywhere. With difficulty, we placed a large 32 French gastric lavage tube; the 18 French Salem sump tube was too narrow to handle the large clots in the patient's stomach. I was sent to get a large basin of ice into which the intern emptied a several-liter bottle of normal saline irrigation solution. I was told the cold saline would stop the bleeding.
We started lavage using a 60 mL syringe and managed to make a big mess. This was before the recognition of HIV and the Universal Precautions era, so the intern and I had blood on our hands and arms. There was blood on the patient, in the bed, and on the floor. Unfortunately, bleeding continued and so, when the second-year resident came by to see how we were doing, he confidently added several vials of norepinephrine to our lavage solution. I was told this would cause vasoconstriction of the mucosal vessels and stop the bleeding.
Well, the bleeding continued. The intern went off to call the surgery resident and left me to continue lavage. Two syringes of saline in, then aspirate. Sometimes fluid returned, and sometimes not. The intern returned with the surgery resident, who looked at us with bemusement. He said the only way to stop serious hemorrhage is with a silk suture. So, off the patient went to the OR.
Much has changed in the past 20 years. Drugs, endoscopic approaches, and interventional radiology have dramatically reduced the need for operative intervention in patients with upper GI bleeding. This issue of Emergency Medicine Reports will provide the emergency physician with a current review of this important topic. While, the emergency physician may not use some of the treatments, it is useful to understand them and be able to communicate with the intensivist, gastroenterologist, interventional radiologist, or general surgeon.
And the patient survived.
—J. Stephan Stapczynski, MD, Editor
Introduction
Very little else evokes the visceral response of watching someone vomit blood. Emergency physicians deal with bleeding in many arenas, but gastrointestinal (GI) bleeding is one type that in the past has offered limited options for treatment in the emergency department. Unlike with other forms of bleeding, direct pressure is simply not an option for GI bleeding.
Bleeding can arise from anywhere in the gastrointestinal tract from the mouth to the anus. It can also originate from any of the structures that empty secretions into the GI tract, such as the liver or the pancreas. This article will focus mostly on upper GI bleeding because lower GI bleeding is caused by a different disorder. The diagnosis and treatment of lower GI bleeding also differs and focuses less on the emergency department management.
The Scope of the Problem
According to the CDC, the rate of upper GI bleeding has decreased slightly from 67/100,00 in 1994 to 47/100,000 in 2000. Upper GI bleeding still accounts for 0.1-0.2% of hospital admissions. Of these, 5-14% will die during that hospitalization.1,2 Gastrointestinal hemorrhage does not make the CDC's list of leading causes of death. In fact, GI hemorrhage rarely is the listed cause of death; rather it contributes to decompensation from other underlying illnesses. Even with all the advances in medicine in the past 30 years, there has been little change in the morbidity and mortality from upper GI bleeding. The emergency department management of gastrointestinal hemorrhage has been evolving for years. From the 1990s onward, there have been many advances, particularly in the availability and usefulness of endoscopy as a diagnostic and therapeutic tool. The advances in the past few years have made this topic worthy of a critical review at this time.
Upper vs. Lower GI Bleeding
Upper GI bleeding is defined as bleeding proximal to the ligament of Treitz. The ligament of Treitz is a fold of peritoneum that suspends the fourth part of the duodenum. Upper GI bleeding encompasses a large list of possible causes that differ in origin and treatment from lower GI bleeding. Upper GI bleeding accounts for about 85% of patients presenting to emergency departments with GI bleeding. It has a higher morbidity and mortality than lower GI bleeding. Its treatment is more often medical. Lower GI bleeding is distal to the ligament of Treitz and treatment is more often surgical.
It is sometime difficult to make the distinction between upper and lower with small bowel bleeding, but lower GI bleeding has a small bowel source 1-5% of the time. These transition area cases may be called "hemorrhage of obscure origin" and sometimes can only be identified with tests not available to the ED physician, such as video capsule endoscope.
Diagnosis
For emergency physicians, diagnosis and treatment often start simultaneously. The initial treatment of GI bleeding mostly involves the treatment for hypovolemic shock. It is important to realize that patients with GI bleeding often have other comorbidities and often take medication that can mask the seriousness of their disease. Patients may appear stable but decompensate suddenly. The astute clinician will be able to recognize these subtle, often partially masked, signs of serious hemorrhage.
Presentations of GI Bleeding
When a patient enters the emergency department with hematemesis (vomiting blood or coffee grounds), hematochezia (passing blood from the rectum) or melena (dark, tarry, heme-containing stool) the diagnosis is easy, but GI bleeding can present in a variety of ways. As with many illnesses presenting to the emergency department, the difficulty usually is not with inaccurate or insensitive diagnostic tests, but rather with deciding to look for the disease in the first place. (See Table 1.)
| Table 1. Common Presentations of Gastrointestinal Bleeding | ||||||||||||||
|
History
As with all patients, the importance of taking a good history cannot be over-emphasized in patients who present with GI bleeding. The first thing to establish is whether the blood is from an upper or a lower source. Hematemesis and melena usually are signs of upper GI bleeding, while hematochezia is associated with lower GI bleeding. There are exceptions, however. In one study, more than 10% of hematochezia was shown to originate from an upper source.3 This makes sense since most GI bleeding is from an upper source, and blood is an intestinal irritant that increases its motility through the GI tract. Melena, on the other hand, can have a lower GI source in up to 25% of cases. To produce melena there should be at least 200 mL of blood that spends at least 8 hours in the intestines. Dark, non-tarry stool can be produced with considerably less blood if it is more distal in the intestine. Another confounding factor is that stool can remain dark for days after the intestinal bleeding has stopped.
