Evaluation of the Acutely Injured Knee in the ED: Diagnosis and Treatment: Part I
Evaluation of the Acutely Injured Knee in the ED: Diagnosis and Treatment: Part I
Authors: Gary D. Hals MD, PhD, Attending Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC; Steven Cruea, MD, Resident Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC; and Dusty Moses, MD, Resident Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC.
Peer Reviewers: Andrew D. Perron, MD, Residency Program Director, Maine Medical Center, Portland; and Timothy C. Stallard, MD, Program Director, Emergency Medicine Residency, Texas A&M Health Sciences Center, Scott & White Hospital, Temple, TX.
Often, the emergency department (ED) evaluation of a painful knee is a quick x-ray followed by discharge with the limb placed a knee immobilizer, the patient walking with crutches, a prescription for analgesics, and a referral to see an orthopedist next week. This tends to be unsatisfying to the patient, who would like more information about what is causing the problem and it should also be intellectually unsatisfying to the emergency physician. An understanding of the knee anatomy, relevant biomechanics, mechanism of injury, and a directed physical examination will lead to a more accurate diagnosis, better communication with the patient, and more appropriate referral. This issue of Emergency Medicine Reports starts a two-part series on knee injuries and conditions relevant to the practice of emergency medicine.
I would like to point out to the reader that arthrocentesis of the knee is a topic for the 2008 Lifelong Learning and Self-Assessment (LLSA) reading list created by the American Board of Emergency Medicine.
—J. Stephan Stapczynski, MD, Editor
Introduction
The knee joint is the largest joint in the body, and its biomechanics are surprisingly complex. The knee must endure substantial stress, up to several times the body's weight, during simple activity such as walking downhill. Athletic activity can push the knee to its limits, and even non-contact sports are a frequent source of knee injury. An estimated 3 million knee injuries occur yearly in North America, resulting in roughly 1 million patients presenting to emergency departments (EDs) annually with acute knee injuries.1 These visits represent approximately 2.5% of the patients seen in EDs every year for injuries, and account for up to 33% of all musculoskeletal complaints in the primary care setting.2
Besides fractures and dislocations, soft-tissue injuries of the knee are common, and include damage to menisci, ligaments, and tendons. Most of these injuries occur in younger patients as a result of sports or other physical activity, and correct diagnosis and treatment is essential to minimize disability, time lost from work, and to preserve function. Unfortunately the emergency physician may not be able to accurately diagnose all of the various soft-tissue injuries, as magnetic resonance imaging (MRI) is necessary to fully define the extent of damage in many cases. Further, knee pain in the pediatric patient may falsely draw the physician's attention away from the true source of pathology: the hip joint.
Knee pain also may present from a variety of systemic diseases that affect its large synovial surface. In some cases knee pain may be the primary issue, such as gout or degenerative arthritis, or it may be part of a larger problem as in sarcoidosis or rheumatoid arthritis. While many of these conditions are not true emergencies, one must remain vigilant for the case of septic arthritis hidden among the other many patients with acutely symptomatic knees. This article will review relevant anatomy and discuss bony as well as soft-tissue injuries commonly seen in ED patients presenting with acute knee pain. Non-traumatic sources of acute knee pain as well as knee pain in the pediatric patient will also be discussed. Be aware that when knee immobilization is recommended as treatment in the text, this refers only to a brief (2-4 day) period of immobilization. Long-term joint immobilization is associated with muscle atrophy, ligament stiffness, lost range of motion, etc., and should be avoided when possible. Ideally, the patient using a knee immobilizer should be re-evaluated by the orthopedist or other appropriate specialist during this initial time period.
Relevant Knee Anatomy
The knee joint is a modified hinge joint whose bones, ligaments, and cartilage all may be a source of knee pain or injury. The healthy knee has a range of motion between 0° and 135° of flexion. At full extension, the healthy knee joint does not rotate, but during flexion, up to 30° of external rotation is possible, generally when the knee is around 90° of flexion.3 The knee joint is composed of three bones: the distal femur, proximal tibia, and the posterior patella. The proximal fibula is not actually part of the knee joint, but does aid joint stability by acting as an attachment base for supporting ligaments (lateral collateral and iliotibial band). The patella is the largest sesamoid bone in the body. The patella acts to both protect the quadriceps tendon and to increase its mechanical advantage during knee extension.
Knee joint stability is provided strictly through the ligaments found in and around the knee. As tension is placed on the ligaments during knee extension, they tighten and provided increasing stability. Of these ligaments, the anterior cruciate (ACL), posterior cruciate (PCL), medial (MCL), and lateral collateral (LCL) ligaments are the primary knee stabilizers. The collateral ligaments stabilize the outer knee and prevent lateral movements, while the cruciate ligaments stabilize the knee from the inside and prevent movement in the anterior/posterior (A/P) directions. The MCL attaches at the medial femoral epicondyle and the medial tibia, and resists valgus stress. Valgus stress refers to lateral movement of the knee by widening of the medial side of the knee joint ("knock-kneed"), where varus stress refers to lateral movement by widening the lateral side of the knee joint ("bow-legged"). Current belief is that the MCL provides most of its stability when the knee is flexed, and thus most MCL injuries occur while the knee is flexed as opposed to extended. The LCL is the corresponding ligament on the lateral knee. It attaches to the lateral femoral epicondyle and the lateral tibia and fibular head. LCL injuries are less common than MCL injuries, but often are more disabling as the lateral stabilizers provide more stability during normal gait than their medial counterparts.
The ACL is probably the most familiar knee ligament, as it is commonly injured necessitating surgical repair for many patients. The PCL is the stronger of the two ligaments with a cross-sectional area 1.5 times that of the ACL as well as higher tensile strength.3 Thus the PCL is roughly twice as strong as the ACL, and injured much less often. The ACL attaches to the anterior tibial plateau and the posterior intercondylar notch of the femur. It limits forward movement of the tibia relative to the femur. The PCL attaches to the posterior tibial plateau and the anterior intercondylar notch. It limits backward movements of the tibia. Together they form an "X" or "cross" inside the joint, hence the name "cruciate" ligaments.
