Feds prioritize population to allocate scarce vaccine
Feds prioritize population to allocate scarce vaccine
Guidelines emphasize limiting spread
Since there have been pandemics and vaccines to fight them, ethicists have wrestled with the question of who should have priority when it comes to distributing vaccine. The federal government has released a draft in which it sets out how health authorities should allocate scarce doses of influenza vaccine in the event of a pandemic.
The interagency working group, made up of members representing all sectors of the federal government, sorted the population into four categories, and then ranked groups within those categories in order of priority. (See "Pandemic vaccine prioritization list," below.)
The draft was prepared from the most current scientific information available, "balanced with the values of our society and the ethical issues involved in planning a phased approach to pandemic vaccination," according to the paper. (The draft guidelines are posted at www.pandemicflu.gov.)
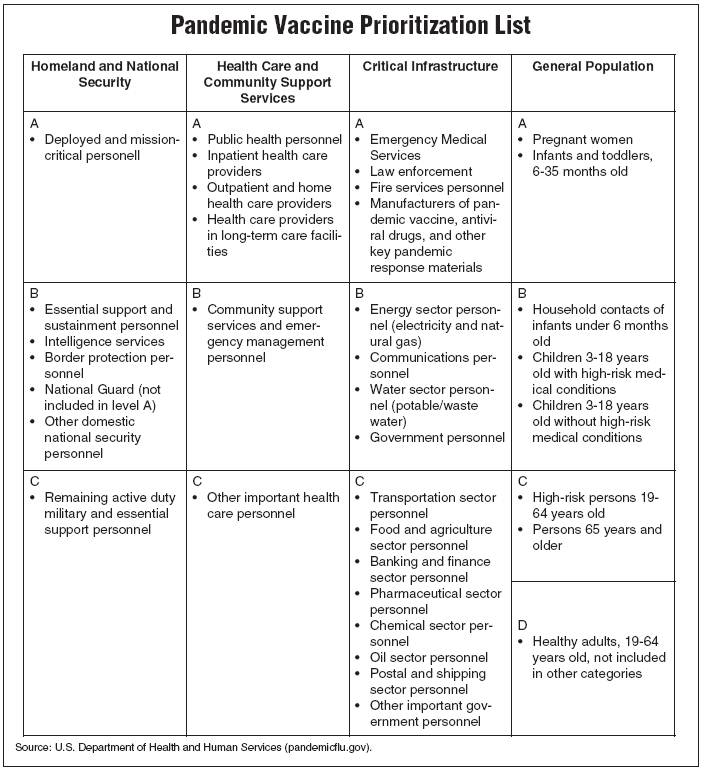
Babies, soldiers top the list
Infants, pregnant women, doctors, and soldiers are among those in the top tier, or Level A, on the pandemic prioritization list. The four categories are Homeland and National Security, Health Care and Community Support Services, Critical Infrastructure, and General Population.
In the lowest-priority category — Level D under General Population — are healthy young adults and the elderly. Listing the elderly in the lowest-priority group and young children in higher-priority groups reflects the working group's conclusion that the most effective use of limited pools of vaccine is to target those who are most likely to spread the flu, not just those most likely to suffer the most — or die — from it.
The working group reports that strong consideration was given to the ethical issues involved in allocating limited supplies of vaccine.
"Vaccinating some people earlier than others to minimize health and societal impacts of a pandemic was considered ethically appropriate," the report explains.
Principles the group considered were:
- fairness and equity (recognizing that all persons have equal value, and providing equal opportunity for vaccination among all persons in a priority group);
- reciprocity, defined as protecting persons who assume increased risk of becoming infected because of their jobs;
- flexibility to assure that vaccine priorities are optimally tailored to the severity of the pandemic and the groups at greatest risk of severe infection and death.
A second ethical focus was the importance of developing guidance through an open and transparent process with multiple opportunities and avenues for input from the public and stakeholders, according to the working group's report. Meetings were held with local representatives in several areas of the country as the draft was prepared, and when finalized, the guidelines will be open to interpretation and adaptation by states.
Estimates are that the federal government will have stockpiled enough vaccine against the current strain of avian flu for 13 million people by the end of 2007, with a goal of stockpiling enough for 20 million people. Twenty million is the estimated number of people in the top levels of the proposed prioritization list.
(Editor's note: Comments on the guidelines are being accepted by the Department of Health and Human Services through Dec. 28, 2007. Electronic responses are preferred and may be addressed to [email protected]. Written responses should be addressed to U.S. Department of Health and Human Services, Room 434E, 200 Independence Avenue, SW, Washington, DC 20201, Attention: Pandemic Influenza Vaccine Prioritization Guidance Comments.)
Since there have been pandemics and vaccines to fight them, ethicists have wrestled with the question of who should have priority when it comes to distributing vaccine. The federal government has released a draft in which it sets out how health authorities should allocate scarce doses of influenza vaccine in the event of a pandemic.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