Estimates of blood loss have been shown to be inaccurate in many aspects of medicine from trauma to GI bleeding. With bleeding in the bowel or in the urine, patients often state that the bleeding had reddened all the water in the toilet bowl. Although this can be quite striking to the patient, it can occur with as little as 2-5 mL of blood.
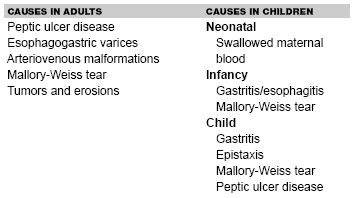
There are few aspects of the history that help with the exact localization of the bleeding. A small number of disorders produce most cases of upper GI bleeding. (See Table 2.) Patients with a history of ulcers or other causes of bleeding will have a similar cause about 60% of the time.4 Patients with a history of esophageal varices should be assumed to be bleeding from the varices.
| Table 2. Causes of Upper GI Bleeding in Adults and Children |
|
Crampy abdominal pain is more common with upper GI bleeding than with lower GI bleeding. Medications, especially NSAIDs, often contribute to gastric and duodenal sources of bleeding. Preceding dyspepsia may indicate a gastric ulcer. A history of alcohol abuse, liver disease, or cirrhosis may point toward esophageal or gastric variceal bleeding. Evidence of postural hypotension, dizziness, or syncope may indicate more severe rapid bleeding.5
Patients with a history of aortic grafts who present with evidence of bleeding should prompt an investigation into a possible aortoenteric fistula. This complication has been reported to occur in 0.4 to 4% of patients with a history of abdominal aortic grafts. Aortoenteric fistula has a high mortality if not detected early.6,7
Physical Examination
The physical examination should focus first on determining if the patient is bleeding and, second, if the patient is in shock. Signs of intra-abdominal bleeding start with the vital signs. The obvious signs of shock are hypotension and tachycardia. Patients who are hypotensive from blood loss of up to 35%, however, often will not be tachycardic. The absence of tachycardia should not reassure the clinician about the absence of significant blood loss.8 (See Table 3.)
| Table 3. Severity Categories of Hemorrhagic Shock |
|
The usefulness of orthostatic testing has been debated for a long time. Although not perfect even in healthy non-medicated people, a pulse increase greater than or equal to 20 bpm has a specificity for hypovolemia of 0.98. A decrease in systolic blood pressure greater than or equal to 20 mmHg has a specificity of 0.97 and a shock index ((heart rate in bpm) / (SBP in mmHg)) increase greater than or equal to 0.2 has a specificity for hypovolemia of 0.99.9 Most clinicians use some combination of these as well as patient reported dizziness on standing as a reasonable assessment of intravascular volume loss. Age, co-morbidities, and medications such as beta-blockers affect the sensitivity of orthostatic testing.
Capillary refill also has been used for evaluation of fluid status. The sensitivity is as low as 10% using a cutoff of 2-3 sec.10 Orthostatic measurements have been shown to have a better sensitivity and specificity than delayed capillary refill.
Physical clues of GI bleeding include the signs of anemia. These include pallor (color of skin, palms, oral and conjunctival mucous membrane, and nail beds), systolic ejection murmur, mild peripheral edema, and venous hums and wide pulse pressure. In the elderly, angina pectoris can be an important clinical manifestation. Although any one or group of these signs may not be pathoneumonic they often can help guide the physician.
Consideration and examination of possible alternative bleeding sources such as pulmonary, urinary, and vaginal sources should be part of the physical evaluation. Almost 10% of patients presenting to emergency departments with histories consistent with gastrointestinal bleeding are found to have another source of bleeding or no detectable bleeding.
It is important to look for the sequelae of chronic liver disease. These include hepatomegaly, splenomegaly, hepatojugular reflux, ascites, jaundice, and signs of collateral circulation such as spider angiomata and facial telangectasia.11 These signs are present in an increased number in patients and can lead one to consider esophageal varices as a possible source for the bleeding.
Laboratory Testing
If significant GI bleeding is suspected, the first and most important laboratory test is a type and cross. Especially if esophageal varices are present, having 4 units of packed red blood cells as well as fresh frozen plasma available can make the difference. An INR as well as an initial PT should help guide therapy, although if massive bleeding is suspected waiting for the results should not delay treatment with coagulation factors.
A spun hematocrit, a point-of-care hemoglobin measurement, or a CBC performed in the clinical laboratory provides the initial assessment of red cell status. There have been questions about the magnitude of the change and how long it takes to equilibrate. Removal of 500 mL of blood has been shown to decrease the hematocrit by about 5-6%. Transfusion with 1 unit increases the hematocrit by about 2.5-3%. In a controlled setting these changes were present within 15-30 minutes and remained constant for 24 hours, suggesting that the hematocrit may equilibrate fast enough to be useful. The initial hematocrit is a point in time that is most useful as when following subsequent values. It is helpful, therefore, to know that the addition of 1 L of normal saline can decrease the hematocrit by as much as 4-4.5%.12,13
Other laboratory values that which may be helpful are basic chemistries including glucose, liver function tests (LFTs), and in specific cases ammonia, and fibrinogen split products or a DIC panel. The blood urea nitrogen (BUN) to creatinine (Cr) ratio has been used to evaluate the severity of the bleeding and whether there is an upper vs. a lower source of bleeding. A BUN/Cr ratio greater than 36 has a sensitivity of 90% and a specificity of 27% for upper GI bleeding.14 A higher ratio makes it more likely that severe bleeding is present, although the sensitivity of this criteria is poor.
