Human Papillomavirus Vaccines
Human Papillomavirus Vaccines
Authors: Cynthia M. Rand, MD, MPH, Assistant Professor, Pediatrics, University of Rochester School of Medicine and Dentistry, Rochester, NY; and Sharon G. Humiston, MD, MPH, Associate Professor, Emergency Medicine and Pediatrics, University of Rochester School of Medicine and Dentistry, Rochester, NY.
Peer Reviewer: William A. Kennedy, MD, Division of Pediatric Research, Loma Linda University, Loma Linda, CA
HPV Epidemiology
Human papillomavirus (HPV) is the most common sexually transmitted infection in the United States, causing an estimated 6 million new genital infections annually.1 An estimated 25 million women (aged 14 through 59) in the United States are currently infected, with the highest prevalence among women aged 20-24.2 Prevalence of HPV in young women varies based on the population studied, but ranges from 14-90%,3 and is usually approximately 30% based on many clinic-based prevalence studies. At least half of sexually active men and women acquire HPV at some point in their lifetime, and models suggest that more than 80% of sexually active women will have acquired genital HPV by age 50 years.4 While the natural history and transmission of HPV in men is much less studied, HPV prevalence in men ranges from 1-73%, depending on anatomic site evaluated and source of the sample.5 The ubiquity of this virus makes it an important one to understand.
Transmission and Risk Factors
Genital HPV is transmitted through genital contact, most often by vaginal or anal intercourse. Infection usually occurs soon after sexual initiation.6 In one study of female college students, more than 50% acquired cervical HPV infection within 48 months after first sexual intercourse.7 Vertical transmission from mother to newborn can occur during delivery, and manifests as recurrent respiratory papillomatosis.
Risk factors for HPV infection include having a higher number of sexual partners (lifetime and recent), younger age (between adolescence and age 25), history of other sexually transmitted infections, and immunosuppression, including human immunodeficiency virus.8 Other factors that may be related, but are less definitive, include cigarette smoking, oral contraceptive use, lack of circumcision of a male partner, younger age at first sexual intercourse, and inconsistent condom use.9
The evidence for condom use and HPV prevention continues to evolve. A study of university women collected cervical and vulvovaginal samples for HPV DNA and Pap testing every 4 months, along with daily recorded diaries of sexual behavior for 12 months.10 Women whose partners used condoms in all incidences of vaginal intercourse were 70% less likely to acquire a new genital HPV infection than were women whose partners used condoms less than 5% of the time. While condoms are not able to prevent all infections with HPV since the virus can be transferred from skin that is not covered, they appear to dramatically reduce its transmission.
Natural History of HPV Infection
Although HPV infections are common, the majority of infections resolve spontaneously; 70% of infections clear within 12 months, and 90% within 24 months.11 The median duration of new infections is roughly 8 months,12 although it is unclear whether the virus is eliminated or present at undetectable levels. High-risk types of HPV have a higher rate of persistence,6 and are the most important risk factor for precancerous cervical lesions and cervical cancer.
HPV Virology and Pathogenesis
HPV is a double-stranded, circular DNA virus in the Papillomaviridae family of viruses. More than 100 HPV types have been identified.13 Their genome contains two functional regions: an early region (E1, E2, E4, E5, E6, and E7), which encodes for proteins that control transcription, replication, and cellular transformation; and a late region (LI and L2), which encodes structural proteins required for viral assembly. In the HPV types that are associated with cancer, E6 and E7 proteins interfere with normal cell cycle regulation and cause uncontrolled cell proliferation by promoting degradation of the p53 tumor suppressor gene product and inactivating the retinoblastoma protein.14 When purified, the major capsid protein (L1) will self-assemble to form virus-like particles (VLPs)—empty shells that resemble virus.