Two types of cartilage are found in the knee: stabilizing and articular cartilage. The medial and lateral menisci are the stabilizing cartilage, while the smooth cartilage lining the surfaces of the patella, tibia, and femur are the articulating cartilages. The menisci lie in the joint and function as padding or "shock absorber" between the two articular surfaces. As the tibial plateau is relatively flat and the femoral condyles more rounded, the menisci are necessary to fill in the space that would otherwise exist between the articular surfaces in the knee. The menisci are stabilized by their attachments to other knee structures, such as the MCL, ACL, and PCL. Meniscal injuries are common and can occur in isolation or in association with injury to knee ligaments.
The neurovascular supply for the knee is found in the popliteal fossa, a depression on the posterior aspect of the knee formed by the muscle attachments of the gastrocnemius, semitendonosis, semimembranosis, and biceps femoris. The popliteal artery is a deep structure in the popliteal fossa, located just outside the posterior knee joint capsule. It is tightly attached at its proximal and distal entrances to the popliteal fossa, which explains the high incidence of arterial injury seen in knee dislocations. The popliteal artery is a continuation of the femoral artery and just below the knee joint, divides into the anterior and posterior tibial arteries. Geniculate branches arise from the popliteal artery, form an anastomosis around the knee, and are the direct blood supply of the knee.
The innervation of the knee is from the tibial nerve and one of its branches, the common peroneal nerve. The tibial nerve arises as a continuation of the sciatic nerve and gives off the common peroneal nerve in the proximal popliteal fossa. The common peroneal nerve travels around the head of the fibula, and can be injured with fractures of the proximal fibula. Common peroneal nerve injury also can occur by prolonged pressure on the lateral knee, as seen with casting that is too tight. Common peroneal nerve injures manifest as foot drop, or weakness of ankle dorsiflexion. Tibial nerve injuries are less common than popliteal artery injuries with knee dislocation, as the nerve is more loosely attached proximally than the artery.
ED Evaluation
Clinical Presentation. When a patient presents with acute knee pain, certain historical features should be sought as they can be indicative of specific injury patterns or diseases, and may help the emergency physician arrive at the correct diagnosis. (See Table 1.) In the case of traumatic knee pain, defining the mechanism of injury may be the most vital information to obtain, as the direction of force applied to the knee produces predictable injury patterns. Although not possible in every case, try to have the patient describe the position of the knee (flexed or extended) and the direction of force applied to the knee. For example, when a football player is struck on the lateral knee (valgus force) with the knee flexed, the MCL is likely to be injured. Direct blows to the anterior knee are common during motor vehicle accidents (knee hitting the dashboard) or from falls onto the knee. Besides obvious patella fractures, these injuries can also produce PCL tears. In most cases of PCL injury, a significant joint effusion will develop in a few hours, whereas isolated patella fractures are usually not associated with large joint effusions. In many cases the injury occurs in the absence of physical contact such as ACL tear with hyperextension, or during a "cutting" maneuver (rapid direction change while running).
| Table 1. History and Physical Examination Features and the Pathology They Suggest |
|
Rapidly evolving effusions are also associated with fractures as well as ACL ruptures. Most patients with fractures or ACL ruptures will report hearing or feeling a "pop" at the time of injury. Thus if the radiograph shows no fracture when swelling and pain suggested its presence, an ACL tear is likely. The time it takes for the effusion to develop can also be helpful. Significant fractures usually will produce effusion in minutes, whereas with smaller fractures or ACL injuries effusions generally develop over a period of 4 hours.4 Be aware, though, that tears of the joint capsule will allow leakage of joint fluid into the surrounding leg tissue, and the patient can present with little or no effusion.
Patients may complain of knee "locking" or inability to fully extend the joint. This often is indicative of meniscal injury, but can also be caused by a loose body in the joint. Meniscal injuries often occur from rotational forces, with medial menisci injuries occurring more frequently than lateral ones. Menisci have no pain fibers, but swelling of the joint will produce pain. Joint effusions in meniscal injuries often occur more slowly, over a period of 12-24 hours.
With non-traumatic knee pain, history of previous episodes, involvement of other joints, past medical history, and medications can all have relevance. Attempts to determine the duration of symptoms, association with swelling, decreases in range of motion, and progression with time should be made. Table 1 summarizes key historical points and the pathology they suggest.
Physical Examination. A thorough knee examination has several components. Palpation to localize tenderness, noting effusion, determining range of motion (ROM), and stability testing are all part of a complete examination. However, there are many cases where one should not perform stability testing without initial imaging and/or pain management first. When fracture is suspected (significant deformity) or pain is great, one should have plain films taken and pain addressed before attempting to manipulate the knee. This can also allow time to pass so the patient may be calmer and more cooperative with the examination. Although ligamentous injury can co-exist with knee fractures, stressing a fractured knee inflicts unnecessary pain and can worsen ligament damage. Even after ruling out fracture and with pain control, many patients will not tolerate full stability testing in the acute situation. When this occurs, the best option is to immobilize the knee and arrange short-term (2-3 days) follow-up with an orthopedist to re-examine the knee. It should be clearly documented in these cases that a complete examination was not possible, that the patient was informed that ligamentous injury has not been ruled out, and the importance of timely follow-up was stressed. In fact, obtaining close follow-up for re-examination is useful in any patient with knee injuries deemed at risk of ligamentous damage whether or not it can be documented in the ED.
Examination of the knee should be done with the patient supine and with both knees exposed. Comparison to the unaffected knee should be done in most cases, as more subtle changes are easier to appreciate when looking at both knees. Smaller effusions are also more evident with the knee fully extended. Some patients initially may be seated with their feet off the side of the bed or in a wheelchair. Although they should be moved to a supine position for examination, this is a good position for determining ROM. The knee's normal ROM is from 0° to 135° of full flexion. ROM testing should include active extension or straight leg raising. Inability to hold the knee in active extension is sometimes the only evidence of a quadriceps or patellar tendon rupture. This should not be confused with locking of the knee where the knee is unable to be extended by the patient or the examiner, indicating meniscal entrapment or loose body in the joint.