Patients with poor liver function are at higher risk of esophageal bleeding. They are also less likely to have adequate glycogen stores and are at risk for hypoglycemia in the stress of acute illness. GI bleeding and the digestion of blood puts a strain on the liver, and those patients with borderline liver function can be at risk for hepatic encephalopathy in the setting of an acute bleed.
Considering that peptic ulcer disease accounts for about 55-60% of GI bleeding presenting to emergency departments, and Helicobacter pylori infections are present in almost 90% of these patients, peptic ulcer disease testing for H. pylori should be considered.15 It is not helpful emergently, but should be tested by the inpatient service or the primary care physician, as it could decrease return visits for pain and bleeding as well as decrease the number of cases of gastric cancer.
Diagnostic Testing
Although most clinicians consider the rectal examination for occult blood using guaiac or other chemical detection as a standard part of the physical examination, understanding its use and limitations is especially important in the diagnosis of gastrointestinal hemorrhage. To get a positive test there must be 8 mg hemoglobin per gram of stool. Although false negative results can occur, and immunochemical fecal occult blood tests have been shown to be more sensitive than stool guaiac cards. For the level of bleeding of interest here, this probably does not matter.16,17 It is possible that in some very acute bleeds, the blood may not have traversed the bowel by the time testing is done. False-positive guaiac tests can be caused by dietary peroxidases, including hemoglobin and myoglobin from meat and foods such as uncooked fruits and vegetables that contain high levels of peroxidase and catalase.18 Therapeutic iron intake has been tested a multitude of studies to see if it can cause false-positive guaiac testing. Although in vitro studies often show interference, most studies in vivo have shown that this is not a common cause of false-positive testing.19 Because the physician is usually looking for significant amounts of hemorrhage, a positive test is usually very evident if bleeding has occurred.
Standard stool guaiac testing should not be used to measure occult blood in gastric fluid because of interferences from low pH, certain medications (antacids and vitamin C lead to false negative results), and metal ions (iron and copper salts lead to false positive results). Gastric specific guaiac testing should be employed for this purpose because gastric specific guaiac testing, unlike standard stool guaiac testing, is not influenced by pH and is less affected by interfering drugs. Testing for gastric pH could be employed to check the validity of the results. However, a positive Hemoccult of stomach contents in the setting of coffee ground or bloody emesis usually can be trusted.20,21
The nasogastric tube can help with clarifying the location of bleeding in hematochezia, decompressing the stomach, and allowing gastric lavage for better visualization during endoscopy. A positive result may change disposition and influence when the patient will have endoscopy. Nasogastric tube placement is considered to be one of the most uncomfortable procedures routinely performed in emergency departments. Although many clinicians feel that NG tube placement is not necessary in all cases of upper GI bleeding, we believe it is still a useful diagnostic tool. Nasal bleeding, vomiting, and pain are not uncommon but there is no evidence that the placement of an NG tube will cause significant morbidity in the setting of varices or cardiac disease.22 Conversely, there are no validated guidelines as to when NG tube placement can be avoided in patients with upper GI bleeding. The sensitivity for active bleeding is about 79%. To perform the procedure properly one is supposed to wait until bile is aspirated to ensure that there is adequate evaluation of the stomach and upper duodenum. The sensitivity and specificity for bile aspiration are 49% and 74%, so this has not proven useful.23
Gastric lavage currently is used to allow better visualization during endoscopy and may help indicate the severity of upper GI bleeding.24 In the past gastric lavage was thought to decrease bleeding or rebleeding particularly with cold water lavage, but the evidence does not support this. In fact, cold water may exacerbate bleeding. Current recommendations indicate that room temperature water in volumes of 200-300 mL is appropriate. For removal, gentle suction should be used as more aggressive suction can damage gastric mucosa or cause misleading endoscopic findings.
Many studies have been devoted to reducing the patient discomfort when placing an NG tube. Lidocaine gel, spray, and nebulized solution have all been shown to be helpful. Anti-emetics like metoclopramide as well as anxiolytics have also been used successfully, as have relaxation techniques. All of these interventions have been shown to reduce discomfort and improve successful placement rate.25 Unfortunately, studies also show that none of these techniques are used routinely.
Treatment
The successful ED management of upper GI bleeding entails rapid assessment, prompt resuscitation, appropriate risk stratification, and timely involvement of intensivists, gastroenterologists, and then if necessary, surgeons and interventional radiologists.
Initial management is consistent with the management of any other potentially sick patient. The patient should be placed on cardiac, blood pressure, and oxygen saturation monitors. Two large bore peripheral IVs need to be inserted to aggressively resuscitate the patient, if necessary. Laboratory tests need to be sent (see above). An electrocardiogram (ECG) and chest x-ray (rule out aspiration and or perforation) may be helpful depending on the severity of illness and co-morbidities.
Accurate assessment of those at greatest risk is essential for managing the upper GI bleed. The factors associated with poor outcome are: increasing age (older than 60 years), coagulopathies, liver failure, cardiac disease, and markers for severe bleeding such as shock, repeat hematemesis, hematochezia, and failure for gastric lavage to clear.3 These predictors indicate the need for a more aggressive approach, including possible airway control, rapid resuscitation with fluids, blood product transfusions (packed red blood cells [PRBC], fresh frozen plasma [FFP], platelets, and even possibly recombinant factor VIIa for patients with severe cirrhosis26), and rapid specialist consultation.
