Special Feature: Analgesic Considerations in the Critically Ill and Injured Patient
Special Feature: Analgesic Considerations in the Critically Ill and Injured Patient
By Charles G. Durbin, Jr., MD
Pain is receiving increased attention from hospital regulators, public entities, and patients. Inadequate pain relief is often reported as a source of dissatisfaction with the health care experience. New standards suggest that pain in hospitalized patients be assessed frequently, to the extent as to be considered the "fifth vital sign" along with pulse rate, blood pressure, respiratory rate, and temperature. While pain management is essential to good care, pain is a subjective experience, something that is difficult to quantitate and especially challenging in the critically ill or injured patient. Opiates (opioids) are the mainstay of treatment of most cases of severe pain. There are a number of significant problems from use of opiate medications in the critically ill, especially when they are used as continuous infusions for sedation. Expanded understanding of pain mechanisms and application of specific techniques used in the operating room to the ICU has contributed to improved patient outcome from pain management in these challenging patients in this difficult environment. In this review I consider some ideas for pain management that apply to the critically ill and injured patient, areas that are often overlooked in more general reviews.
Pain and Agitation
Agitation is a common condition in the critically ill. More than 70% of ICU patients experience some degree of agitation during their ICU stay.1,2 Altered mentation, drug use, as well as the underlying disease, often render ICU patients unable to comprehend the purpose of the therapies or monitoring devices that are a normal part of their care. The causes of agitation in the ICU are numerous. Medical and surgical conditions resulting in ICU admission may by themselves trigger confusion and agitation; for example, sepsis and the systemic inflammatory response syndrome (SIRS) often alters mentation and produces agitation. Other issues resulting in agitation include the discomfort associated with endotracheal intubation, mechanical ventilation, therapeutic surgical and diagnostic procedures, injuries, anxiety, sleep deprivation, and delirium.3
Agitation itself can have deleterious consequences in the critically ill, including interfering with effective mechanical ventilation, causing acute myocardial stress and failure, and aggravating cerebral ischemia.4,5 Agitated patients are often unable to cooperate with diagnostic evaluations (ie, pain assessment), and may interfere with the performance of therapeutic procedures. Of particular importance is the self-removal or disruption of devices used for the diagnosis, treatment, or monitoring of the critically ill. This occurrence may cause injury or death, increase length of stay, and increase costs.6,7 Patient agitation frequently contributes to the stress and discomfort experienced by family and friends, and may affect overall satisfaction with health care delivery.8
Treatment of Agitation and Use of "Chemical Restraints"
In many ICU patients, agitation is a major clinical problem. While classic treatment of agitation consists of identification and elimination of its cause, due to the significance of uncontrolled behaviors associated with agitation in these patients, restraints or pharmacologic agents are often needed to prevent harm prior to specific therapies. In fact, agitation associated with delirium is so common that it could be considered a disease or condition in its own right. Indiscriminant use of physical and chemical restraints is rightfully under attack by regulatory agencies. Confusion exists in the ICU surrounding use of restraints. While the same devices and drugs may be used, these are used "therapeutically" in the ICU, not to reduce the need for careful observation and attention by caregivers. As with chemical and physical restraints used elsewhere, appropriate patient assessment, frequent close observation, and time-limited orders in the ICU will help prevent unnecessary limitation of liberty and morbidity.
Pharmacologic restraints are those medications that are primarily used to control extreme behavior emergently or to restrict the patient’s freedom of movement. Under this definition, a number of pharmaceutical agents commonly used in the ICU could be included in this category. Among them are analgesics, particularly opioid analgesics, sedative agents, particularly benzodiazepines, neuroleptic agents and other major tranquilizers, dissociative agents, and neuromuscular blocking agents. Since most agitated patients in the ICU are treated with one or more of these medications, some people contend that these patients are having chemical restraints applied. The usual goal of therapy with these agents is to treat the underlying cause of the patient’s agitation, and not to simply prevent the patient’s movements. The use of these therapeutic modalities should not be considered synonymous with application of chemical restraints, even though an expected outcome of the therapy will be a decreased level of inappropriate movement and greater cooperation with other needed therapies. Use of these pharmacologic treatments (other than neuromuscular blockers) is analogous to the use of oxygen in treating an agitated patient who is hypoxic. Thus, the use of sedatives, analgesics and neuroleptics in agitated ICU patients is often an appropriate therapy rather that a chemical restraint.