Approximately 40 HPV types cause mucosal infection, and these are classified on the basis of their oncogenic potential. Low risk HPV types, which include types 6 and 11 among others, are associated with anogenital warts, recurrent respiratory papillomatosis, and mild cervical dysplasia. High risk types are associated with anogenital cancers, including cervical, vulvar, penile, and anal cancers as well as low- and high-risk dysplasias. High-risk types include 16, 18, 31, 33, and 35, among others.15 (See Table 1.)
| Table 1. HPV Viral Type and Associated Disease16 |
|
HPV infections occur at the basal layer of the epithelium. The virus accesses this layer through thin epithelial layers, such as those in the transformation zones of the cervix or anus, through microabrasions in the epithelium produced during sexual activity.17 The virus then uses host cell machinery to replicate and express viral proteins, which inhibit cellular differentiation and stimulate continued cellular proliferation.16 The relatively larger transformation zone of the adolescent cervix compared with older women makes young women particularly susceptible to the HPV virus.18
Diseases Caused by HPV
Cervical cancer. High risk HPV types are responsible for most high grade precancerous cervical lesions and 99.7% of cervical cancers.19 In the United States, about 75% of all cervical cancers are squamous cell, and the rest are adenocarcinomas. Among cases of cervical cancer worldwide, HPV types 16, 18, 45, 31, and 33 account for 80% of the type distribution in squamous cell carcinomas, and HPV types 16, 18, 45, 59, and 33 account for 94% of the type distribution in adenocarcinomas.20 The other 20% of squamous cell carcinomas and 6% of adenocarcinomas are caused by 20-23 other types, so that each one of the other types accounts for only 3-0.1%. HPV 16 alone causes 50-60% of invasive cervical cancer, with HPV 18 causing another 10-15%, for a total of 70% between the two types.
Since the introduction of Pap testing in 1949, cervical cancer mortality rates in the United States have decreased by more than 70%.21 Despite such improvements, an estimated 11,150 cases of cervical cancer are expected in the United States in 2007, causing an estimated 3,670 deaths.22 The incidence of cervical cancer in black women in the United States is roughly 1.5 times higher than that of white women, and mortality is twice as high. Hispanic, Vietnamese, and Korean women also have notably higher rates compared to white women.23 Worldwide, cervical cancer is the second most common cause of death from cancer in women, with an estimated 510,000 new cases causing 288,000 deaths annually.24
Generally, cervical HPV infections are asymptomatic, but can result in histologic changes that lead to cancer. These are classified as cervical intraepithelial neoplasias (CIN) grades 1, 2, or 3, based on degree of abnormality, or if more advanced, adenocarcinoma in situ (AIS). Roughly 500,000 precancerous lesions (CIN 2 or CIN 3) are diagnosed annually in the United States, and 50-60% are due to HPV 16 and 18.25 CIN 1 can be caused by multiple HPV types, including 6, 11, 16, and 18. CIN 1 clears on its own without treatment in 60% of cases, whereas only 30-40% of CIN 2 and 3 clear spontaneously.26
Using the Pap test, clinicians can detect cytological changes that indicate which histologic change is occurring. Abnormalities detected by Pap testing include ASC-US (atypical squamous cells of undetermined significance), low grade and high grade squamous intraepithelial lesions (LSIL and HSIL), and AIS.
Anogenital Warts. Anogenital warts (condyloma), 90% of which are caused by HPV types 6 and 11,27 are benign growths that often recur, requiring repeat treatments. In 20-30% of cases, they regress spontaneously. In rare instances, they can become locally invasive. Genital warts are estimated to occur in 1% of the population, and result in an estimated 240,000 visits to physicians’ offices, and 5% of all visits to sexually transmitted disease clinics.28 An estimated 10% of men and women will develop anogenital warts at some point in their lifetimes.29
Vaginal and Vulvar Cancer. Some, but not all vaginal and vulvar cancers, which are less common than cervical cancer, are associated with HPV. In 2007, 2,140 cases of invasive vaginal cancer and 3,490 cases of vulvar cancer are expected, resulting in 790 and 880 deaths, respectively.22 When found, HPV 16 is the most common type associated with vaginal cancers, and types 16 and 18 have been detected in 76% of vaginal intraepithelial neoplasia (VIN) 2 and 3 and 42% of vulvar carcinoma.26 While rates of vaginal cancer are decreasing in the United States, rates of vulvar cancer are increasing, possibly in part due to improved detection or reporting.
