Acute Infectious Diarrhea
Acute Infectious Diarrhea
Author: John Kahler, MD, FAAEM, Clinical Instructor, Department of Emergency Medicine, University of Michigan Health System, Ann Arbor, MI.
Peer Reviewers: Gregory A. Volturo, MD, Chairman, Department of Emergency Medicine, Professor of Emergency Medicine, University of Massachusetts Medical School, Worcester, MA; and David Kramer, MD, FAAEM, FACEP, Program Director and Vice Chair, Department of Emergency Medicine, York Hospital, Associate Professor of Clinical Emergency Medicine, Penn State College of Medicine.
The prologue of Robin Cook's novel "Toxin" contains the following: "Within the pen an obviously sick cow was lying in its own diarrhea." What follows is an intriguing detective story of suspense and devastation caused by society's failure to control a tiny microbe called Escherichia coli.
Fiction is hardly stranger than fact. A recent outbreak of E. coli in Indiana has sickened at least 10 people including 7 school children suffering renal failure requiring dialysis. As a result of an E. coli outbreak that has infected at least 30 people in eight states, Topps Meat Co. has just recalled 21.7 million pounds of frozen hamburgers because of possible E. coli contamination traced to its Elizabeth, New Jersey plant.
The challenge of diarrhea is that although it is often a self-limited condition, the clinician needs to be wary of potentially life-threatening etiologies. This issue provides a broad and practical overview of the burden of diarrhea diseases, their etiologies, appropriate diagnostic work-up, and empiric and definitive treatment courses.
—The Editor
This article was originally published in the October 1, 2007 issue of Emergency Medicine Reports.
Introduction
Diarrhea has plagued mankind since prehistoric times. While nearly a universal experience, it can range from a mild inconvenience to a rapidly fatal demise. Its significance as a public health threat didn't grow until the Paleolithic hunter-gatherer communities converted to Neolithic farming communities.1 Urbanization brought larger groups of people together in communities to live and share resources. In modern times, improvements in sanitation and recognition of sanitary practices have lessened the impact of infectious diarrhea on close-knit communities. Despite this trend, there are still areas of the world where the community infrastructure is underdeveloped, overloaded, or broken, and infectious diarrhea remains a leading cause of death in the world today. Nearly every human being will be affected by this condition in their lifetime. In developed countries where sanitation is better, individuals have been shown to average two bouts of diarrhea annually. Diarrhea cases are due to locally endemic inoculums; whereas periodically outbreaks and epidemics of various organisms occur when sanitation processes break down. Underdeveloped countries are hit the hardest due to poorly developed sanitation, overcrowding, and lack of medical care, financial, and other resources.
Acute diarrhea is a frequent complaint among patients and can be divided into infectious (85%) and non-infectious causes (15%). Various estimates exist but infectious causes occur with the following relative frequencies: viral 50-70%, bacterial 15-20%, parasitic 10-15%, and 5-10% from unknown causes.2
A systematic approach to the diagnosis and management of infectious diarrhea can lead to quicker resolution and avoidance of adverse outcomes. With the ease of world travel today and the ability to fly from one side of the globe to the other in a day, physicians must keep an open mind to the many infectious etiologies. Fluid management and supportive care are the mainstays of therapy, yet antibiotics have a clearly beneficial role in properly selected patient populations.
Epidemiology
Few infectious diseases have caused more morbidity and mortality throughout history than those that infect the gastrointestinal tract. Wars have been lost, cities devastated, and country borders decided by epidemics of infectious diarrhea. In some wars, morbidity and mortality due to diarrhea outpaced injuries from fighting.1
Infectious diarrhea is the second most common cause of death worldwide with nearly 5 million deaths annually.3 It remains one of the leading causes of death in children.2,4,5 Diarrhea is estimated to cause the deaths of up to 10,000 children (younger than age 5) daily in underdeveloped countries.6 This statistic is in stark contrast to the 300 children younger than age 5 who die in the United States annually and illustrates the role of sanitation in curbing infectious diarrhea. Surveys of the U.S. population have demonstrated in excess of 135 million cases per year of non-food-borne gastroenteritis vs. 76 million cases of food-borne gastroenteritis.7 These episodes account for more than 6000 deaths and 900,000 hospitalizations annually.5 Detailed and accurate data do not exist on the full economic impact of these illnesses, but the cost is estimated to be in billions of dollars when taking into account lost work productivity, and direct and indirect health care costs. Diarrhea is second only to the common cold in number of work days lost.6
Diarrhea is the most common illness affecting those that travel internationally.8 It is more common in developing countries, but with the ease of global travel, seeing patients in the United States who have acquired an infection elsewhere is common. Thus, it is imperative to maintain a broad differential diagnosis and take a thorough history.