The primary goal in using analgesics is the reduction of pain; sedative agents, the relief of anxiety; and neuroleptic agents, the normalization of thought processes. The appropriate use of these medications during treatment of the agitated patient will not only alleviate pain and anxiety, but will also ameliorate the physiological derangement engendered by the underlying disorder. Thus, these agents in the agitated patient are considered as alternatives to physical and chemical restraints, rather than chemical restraints themselves.
Neuromuscular Blockers and Sedation
Neuromuscular blockers should never be used solely to restrain a patient but only as part of a therapeutic plan. For instance, muscle relaxants may be a necessary part of a low tidal volume ventilation plan with permissive hypercapnia. Sedation and analgesia must also be part of the therapeutic medication plan. The analgesic and sedative medications are more difficult to titrate to appropriate end points if the patient is paralyzed.
Sedation vs. Analgesia
While I wish to confine this discussion primarily to analgesia, some analgesics are used for sedation and often sedatives are used in conjunction with analgesics. Opiates, the most commonly used analgesics in the ICU, are often inappropriately used for their sedative side effects. This is true mostly in poorly responsive, nonverbal, and delirious patients in whom it is anticipated that pain is an expected consequence of therapy or injury. Clinician belief that the patient is in pain drives therapy in many of these cases. Since opiates are well tolerated hemodynamically, they are often used for sedation in agitated patients who may or may not be in pain. Several are short acting (fentanyl), and all opiates may be reversed with a specific antagonist (naloxone) should inadvertent overdose occur. If the patient remains agitated despite a bolus of what is expected to produce adequate analgesia, then small doses of a drug in a different category should be titrated in order to achieve the desired sedation. This approach, although not absolutely perfect, should produce a low incidence of drug induced delirium and inappropriate opiate dependence.
Opiates: Good, Bad, and Ugly
Continuous infusion of opiates in the critically ill and injured patient is common. When sedation or treatment of agitation is the goal, often this is the wrong approach. Bolus dosing helps prevent overdose and tolerance. Analgesia should be measured with a visual or numerical analogue scale and drug dosages titrated or administered to achieve an acceptable reduction in or level of severity. If patients cannot cooperate with a pain assessment process, it is dangerous to place them on a continuous infusion of opiate. Because tolerance to the effects of opiates occurs rapidly, especially the sedative side effects, the dose needs to be increased frequently to achieve the same initial effect. If patients can be evaluated with a visual analogue scale, there is little risk of inappropriate drug escalation. There is no ceiling effect on most opiates and the dose can be advanced until the desired level of analgesia is achieved. If a patient is maintained on the same continuous infusion rate for a period of days, it is not likely that pain is the reason that the opiate drip is being used.
The routine use of continuous opiate infusions to provide sedation is a practice that should be abandoned. Patients rapidly develop tolerance. A major problem with using opiates is that painful withdrawal symptoms will occur when opiate is discontinued. Other problems and side effects of the opiates are listed in Table 1. After prolonged continuous infusion (2-3 days), a patient must be slowly withdrawn from these agents to avoid these extremely unpleasant and potentially dangerous symptoms. Length of stay and cost of care are greater with continuous infusions.9,10 It is better to not start the drip in the first place than to have to treat the withdrawal syndrome that follows removal.