Anal Cancer. About 4,000 people annually develop anal cancer in the United States, with 80-90% of cases caused by HPV 16 or HPV 18.22 The incidence of anal cancer in men has increased in the past 30 years, and men who have sex with men and those with HIV are particularly at risk. Penile, urethral, and head and neck cancers have also been associated with carcinogenic HPV types.23
Recurrent Respiratory Papillomatosis. About 1 in 200,000 children under 18 years have juvenile laryngeal papillomatosis, which involves benign laryngeal tumors that can lead to obstruction.23 Transmission is thought to occur from mother to baby during delivery. These tumors often require repeated resection. HPV 6 and HPV 11 are most commonly involved, and in rare cases, these papillomas can become invasive carcinoma.30
Cervical Cancer Screening
As mentioned above, Pap testing has remarkably reduced the incidence of cervical cancer in the United States. Screening is effective because lesions are usually slow to progress from mild dysplasia to invasive carcinoma, cytological findings can identify abnormalities before more serious disease occurs, there is good treatment for mild and moderate dysplasia, and in the United States there has been close follow-up of abnormal tests.31 American Cancer Society (ACS)21 and American College of Obstetricians and Gynecologists (ACOG)32 Guidelines state that women should have Pap testing performed within 3 years of beginning sexual activity or by age 21, whichever occurs first. However, due to imperfect cytological testing and some error in follow-up of abnormal Pap smears, roughly 2-3 per 100,000 women can still be expected to develop cervical cancer.23
As added security in diagnosis, several organizations (including ACS21 and ACOG33) have recommended HPV DNA testing simultaneously with cytological testing for women over age 30. The Digene Hybrid Capture® 2 (HC2) High-Risk DNA Test is the only U.S. Food and Drug Administration (FDA) approved test for HPV. The test uses liquid nucleic acid hybridization and detects 13 HPV types (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and 68). The test is used to triage women with equivocal Pap test results and as an adjunct to regular Pap screening.23
HPV Vaccines
Two prophylactic HPV vaccines have been developed: one vaccine was licensed in June 2006, and the second was submitted to the FDA in March 2007 and is pending licensure. The licensed vaccine is a quadrivalent vaccine (Gardasil, Merck & Co., Inc.) that protects against HPV types 6, 11, 16, and 18. The second is a bivalent vaccine (Cervarix, GlaxoSmithKline) that protects against HPV types 16 and 18. Both vaccines are composed of recombinant L1 capsid protein that self-assembles into virus like particles (VLPs), which contain no DNA and consequently are unable to replicate. The vaccines do not contain live viruses.
Quadrivalent HPV Vaccine. Gardasil is given in 3 separate 0.5 mL intramuscular doses.34 The second dose should be given 2 months after the first dose, and the third dose 6 months after the first dose. Each dose contains 20 mcg HPV 6 L1 protein, 40 mcg HPV 11 L1 protein, 40 mcg HPV 16 L1 protein, and 20 mcg HPV 18 L1 protein. Each dose also contains an aluminum-based adjuvant. The vaccine contains no thimerosal, preservative, or antibiotics. The vaccine should be stored at 2°C-8°C and not frozen.