Many cases of infectious diarrhea occur in outbreaks or epidemics and can often be traced to a single source (water or food). In other settings infections are acquired from pathogens endemic to an area (e.g., enterotoxigenic E. coli in Mexico)
Perhaps the most concerning issue is the emerging strains of bacterial pathogens resistant to commonly used antibiotics. Quinolone usage in the animal food industry during the 1990s in both the United States and abroad has been strongly associated with the rise in antibiotic resistant Campylobacter strains.9 Quinolone usage in the poultry and dairy food industry has since been banned in the United States. Human usage of antibiotics is a known contributing factor to growing antibiotic resistance in general but felt to be a lesser cause of rising Campylobacter resistance to quinolones and macrolides.9 In countries such as Thailand, emerging resistance to all commonly used antibiotics (trimethoprim-sulfamethoxazole, ciprofloxacin, azithromycin) for diarrheal illness has been documented.10
Pathophysiology
Stool is mostly water (60-90%), with normal daily volumes 100-300 mL. This amount varies depending on diet, particularly unabsorbable fiber. Diarrhea is defined as an increase in the volume, frequency, and fluidity of stool. Various criteria exist in the literature with greater than 3 loose stools in a 12-hour period being the most common. Acute diarrhea is defined as diarrhea lasting less than 14 days.
Diarrhea is the result of excess water in the stool. Normally the colon is able to regulate stool composition. Diarrhea occurs when the colon is unable to perform this function. This may be the result of increased transit time, increased osmotic load, or infection. Most of the morbidity and mortality of diarrhea is due to volume loss leading to dehydration, electrolyte depletion (Na, K, Cl, Mg), and in some cases systemic illness, sepsis, and death.
There are 4 major types of diarrhea based on pathophysiology: secretory, inflammatory, osmotic, and abnormal motility. Most acute infections fall under the inflammatory or secretory mechanisms.
 |
Inflammatory diarrhea occurs when there is cellular damage to the intestinal mucosa, resulting in loss of proteins, blood, water, and electrolytes. When inflammatory diarrhea is caused by infection, the invading organism must adhere to the intestinal lining. This is commonly referred to as dysentery, which classically manifests as loose stools with blood and mucous. Fecal leukocytes and erythrocytes are often present and can be detected for diagnostic purposes.
In contrast, secretory diarrhea involves the secretion of water and electrolytes (mostly chloride) into the lumen due to increased cellular permeability. This increased cellular permeability is a result of the direct action of secretagogues or pre-formed toxins (cholera toxin, Clostridium difficile toxin) or a direct action of enteropathogenic viruses (rotavirus). Most cases of acute infectious diarrhea fall into this category. Blood may be present to a small degree but the stools are mostly non-bloody and do not contain large amounts of mucous. Electrolyte disturbances may be severe resulting in weakness, paresthesias, cardiac conduction abnormalities, and even cardiovascular collapse.
Diagnosis
A thorough history and physical examination frequently provides most of the diagnostic workup. The history should focus on: recent travel, work and vocational exposure, sexual history (anal intercourse), recent antibiotic usage, contact exposure, vomiting, and the presence of other symptoms. Dietary history is often provided by helpful patients but is rarely beneficial in determining the cause or treatment. Patients' memories may be inaccurate, and reports are often biased by restaurant visits, most recent meals, and dietary variation. The types of high-risk foods that should be sought during history taking are: undercooked meat (rare, medium rare) or fish, unpasteurized dairy products, improperly refrigerated foods, or foods with a prolonged stay in the refrigerator. The differential diagnosis of acute infectious diarrhea also includes non-infectious etiologies such as ischemic colitis, ulcerative colitis, Crohn's disease, and radiation colitis among others.
A thorough history of stool patterns including frequency, consistency, presence of blood or mucous, or foul odor are important. Patients with infectious diarrhea may have a variety of other symptoms other than loose stools. Common symptoms include abdominal pain, bloating, cramping, tenesmus, fever, nausea, and vomiting. The history should also focus on determining the patient's volume status. Decreased urination, dark urine, weakness, fatigue, and thirst all suggest dehydration.
The physical examination should focus on identifying those patients with signs of toxicity or moderate to severe dehydration. The vital signs may show signs of severe volume depletion with the presence of tachycardia, hypotension, or orthostasis. Fever supports the diagnosis of infectious diarrhea. Other findings on physical examination involved in the assessment of volume status are jugular venous pressure, skin turgor, mucosal membranes, and capillary refill. A careful abdominal exam is crucial to detect the presence of surgical emergencies or peritonitis and may suggest a more severe invasive enteric pathogen. A rectal examination should be performed in all patients presenting with acute infectious diarrhea.