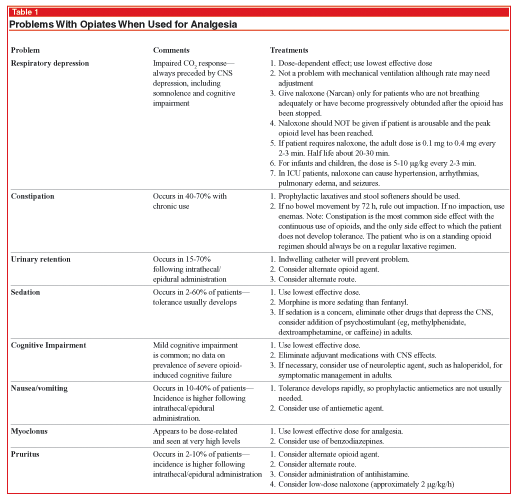
A more logical approach to sedation in patients without severe pain is to use a nonopiate analgesic (if necessary) and a sedative agent. There are sedative scales that can be used to assess the degree sedation and one of these should be employed to avoid over use and to document the success of the therapy. A typical sedation scale is shown in Table 2. Benzodiazepines are the agents of choice for sedation in the crucially ill due to their lack of organ toxicity, lack of cardiovascular effects, predictable effects, and reduction of memory formation. They may cause disorientation and confusion in some patients. They do accumulate in the body and a withdrawal syndrome is now recognized. Symptoms are usually mild with agitation and anxiety being common. Less frequently patients will experience profound withdrawal symptoms, which include: agitation, anxiety, panic, and even seizures in some cases. Withdrawal symptoms can be minimized and seizures prevented by slowly withdrawing the benzodiazepine over a period of weeks. Benzodiazepines can be given by bolus or continuous infusion. Midazolam, a short acting agent, is often administered by continuous infusion, titrating the infusion rate to signs of agitation or complaints of anxiety.
|
Table 2 |
|
| Sedation Scale* | |
| Level |
Patient Response |
| 1 | Anxious, agitated, or restless |
| 2 | Cooperative, oriented, tranquil |
| 3 | Quiet, responds to verbal commands |
| 4 | Asleep, brisk response to forehead tap or loud verbal stimulus |
| 5 | Asleep, sluggish response to forehead tap or loud verbal stimulus |
| 6 | Unresponsive |
|
* A sedation scale such as this can be used to assess and document the patient’s degree of sedation. |
|
Propofol
A better choice for sedation may be propofol. This agent is an anesthetic-amnestic drug dissolved in a fat emulsion. It has a short half-life, does not accumulate, and must be given by continuous infusion. At low infusion rates (10-30 mg/kg/min) it is primarily a sedative. Patients not receiving simultaneous opiates will be able to communicate. However, they are usually calm, often falling asleep when not stimulated. At higher dosage levels (eg, 50-200 mg/kg/min), general anesthesia is produced. The effects on hemodynamics are related to volume status and cardiac reserve. In general, blood pressure falls due to the reduced sympathetic tone resulting from loss of consciousness. Bolus doses of propofol often cause hypotension due to this mechanism as well as through a direct (although minor) cardiac depressive effect.
There are several other drawbacks and problems with propofol. It is an ideal bacterial growth medium. Bottles must be discarded after 12 or maybe 24 hours after being opened depending on the preservation technique, and deaths due to infusion of contaminated propofol have been reported. A major benefit of this drug is its short duration of action. Prolonged infusions, for periods up to weeks, are still followed by rapid awakening when the drug is stopped. The major disadvantage is drug cost. This changes with patent protection and will not always be as much of a concern. Accumulation of lipids in the blood is possible with prolonged infusions at high rates, as is calorie overload. The rare association of propofol infusion and the development of fatal lactic acidosis is suspected, but not proven. Use of this agent in children is controversial. Tachyphylaxis does occur with propofol but rarely is more than a doubling of the rate needed to achieve the same sedation end point.
Midazolam, a short-acting benzodiazepine, has been suggested as an alternative to propofol. After prolonged continuous administration or repeated doses, its duration of action increases. Wake up time is prolonged when compared to propofol. When used as a continuous infusion, level of consciousness cannot be used as the end point as with propofol. The benzodiazepine effects on mood (sedation, anxiolysis) and the prevention of memory formation are useful properties of this class of drugs. However, these beneficial effects can easily be achieved with bolus administration of midazolam or a less expensive, longer acting agent such as lorazepam.