Efficacy (Per Protocol). Since it takes 20 years on average for cervical lesions to progress to cervical cancer, and effective treatments exist for precursor lesions, it would be unethical to use cervical cancer as an endpoint for vaccine efficacy. Instead, CIN 2/3 and AIS are considered acceptable intermediate disease endpoints for cervical cancer. Double-blind, placebo-controlled studies have shown high efficacy of vaccines against HPV 16 (monovalent),35,36 and 6, 11, 16, and 18 (quadrivalent)37-39 vaccines for prevention of respective HPV type persistent infection, CIN and CIN 2/3 related to these vaccine types, and genital warts when analyses are restricted to those who received 3 doses of vaccine, had no protocol violations, and no evidence of HPV type specific infection through 1 month past dose 3.34 If patients have vaccine type-specific infection at baseline (based on PCR), there is no evidence of protection from the vaccine against that type. In phase II trials, four cases of CIN occurred in the vaccine group, all of which were CIN 1. Overall efficacy of the quadrivalent vaccine in prevention of cervical disease was 95%, and 99% in prevention of external genital warts. In phase III trials, the vaccine is 98% effective in preventing HPV-16 and 18 related CIN 2/3 and AIS.40 Efficacy against HPV 16 or 18 related VIN 2/3 was 100% in phase II and III trials.41
Efficacy (Intent to Treat). Intent to treat analyses were performed to determine the efficacy of the quadrivalent vaccine in all women who were randomized into the trial and received at least one dose of the vaccine, regardless of follow-up vaccination and baseline HPV status.34 Overall, there was a 39% reduction in HPV 16 and 18 related CIN 2/3 or AIS, and a 46% reduction in HPV 6, 11, 16, and 18 related CIN or AIS in the vaccinated group compared to placebo. This group also had a 69% reduction in HPV 6, 11, 16, and 18 related genital warts, and a 69% reduction in HPV 16 or 18 related VIN 2/3. Individuals were only eligible for the clinical trials if they reported no more than 4 lifetime partners, so actual efficacy may be lower in the general population. Clearly, vaccinated women remain at risk for cervical and genital disease from HPV types that are present prior to vaccination.
Immunology. Immunogenicity of the quadrivalent vaccine is measured by quantifying IgG antibody to HPV L1 (geometric mean titers). In all studies, vaccination provides higher antibodies than natural infection for all 4 HPV types.38 Women who were HPV-positive to a specific HPV type at the beginning of the study developed an even higher response than those who were HPV negative. A study of young women 9-15 years old showed even higher antibody responses than women aged 16-26 years.42 It is unclear what level of antibody response is needed to protect against HPV infection, since the vaccine has been so efficacious. Follow-up studies may help determine the minimal protective antibody level.
Duration of Protection. Some individuals from phase II trials have been followed for 60 months, and phase III trials have followed women for 3 years after their final dose. Through 5 years, vaccine-induced anti-HPV antibody titers have remained at or above those following natural infection.39 Thus far, cumulative efficacy against HPV type persistent infection or disease is 96%, and efficacy against vaccine type CIN or external genital lesions is 100%.39 The vaccine manufacturer is planning to follow 5,500 women in northern Europe for 15 years postvaccination to further examine duration of protection.43
Safety. Clinical trial participants experienced side effects from the quadrivalent HPV vaccine that were similar to other immunizations. Pain at the injection site was the most common side effect reported from vaccine recipients, and was slightly more likely in those receiving vaccine (84%) compared with those receiving an aluminum-containing placebo (75%).34 The other common side effects included localized swelling and erythema at the injection site. Fever was reported as an adverse reaction in 13% of those receiving quadrivalent vaccine, compared to 11% of those receiving placebo. Other systemic adverse reactions (nausea, nasopharyngitis, etc.) were similar among both groups. There was no mortality during vaccine trials thought to be a result of vaccine administration. Out of 21,464 subjects, there were 102 serious adverse experiences in the 1-15 day period after vaccination. These included headache, gastroenteritis, appendicitis, and pelvic inflammatory disease, as well as one case of bronchospasm and 2 cases of asthma exacerbation. Rates of these symptoms in placebo recipients were similar.
Simultaneous Administration with Other Vaccines. Administration of the quadrivalent HPV vaccine was studied with concomitant hepatitis B vaccine administration in a placebo controlled trial. Rates of injection site and systemic adverse experiences were the same in both the vaccine and placebo groups. Studies are planned to examine simultaneous administration of meningococcal conjugate vaccine, tetanus, diphtheria and acellular pertussis (Tdap) vaccine, and Gardasil.26
Pregnancy and Lactation. Pregnant women were excluded from the beginning of the quadrivalent vaccine trials, and if they became pregnant during the trial, the next vaccine dose was delayed until after delivery. However, 1,115 pregnancies occurred in the vaccine group and 1,151 in placebo group.34 There were 15 cases of congenital anomaly in subjects who had received the vaccine versus 16 anomalies in subjects who received placebo. Sub-analyses examined infants born to women who received vaccine or placebo within 30 days of estimated pregnancy onset. Within this subset, in the infants born to vaccinated subjects there were five congenital anomalies compared to zero in the placebo group. An expert panel determined these were unrelated to vaccination.26 The quadrivalent vaccine has been classified as pregnancy category B, based on no evidence of harm to the fetus. Merck & Co. is maintaining a pregnancy registry to monitor fetal outcomes of pregnant women who are exposed to Gardasil.