Palpation of the knee should begin, as with any painful area, as far away as the reported site of pain. Locating precise areas of tenderness can help one raise suspicion for injury to a particular structure. Tenderness medially suggests MCL damage or medial meniscus pathology; patellar tenderness indicates possible fracture, where infrapatellar tenderness may suggest patellar tendon injury or tendinitis, and so on. More subtle effusions can be detected by noting fullness in the normally hollow areas just on either side of the patella. Ballottement of the patella can also be performed to detect smaller effusions. To accomplish this, one applies pressure to the tissues just superior to the patella. While doing so, use the other hand to push directly down on the patella. When no effusion is present, the patella will be sitting directly over the femoral condyle and will not move with downward pressure. With joint effusion, the pressure applied proximal to the patella will lift it off the condyle and one will feel the patella tapping against the femoral condyle. Table 1 summarizes key physical findings and the pathology they suggest.
Stability Testing. A variety of different maneuvers have been used to assess ligament stability. They allow one to detect tears in the primary stabilizing ligaments of the knee, the cruciate and collateral ligaments, as well as for meniscal injury. These tests involve applying gentle, firm pressure to identify excessive joint motion. Healthy, intact ligaments will give hard end points during these tests and resist abnormal joint motion. Soft or vague end points in motion or excessive motion indicate positive test results. Tests should always be performed on both injured and uninjured knees to control for natural variability in each patient. Do not use sudden forceful movements in these testing maneuvers, as they may cause reflexive muscular contraction that produces false negative results. As mentioned above, they may be inconclusive when performed in the acute setting when pain and initial swelling make stability maneuvers more difficult. If the patient cannot tolerate the maneuvers after adequate pain control, they are of little benefit as treatment of immobilization and follow-up will not be altered. They are also contraindicated and may be damaging in patients with acute knee fractures. However, they can be useful when patients present with sub-acute or more chronic problems from previous trauma as they can identify patients with significant ligament damage without expensive magnetic resonance imaging (MRI).
Stability Tests: ACL/PCL. The Lachman's test, anterior drawer test, and pivot shift test all assess ACL stability, while the posterior drawer examines PCL integrity. The Lachman's test is the only ACL stability test that should be used in the acute setting.1 One study found the Lachman's test was 99% accurate compared to 70% for the anterior drawer test in the acute setting.5 The anterior drawer test is more useful for evaluating chronic injuries, as joint effusion and hamstring spasm can cause many false-negative results.6 Although the pivot shift test is very specific for ACL injury6 it is not very sensitive. It generally is not recommended in the acute setting as it can actually worsen ligament tears.1
Lachman's Test. This test was introduced in the 1970s7 and again, is the only test of ACL stability recommended for use in setting of acute knee injury.1 However, the test may not reliably detect partial ACL tears. To perform this test, place the patient supine and flex the knee to between 20°-30°. Use one hand to stabilize the patient's thigh just superior to the patella. Place the other hand above the calf and gently but firmly try to move the tibia forward. With an intact ACL, a firm endpoint to anterior pull of the tibia is felt, where a torn ACL will allow noticeably more movement of the tibia anteriorly compared to the unaffected knee and/or produce a soft end point. Subtle movement of the tibia also can be seen when the normal concave profile of the knee between the patella and anterior tibial tuberosity is lost during the test. A grading system can be used with 1+ (0-5 mm displacement greater than the normal side), 2+ (5-10 mm), and 3+ (> 10 mm) levels. False-positive results can be produced by PCL rupture, which allows the tibia to start at an abnormal posterior position and the anterior movement detected is just the return to normal joint position. False negatives also can be meniscal tears, hamstring spasm, or third-degree MCL tears.1 Also one must stabilize the femur to detect the forward tibial motion, and in patients with large thighs this may be difficult.
Anterior Drawer Test. Use this test to confirm ACL instability in the subacute (> 2 weeks post-injury) or chronic setting. As with the Lachman's test, the patient is placed supine, but the hip is held in 45° flexion and the knee in 90° flexion. The examiner sits on the patient's foot to stabilize the position of the leg. Grasp behind the knee with both hands but align both thumbs along the anterior knee joint line, and gently pull forward. The amount of movement is compared to the unaffected knee. As with the Lachman's test, false positives can arise from PCL rupture and false negatives from hamstring muscle contraction due to pain and effusions that can limit flexion to less than 90°.
Pivot Shift Test. Although the pivot shift test or "jerk test" is the most sensitive test to detect ACL tears, it generally is not recommended for evaluation of acute injuries as one can further damage the ACL by performing this test. It is performed with the patient supine and the knee in full extension. The tibia is internally rotated and valgus stress is applied to the knee. In patients with ACL damage, this will cause mild subluxation of the knee. The knee is then flexed to 20-30°, which causes relocation.
Posterior Drawer Test. The posterior drawer is the most accurate test of PCL integrity, with a sensitivity of 90% and specificity of 99%.3 The patient is positioned in similar fashion to the anterior drawer test with hip flexed to 45° and knee flexed to 90°. The examiner sits on the top of the patient's foot to stabilize it. Place one hand on each side of the knee with the thumbs on the joint line. Gentle, firm force is directed backward on the proximal tibia with greater than 5 mm posterior displacement of the tibia constituting a positive test. As with the anterior drawer test performed on a knee with PCL instability, false-positive results can be obtained from the posterior drawer test in patients with ACL tears. Care should be taken to compare the results to the opposite knee.
Posterior Sag Test (or Godfrey Test). This maneuver can help distinguish which cruciate ligament is damaged in cases where knee instability makes results of drawer tests inconclusive. The patient is placed supine with both hips and knees flexed to 90°. The examiner supports both heels with their hands and posterior translation or "sag" compared to the opposite side confirms PCL instability.
Stability Tests: MCL/LCL. MCL and LCL stability can be established as follows. Place the patient supine and examine the knee in both 0° and 30° of flexion. The knee is stressed with both valgus and varus pressure, and one looks for opening of the side of the knee joint. Abnormal joint opening will be palpable as a space appearing between the femur and tibia. Joint opening is estimated in millimeters or by mild, moderate, and complete laxity (grades 1-3). Instability in full extension implies not only full collateral ligament tear, but also damage to other knee stabilizers (ACL and/or PCL). If instability is only seen at 30° flexion, this implies that only the collateral ligaments are affected as stability from cruciate ligaments is absent in flexion. To assess the MCL, place one hand on the lateral knee and grasp the ankle with the other hand. Apply lateral force on the ankle and attempt to open the medial aspect of the knee (valgus stress). To assess the LCL move the hand to the medial knee. Apply force in the opposite direction on the ankle trying to open the lateral aspect of the knee (varus stress).