The most useful diagnostic step in the workup of diarrhea is determining whether it is an invasive or non-invasive organism. This differentiation can help guide the clinician in deciding whether further workup is necessary (stool cultures, blood work). Two major methods exist for this: Hemoccult testing of stool for hemoglobin and microscopic evaluation of stool for leukocytes. Hemoccult testing is the quickest and can be performed in seconds at the bedside. Testing stool for blood helps to categorize likely infectious agents and to identify infections more likely to require antibiotic treatment. Most of the infectious agents amenable to treatment with antibiotics fall under the invasive category and are heme positive.
 |
The simplest test to direct therapy is testing stool for heme. This is done most simply by obtaining a stool sample from digital rectal exam and using a Hemoccult developer card with developer. These tests are capable of detecting as little as 6 mg of hemoglobin in 1 gram of feces. The clinician must be aware of false positives and false negatives that can occur with occult blood testing. Diets heavy in red meat as well as peroxidase-rich vegetables can cause false positive tests for occult blood. Many drugs such as aspirin, steroids, colchicines, and NSAIDs can cause microscopic blood loss at therapeutic doses, which can affect interpretation of positive results.
 |
 |
Sending stool samples for fecal leukocytes evaluation can help, but is more time consuming and expensive.11 The presence of fecal leukocytes has been shown to be a predictor of positive stool culture.12 It is most useful when a rectal examination reveals no stool for testing, results of occult blood testing are in question (false positives), or a rectal examination cannot be performed. It is important to note that stool specimens positive for fecal leukocytes do not accurately predict the presence of C. difficile.13
The history and physical examination dictate the need for further workup, which can include: metabolic panel, CBC, U/A, stool for fecal leukocytes, stool C & S, stool for O & P, C. difficile assay. Indiscriminate laboratory testing in the setting of uncomplicated diarrhea rarely changes management. Focused testing is most helpful in patients who are sick, febrile, or have a prolonged course of illness.
Patients who have been treated with antibiotics in the last 2 months or who have a recent past history of C. difficile colitis (even if fully treated) warrant stool studies to detect the presence of C. difficile toxin. This is often done with a rapid ELISA on stool samples to detect C. difficile toxins A and B. Recovery of C. difficile in the stool in itself does not indicate a cause for diarrhea unless the stool is positive for toxin production.
The CBC helps assess whether the patient complaining of bloody diarrhea has lost a significant amount of blood. In most cases blood loss is minimal in dysentery. The hemoglobin and hematocrit are also frequently elevated in dehydration, offering clues to volume status. The clinician must interpret this with caution, however, as many other factors can elevate hematocrit such as living at higher elevations, smoking, and hematologic disease. The white blood cell count (WBC) can be useful in evaluating the invasiveness of infection. Although the degree of elevation may not help reveal which organism is responsible for infection, it may suggest a more invasive illness. The band count has traditionally been used to assess severity of infection but has never been shown in the literature to be a good measure of illness severity.14
Measurement of electrolytes is important, especially in secretory (watery) diarrhea illness and/or if signs of significant volume depletion are present. Blood urea nitrogen (BUN) and creatinine (Cr) are vital to rule out renal insufficiency and may suggest volume depletion with an elevated BUN/Cr ratio.
Stool cultures are the gold standard for diagnosis of dysentery. Not all patients presenting to the clinician with acute infectious diarrhea require cultures.15 Overuse of cultures is not cost effective and should be avoided. Stool cultures should be considered in the following circumstances: fever, bloody diarrhea, toxic appearing patient, patient with significant abdominal pain, possible acute flare of inflammatory bowel disease, recent antibiotic usage, immunosuppressed patients, employees in the food handling industry, day care workers, travel to an endemic area, history of anal intercourse.
The ordering of routine stool cultures in most hospitals will automatically initiate testing for salmonella, shigella, and Campylobacter. Be aware of the testing your laboratory routinely performs. Specific requests may need to be made if Yersinia, Vibrio, or E. coli strains are suspected. It is optimal practice to indicate sought-after pathogens on the laboratory request.
Stool samples can be sent to the laboratory for ova and parasite evaluation if the clinical suspicion is high for parasites (foreign travel, recurrent disease). Flexible sigmoidoscopy and/or colonoscopy with biopsy are indicated in some cases of severe diarrhea when the etiology cannot be ascertained with normal testing and the clinician needs to distinguish between infection, ischemia, and inflammatory bowel disease.
Clinical Management
The most important aspect of managing the patient with an acute diarrheal illness is assessing and replacing fluid volume. Laboratory studies can also assist with evaluation of the BUN/Cr ratio. The BUN is often elevated in cases of moderate to severe dehydration. If severe enough, an elevated Cr may also suggest early renal failure. The CBC can also be suggestive of volume contraction if the hemoglobin and hematocrit are elevated. An elevated urine specific gravity suggests the kidney is conserving body water due to dehydration. A dilute urine or low specific gravity suggests a more euvolemic state.
