2007 Salary Survey Results: New jobs are opening up and existing ones compete for workers in clinical research industry
2007 Salary Survey Results
New jobs are opening up and existing ones compete for workers in clinical research industry
Experts say more education is latest trend
It's a very good time to be an experienced clinical trials professional, according to experts and the 2007 Clinical Trials Administrator's salary survey.
"I definitely think this is a wonderful industry to be in," says Tamara Dowd Owens, RN, MSN, MBA, director of clinical trials for Pinehurst Medical Center in Pinehurst, NC. Owens also is an editorial advisory board member for Clinical Trials Administrator.
"I have had several friends and colleagues in the industry tell me they are getting calls left and right from job recruiters, and it sounds like there are numerous jobs in the field," Owens says. "And the pay sounds good too."
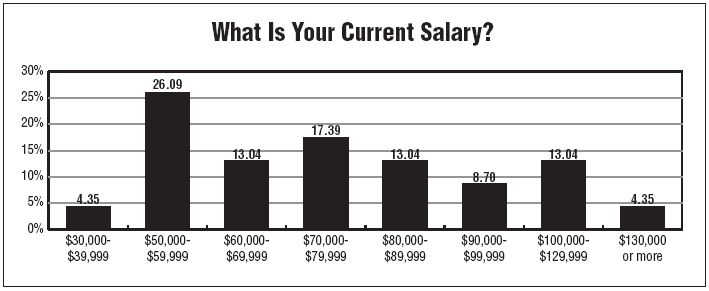
The CTA salary survey has found that nearly all respondents to the survey report having annual salaries of greater than $50,000 per year, with close to 70% reporting salaries greater than $60,000.
The survey's 23 respondents hold a variety of positions within the clinical research world including these: senior director of clinical research operations, IRB administrator, clinical research specialist, director of clinical trials protocol management office, medical director, certified clinical research coordinator, quality assurance/site trainer, and research manager.
Clinical research professionals, whether they come from nursing or other medical backgrounds, have a wider variety of career options in the current clinical research environment than they have had in the past.
For example, while many have gained experience working for clinical research sites, there are new jobs opening up at clinical research organizations (CROs), particularly in the relatively new field of working in site start-up groups, Owens says.
New job trends
CROs are creating lists of preferred CR sites, offering pharmaceutical companies value by saying they can provide a group of sites that are able to recruit X number of trial participants, Owens explains.
Once a CRO receives a trial contract, the organization goes to the site start-up group and tries to identify sites that may fit well with the protocol's recruitment needs. The site start-up group facilitates the recruitment and start-up process by having a site start-up specialist assist research sites with contract work, regulatory work, and other tasks, Owens says.
This relatively new position of start-up specialist is a great career move for clinical research professionals, Owens adds.
"This is absolutely creating new jobs," she says.
Another new job trend is in the area of pharmacovigilence, which was off the radar screen for nurses looking at career changes until recently.
"Pharmacovigilence is booming," Owens says. Nurses who decide to switch from hospital and clinical work are moving into this field where they become drug safety associates, she explains.
"If a clinical trial site has a serious adverse event occur, and it's sent to the CRO, then the event is reviewed by the site safety associate," Owens says. "The site safety associate makes sure all information in the initial report is done and puts the information into the FDA's MedWatch form."
The site safety associate is a separate career path than clinical research organization, but both types of jobs are drawing from the nursing pool of applicants, which has helped to make the clinical research job market challenging from an employer's perspective.
It's hard to find good people to work in clinical trials research, notes Barbara J. LoDico, BS, CIP, director of the human subjects research at The Children's Hospital of Philadelphia in Philadelphia, PA. LoDico is on the editorial advisory board of Clinical Trials Administrator.
The pay and work are rewarding, so the field is continuing to attract new candidates, but many are coming from fields other than nursing, LoDico says.
"They used to recruit just nurses, but now they're recruiting PhDs, MDs, clinical and advanced practice nurse practitioners, so I think what they're looking for has changed a little bit," LoDico says.
Research-specific curriculum
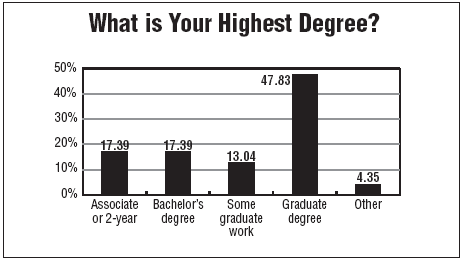
Also, some universities have begun to offer specific courses in clinical research with the goal of providing new graduates with backgrounds in either nursing or other fields who can find a job in clinical research and have training that replaces experience, LoDico says.
For example, Duke University has clinical research management programs for both nurses and professionals with other college degrees, says Elizabeth E. Hill, RN, DNSc, an assistant professor, nurse planner, and director of the Clinical Research Management program at the Duke University School of Nursing in Durham, NC. Hill also is on the editorial advisory board of Clinical Trials Administrator.
The clinical research management program includes courses in regulatory affairs and how to make an IRB submission, Hill says.
Traditionally, nurses might apply to work at a research site at their medical institution, and they acquire on-the-job training and experience, Hill says.
But the latest trend is for nurses and other professionals to acquire coordinated education about how to perform study coordinator tasks, she says.
The master's degree program in clinical research management at Duke is more for an advanced position in the research industry, but the principle serves the same goal of giving people with experience in other fields—mostly medical fields—education and training in clinical research for the purpose of helping them make a career change.
"We're the only group in the school of nursing that has something to offer people who are not nurses," Hill says. "You can apply if you have a graduate degree of some sort."
For example, some of the recent students have been Chinese physicians who are unable to practice medicine here, but who want to be involved in some aspect of science and medicine, Hill says.
Others are professionals who have earned a PhD in another field.
"We have a lot of people who have been in the industry for a while, and they feel they need a graduate degree to advance," Hill adds.
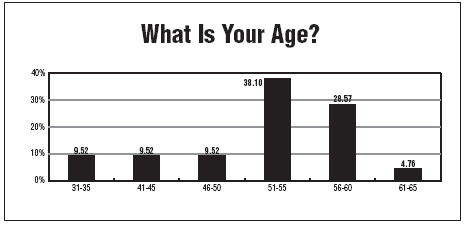
Since clinical research work often draws people who have started out in other fields first, it's a field that has mostly middle-aged and older workers, according to the experts and the salary survey.
More than 90% of those surveyed are age 41 or older, and more than 70% are age 51 and older.
"Really young nurses just coming out of school are not aware of clinical research and are not interested in it," Hill says. "It's better if they have a little experience anyway."
People don't start their career thinking they'd like to be a study coordinator, LoDico says.
What typically has happened is that a physician sees someone who is very bright and recruits that nurse, or a nurse decides he or she doesn't want to do floor duty anymore, LoDico says.
Clinical research is a good field for a second career, Owens says.
Salary-wise it's comparable to nursing, but the benefit is that the hours are more regular and it's a salaried position so there's more potential for advancement, Owens says.
Salaries likely will remain high due to the growth in jobs for people with research experience, the experts say.
"Everything is on the upswing," Owens says. "Small biotech companies and start-up companies and spin-offs from universities are doing clinical research and need staffs."
Also, more research is moving into the community where physician offices need experienced people to assist with clinical research, Owens adds.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