Aggressive resuscitation is the key to appropriate emergency treatment. Initial resuscitation should start with volume replacement with crystalloid (normal saline or lactated Ringers). Volumes used for adults are 2 liters wide open while pediatrics should start with 20 mL/kg. The need for additional fluids should be based on blood loss (can estimate crystalloid need as 3 times blood loss volume), continued hemodynamic instability, and continued bleeding. These factors will also help indicate the need for blood product transfusions.
It may be prudent to consider giving coagulation factors as soon as possible to those patients who are sickest and those patients on warfarin, heparin, or heparin analogs. Reversal of warfarin should be undertaken with an understanding of the risks, but early reversal should be considered especially because these patients tend to be older with more co-morbidities. Use fresh frozen plasma (FFP) (2 units), or prothrombin complex concentrate (PCC) and or vitamin K (10 mg PO or SQ). Using vitamin K IV or IM can be associated with a severe allergic reaction and should be considered only when the other routes are unavailable. There are case reports about the use of activated factor VIIa in the setting of GI bleeding, but this use has not been well studied. Protamine sulfate can be used to reverse heparin and the heparin analogs. Aspirin and clopidogrel use is also on the rise, but platelet transfusions have not been shown to reduce bleeding due to aspirin- or clopidogrel-induced hemorrhage.
Pharmacologic Therapy
After initial resuscitation the emergency physician has several additional pharmacologic tools for the treatment of upper GI bleeding depending of the suspected source of bleeding and associated comorbidities. These modalities are particularly important in the emergency department as they do not require the involvement of specialty services.
Acid suppression has long been studied in peptic ulcer disease. Increasing pH has been theorized to improve clot formation and prevent clot breakdown.27 Many of the findings have been inconsistent but a recent meta analysis showed proton pump inhibitors (PPI, intravenous and oral) significantly reduce the risk of re-bleeding and the need for surgery, but with no change in mortality.28 The benefit of intravenous PPI versus oral has not been well studied but the existing literature supports that oral PPIs have benefit in stronger doses (omeprazole 40 mg PO BID).29 Pantoprazole is the only PPI that has FDA approval for the treatment of upper GI bleeding. The findings with PPIs have not been reproducible with H2 blockers and therefore H2 blockers are not recommended for treatment of upper GI bleeding due to peptic ulcer disease.27
Somatostatin and its analogs have long been used for the treatment of variceal bleeding. The mechanism is theorized to be due to a decrease of portal blood pressure and a decrease of splanchnic blood flow in response to food or blood in the GI tract.30 Several studies have evaluated somatostatin and its analogs in the treatment of active bleeding and the results have varied, but more recent literature comparing somatostatin with vasopressin has shown improved control of bleeding and less side effects.30,31 Somatostatin analogs have also been compared with sclerotherapy and found to have similar control of bleeding and rates of rebleeding.32 The best results in the control of bleeding esophageal varices have been achieved with the combination of both somatostatin analog and endoscopy.30,33-35 In the United States octreotide (a long-acting analog) is the only drug FDA approved for the treatment of bleeding esophageal varices. The usual octreotide dose in studies was 50-100 mcg bolus followed by 25-50 mcg per hour for several days. The side effects of octreotide are usually only mild hyperglycemia and abdominal cramping.30 Somatostatin analogs have also been evaluated in the treatment of bleeding peptic ulcers and have been associated with a mild reduced risk of continued bleeding [relative risk 0.53 (95% CI, 0.43-0.630], but in general are used only as additional therapy and when endoscopy is not available or is contraindicated.36
Vasopressin also has been used in the past to help control bleeding from esophageal varices. Vasopressin decreases portal pressures by constricting the mesenteric arterioles.30 The literature demonstrates that vasopressin is able to stop active bleeding in approximately 60-80% of patients, but with no change in rebleeding and possibly increased mortality due to frequent complications.37 The complications seen with vasopressin include myocardial, cerebral, bowel, and limb ischemia.30 To combat some of the complications, some authors have added intravenous nitroglycerin, which theoretically may also decrease portal pressures, to intravenous vasopressin. The results have shown improved control of variceal bleeding and reduced incidence of major complications.37 The use of vasopressin is limited because of high side effect profile and lack of benefit over the somatostatin analogs. One vasopressin analog, terlipressin, has been shown to also have an improvement in mortality, but at this time is not available in the United States.38
DDAVP (1-Deamino-8-D-arginine vasopressin) has been used in the past to control bleeding in patients taking aspirin. It has been shown to help normalize bleeding time and to normalize other surrogate makers in the setting of cardiac surgery but has not been shown to decrease the amount of blood products needed or affect mortality.39 One study comparing terlipressin with or without DDAVP was stopped early due to increased mortality in the DDAVP group. At this time, its routine use is not recommended.40
Additional pharmacologic interventions for upper GI bleeding include antibiotics. Antibiotics are often used for different reasons in patients with upper GI bleeding. Gastroenterologists often request that patients be given intravenous erythromycin prior to endoscopy based on studies that show improved quality of the endoscopy because of erythromycin's promotility effects, and reduced need for repeat endoscopy.41 Several studies have shown that prophylactic antibiotics in cirrhotic patients with upper GI bleeding can reduce infectious complications, provide a mortality benefit, and may reduce rebleeding.30,42,43 The antibiotics traditionally used in the past are fluoroquinolones but recent resistance patterns have encouraged the use of third-generation cephalosporins.30,44 Antibiotics that decrease urease-producing bacteria in the gastrointestinal tract [neomycin (FDA approved for this use) and metronidazole (Flagyl) (off-label use)] have also been used for the treatment of hepatic encephalopathy.45 The timing of these interventions has not been clarified. There is no evidence to suggest that these need to be started in the ED.