Rehydration can be done orally or intravenously depending on the clinical situation. Oral rehydration can be effective if the patient is able to hold down liquids and the proper replacements are chosen.16 It is the preferred method for mild to moderate fluid losses. It is important that not only body water but also electrolytes are replaced. There are many different strategies to accomplish this. Various sports drinks on the market have sodium and potassium replacement but usually contain simple sugars that may worsen diarrhea osmotically. Other products such as HEED or Pedialyte provide electrolytes without the sucrose and fructose. Pedialyte comes in many forms including popsicles for children. Salt tablets also can be of use. One to two tablets with 8-12 ounces of water can help replace lost sodium. Diluted fruit juices, non-caffeinated soft drinks, clear soups, broths, and crackers can all provide fluid and sodium replacement. Dairy products should be avoided as many infectious agents induce a transient lactase deficiency, which causes symptomatic lactose intolerance and thus may worsen symptoms. Food is encouraged along with liquids; however, dairy, sugar containing foods, and high fat foods may worsen symptoms. The BRAT diet (bananas, rice, apples, toast) has been well described and considered safe despite lack of proven efficacy.6
Intravenous rehydration may be indicated in those patients who are unstable or unable to drink. Isotonic fluids such as lactated ringers or normal saline should be used alone or in combination with oral rehydration in select instances. In children a 20 cc/kg bolus is initiated whereas in adults boluses of 250 cc to 1000 cc are indicated initially, depending on the patient's age, underlying cardiac status (history of CHF, ejection fraction), and renal status. The endpoint should be the improvement of the patient's hydration as evidenced by the patient making clear urine, improvement of symptoms, and resolution of laboratory abnormalities.
Electrolyte replacement can occur either intravenously or orally as well. Check for magnesium depletion in patients with hypokalemia. In the setting of severe magnesium deficiency, it is difficult to correct hypokalemia without also correcting magnesium levels.
Antibiotics or No Antibiotics?
There are few things in patient care that are as controversial as use of antibiotics in the patient with diarrhea. Although most physicians recognize the benefit of treating positive cultures, antibiotic therapy has been found to be most effective if instituted early, thus precluding culture-guided decision. In most cases of infectious diarrhea, a specific pathogen cannot be identified on that visit. Empiric antibiotics for bacterial diarrhea have been shown to be superior over placebo in select circumstances, however there are a number of downsides. First of all, most cases of enteritis resolve without intervention regardless of whether the source is bacterial or viral. Traditional teaching has been to avoid antibiotic therapy if Salmonella is suspected due to the risk of prolonged shedding of the organism. However, studies looking at this topic have revealed conflicting evidence, and the clinical significance of prolonged excretion is not clear.
 |
Evolving bacterial resistance to common antibiotics is a serious problem worldwide. In the United States, fluoroquinolones have been used in food-producing animals, which has been associated with a rising incidence of antibiotic resistance to Campylobacter that is isolated from a large percentage of chickens and beef.17 The highest rates of antibiotic resistance to Campylobacter have been found in Spain and Thailand, and these rates continue to rise.18
The preferred empiric antibiotic depends on the clinical situation. In most instances treatment with a fluoroquinolone such as ciprofloxacin is the best choice. It has been shown to be superior to trimethoprim-sulfamethoxazole and is given as 500 mg twice daily for 3-7 days.19 It should not be used in pregnant patients or children. If, however, the clinical suspicion exists for C. difficile infection (due to recent antibiotic use) or amebic dysentery, then metronidazole is a superior choice. Macrolides such as erythromycin and azithromycin have been tested and found effective for bacterial dysentery. Due to the high incidence of Campylobacter resistance to quinolones (as high as 85%) in Thailand, travelers to this country may respond better to macrolides. Azithromycin can be dosed in a 3-day course or single-dose therapy, and is also the preferred choice for pediatric patients since quinolones are contraindicated.20
 |
 |
Use of anti-diarrheal agents is controversial; however, most preparations are over-the-counter and patients have access to them regardless of physician recommendations. Loperamide is the most commonly used over-the-counter agent by patients.21 A variety of different preparations exist to help decrease the frequency of diarrhea. Most of the agents are effective at reducing symptoms and are often combined with empiric antibiotics for traveler's diarrhea with good results in selected patient populations. It is imperative to warn those patients who might be at risk for adverse outcomes from such treatment.22,23 Prolonged fever, toxic megacolon (C. difficile colitis), and hemolytic-uremic syndrome (E. coli 0157:H7) have all been observed in the setting of anti-motility usage. Fulminant amoebic colitis has even been observed with loperamide usage.24 Anti-diarrheal drugs can be divided into 2 major groups: anti-motility agents and bulk forming/toxin binders. Opium products and loperamide (Imodium) impair peristalsis, thus very effectively reducing the number of stools. In patients with toxic and/or invasive infections, anti-motility agents may increase the risk of worsening infection, toxic megacolon, and sepsis. These drugs are not recommended in pediatric patients or those with antibiotic-associated colitis, inflammatory bowel disease, or toxic dysentery. Bulk forming/toxin binding drugs such as kaolin/pectin (Kaopectate) or bismuth subsalicylate (Pepto-Bismol) do not impair peristalsis and are considered safer therapies.
 |
Disposition
The majority of patients with acute diarrhea can be managed as outpatients with supportive care. Exceptions to this are those with severe dehydration that cannot be reversed quickly such as in the elderly, those with poor ejection fractions, hemodynamic instability, children, acute renal insufficiency or renal failure, poor social support, mental impairment, and those failing outpatient therapy. Also, patients demonstrating systemic toxicity or immunosuppression due to chemotherapy, autoimmune disease, or organ transplants should be considered for admission to the hospital.