Nonopioid Analgesic Medications
One of the frequently used analgesic classes outside the ICU, nonsteroidal anti-inflammatory drugs (NSAIDs), is used to treat moderate pain. NSAIDs work by affecting the hypothalamus and by inhibiting the production of inflammatory mediators, primarily prostaglandins, at the peripheral site of the painful stimulus. The salicylates are the oldest member of this class, and have been known for over 100 years for their effects as antipyretics. Aspirin decreases the synthesis of prostaglandin by irreversibly inhibiting both of the cyclooxygenase enzymes, COX-1 and COX-2. COX-1 is primarily located on tissues, including blood vessels, the kidney, and gastric mucosa, while COX-2 is primarily associated with inflammation. Unlike aspirin, other NSAIDs reversibly inhibit these enzymes, and may have fewer or less severe side effects. Selective COX-2 agents appear to have similar efficacy and a better safety profile.
Gastric irritation and ulceration are major problems with administering NSAIDs. Renal injury can result from prolonged use and high dosage of these medications. They also inhibit platelet aggregation; and this compounds the problem of gastrointestinal bleeding. The anti-platelet effects are used therapeutically following or to prevent cardiac thrombosis. Aspirin use in childhood febrile illness has been associated with an increased incidence of Reye’s syndrome, an often-fatal rise in intracranial pressure with massive hepatic dysfunction.11,12 The use of aspirin in critically ill children is controversial. Allergic reactions, although usually minor, to this class of drugs, are common. Rashes, urticaria, angioneurotic edema, asthma, and anaphylaxis can also occur.
Acetaminophen, although a weak inhibitor of the cyclooxygenase system, has no significant anti-inflammatory effects, although it is effective in relieving mild-to-moderate pain. It does not inhibit platelets or cause gastric ulcers. In large doses, greater than 4 g/d, it can cause lethal hepatic necrosis.
While all the NSAIDs are rapidly absorbed from the gastrointestinal tract, the effects of administration by this route are unpredictable in the critically ill. One NSAID, ketorolac, is available in an IV form. It is as effective as others in this group, and probably no safer. Recently, selective COX-2 inhibitors have been released into clinical practice. They should have fewer gastrointestinal side effects and fewer effects on platelet and white blood cell function. Their role in treatment of pain in the critically ill patients is yet to be determined. Patients with sulfa allergies and asthma need to be cautious with these as well as all of the NSAIDs.
Critically ill patients reporting moderate to severe pain who can receive one of these agents should have a baseline administration of a NSAID. They may also require intermittent administration of an opiate based on their visual analogue pain scale changes.
Regional and Neuraxial Anesthesia
Another approach to pain management is to block or modify the pain transmission pathway. This can be done with local anesthetics delivered in a variety of ways. The most common technique is epidural administration. Continuous epidural infusions for analgesia have improved postoperative and traumatic pain treatment. There is even evidence that patient survival is improved with epidural infusions, especially in the very ill.13-15 The quality of the analgesia and the ability to eliminate pain in many body areas is superior with local anesthetic infusion when compared to systemic analgesics. Minimal effects on normal sensory and motor function can be achieved with very dilute local anesthetics. Addition of opioids to the mixture permits even less local anesthetic to be infused. If sympathetic blockade produces unacceptable hypotension, local anesthetics can be eliminated completely with significant analgesia obtained from narcotic infusion alone.