A total of 995 nursing mothers were given the quadrivalent vaccine or placebo during the clinical trials. Antibody response was similar in both groups. No adverse events were thought to be vaccine-related (17 vaccine, 9 placebo; most respiratory infections and gastroenteritis), and lactating women can receive the quadrivalent HPV vaccine.
Bivalent Vaccine (Cervarix). As mentioned above, Cervarix (GlaxoSmithKline) is a bivalent vaccine that protects against HPV types 16 and 18. This vaccine is delivered on a schedule of 0, 1, and 6 months. In placebo-controlled, randomized clinical trials that included women aged 15-25 years, vaccine efficacy was 100% in preventing HPV 16 or 18 related CIN in a follow-up period of 4.5 years.44 This vaccine also offered broad protection against incident infection HPV 45 and HPV 31, despite those types not being included in the vaccine, implying that serologic cross-reactivity occurs. This vaccine uses an AS04 adjuvant that contains aluminum hydroxide and 3-deacylated mono-phosphoryl lipid A. Clinical trials have shown higher antibody titers when AS04 is used as an adjuvant rather than aluminum by itself.45
The bivalent vaccine has been shown to be safe and well-tolerated. Vaccine recipients had a slightly higher rate of local reaction, including pain, redness, and swelling (94%) compared to placebo recipients (88%). Approval for this vaccine has been submitted to the FDA and is pending.
Recommendations for Vaccination
Both the Centers for Disease Control and the American Cancer Society have developed recommendations for vaccination with the FDA-approved quadrivalent vaccine.9,23 (See Table 2.) Based on evidence that the vaccine provides prophylactic protection for HPV types 6, 11, 16, and 18 prior to natural infection, but is not therapeutic if already infected, both recommendations emphasize the continued need for routine cervical cancer screening in vaccinated individuals. In addition, 30% of HPV types that result in cervical cancer are not covered by the vaccine. To achieve the greatest benefit from vaccination, both recommendations advise vaccinating young women, since it is more likely that they will not have been exposed to HPV virus.
| Table 2. Centers for Disease Control (CDC) and American Cancer Society (ACS) Recommendations for Quadrivalent Human Papillomavirus Vaccine Use23,26 |
|
Pap testing and screening for HPV are not needed before vaccination at any age. The minimal interval between doses 1 and 2 is 4 weeks, and the minimal time between doses 2 and 3 is 12 weeks. The vaccine series does not need to be restarted if a longer interval than recommended has elapsed between doses. It can be given simultaneously with any other recommended adolescent vaccines, including meningococcal conjugate, Tdap, and hepatitis B vaccines. While women should not receive the vaccine if they are pregnant, they can receive it if they are lactating. There is no contraindication to vaccinating immunocompromised individuals, since there is no live component to the vaccine. The vaccine has not yet been approved for males, although studies are ongoing.
HPV Vaccine Acceptance
Several studies examined acceptability of HPV vaccine among adolescents, parents, and physicians prior to vaccine availability, and generally found that acceptability of the vaccine is high. Two studies have reported parental concern that offering the HPV vaccine would increase unsafe sexual behavior,46,47 but another study found that severity of infection and vaccine efficacy were much more critical factors for parents in making vaccination decisions for their children.48 In the past, knowledge about HPV has been low,49 but studies vary on whether providing parents educational interventions change their opinions about the vaccine.46,50 In general, parents look to their physicians for vaccination information and recommendations, so providers have a critical role in explaining the usefulness to vaccinate prior to sexual debut.