Stability Tests: Menisci. McMurray's Test. McMurray's test can identify meniscal injuries, but in the acute setting the patient may not tolerate the ROM needed and the test may produce false-negative results. Position the patient supine with the knee in maximum flexion. To examine the medial meniscus, hold the knee over the medial meniscus with one hand and grasp the foot with the other. Externally rotate the knee as far as possible and then carefully extend the knee. As the femur moves over the medial meniscus, tears will produce audible/palpable clunks or clicks along with pain. Place one hand over the lateral meniscus and internally rotate the knee to test the lateral meniscus.
Apley's Test. Apley's test also evaluates meniscal injuries. Place the patient prone with the knee flexed to 90°. Internally and externally rotate the knee while applying pressure to the heel. Pain and/or crepitus with heel pressure and rotation indicates meniscal damage.
Imaging Tests. The knee can be imaged with plain films, computed tomography (CT) scan, MRI, and ultrasound. Each modality has its advantages. Plain films can be helpful to quickly screen for fractures, CT scans provide detailed information on bony injuries, MRI provides the best soft-tissue imaging, and ultrasound can evaluate vascular integrity. Laboratory tests used in evaluating other causes of knee pain (i.e., infectious, systemic diseases) will be discussed in the section on acute non-traumatic knee pain.
Plain Radiographs. In the past, the standard was to obtain plain films on virtually every patient with acute knee trauma. A large number of films are taken, but only on average only 15% show positive findings.8 Like with ankle injuries, criteria have been sought that allow one to minimize the number of unnecessary studies without meaningful risk of "missing a fracture." The Ottawa9 and Pittsburgh10 criteria are the two most familiar and well studied of these decision aids. (See Table 2.) Both have been shown to detect nearly 100% of fractures.8,11,12 Likewise, they have been shown to reduce the number of knee films taken in the ED by at least 23% (Ottawa) and 53% (Pittsburgh).13 However accurate, both criteria will miss about 1% of fractures on average.13 The most commonly missed fractures include fractures of the patella, non-displaced tibial plateau, and fibular head, especially in elderly patients. Several authors pointed out malpractice lawsuit fears as a difficulty in implementing these criteria in practice.14,15 Many patients expect to have an x-ray if injured, and physicians often comply with this request even when no indication exists.15 Given that a "missed fracture" is one of the most common reasons for lawsuits in emergency medicine, without legal protection from "missed fractures" that such criteria will cause, true reduction in ED x-rays using these criteria will remain variable.
| Table 2. Ottawa and Pittsburgh Knee Criteria for Patients with Acute Knee Trauma9,10 |
|
Besides confirming obvious fractures, plain films can provide evidence for other pathology as well. An anteroposterior (AP) and lateral view are standard for most plain knee films, but sunrise views can be added to isolate the patella. Unilateral joint widening is a sign of ligamentous instability. Oblique films are useful to highlight tibial plateau fractures when not seen on AP views or to show loose bodies in the joint. Effusions can be seen on lateral films and can be a clue for hard-to-see tibial plateau fractures or for ACL tears. A high (patella ala) or low-riding patella (patella baja) can be more easily seen on lateral films and indicates patellar or quadriceps tendon injury. Ligamentous injuries are not seen on plain films, but sometimes one can see bony fragments associated with avulsion of the ligament attachment (tibial spine, femoral condyles).
CT Scan. CT scan of acute knee injuries often is performed in consultation with the orthopedist in preparation for surgical repair of knee fractures, most often for distal femur or tibial plateau fractures. CT scan also may be used to detect subtle tibial plateau fractures when suspicion is high and plain films are non-diagnostic.
MRI. MRI has replaced arthrograms as the standard evaluation for ligament injury. Arthrograms are performed by injection of radiopaque dye (and usually air called "double contrast") into the knee joint to better define injury of menisci, cruciate ligaments, and articular cartilage. In cases where MRI is contraindicated or is unable to be performed (i.e., claustrophobia, obesity), they still may be used. Recently arthrography combined with CT scan has been suggested as an alternative to MRI and provides a more detailed study than with plain films.16
MRI has the advantage that one study can evaluate all the knee ligaments as well as the menisci and articular cartilages. MRI is highly sensitive and specific (both around 95%)3 for ACL or PCL injury. Meniscal tears, collateral ligament damage, occult tibial plateau fractures, bone contusions, and avulsion of iliotibial band attachments can also all be identified on MRI. Thus MRI is very useful in pre-operative planning as all injuries are known beforehand. Further advantages of MRI are that it is non-invasive and does not expose the patient to radiation.
While MRI is essential for complete diagnosis and surgical planning of ligamentous injuries, in most cases it will not be part of the ED work-up. If no fractures are present, many ligament injuries can be diagnosed clinically through stability testing. However, on the day of the injury, acute pain and swelling often preclude full stability testing of the injured knee. Regardless of whether one can establish ligament injury in the ED at the time of injury, the patient likely will be sent home in an immobilizer and using crutches for orthopedic follow-up. Thus benefits of obtaining an MRI in the ED on the day of the injury will not normally outweigh the added cost of an "emergent" MRI. Further, using MRI as a screening tool for ligament injury in the ED will undoubtedly result in excessive scans and avoidable expenses for the patient. This is supported by a recent study designed to test the idea of using a truncated MRI study instead of plain film studies on patients with acute knee injuries. As expected, MRI accurately identified injuries requiring further treatment, but did not improve identification of patients who can be discharged without further follow-up.17
Ultrasound. While ultrasound has long been used in identification of Baker's cyst rupture and popliteal artery aneurysm, it recently has been expanded for diagnosis of ligamentous and meniscal injury.18 Ultrasound also has been used to characterize effusions as hemorrhagic or inflammatory, as well as to identify intra-articular bodies. It has been used to detect quadriceps or patellar tendon rupture,19 and except for tendon attachment injuries, ultrasound is believed by some to be superior to MRI.18 Although the menisci and cruciate ligaments sometimes may be evaluated with ultrasound, MRI remains the gold standard for diagnosis of these injuries.18
Arthrocentesis. As the knee is the largest joint in the body, it easily contain 50-70 mL or more of fluid. Thus it is the least difficult joint of the body from which to obtain fluid. Aspiration of fluid can be diagnostic as bloody fluid will confirm intra-articular injury from ligament tears, or confirm joint penetration from gun-shot wounds that may not appear to have entered the joint space. The presence of fat droplets (presence of bone marrow) will also confirm subtle intra-articular fractures. In the case of septic arthritis, joint fluid cultures are essential to accurately identify bacteria and guide antibiotic therapy.