Special Populations
U.S. Military Personnel. As of January 2003, there were 1.4 million active-duty men and women in the U.S. armed services. Since 2001, hundreds of thousands of American service men and women have deployed to either Afghanistan or Iraq. Given the large numbers of personnel involved and the broad geographical range from which they originate, physicians should be familiar with this aspect of travel medicine. Physicians may provide care to military personnel while they are home on leave, or after they have finished their deployment.
 |
Although injuries predominate health care concerns in returning soldiers, infections of various sorts are common. Diarrhea is a common ailment and presenting complaint of overseas military personnel.25 They are especially vulnerable due to the lack of ability to secure "safe food." Attack rates are directly related to consumption of locally procured foods like raw vegetables, fruit, undercooked meat products, water, and ice. A 1979 study of U.S. army soldiers noted a nearly 50% incidence of diarrhea upon arrival to South Korea, most of which was from ETEC.26 Of the troops deployed to Thailand in 1993, 28% experienced diarrheal illness attributed to Campylobacter (25%), E. coli (13%), nontyphoidal salmonella (8%) and rotavirus (4%).27 More recently U.S. and British troops deployed to Afghanistan and Iraq have reported outbreaks of Shigella and norovirus.28 Aside from these outbreaks, surveillance cultures in these troops have revealed a 60% incidence of diarrhea with 50% of troops having multiple episodes. Aside from outbreaks, the most common offending pathogen again was found to be ETEC.28-30
Adequate hydration in soldiers during active duty can be difficult. Soldiers wearing full gear in hot climates already have the tendency toward dehydration prior to the onset of diarrhea. Aggressive hydration should occur in this otherwise usually healthy population.
Traveler's Diarrhea. Acute infectious diarrhea affects up to 55% of travelers.31 The number of people involved is in excess of 250,000 million annually. Illness can occur while traveling and after returning home. The exact incidence and etiologies vary depending on the geographic location traveled.
Most cases of traveler's diarrhea occur within the first 2 weeks of travel and usually last around 4 days without treatment.32,33 Most cases are non-life threatening but may involve severe symptoms and days lost from work.
Destination has the strongest association with occurrence of traveler's diarrhea with the highest risk areas being: Latin America, Africa, South Asia, and the Middle East. This is in contrast with the lowest risk destinations: northern and central Europe, United States, Canada, Japan, and Australia.31,34,35
Regardless of the destination, some patients are at higher risk when traveling such as those on acid reduction therapy (proton pump inhibitors, H2 blockers), and immunosuppressed patients. In contrast to developed countries where viral illness predominates, traveler's diarrhea obtained in developing countries is usually bacterial. Bacterial pathogens are spread through poor sanitation and mishandling of food and water.
When traveling, there are certain dietary sources that have demonstrated the highest risk such as salads, raw or poorly cooked meats and seafood, unpasteurized dairy products, tap water, and unpeeled fruits. Thus, eating in restaurants increases one's chance of contracting diarrhea, especially if consuming cold sauces or salsas.
The most common offender is enterotoxigenic E. coli (ETEC) with other common pathogens close behind such as: Campylobacter, Shigella, other E. coli species, Salmonella, Aeromonas, and Vibrio. Parasitic infections are not uncommon and include Giardia lamblia, Entamoeba histolytica, Cyclospora, and Cryptosporidium. Enteropathogenic viruses like rotavirus and norovirus occur with increased frequency in developing countries.
Recent outbreaks of infectious diarrhea on cruise ships have revealed norovirus as the culprit. This poses unique diagnostic challenges to the physician on cruise ships due to limited diagnostic capabilities and may involve the need to send cultures to nearby port towns for evaluation. Empiric antibiotics are not likely to be helpful in this setting unless the physician can confirm the presence of a bacterial pathogen.
Treatment. At this time, the Centers for Disease Control and Prevention (CDC) does not recommend prophylactic antibiotics. Despite this, it may be considered for immunosuppressed patients or other high-risk patients traveling to high-risk destinations. If antibiotic prophylaxis is given, fluoroquinolones are the treatment of choice with up to 90% effectiveness.32,33,36 Rifampin may be another option but further testing is needed.31
 |
Prevention. Prophylaxis in the form of probiotics (Lactobacillus) and bismuth subsalicylate (Pepto-Bismol) has been shown to have protection rates of up to 47% and 60% respectively but can be cumbersome due to frequent dosing.32,33 Bismuth preparations should not be used in patients taking anticoagulants, other salicylates, or in patients taking doxycycline for malaria prophylaxis.