The in-hospital use of beta-blockers have also been shown to aid with reducing rebleeding episodes in the setting of esophageal varices. Its long-term use has been shown to reduce the risk of bleeding in those patients with portal hypertension and should be considered in those patients with portal hypertension who may be discharged.46
Endoscopy
Upper GI endoscopy is not only the preferred diagnostic tool, it is now also the definitive treatment for upper GI bleeding. It can be used to treat most causes of upper GI bleeding and has been shown to reduce hospital costs, improve mortality, and decrease the number of patients requiring surgery.27,47,48 Multiple different endoscopic techniques have been used for the treatment of various causes of upper GI bleeding and will be discussed in relation to the bleeding source most amenable to that modality.
Endoscopy has revolutionized the treatment of peptic ulcer disease. It allows not only the treatment but is also a valuable tool in enabling the risk stratification of patients. The appearance of lesions on endoscopy enables the gastroenterologist to better predict which lesions are more likely to rebleed in the near future. Lesions with a high likelihood of rebleeding include active bleeding during the endoscopy, a visible vessel protruding, an adherent clot, and ulcers greater than 2 cm. Lesions that are described as a flat pigmented spot or an ulcer with a clean base are at low risk for rebleeding. This risk stratification becomes important because ulcers with a clean base and possibly a flat pigmented spot may be managed with outpatient management after the endoscopy.27 Interestingly, the use of ED endoscopy vs. in-hospital endoscopy for non-variceal bleeding has not proven to impact morbidity or resource utilization, although it often is used for more critically ill patients and to aid in the triage decisions.49
There are several endoscopic methods used for the treatment of peptic ulcer disease: injection therapy, thermal coagulation, argon plasma coagulation, hemostatic clips, and combination therapy. The body of literature comparing the different methods does not provide a clear consensus, but in general combination therapy involving epinephrine injection followed by either thermal coagulation or hemoclips is better for ulcers that are actively bleeding, have a visible vessel, or have an adherent clot.27,50-52 Ulcers with a clean base and/or flat pigmented spots may need no endoscopic intervention or only one of the above methods. Complications from the endoscopic procedures include perforation and increased or continued bleeding. Treatment failure is defined as continued bleeding or rebleeding and the limited literature supports a second endoscopy which, in one study, was successful in stopping the bleeding in 73% of initial failures.53
Endoscopic treatment for esophageal variceal hemorrhage is critical because only approximately 50% of variceal bleeds stop spontaneously.54 As with peptic ulcer disease, endoscopy is the most widely used treatment for active variceal bleeding and occasionally can be used for prevention as well.30 In general there are two endoscopic modalities used for treatment: sclerotherapy and band ligation. Sclerotherapy and band ligation have similar results for the control of bleeding and preventing rebleeding, but band ligation has fewer complications, particularly less esophageal stricture, and thus probably better long-term outcome.55 The general consensus is that sclerotherapy or band ligation plus a somatostatin analog is probably the most effective for control of bleeding and preventing rebleeding, although it is not clear whether there is any improvement in mortality.33-35 Endoscopic complications include ulceration, bleeding, dysmotility, stricture, perforation, mediastinitis, aspiration, and worsening of portal hypertensive gastropathy. Initial endoscopy with somatostatin analog will fail in approximately 10-20% of patients either in control of initial bleeding or rebleeding soon after endoscopy.30 Continued bleeding may not be reflected by gross bleeding alone but also by the need for additional transfusions or a lack of improvement in vital signs. In these cases, guidelines support a second endoscopic procedure to control the bleeding.56
Balloon Tamponade
Although now rarely needed, balloon tamponade uses an inflatable gastric balloon to apply pressure at the gastroesophageal junction to halt hemorrhage from bleeding esophageal varices. Some devices have a separate esophageal balloon and aspiration ports to handle gastric and esophageal secretions. There are three different types: the Sengstaken-Blakemore tube, the Minnesota tube, and the Linton-Nachlas tube. The balloons are inflated starting with the gastric balloon to tamponade any bleeding. Control of initial bleeding has been documented to range from 30-90%.57 The wide variation is thought to be due to multiple factors, including additional treatment modalities used and physician inexperience. In studies comparing balloon tamponade to somatostatin analogs the rate of initial bleeding control is equal or higher than the somatostatin analogs, but the rate of complications is also much higher.58,59 One of the major considerations when contemplating balloon tamponade is the need for a secure airway as patients may have difficulty clearing oral secretions. Rebleeding is a common problem particularly upon balloon deflation, and because of this balloon tamponade is used in general as a temporizing procedure until the patient can have more definitive treatment of either endoscopy or, if endoscopy has failed, surgery.30 Complications include mucosal ulceration, tracheal compression, aspiration pneumonia, esophageal and gastric rupture, and asphyxiation.