Arthrocentesis also may be therapeutic to relieve the pain of a large, tense joint effusion, or to provide pain relief from intra-articular injection of analgesic compounds. Traditionally bupivacaine (10 mL of 0.25%) was injected for pain relief, but morphine (1-5 mg in 30 mL normal saline) has been shown to provide relief for up to 24 hours.20 More recently ketorolac (Toradol 60 mg in 10 mL normal saline) has been shown to be more effective than either bupivacaine or morphine alone in postoperative cases.21
While therapeutic arthrocentesis is a widely used practice and the benefits of pain relief can be substantial, there is a risk of introducing infection when performing arthrocentesis. Further, the literature is unclear on whether this procedure benefits healing. Some suggest hemarthrosis in an otherwise healthy joint will spontaneously clear without complication while others suggest that drainage will improve healing and should be performed.22 Since no clear consensus exists, one approach would be to use joint aspiration for pain relief in patients with large hemarthroses or those who do not have adequate pain control with analgesics. Strict sterile technique always should be used to help reduce risk. Consultation with the orthopedist who will see the patient in follow-up also can be helpful if one is unsure whether to perform arthrocentesis for pain control.
To perform arthrocentesis, the knee can be in full extension or the joint space opened by flexing to about 15°-20° with a pad underneath the knee. Take care to have the patient relax the quadriceps muscle, as this makes entering the joint space easier. Thoroughly clean the skin with Betadyne or other antiseptic, and drape with sterile towels to reduce risk of infection. Use of lidocaine to anesthetize the skin and joint capsule before aspiration can greatly ease pain of the procedure and aid patient relaxation.
The most common entry point is found by palpating the middle or superior edge of the patella on the medial side of the knee. Insert the needle about 1 cm medial to the edge of the patella. When no effusion is present, this area corresponds to the small depression on the medial knee felt just below the mid-patella. When an effusion is present, the small depression will be replaced by a bulge as the effusion fluid fills this space. Alternatively, the superolateral approach can be used. Here, one enters the suprapatellar pouch from the lateral aspect of the superior patellar margin. This pouch is greatly expanded by large effusions and it can be easier to obtain fluid using this approach.
A plain 18 gauge needle (or IV catheter/needle combination) is inserted until fluid is obtained. Take care not to touch articular surfaces with the needle. As this cartilage contains no pain fibers, one can damage the articular cartilage but not be aware of the injury. Even a 20 or 30 mL syringe will not be large enough to drain larger effusions, and one should be prepared to change syringes to fully drain the effusion. Take care to avoid movement of the needle inside the joint space during syringe changes. Have an assistant use a hemostat to hold the needle hub still while changing syringes, or use a stopcock to aid syringe changes. Synovial fluid can be sent for a variety of tests depending on the clinical situation. (See Table 3.) When a hemarthrosis is drained, no laboratory testing of the fluid is necessary. General laboratory studies on synovial fluid include: cell count with differential, crystal analysis, Gram stain, bacterial culture, and glucose level.
Trauma
Fractures. Distal Femur Fracture. The majority of femur fractures involve the proximal end (hip fracture) or shaft of the femur, while only 4% of femur fractures are distal femur fractures.1 These can be devastating injuries, as they often are comminuted, unstable, and complications of malunion/nonunion and infection are not rare.23 Further, distal femur fractures typically are not isolated problems for the patient. These fractures are most common in elderly women (> 75 years) whose co-morbid conditions make treatment difficult, or in younger males (15-24 years) with multiple trauma.23 In the older patient, a simple fall on the flexed knee while walking is a common mechanism of injury. The younger patient often is the victim of high-energy trauma, such as high-speed motor vehicle accident (MVA) or fall from a height.
Patients present with knee deformity and history of trauma, and plain films normally are adequate to identify the fracture and rule out other diagnoses such as knee dislocation. Any open wound other than abrasions near a fracture should prompt evaluation for an open joint injury. A common approach is to inject methylene blue into the knee joint and look for any leakage of methylene blue from the wound(s). Typically, 1-2 mL of methylene blue is mixed with 60 mL of sterile saline and injected into the joint using a similar technique to joint aspiration. (See section on arthrocentesis.) One can substitute a similar volume of 1% lidocaine for 10-20 mL of the sterile saline to aid in pain control. The larger volume of fluid is necessary to ensure accurate detection of smaller joint penetrations. Detection of blue fluid in any wound around the knee joint confirms intra-articular communication. In cases where no open joint injury is found, one should try to pull out as much fluid as was injected, although the full amount often may not be aspirated.
Distal femur fractures can be classified as supracondylar (metaphyseal) or intracondylar (epiphyseal). The most commonly used classification system is by Müller,24 which divides the fractures into three types: type A are extra-articular fractures, type B are unicondylar, and type C are bicondylar. As with most medical classifications, type A are less severe fractures with better prognosis, and type C are worse fractures with the least hope of a good outcome. The more comminuted the fracture the more difficult it will be to restore anatomic alignment. Patients are admitted and most are treated with open reduction and internal fixation (ORIF) using a combination of screws and plates. (See Table 4.)
| Table 4. Conditions that Require Orthopedic Consultation in the ED |
|
As one might expect, ligamentous injuries commonly complicate these fractures and are seen in at least 20% of cases.25 However, they are seldom diagnosed in the ED as the unstable nature of the fracture precludes ligament stability testing. Proximal tibia injuries (plateau fractures) and popliteal artery injuries also can complicate distal femur fractures. Dislocations can be present with distal femur fractures. (See section on complex knee fractures.) Any patient with suspected arterial injury resulting from distal femur fracture requires immediate orthopedic consultation. (See section on popliteal artery injury in knee dislocation.)