Medical counseling of travelers and non-antibiotic prophylaxis do not eliminate the risk of traveler's diarrhea. Access to good quality medical care may be limited, therefore it is considered acceptable practice to provide reliable travelers with means for empiric self-treatment, which can significantly reduce morbidity.31
Immunosuppressed Patients. Organ Transplants. In the United States there are up to 27,000 organs transplanted annually. Diarrhea is a common complication of organ transplantation, which can result in significant morbidity and mortality.37 The differential diagnosis includes infection, medication effect (immunosuppressive agents, antibiotics, laxatives), and the surgical procedure itself. Bone marrow transplant patients may develop graft-versus-host disease, which manifests as diarrhea and can present as an acute infection. Traditionally, in the first 6 months after transplantation, opportunistic and viral infections are the rule.37 The most common opportunistic organisms causing diarrhea in the transplant patient are cytomegalovirus (CMV), microsporidium, cryptosporidium, Isospora belli, cyclospora, and Giardia lamblia. One study reviewed post transplanted kidney and liver patients and found Giardia, Cryptosporidia, and CMV the most commonly identified pathogens.38 Cryptosporidia is sometimes difficult to detect and intestinal biopsy may be needed to make the diagnosis. It is also very difficult to eradicate and may require reduction of immunosuppression. The nematode Strongyloides stercoralis has been shown to re-activate in the post-transplant patient as well.
 |
After 6 months if the transplanted organ takes well, normal community-acquired organisms predominate. Bacteria most commonly isolated from transplant patients with diarrhea include: Clostridium difficile, Yersinia enterocolitica, Campylobacter jejuni, Salmonella, and Listeria monocytogenes.39 Up to 50% of transplant patients receiving a therapeutic course of antibiotics develop C. difficile-associated diarrhea.39 These patients with enteritis may present with simple diarrhea, febrile enterocolitis, and toxic megacolon. Thus x-rays are indicated in the transplant patient with diarrhea and abdominal pain.
Opportunistic organisms are again found in high incidence during times of rejection and other periods of aggressive immunosuppression. CMV infection is the most common viral infection causing clinical symptoms after transplantation. The CMV virus after initial infection produces a life-long latent infection. Intestinal transplants are associated with the highest risk of latent virus reactivation. Other viruses commonly isolated from stool of post-transplant patients include adenovirus, calicivirus, and herpes virus.39
Transplant patients with diarrhea must be screened carefully for any signs of toxicity. Fluid status must be monitored carefully and a low threshold maintained for hospital admission. It is imperative that the physician communicate directly with the transplant service that manages the patient. Anti-motility agents should be used with great caution and are best avoided.
HIV Infected and AIDS Patients. Diarrhea, both acute and chronic, is a common problem affecting HIV-infected patients. Diarrhea has been shown to be an independent predictor of a lesser quality of life.40 It is most commonly caused by infection, drug therapy (nelfinavir, ritonavir), and HIV-associated enteropathy.41 Diarrhea can often be the presenting symptom that leads to the diagnosis of HIV infection, especially in underdeveloped countries. In one study up to 40% of HIV-infected adults reported at least one episode of diarrhea in the preceding month.42,43 It is important to realize that diarrhea may be due to infiltrative diseases such as lymphoma and Kaposi's sarcoma as well.
The clinical picture varies depending on many factors such as sexual habits and immune status. Patients who are HIV positive but who have low viral loads and higher CD4 counts generally experience the same community-acquired infections as the rest of the population. However, once the cellular immunity drops (CD4 < 100), a number of opportunistic pathogens enter the differential diagnosis. Opportunistic pathogens such as cyto-megalovirus, cryptosporidium, and microsporidium infect normal hosts but usually cause self-limited disease. In HIV-infected patients with lower immunity, the infection persists and becomes a chronic debilitating illness.
In some cases the etiology of infectious diarrhea can be inferred by the type and location of symptoms.41 For example, upper-mid abdominal cramping, bloating, and nausea suggest upper GI involvement, which favors MAC, Cryptosporidium, Giardia, or Isosporabelli infection. Patients with lower abdominal cramps, tenesmus, and hematochezia usually have colonic involvement with such pathogens as CMV, HSV, or bacterial pathogens such as Salmonella, Shigella, Yersinia, or Campylobacter.44
HIV patients also have a high incidence of C. difficile colitis. One retrospective review identified C. difficile as the most common pathogen, composing 54% of all bacterial pathogens.45
Males involved in anal intercourse may experience what has been referred to as Gay Bowel Syndrome. Along with the normal enteric pathogens, sexually acquired infections such as Chlamydia, Gonorrhea, LGV, condyloma accuminatum, syphilitic chancres, and Herpes simplex occur in patients who engage in anal intercourse. Many of these patients have concurrent infections with more than one pathogen.46
Many cases of acute infectious diarrhea in HIV-infected patients become chronic and are difficult to eradicate. Many of the opportunistic enteric pathogens in the AIDS patient can lead to malabsorption (MAC, microsporidia). This can be severe and may warrant hospital admission for nutritional support and aggressive GI workup.47
Care should focus on obtaining specimens for testing after hydration status has been addressed. In more than 50% of diarrhea illness in HIV-infected individuals, no identifiable pathogen can be found.48 Consultation with an infectious disease specialist may be indicated.