State Mandates
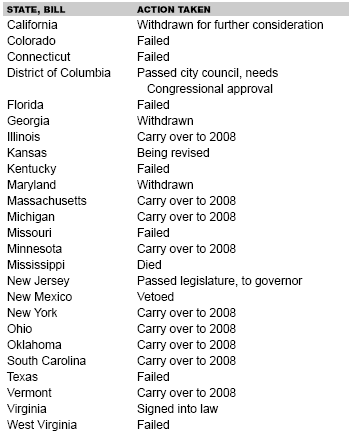
Each state decides independently which vaccines to mandate for school entry. The issue of states requiring a vaccine that prevents a sexually transmitted infection has engendered much controversy in recent months. The Michigan senate was the first to introduce legislation in September 2006 to require HPV vaccine for girls entering sixth grade, but the bill was revised to only require education about HPV. Much controversy surrounded the February 2007 executive order from the governor of Texas to mandate the vaccine for girls entering sixth grade. Legislators in that state passed a bill to override the executive order, and the governor did not veto that bill. In March 2007, Virginia became the first state in the nation to enact a school HPV vaccination requirement, with an opt-out policy for parents. Legislators in 41 states and the District of Columbia have introduced legislation to require, fund, or educate the public about HPV vaccine, and 17 states have enacted this legislation.51 In the past year, 24 states and the District of Columbia introduced legislation to specifically mandate the vaccine for sixth grade school entry, with parent exemptions included. (See Table 3.) Many states have withdrawn their initial bills that mandated the vaccine in favor of required HPV education, which is most needed. The CDC, American Academy of Pediatrics, and American Academy of Family Physicians oppose mandates at this point, and consider them premature, since vaccine finance issues, reimbursement, and long-term safety with widespread use have not yet been established.
| Table 3. State Legislation for School Mandates Requiring HPV Vaccine for 6th Grade School Entry51 (As of August 1, 2007) |
|
Cost and Cost-Effectiveness
The quadrivalent HPV vaccine is the most expensive vaccine to date, at $120 per dose, and $360 for the entire series. Several studies have used mathematical modeling to determine the cost-effectiveness of vaccinating 12-year-olds. In those studies, the cost of each quality-adjusted life year gained ranges from $3,000 to $24,300, and varies based on assumption of duration of protection, need for boosters, cost, and vaccine coverage.26 Overall these values are considered cost-effective. Modelers point out that if the rate of vaccination is high for women, the incremental benefit of vaccinating men is small.23
Summary
HPV infection is extremely common, and high risk HPV types are associated with the development of cervical and other anogenital cancers. The quadrivalent HPV vaccine is efficacious in preventing infection with HPV types included in the vaccine. The vaccine is not effective if individuals have already been exposed to those types of the virus, so vaccinating at early adolescent ages has been recommended. In the coming year, a bivalent vaccine is expected to reach the U.S. market, and individual states are likely to resolve legislation related to the vaccine. In most states, it is likely that school mandates will not result, but rather there will be much-needed programs for vaccine financing and mandatory education about the relationship between HPV and cervical cancer.
References
1. Weinstock H, Berman S, Cates W, Jr. Sexually transmitted diseases among American youth: Incidence and prevalence estimates, 2000. Perspectives on Sexual & Reproductive Health 2004;36(1):6-10.
2. Dunne EF, Unger ER, Sternberg M, et al. Prevalence of HPV infection among females in the United States. JAMA 2007;297813-819.
3. Revzina NV, Diclemente RJ. Prevalence and incidence of human papillomavirus infection in women in the USA: A systematic review. Int J STD AIDS 2005;16:528-537.
4. Myers ER, McCrory DC, Nanda K, et al. Mathematical model for the natural history of human papillomavirus infection and cervical carcinogenesis. Am J Epidemiol 2000;151:1158-1171.
5. Dunne EF, Nielson CM, Stone KM, et al. Prevalence of HPV infection among men: A systematic review of the literature. J Infect Dis 2006;194:1044-1057.
6. Trottier H, Franco EL. The epidemiology of genital human papillomavirus infection. Vaccine 2006;24 Suppl 1:S1-15.
7. Winer RL, Lee SK, Hughes JP, et al. Genital human papillomavirus infection: Incidence and risk factors in a cohort of female university students. Am J Epidemiol 2003;157:218-226.