Tibial Plateau Fractures. The proximal tibia flattens out into a "plateau" formed by medial and lateral condyles. Tibial plateau fractures account for only 1% of all fractures,26 but like distal femur fractures they occur predominately in two groups of patients: young adults and the elderly. The mechanism of injury in young adults is from higher energy forces, such as motor vehicle accident or fall from significant height. They also are seen resulting from lower energy causes, such as sports injuries and by a car bumper striking the leg at low speeds. As expected, the young adult often has multiple injuries and plateau fractures may be open. In elderly patients, less violent trauma such as simple falls while walking is the more common cause. However lower bone density and strength puts elderly patients at much higher risk for plateau fractures, and they make up 8% of fractures in the elderly.26
Plateau fractures can vary from a subtle depression of one condyle to comminution and displacement of both condyles. The Schatzker classification is used by most consultants to describe plateau fractures. Lateral plateau fractures occur more commonly and are seen in up to 70% of cases, whereas medial injuries occur in up to 23% of cases.26 Both plateaus are fractured in 11-31% of cases.26 The Segond fracture occurs when the lateral capsular ligament avulses a small portion of the lateral tibial plateau. The injury often is seen in children and younger adults resulting from sports-related trauma. It is important to recognize as it is a marker of ACL disruption,27 but often can be missed on plain films when the avulsion fragment is small. Although much less common, a similar injury of the medial plateau, called the "reverse Segond," also occurs and is associated with PCL tears.28
Patients with plateau fractures often present in a similar fashion to those with pure ligament injuries: history of trauma, inability to bear weight, rapidly developing effusion (hemarthrosis), and decreased range of motion. Neurovascular integrity should be documented in every case, as popliteal artery and peroneal nerve injuries are not rare. Plain films will diagnose displaced fractures accurately, but non-displaced fractures can easily be missed. Be aware that non-displaced plateau fractures (in addition to tibial spine fractures) were the most commonly missed fracture using Ottawa or Pittsburgh rules.13 CT or MRI scan can be used to identify non-displaced fractures in cases with high suspicion of injury. CT scan will also be useful in more severe injuries to guide pre-operative planning.
Treatment of tibial plateau fractures varies from long leg casting with non-weight bearing for 8-10 weeks, to open reduction internal fixation (ORIF). As expected, stable non-displaced fractures are treated conservatively. Displaced or unstable fractures will require operative repair to restore the joint surface, or a combination of screws and plates in complex injuries. However, there remains some disagreement on how much depression of the plateau is acceptable or requires repair. Current orthopedic texts recommend that patients with greater than 10 mm of depression involving an articular surface are treated surgically and those with less than 5 mm depression are not.23 Patients with between 5-10 mm depression remain controversial and are treated individually based on age and activity levels.
Complications seen with tibial plateau fractures can be similar to those seen with high-energy injuries and include neurological, vascular, and ligamentous injuries as well as compartment syndrome. Complications are much more common in cases with comminution of the tibia, or where the distal femur is also fractured. (See section on complex knee fractures.) The popliteal artery is fixed in position at the level of the tibial plateau, and also braches here to form the anterior and posterior tibial artery. Thus, vascular injuries occur frequently in bicondylar fractures or highly comminuted fractures. (See section on popliteal artery injury in knee dislocation). The peroneal nerve's location on the posterior aspect of the fibular head makes it especially vulnerable. Loss of sensation on the dorsal foot and foot drop can result. Be aware that meniscal cartilage, cruciate ligament, and/or MCL injuries are common with plateau fractures, but will not typically be diagnosed in the ED.
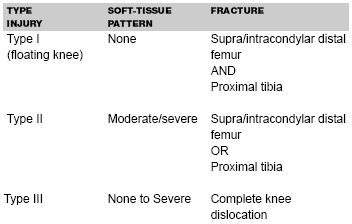
Complex Knee Fractures. The term "complex knee trauma" arose to describe patients whose injuries were not limited to just the distal femur or proximal tibia. Complex knee injuries are divided into three categories. (See Table 5.) Type 1 injuries have supracondylar or intracondylar distal femur fractures and proximal tibia fractures. These are also referred to as "floating knee injuries," as the knee joint has lost both proximal and distal bony attachments. Type 2 injuries have moderate/severe soft-tissue injuries in combination with either distal femur fracture or proximal tibia fractures. Type 3 injuries refer to complete knee dislocations. (See section on knee dislocation.)
| Table 5. Complex Knee Trauma Defined31 |
|
Typically, patients with complex knee trauma have multiple systemic injuries in addition to their knee injury, and they usually are referred to Level 1 trauma centers for treatment. Vascular injuries are a common complication of complex knee trauma, and perfusion of the affected limb should be ascertained early in the patient's evaluation. Type 3 injuries, or complete knee dislocations, carry the highest risk of vascular injury with up to a 32% incidence of major popliteal artery injury.29 (See section on popliteal artery injury in knee dislocation.) The incidence of either compartment syndrome or vascular injury in Type 1 injuries (floating knee) is as high as 25% each.30 These rise to 31% for either vascular injury or compartment syndrome for Type 2 injuries (fracture plus severe soft-tissue damage), and peroneal nerve injury is seen in an additional 23%.30
Care of these injuries often requires a team approach, including consultation with vascular and plastic surgeons in addition to orthopedic surgeons. Needless to say, surgical repair can be technically challenging and rehabilitation prolonged. Thus, in cases where the complex knee injury is the patient's only problem, this injury alone is indication for transfer to trauma centers with experience treating these injuries.
Tibial Spine Fractures. The tibial spine is the small protrusion of bone on the central tibial plateau that fits between the femoral condyles, and is composed of the medial spine (or tubercle) and lateral spine. The tibial spine is of significance as the spines serve as attachment points for knee ligaments and menisci. The medial tibial spine is the larger of the two, and along with the anterior portion of the intercondylar fossa, is the attachment point for the ACL and the anterior portion of the medial and lateral menisci. Likewise, the PCL and posterior portion of the medial and lateral menisci attach to the lateral tibial spine and the posterior portion of the intercondylar fossa.