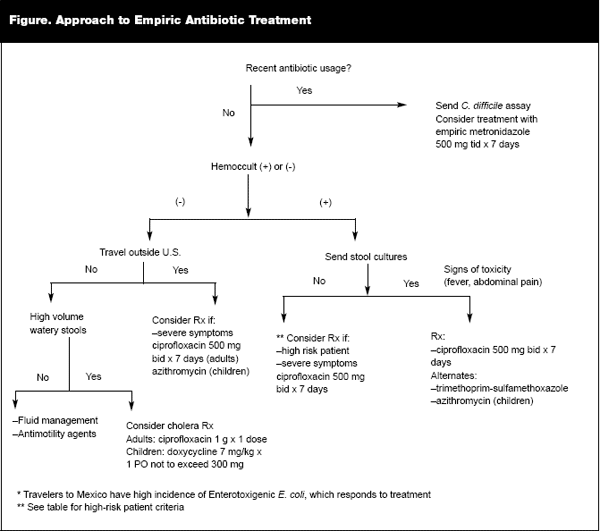 |
Summary
Acute infectious diarrhea is one of the most common clinical problems. A thorough history and physical examination suggests the diagnosis and treatment in most cases. It also avoids the unnecessary use of diagnostic testing. Although the majority of patients who present will have self-limited and mild disease, many will have conditions that warrant use of empiric antibiotics and anti-motility agents. Judicious use of antibiotics limits the spread of resistant bacteria and has been shown to be effective in treating patients' symptoms in select cases. The top priority in management of the diarrhea patient is to evaluate and manage dehydration. This can be done safely by oral administration of fluids and electrolytes in most patients. Intravenous fluids are reserved for those patients who are unable to drink, have severe dehydration, or have hemodynamic instability. The majority of patients can be managed and sent home; however, a low threshold for admission of immunosuppressed patients is important.
References
1. Lim MI, Wallace MR. Infectious diarrhea in history. Infect Dis Clin North Am 2004;18:261-274.
2. Gore JI, Surawicz C. Severe acute diarrhea. Gastroenterol Clin North Am 2003;32:1249-1267.
3. Ilnyckyj A. Clinical evaluation and management of acute infectious diarrhea in adults. Gastroenterol Clin North Am 2001;30:599-609.
4. Dennehy P. Acute diarrheal disease in children: Epidemiology, prevention, and treatment. Infect Dis Clin North Am 2005;19:585-602.
5. Thielman NM, Guerrant RL. Acute infectious diarrhea. N Engl J Med 2004;350:38-47.
6. Marx J, Hockberger R, Walls R. Rosen's Emergency Medicine: Concepts and Clinical Practice, 6th ed. St. Louis: Mosby;2006.
7. Sandler RS, Everhart JE, Donowitz M, et al. The burden of selected digestive diseases in the United States. Gastroenterology 2002;122:1500-1511.
8. Sanchez JL, Gelnett J, Petruccelli BP, et al. Diarrheal disease incidence and morbidity among United States military personnel during short-term missions overseas. Am J Trop Med Hyg 1998;58:299-304.
9. Engberg J, Aarestrup F, Taylor DE, et al. Quinolone and macrolide resistance in Campylobacter jejuni and E. coli: Resistance mechanisms and trends in human isolates. Emerging Infect Dis 2001;7:24-34. Available at www.cdc.gov.
10. Hoge CW, Gambel JM, Srijan A, et al. Trends in antibiotic resistance among diarrheal pathogens isolated in Thailand over 15 years. Clin Infect Dis 1998;26:341-345.
11. Turgeon DK, Fritsche TR. Laboratory approaches to infectious diarrhea. Gastroenterol Clin North Am 2001;30:693-707.
12. Savola K, Baron E, Tompkins L, et al. Fecal leukocyte stain has diagnostic value for outpatients but not inpatients. J Clin Microbiol 2001y;39:266-269.
13. Reddymasu S, Sheth A, Banks DE. Is Fecal Leukocyte Test a good predictor of Clostridium difficile associated diarrhea? Ann Clin Microbiol Antimicrob 2006;5:9.
14. Cornbleet P. Clinical utility of the band count. Clin Lab Med 2002:22:101-136.
15. Thielman N, Guerrant R. Acute infectious diarrhea. N Engl J Med 2004;350:1:38-47.
16. Duggan C, Santosham M, Glass R, et al. The management of acute diarrhea in children: Oral rehydration, maintenance, and nutritional therapy. MMWR 1992;41:001.
17. Food and Drug Administration—Center for Veterinary Medicine (CVM); press release; Sept 2001.
18. Hakanen A, Jousimies-Somer H, Siitonen A, et al. Fluoroquinolone resistance in Campylobacter jejuni isolates in travelers returning to Finland: Association of ciprofloxacin resistance to travel destination. Emerg Infect Dis 2003;9:267-270.
19. Oldfield EC 3rd, Wallace MR. The role of antibiotics in the treatment of infectious diarrhea. Gastroenterol Clin North Am 2001;30:817-836.
20. Tribble DR, Sanders JW, Pang LW, et al. Traveler's diarrhea in Thailand: Randomized, double-blind trial comparing single-dose and 3-day azithromycin-based regimens with a 3-day levofloxacin regimen. Clin Infect Dis 2007;44:338-346.