Surgery
Surgery is, in most cases, a last resort for treatment of upper GI bleeding, whether due to varices, peptic ulcer disease, or Mallory-Weiss tears. Patients who warrant early surgical consultation are those who present with clinical predictors of poor outcome (increasing age, co-morbid conditions, and markers for severe bleeding) and those who have failed pharmacological and endoscopic treatment. Surgical options for peptic ulcer disease are over-sewing of the artery plus truncal vagotomy and pyloroplasty, antrectomy, and gastrojejunostomy. Limited literature suggests that the elderly may benefit from earlier surgical intervention.27 Surgical options for varices are shunt procedures that partially or completely decompress the portal system, esophageal transaction, or devascularization of the gastroesophageal junction. Unfortunately, surgery, particularly in patients with severe cirrhosis, has a very high mortality approaching 80%. The definitive treatment for many patients with cirrhosis is liver transplant, but many people are not appropriate candidates, and with acute bleeding episodes, transplantation rarely is an option.30
Interventional Radiology
Another option for patients who have failed pharmacologic and endoscopic treatment and have a high risk of mortality with surgery due to comorbid conditions and severe cirrhosis is the transjugular intrahepatic portosystemic shunt (TIPS). The TIPS procedure involves the placement of a shunt to create a low-resistance channel between the hepatic vein and the intrahepatic portion of the portal vein using angiographic techniques. This reroutes blood flow in the liver and reduces portal hypertension. One study on patients who were poor surgical candidates used balloon tamponade as a bridge until the TIPS procedure could be performed. This study demonstrated that the TIPS procedure was able to stop all bleeding and had a six-week survival of 60% compared to only 10% expected survival for historical controls with surgery.60 When comparing TIPS with surgery in patients who are good or average surgical candidates, the limited studies have shown that the rate of rebleeding and long-term mortality was higher with TIPS.61 In general, TIPS is effective in stopping variceal bleeding that has failed more conservative treatment but there may be an increase in mortality. In addition, TIPS is associated with several complications including hepatic encephalopathy, TIPS thrombosis, stent retraction, and stent stenosis.30,33,55 As a result, the general consensus is that TIPS is probably best used as a temporizing measure until transplantation can be arranged.30
An alternative for patients with GI bleeding who are poor surgical candidates that continue to bleed despite pharmacologic and endoscopic treatment is the use of angiography with either intraarterial vasopressin or embolization. Intraarterial vasopressin is indicated for hemorrhagic gastritis, bowel with poor collateral blood supply, bleeding from endoscopy biopsy sites, and bleeding from anastomotic ulcers.62 Success rates for the use of vasopressin are approximately 70% for Mallory-Weiss tears and severe gastritis and 60% for other gastric lesions.62,63 Complications from vasopressin include bradycardia, water retention, hyponatremia, and rebleeding.62 Indications for embolization include failed intraarterial vasopressin, pyloroduodenal bleeding, and bleeding from endoscopy treatment sites. Embolization is a little more successful than vasopressin with approximately 80% control for Mallory-Weiss tears and gastritis and 65% for other gastric and duodenal lesions.62,64 Complications associated with embolization are hematomas, arterial thrombosis, dissection, embolism, pseudoaneurysm, and bowel infarction.
Gastric Varices
Gastric varices are discussed separately from esophageal varices because they tend to bleed more aggressively and their treatment is different mainly because endoscopic treatment is often not successful.30 There have been limited studies assessing injecting gastric varices with sclerosant and cyanoacrylate with cyanoacrylate showing some success, but it is not available in the United States.65 The general management for gastric varices is to treat with octreotide and balloon tamponade until either TIPS or surgery can be done.60,65
Conclusion
The diagnosis and management of upper GI bleeding is a complex process often involving multiple decisions and specialists. It important to keep ahead of the disease process and have a low threshold for resuscitation. The use of blood and blood products should be considered for those suspected of any major bleeding. It is important to be aware that those patients who are older and have multiple medical problems as well as those with previous or suspected liver disease are at greatest risk. Most non-esophageal bleeding will be self-controlled, although there is a significant risk of rebleeding. Esophageal bleeding should prompt the use of somatostatin analogs, and consideration of endoscopic treatment, if available. In the most severe cases when bleeding cannot be controlled by these efforts the patient may require balloon tamponade until surgical or interventional radiological intervention can be arranged.
References
1. Lewis JD, Bilker WB, Brensinger C, et al. Hospitalization and mortality rates from peptic ulcer disease and GI bleeding in the 1990s: Relationship to sales of nonsteroidal anti-inflammatory drugs and acid suppression medications. Am J Gastroenterol 2003;98:1896.
2. van Leerdam ME, Vreeburg EM, Rauws EA, et al. Acute upper GI bleeding: Did anything change? Time trend analysis of incidence and outcome of acute upper GI bleeding between 1993/1994 and 2000. Am J Gastroenterol 2003;98:1494-1499.
3. Jensen DM, Machicado GA. Diagnosis and treatment of hematochezia. Gastroenterology 1988;95:1569-1574.
4. Palmer ED. A vigorous diagnostic approach to upper gastrointestinal hemorrhage: A 23 year prospective study of 1400 patients. JAMA 1969;207:1477.
5. Jutabha R, Jenson DM. Management of sever upper gastrointestinal bleeding in the patient with liver disease. Med Clin North Am 1996;80:1035.
6. Hickey NC, Downing R, Hamer JD, et al. Abdominal aortic aneurysms complicated by spontaneous ileocaval or duodenal fistulae. J Cardiovasc Surg 1991;32:181-185.
7. Olcott C IV, Holcroft JW, Stoney RJ, et al. Unusual problems of abdominal aortic aneurysms. Am J Surg 1978;135:426-431.
8. Gregory P, Victorino MD, Felix D, et al. Does tachycardia correlate with hypotension after trauma? J Am Coll Surg 2003;196:679-684.