Therefore fractures of the tibial spine are basically avulsion fractures of ACL or PCL ligament attachments. As growing bone often gives way in children instead of the stronger ligament tearing, tibial spine fractures are more commonly seen in children and adolescents than in adults. However, one study still found that 40% of tibial spine fractures were seen in adults and 60% in children.32 As tibial spine fractures in children are the equivalent of an ACL rupture in adults, they usually are isolated injuries often resulting from sports activity. (See section on pediatric knee fractures.) In adults, tibial spine fractures more often result from high-energy mechanisms (MVAs), and in 68% of cases involve other injuries with 58% of these other injuries involving the same knee.32 Most of these other knee injuries are intra-articular fractures.
Patients present with a history of knee trauma and the expected signs: effusion due to hemarthrosis, inability to bear weight, and decreased range of motion. Signs of ACL instability will be present (positive Lachman's test), but in the acute setting pain and swelling may preclude accurate stability testing. While tibial spine fractures can be seen on standard plain films of the knee, the tunnel view helps highlight this fracture and may be needed to adequately visualize it. One should not be confused on the plain film by the presence of a fabella. The fabella is a sesamoid bone located in 10-20% of patients at the gastrocnemius muscle head, and will be seen on tunnel views in a similar location to tibial spine fracture fragments. The fabella can be distinguished as it has rounded edges and may also be confused with an intra-articular loose body. One might expect a fabella to be a bilateral finding, but at least one study suggested that comparison views may not be useful as the fabella was bilateral in only 63% of cases.33 Tibial spine fractures are classed in three types: type 1 is an incomplete fracture with no displacement, type 2 shows minimal displacement, and type 3 has significant displacement. Type 3 injuries carry the worst prognosis due to associated collateral ligament and meniscal cartilage injuries. Type 1 and 2 fractures are treated with simple casting in full extension for 6 weeks, while displaced fractures require operative repair to restore ACL function.
Patella Fractures. The patella is located inside the patellar (or quadriceps) tendon, and is the largest sesamoid bone in the body. Sesamoid bones form inside tendons where the tendon passes over a joint. They serve to protect the tendon from wear and to increase mechanical advantage of the tendon around the joint. The sesamoid bone lifts the tendon slightly away from the joint center, thereby increasing the moment arm, and also helps maintain the tendon's shape as mechanical load increases. Besides the patella, they are commonly found at the first metatarsal and at the base of the thumb.
Patella fractures occur in all age groups and make up 1% of all skeletal injuries.23 The subcutaneous location of the patella on the anterior knee makes it vulnerable to direct trauma from falls or striking the dashboard in MVAs. Patella fractures can also occur indirectly from forceful contractions of the quadriceps in a flexed knee. Indirect trauma is actually the most common mechanism for patella fractures, causing 50-80% of fractures.23
Patella fractures can be classified in several ways. First, they are either displaced or non-displaced. Displacement is defined as greater than 3 mm of bony separation.34 They are further divided as transverse, vertical (longitudinal), or stellate (comminuted). Transverse fractures are the most common and typically are seen in young adults resulting from indirect trauma (forceful quadriceps contraction). Transverse fractures usually are displaced with complete disruption of the patellar retinacula, resulting in complete loss of knee extension. Non-displaced transverse fractures also occur, but are less common and normally result from a direct blow to the patella. As the retinacula remains intact some knee extension is preserved. More severe direct knee trauma will produce stellate or comminuted fractures, and these make up 30-35% of patella fractures.23 Vertical fractures usually occur on the middle or lateral third of the patella, and are rarely displaced as the retinacula remains intact. These patients also retain some knee extension. They also result from direct trauma and constitute 12-17% of patella fractures.23 Small vertical avulsions of the patellar edge are called marginal fractures, and are the only patella fractures considered not to be intra-articular injuries.
Patients present with complaints of knee pain/injury, as well as swelling, tenderness, and ecchymosis over the patella. Displaced fractures are associated with complete tears of the retinacula, and therefore a palpable defect in the patella can be felt. The proximal portion is usually displaced several inches above its normal location. While these patients will have loss of active knee extension, be aware that patients with non-displaced fractures will retain some knee extension. Besides the typical AP and lateral knee films, tangential views (sunrise view) can be useful to isolate the patella on the film. Vertical patella fractures are commonly not visible on lateral knee views.35 Be careful not to confuse normal variations of bipartite or multipartite patella with acute fractures. The smooth, sclerotic margins of these normal variants help distinguish them from acute fractures.
All patella fractures (except marginal ones) are intra-articular, and thus any break in the skin over a patella fracture may represent an open joint injury. Examine the knee carefully for skin wounds, as subcutaneous location of the patella makes it easy for even small skin wounds to be significant. Open joint injuries are surgical emergencies, and require orthopedic consultation in the ED. Also be aware that open patella fractures are indicative of higher-energy injuries. In one series, 80% of patients with open patella fractures had other significant injuries.36 Given that MVAs were often the cause of open fractures, common associated orthopedic injuries included hip dislocation and femoral neck/acetabulum fractures.
Patients with non-displaced fractures and preserved knee extension (i.e., an intact retinacula) can be safely discharged, using a knee immobilizer to maintain full extension, for outpatient follow-up. The patient should use crutches to perform partial weigh-bearing activity as tolerated until orthopedic follow-up. Displaced fractures (3 mm or greater bony separation) or those with loss of knee extension are treated with ORIF. As long as there is no question of open joint injury, these patients can be discharged after phone consultation for outpatient repair in a knee immobilizer or long-leg posterior splint. In most cases, repair of the patella with tension bands and/or Kirschner wires is the goal. However, in the case of severely comminuted injuries, partial or total patellectomy may result. Most surgical cases result in preserved range of motion, but 55% of patients report pain and/or arthritic complications later.37
References
References
1. Lyn E, Pallin D, Antosia RE. Knee and lower leg. In: Marx, JA ed. Rosen's Emergency Medicine: Concepts and Clinical Practice, 6th ed. St. Louis: Mosby; 2006: 770-796.