8. Manhart LE, Holmes KK, Koutsky LA, et al. Human papillomavirus infection among sexually active young women in the United States: Implications for developing a vaccination strategy. Sex Transm Dis 2006;33:502-508.
9. Dunne EF, Markowitz LE. Genital human papillomavirus infection. Clin Infect Dis 2006;43:624-629.
10. Winer RL, Hughes JP, Feng Q, et al. Condom use and the risk of genital human papillomavirus infection in young women. N Engl J Med 2006;354:2645-2654.
11. Franco EL, Villa LL, Sobrinho JP, et al. Epidemiology of acquisition and clearance of cervical human papillomavirus infection in women from a high-risk area for cervical cancer. J Infect Dis 1999;180:1415-1423.
12. Ho GY, Bierman R, Beardsley L, et al. Natural history of cervicovaginal papillomavirus infection in young women. N Engl J Med 1998;338:423-428.
13. de Villiers EM, Fauquet C, Broker TR, et al. Classification of papillomaviruses. Virology 2004;324:17-27.
14. Munger K, Howley PM. Human papillomavirus immortalization and transformation functions. Virus Res 2002;89:213-228.
15. Munoz N, Bosch FX, de Sanjose S, et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med 2003;348:518-527.
16. Frazer IH, Cox JT, Mayeaux EJ, Jr., et al. Advances in prevention of cervical cancer and other human papillomavirus-related diseases. Pediatr Infect Dis J 2006;25:S65-81, quiz.
17. Schiffman M, Kjaer SK. Chapter 2: Natural history of anogenital human papillomavirus infection and neoplasia. J Natl Cancer Inst Monogr 2003;14-19.
18. Moscicki AB. Human papillomavirus infection in adolescents. Pediatr Clin North Am 1999;46:783-807.
19. Walboomers JM, Jacobs MV, Manos MM, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol 1999;189:12-19.
20. Bosch FX, de Sanjose S. Chapter 1: Human papillomavirus and cervical cancer—burden and assessment of causality. J Natl Cancer Inst Monogr 2003;3-13.
21. Saslow D, Runowicz CD, Solomon D, et al. American Cancer Society guideline for the early detection of cervical neoplasia and cancer. CA Cancer J Clin 2002;52:342-362.
22. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2007. CA Cancer J Clin 2007;57:43-66.
23. Saslow D, Castle PE, Cox JT, et al. American Cancer Society Guideline for human papillomavirus (HPV) vaccine use to prevent cervical cancer and its precursors. CA Cancer J Clin 2007;57:7-28.
http://www.who.int/vaccine_research/diseases/hpv/en/. Accessed July 9, 2007.
25. Clifford GM, Smith JS, Aguado T, et al. Comparison of HPV type distribution in high-grade cervical lesions and cervical cancer: A meta-analysis. Br J Cancer 2003;89:101-105.
26. Markowitz LE, Dunne EF, Saraiya M, et al. Quadrivalent human papillomavirus vaccine: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2007;56:1-24.
27. Greer CE, Wheeler CM, Ladner MB, et al. Human papillomavirus (HPV) type distribution and serological response to HPV type 6 virus-like particles in patients with genital warts. J Clin Microbiol 1995;33:2058-2063.
28. Strauss MJ, Khanna V, Koenig JD, et al. The cost of treating genital warts. Int J Dermatol 1996;35:340-348.
29. Munk C, Svare EI, Poll P, et al. History of genital warts in 10,838 women 20 to 29 years of age from the general population. Risk factors and association with Papanicolaou smear history. Sex Transm Dis 1997;24:567-572.
30. Reeves WC, Ruparelia SS, Swanson KI, et al. National registry for juvenile-onset recurrent respiratory papillomatosis. Arch Otolaryngol Head Neck Surg 2003;129:976-982.
31. Kitchener HC, Castle PE, Cox JT. Chapter 7: Achievements and limitations of cervical cytology screening. Vaccine 2006;24 Suppl 3:S63-S70.