9. Witting MD, Gallagher K. Unique cutpoints for sitting-to-standing orthostatic vital signs. Am J Emerg Med 2003;21:45-47.
10. Schriger DL, Baraff LJ. Capillary refill—is it a useful predictor of hypovolemic states? Ann Emerg Med 1991;20:601-605.
11. De Bruyn G, Graviss E. A systematic review of the diagnostic accuracy of physical examination for the detection of cirrhosis. BMC Med Inform Decis Mak 2001;1:6.
12. Kass LE, Tien IY, Ushkow BS, et al. Prospective crossover study of the effect of phlebotomy and intravenous crystalloid on hematocrit. Acad Emerg Med 1997;4:198-201.
13. Elizalde JI, Clemente J, Marin JL, et al. Early changes in hemoglobin and hematocrit levels after packed red cell transfusion in patients with acute anemia. Transfusion 1997;37:573-576.
14. Ernst AA, Haynes ML, Nick TG, et al. Usefulness of the blood urea nitrogen/creatinine ratio in gastrointestinal bleeding. Am J Emerg Med 1999;17:70-72.
15. Rautelin H, Lehours P, Megraud F. Diagnosis of Helicobacter pylori infection. Helicobacter 2003; 8 Suppl 1:13-20.
16. Ransohoff DF, Lang CA. Screening for colorectal cancer. N Engl J Med 1991;325:37-41.
17. Fleischer DE, Goldberg SB, Browning TH, et al. Detection and surveillance of colorectal cancer. JAMA 1989;261:580-585.
18. Gogel H K, Tandberg D, Strickland RG. Substances that interfere with guaiac card tests: Implications for gastric aspirate testing. Am J Emerg Med 1989;7:474-480.
19. McDonnell WM, Ryan JA, Seeger DM, et al. Effect of iron on the guaiac reaction. Gastroenterology 1989;96:74-78.
20. Rosenthal P, Thompson J, Singh M. Detection of occult blood in gastric juice. J Clin Gastroenterol 1984;6:119-121.
21. Holman JS, Shwed JA. Influence of sucralfate on the detection of occult blood in simulated gastric fluid by two screening tests. Clin Pharm 1992;11:625-627.
22. Cappell MS. Safety and efficacy of nasogastric intubation for gastrointestinal bleeding after myocardial infarction: An analysis of 125 patients at two tertiary cardiac referral hospitals. Digestive Diseases and Sciences 2005;11:2063-2070.
23. Cuellar RE, et al. Gastrointestinal tract hemorrhage. The value of a nasogastric aspirate. Arch Intern Med 1990;150:1381-1384.
24. SD Lee, DJ Kearney. A randomized controlled trial of gastric lavage prior to endoscopy for acute upper gastrointestinal bleeding. J Clin Gastroenterol 2004;38:861-865.
25. Gallagher EJ. Nasogastric tubes: Hard to swallow. Ann Emerg Med 2004;44:138-141.
26. Bosch, J, Thabut, D, Bendsten, F, et al. Recombinant factor viia for upper gastrointestinal bleeding in patients with cirrhosis: A randomized, double-blind trial. Gastroenterology 2004;127:1123-1130.
27. Laine L, Peterson WL. Bleeding peptic ulcer. N Engl J Med 1994;331:717-727.
28. Leontiadis GI, Sharma VK, Howden CW. Systemic review and meta-analysis of proton pump inhibitor therapy in peptic ulcer bleeding. BMJ 2005;330:568-570.
29. Khuroo MS, Yatoo GH, Javid G, et al. A comparison of omeprazole and placebo for bleeding peptic ulcer. N Engl J Med 1997;336:1054-1058.
30. Sharara AI, Rockey DC. Gastroesophageal variceal hemorrhage. N Engl J Med 2001;345:669-681.
31. Imperiale TF, Teran JC, McCullough AJ. A meta-analysis of somatostatin versus vasopressin in the management of acute esophageal variceal hemorrhage. Gastroenterology 1995;109:1289-1294.
32. D'Amico G, Pietrosi G, Tarantino I, et al. Emergency sclerotherapy versus vasoactive drugs for variceal bleeding in cirrhosis: A Cochrane meta-analysis. Gastroenterology 2003;124:1277-1291.
33. Besson I, Ingrand P, Person B, et al. Sclerotherapy with or without octreotide for acute variceal bleeding. N Engl J Med 1995;333:555-560.
34. Villanueva C, Ortiz J, Sabat M, et al. Somatostatin alone or combined with emergency sclerotherapy in the treatment of acute esophageal variceal bleeding: A prospective randomized trial. Hepatology 1999;30:384-389.
35. Sung JJ, Chung SC, Yung MY, et al. Prospective randomized study of effect of octreotide on rebleeding from esophageal varices after endoscopic ligation. Lancet 1995;346:1666-1669.
36. Imperiale TF, Birgisson S. Somatostatin or octreotide compared with H2 antagonists and placebo in the management of acute nonvariceal upper gastrointestinal hemorrhage: A meta-analysis. Ann Intern Med 1997;127:1062-1071.
37. Gimson AE, Westaby D, Hegarty J, et al. A randomized trial of vasopressin and vasopressin plus nitroglycerine in the control of active variceal hemorrhage. Hepatology 1986;6:410-413.
38. Ioannou G, Doust J, Rockey DC. Terlipressin for acute esophageal variceal hemorrhage. Cochrane Database Syst Rev 2003;CD002147.