2. Calmbach WI, Hutchens M. Evaluation of patients presenting with knee pain: part I. History , physical examination, radiographs and lab tests. Am Fam Physician 2003;68:907-912.
3. Brown JR, Trojian TH. Anterior and posterior cruciate ligament injuries. Prim Care Clin Office Prac 2004;31:925–956.
4. Maffulli N. Acute haemarthrosis of the knee in athletes. A prospective study of 106 cases. J Bone Joint Surg Br 1993;75:945–949.
5. Sonzogni JJ. Examining the injured knee. Emerg Med 1996;28:76–83.
6. Benjaminse A, Gokeler A, van der Schans CP. Clinical diagnosis of an anterior cruciate ligament rupture: a meta-analysis. J Orthop Sports Phys Ther 2006;36:267–288.
7. Wroble RR, Lindenfeld TN. The stabilized Lachman test. Clin Orthop 1988;237:209–212.
8. Seaberg DC, Jackson R. Clinical decision rule for knee radiographs. Am J Emerg Med 1994;12:541–543.
9. Steil IG, Wells GA, McDowell I, et al. Use of radiography in acute knee injuries: need for clinical decision rules. Acad Emerg Med 1995;2:966–973.
10. Bauer SJ, Hollander JE, Fuchs SH, et al. A clinical decision rule in the evaluation of acute knee injuries. J Emerg Med 1995;13:
611–615.
11. Imparanza JI, Aginaga JR. Validation of the Ottawa knee rules. Ann Emerg Med 2001;38:364–368.
12. Stiell IG, Greenberg GH, Wells GA, et al. Prospective validation of a decision rule for the use of radiography in acute knee injuries. JAMA 1996;275:611–615.
13. Seaberg DC, Yealy DM, Lukens T, et al. Multicenter comparison of two clinical decision rules for the use of radiography in acute, high-risk knee injuries. Ann Emerg Med 1998;32:8–13.
14. Verma A. A screening method for knee trauma. Acad Radiol 2001;8:392-397.
15. O'Sullivan MJ, O'Sullivan I. Application of the Ottawa Knee Rules in assessing acute knee injuries. Ir Med J 2006;99:199–200.
16. Buckwalter KA. CT arthrography. Clin Sports Med 2006;25:899–915.
17. Oei EH, Nikken JJ, Ginai AZ, et al. Acute knee trauma: value of a short dedicated extremity MR imaging examination for prediction of subsequent treatment. Radiology 2005;234:125–133.
18. Finlay K, Friedman L. Ultrasonography of the lower extremity. Orthop Clin North Am 2006;37:245-275.
19. Friedman L, Finlay K, Jurriaans E. Ultrasound of the knee. Skeletal Radiol 2001;30:361–377.
20. VanNess SA, Gittins ME. Comparison of intra-articular morphine and bupivacaine following knee arthroscopy. Orthop Rev 1994;23:743–747.
21. Camlet J, Esteve C, Boada S, et al. Analgesic effect of intra-articular ketorolac in knee arthroscopy: comparison of morphine and bupivacaine. Knee Surg Sports Traumatol Arthosc 2004;12:552–555.
22. Parrillo SJ, Fisher J. Arthrocentesis. In: Roberts JR, Hedges JR, et al, eds. Clinical Procedures in Emergency Medicine, 4th ed. Philadelphia: Saunders, 2004:1042-1056.
23. Whittle P, Wood GW. Fractures of the lower extremity. In: Canale ST ed. Canale: Campbell's Operative Orthopaedics, 10th ed. St. Louis: Mosby; 2003:2805–2873.
24. Müller ME, Nazarian S. Classification of fractures of the femur and its use in the A.O. index. Rev Chir Orthop Rep Appar Mot 1981;76:297–309.
25. Walling AK, Seradge H, Spiegel PG. Injuries to the knee ligaments with fractures of the femur. J Bone Joint Surg Am 1982;64:1324–1327.
26. Wiss DA, Watson JT, Johnson EE. Fractures of the knee. In: Rockwood CA, ed. Rockwood and Green's: Fractures in Adults, 5th ed. St Louis: Lippincott, Williams & Wilkins; 2001:1843–1928.
27. Davis DS. Segond fracture: lateral capsular ligament avulsion. J Orthop Sports Phys Ther 1997;25:103–106.
28. Escobedo RM. The "reverse Segond" fracture: association with a tear of the PCL and medial meniscus. AJR Am J Roentgenol 2002;178:979–983.
29. Varnell RM, Coldwell DM, Sangeorzan BJ, et al. Arterial injury complicating knee disruption. Third place winner: Conrad Jobst award. Am Surg 1989;55:699–704.
30. Tscherne H, Lobenhoffer P. Tibial plateau fractures. Management and expected results. Clin Orthop Relat Res 1993;292:87–100.
31. Krettek C, Tscherne H. In: Fu FH, et al., eds. Knee Surgery. Baltimore, Williams & Wilkins, 1994, pp. 1027–1035.
32. Kendall NS, Hsu SY, Chan KM. Fracture of the tibial spine in adults and children. A review of 31 cases. J Bone Joint Surg Br 1992;74:848–852.
33. Houghton-Allen BW. In the case of the fabella a comparison view of the other knee is unlikely to be helpful. Australas Radiol 2001;45:318-319.
34. Roberts DM, Stallard TC. Emergency department evaluation and treatment of knee and leg injuries. Emerg Med Clin North Am 2000;18:67–84.
35. Miller MD. Commonly missed orthopedic problems. Emerg Med Clin North Am 1992:10;151–161.
36. Catalano JB, Iannacone WM, Marczyk S, et al. Open fractures of the patella: long-term functional outcomes. J Trauma 1995;39:439–444.
37. Levack B, Flannagan JP, Hobbs S. Results of surgical treatment of patellar fractures. J Bone Joint Surg Br 1985;67:416–419.
Often, the emergency department (ED) evaluation of a painful knee is a quick x-ray followed by discharge with the limb placed a knee immobilizer, the patient walking with crutches, a prescription for analgesics, and a referral to see an orthopedist next week.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.