21. McMahan ZH, Dupont HL. Review article: The history of acute infectious diarrhea management—from poorly focused empiricism to fluid therapy and modern pharmacology. Aliment Pharmacol Ther 2007:25:759-769.
22. Ericsson CD, DuPont HL, Matthewson JJ. Optimal dosing of ofloxacin with loperamide in the treatment of non-dysenteric travelers' diarrhea. J Travel Med 2001;8:207.
23. Sanders J, Frenck R, Putnam S, et al. Azithromycin and loperamide are comparable to levofloxacin and loperamide for the treatment of traveler's diarrhea in United States military personnel in Turkey. Clin Infect Dis 2007;45:294-301.
24. McGregor A, Brown M, Thway K, et al. Fulminant amoebic colitis following loperamide use. J Travel Med 2007;14:61-62.
25. Sanchez JL, Gelnett J, Petruccelli BP, et al. Diarrheal disease incidence and morbidity among United States military personnel during short-term missions overseas. Amer J Trop Med Hyg 1998;58:299-304.
26. Echeverria P, Ramirez G, Blacklow NR, et al. Travelers' diarrhea among U.S. Army troops in South Korea. J Infect Dis 1979;139:215-219.
27. Echeverria P, Jackson LR, Hoge CW, et al. Diarrhea in U.S. troops deployed to Thailand. J Clin Microbiol 1993;31:3351-3352.
28. Aronson NE, Sanders JW, Moran KA. In harm's way: Infections in deployed American military forces. Clin Infect Dis 2006:43:1045-1051.
29. Monteville M, Riddle M, Baht U, et al. Incidence, etiology, and impact of diarrhea among deployed U.S. military personnel in support of Operation Iraqi Freedom and Operation Enduring Freedom. Amer J Trop Med Hyg 2006;75:4:762-767.
30. Wolf MK, et al. Characterization of enterotoxigenic Escherichea coli isolated from troops deployed to the Middle East. J Clin Microbiol 1993;31:4.
31. Yates J. Travelers' diarrhea. Am Fam Physician 2005;71:11.
32. Kamat D, Mathur A. Prevention and management of traveler's diarrhea. Dis Mon 2006;52:289-302.
33. Diemert D. Prevention and self-treatment of travelers' diarrhea. Prim Care 2002;29:843-855.
34. Al-Abri SS, Beeching NJ, Nye FJ. Traveller's diarrhoea. Lancet Infect Dis 2005;5:349-360.
35. DuPont H. New insights and directions in travelers' diarrhea. Gastroenterol Clin North Am 2006;35:337-353.
36. Rendi-Wagner P, Kollaritsch H. Drug prophylaxis for travelers' diarrhea. Clin Infect Dis 2002;34:628-633. ABSTRACT
37. Gautam A. Gastrointestinal complications following transplantation. Surg Clin North Am 2006;86:1195-1206.
38. Arslan H, Inci EK, Azap OK, et al. Etiologic agents of diarrhea in solid organ recipients. Transpl Infect Dis 2007; May 19: [ePub].
39. Sellin JH. The pathophysiology of diarrhea. Clin Transplant 2001;15:2-10. ABSTRACT
40. Watson A, Samore MH, Wanke CA. Diarrhea and quality of life in ambulatory HIV-infected patients. Dig Dis Sci 1996;41:1794-1800.
41. Wilcox C, Wanke C. Evaluation of the HIV-infected patient with diarrhea. October 2006; version 15.2. Available at www.uptodate.com.
42. Cohen J, West AB, Bini EJ. Infectious diarrhea in human immun-odeficiency virus. Gastroenterol Clin North Am 2001;30:637-664.
43. Knox TA, Spiegelman D, Skinner SC, et al. Diarrhea and abnormalities of gastrointestinal function in a cohort of men and women with HIV infection. Am J Gastroenterol 2000;95:3482.
44. Bonfim D, Jurado E, Favoreto CA, et al. Diarrhea in AIDS patients. Int Conf AIDS 1990;6:358. ABSTRACT #2017.
45. Sanchez TH, Brooks JT, Sullivan PS, et al. Bacterial diarrhea in persons with HIV infection, United States, 1992-2002. Clin Infect Dis 2005;41:1621.
46. Kazal HL, Sohn N, Carrasco JI, et al. The gay bowel syndrome: Clinicopathologic correlation in 260 cases. Ann Clin Lab Sci 1976;6:184-192.
47. Bonfim D, Jurado E, Ribeiro V, et al. Colonoscopic etiological diagnosis of diarrhea in AIDS patients. Int Conf AIDS 1991;7:244. ABSTRACT #2249.
48. Mayer H, Wanke CA. Diagnostic strategies in HIV infected patients with diarrhea. AIDS 1994;8:1639.
The prologue of Robin Cook's novel "Toxin" contains the following: "Within the pen an obviously sick cow was lying in its own diarrhea." What follows is an intriguing detective story of suspense and devastation caused by society's failure to control a tiny microbe called Escherichia coli.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