39. Pleym H, Stenseth R, et al. Prophylactic treatment with desmopressin does not reduce postoperative bleeding after coronary surgery in patients treated with aspirin before surgery. Anesth Analg 2004;98:578-584.
40. de Franchis R, Arcidiacono PG, et al. Randomized controlled trial of desmopressin plus terlipressin vs. terlipressin alone for the treatment of acute variceal hemorrhage in cirrhotic patients: A multicenter, double-blind study. Hepatology 1993;18:1102-1107.
41. Frossard JL, Spahr L, Queneau PE, et al. Erythromycin intravenous bolus infusion in acute upper gastrointestinal bleeding: A randomized, controlled, double-blind trial. Gastroenterology 2002;123:17-23.
42. Bernard B, Grange JD, Khac EN, et al. Antibiotic prophylaxis for the prevention of bacterial infections in cirrhotic patients with gastrointestinal bleeding: A meta-analysis. Hepatology 1999;29:1655-1661.
43. Hou MC, Lin HC, Liu TT, et al. Antibiotic prophylaxis after endoscopy therapy prevents rebleeding in acute variceal hemorrhage: A randomized trial. Hepatology 2004;39:746-753.
44. Fernandez J, Navasa M, Gomez J, et al. Bacterial infections in cirrhosis: Epidemiological changes with invasive procedures and norfloxacin prophylaxis. Hepatology 2002;35:140-148.
45. Riordan MD, Williams R. Treatment of hepatic encephalopathy. N Engl J Med 1997;337:473-479.
46. Mela M, Mancuso A, Burroughs A. Drug treatment for portal hypertension. Ann Hepatol 2002;1:102-120.
47. Jiranek GC, Kozarek RA. A cost effective approach to the patient with peptic ulcer bleeding. Surg Clin North Am 1996;76:83-103.
48. Grace ND. Diagnosis and treatment of gastrointestinal bleeding secondary to portal hypertension. American College of Gastroenterology Practice Parameters Committee. Am J Gastroenterol 1997;92:1081-1091.
49. Bjorkman DJ, Zaman A. Urgent vs. elective endoscopy for acute non-variceal upper-GI bleeding: An effectiveness study. Gastrointest Endosc 2004;60:1-8.
50. Bleau BL, Gostout CJ, Sherman KE, et al. Recurrent bleeding from peptic ulcer associated with adherent clot: A randomized study comparing endoscopic treatment with medical therapy. Gastrointest Endosc 2002;56:1-6.
51. Park CH, Joo YE, Kim HS, et al. A prospective, randomized trial comparing mechanical methods of hemostasis plus epinephrine injection to epinephrine injection alone for bleeding peptic ulcer. Gastrointest Endosc 2004;60:173-179.
52. Bianco MA, Rotondano G, Marmo R, et al. Combined epinephrine and bipolar probe coagulation vs. bipolar coagulation alone for bleeding peptic ulcer: A randomized controlled trial. Gastrointest Endosc 2004;60:910-915.
53. Lau JY, Sung JJ, Lam YH, et al. Endoscopic retreatment compared with surgery in patients with recurrent bleeding after initial endoscopic control of bleeding ulcers. N Engl J Med 1999;340:751-756.
54. Prandi D, Rueff B, Roche-Sicot J, et al. Life-threatening hemorrhage of the digestive tract in cirrhotic patients. Am J Surg 1976;131:204-209.
55. Steigmann GV, Goff JS, Michaletz-Onody PA, et al. Endoscopic sclerotherapy as compared with endoscopic ligation for bleeding esophageal varices. N Engl J Med 1992;326:1527-1532.
56. Grace ND, Groszmann RJ, Garcia-Tsao G, et al. Portal hypertension and variceal bleeding: An AASLD single topic symposium. Hepatology 1998;28:868-880.
57. Hunt PS, Korman MG, Hansky J, et al. An 8-year perspective experience with balloon tamponade in emergency control of bleeding esophageal varices. Dig Dis Sci 1982;27:413-416.
58. Avgerinos A, Klonis C. A prospective randomized trial comparing somatostatin, balloon tamponade and the combination of both methods in the management of acute variceal haemorrhage. J Hepatol 1991;13:78-83.
59. Zaman A. Current management of esophageal varices. Current Treatment Options in Gastroenterology 2003;6:499-507.
60. Sanyal AJ, Freedman AM, Luketic VA, et al. Transjugular intrahepatic portosystemic shunts for patients with active variceal hemorrhage unresponsive to sclerotherapy. Gastroenterology 1996;111:138-146.
61. Rosemurgy AS, Bloomston M, Clark WC, et al. H-Graft portacaval shunts versus TIPS: Ten-year follow-up of a randomized trial with comparison to predicted survivals. Ann Surg 2005;241:238-246.
62. Kruskal J. Angiographic control of gastrointestinal bleeding. Up to Date 2005;14.3.
63. Rahn NH, Tishler JM, Han SY, et al. Diagnostic and Interventional angiography in acute gastrointestinal hemorrhage. Radiology 1982;143:361-366.
64. Lieberman DA, Keller FS, Katon RM, et al. Arterial embolization for massive upper gastrointestinal tract bleeding in poor surgical candidates. Gastroenterology 1984;86:876-885.
65. Sanyal AJ. Treatment of active variceal hemorrhage. Up to Date 2006; 14.3.
Very little else evokes the visceral response of watching someone vomit blood. Emergency physicians deal with bleeding in many arenas, but gastrointestinal (GI) bleeding is one type that in the past has offered limited options for treatment in the emergency department.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.