32. ACOG Practice Bulletin: Clinical management guidelines for obstetrician-gynecologists. Number 45, August 2003. Cervical cytology screening (replaces committee opinion 152, March 1995). Obstet Gynecol.2003;102:417-427.
33. ACOG Practice Bulletin. Clinical Management Guidelines for Obstetrician-Gynecologists. Number 61, April 2005. Human papillomavirus. Obstet Gynecol 2005;105:905-918.
http://www.merck.com/product/usa/pi_circulars/g/gardasil/gardasil_pi.pdf. Accessed July 22, 2007.
35. Koutsky LA, Ault KA, Wheeler CM, et al. A controlled trial of a human papillomavirus type 16 vaccine. N Engl J Med 2002;347:1645-1651.
36. Mao C, Koutsky LA, Ault KA, et al. Efficacy of human papillomavirus-16 vaccine to prevent cervical intraepithelial neoplasia: a randomized controlled trial. Obstet Gynecol 2006;107:18-27.
37. Villa LL, Costa RL, Petta CA, et al. Prophylactic quadrivalent human papillomavirus (types 6, 11, 16, and 18) L1 virus-like particle vaccine in young women: A randomised double-blind placebo-controlled multicentre phase II efficacy trial. Lancet Oncol 2005;6:271-278.
38. Villa LL, Ault KA, Giuliano AR, et al. Immunologic responses following administration of a vaccine targeting human papillomavirus Types 6, 11, 16, and 18. Vaccine 2006;24:5571-5583.
39. Villa LL, Costa RL, Petta CA, et al. High sustained efficacy of a prophylactic quadrivalent human papillomavirus types 6/11/16/18 L1 virus-like particle vaccine through 5 years of follow-up. Br J Cancer 2006;95:1459-1466.
40. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med 2007;356:1915-1927.
41. Garland SM, Hernandez-Avila M, Wheeler CM, et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med 2007;356:1928-1943.
42. Block SL, Nolan T, Sattler C, et al. Comparison of the immunogenicity and reactogenicity of a prophylactic quadrivalent human papillomavirus (types 6, 11, 16, and 18) L1 virus-like particle vaccine in male and female adolescents and young adult women. Pediatrics 2006;118:2135-2145.
43. Lehtinen M, Herrero R, Mayaud P, et al. Chapter 28: Studies to assess the long-term efficacy and effectiveness of HPV vaccination in developed and developing countries. Vaccine 2006;24 Suppl 3:S233-S241.
44. Harper DM, Franco EL, Wheeler CM, et al. Sustained efficacy up to 4.5 years of a bivalent L1 virus-like particle vaccine against human papillomavirus types 16 and 18: Follow-up from a randomised control trial. Lancet 2006;367:1247-1255.
45. Giannini SL, Hanon E, Moris P, et al. Enhanced humoral and memory B cellular immunity using HPV16/18 L1 VLP vaccine formulated with the MPL/aluminium salt combination (AS04) compared to aluminium salt only. Vaccine 2006;24:5937-5949.
46. Davis K, Dickman ED, Ferris D, et al. Human papillomavirus vaccine acceptability among parents of 10- to 15-year-old adolescents. J Low Genit Tract Dis 2004;8:188-194.
47. Olshen E, Woods ER, Austin SB, et al. Parental acceptance of the human papillomavirus vaccine. J Adolesc Health 2005;37:248-251.
48. Zimet GD, Mays RM, Sturm LA, et al. Parental attitudes about sexually transmitted infection vaccination for their adolescent children. Arch Pediatr Adolesc Med 2005;159:132-137.
49. Waller J, McCaffery K, Forrest S, et al. Awareness of human papillomavirus among women attending a well woman clinic. Sex Transm Infect 2003;79:320-322.
50. Dempsey AF, Zimet GD, Davis RL, et al. Factors that are associated with parental acceptance of human papillomavirus vaccines: A randomized intervention study of written information about HPV. Pediatrics 2006;117:1486-1493.
http://www.ncsl.org/programs/health/HPVvaccine.htm#hpvlegis. Accessed July 21, 2007.
Human papillomavirus (HPV) is the most common sexually transmitted infection in the United States, causing an estimated 6 million new genital infections annually.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.