Pain modulation from epidural opioids occurs at receptors at the spinal cord segmental level. In fact, even systemic opioids probably have their major analgesic affects at the spinal cord level. The central nervous system effects are primarily modification of the pain perception. The technique of epidural catheter placement in the critically ill is complicated due to mental status changes, patient positioning, and coagulopathy. The major side effects and concerns of providing epidural analgesia in the critically ill are listed in Table 3.
| Table 3 | |
|
Technical Issues, Concerns, Side Effects, and Complications from Epidural and Spinal Analgesia |
|
| Placement | |
| Patient positioning | Sitting is best, lateral possible |
| Level of consciousness | Patient must be awake and responsive to avoid nerve root injury (pain on injection) identify correct placement |
| Coagulation |
Normal coagulation is mandatory including platelet count at least 50K, INR < 1.4 if catheter to be inserted or removed |
|
Drug Considerations |
|
|
Local Anesthetics |
Concentration dependent |
| Numbness | Concentration dependent |
| Hypotension | Concentration and dose dependent |
| Difficulty diagnosing epidural hematoma | The severe back pain from an epidural hematoma may not be apparent in a heavily sedated patient. |
| Opioid Side Effects | |
| Respiratory depression | Equal or less than systemic opioids |
| Reduced GI motility | Greater than systemic opioids |
| Nausea and vomiting | Equal to systemic opioids |
| Difficult micturition | Greater than systemic opioids |
| Pruritus | Much greater than systemic opioids |
Spinal placement of narcotics is an alternative to epidural analgesia. The technique is less complicated, but analgesia is limited since no catheter is left in place. When morphine (eg, 100-300 mg) is placed in the lumbar intrathecal space, analgesia for up to 24 hours is usually obtained.
If major neuraxial analgesia is contraindicated or technically difficult, peripheral nerve block may eliminate pain and reduce the need for systemic analgesics.
Summary and Conclusions
Opioids are poor analgesics. They mainly alter pain perception and cause sedation. Opioid infusions are often used for sedation in the critically ill patient. Tolerance to the sedative effects of opiates leads to increased infusion rates. Withdrawal of opiate infusions leads to severe, painful symptoms and must be done slowly. Prolonged ICU stay can result as a consequence of these inappropriate opiate infusions. Opioids should be give intermittently in response to reported pain.
Sedatives, such as propofol or the benzodiazepines, are alternatives to opiates for sedation. Sedatives and analgesics in delirious patients are not "chemical restraints": they are treatment for the delirium.
Pain should be measured and documented in the critically ill. The use of analgesics other than opiates should be considered to decrease the patients’ reported pain. Epidural, spinal, and regional analgesia with local anesthesia are reasonable and possibly superior alternatives for pain relief in some critically ill patients and may actually improve survival as well as the quality of analgesia.
References
1. Fraser GL, et al. Frequency, severity, and treatment of agitation in young versus elderly patients in the ICU. Pharmacotherapy. 2000;20:75-82.
2. Ely EW, et al. Evaluation of delirium in critically ill patients: validation of the confusion assessment method for the intensive care unit (CAM-ICU). Crit Care Med. 2001;29:1370-13791.
3. Treggiari-Venzi M, et al. Overnight sedation with midazolam or propofol in the ICU: Effects on sleep quality, anxiety, and depression. Intensive Care Med. 1996;22:1186-1190.
4. Mazzeo AJ. Sedation for the mechanically ventilated patient. Crit Care Clin. 1995;11:937-955.
5. Bruder N, et al. Energy expenditure and withdrawal of sedation in severe head-injured patients. Crit Care Med. 1994;22:1114-1119.
6. Fraser GL, et al. The frequency and cost of patient-initiated device removal in the ICU. Pharmacotherapy. 2001;21:1-6.
7. Boulain T. (Association des Reanimateurs du Centre-Ouest). Unplanned extubations in the adult intensive care unit: a prospective multicenter study. Am J Respir Crit Care Med. 1998;157:1131-1137.
8. Osterman ME, et al. Sedation in the intensive care unit: A systematic review. JAMA. 2000;283:1451-1459.
9. Kollef MH, et al. The use of continuous i.v. sedation is associated with prolongation of mechanical ventilation. Chest. 1998;114:541-548.
10. Kress JP, et al. Daily interruption of sedative infusions in critically ill patients undergoing mechanical ventilation. N Engl J Med. 2000;342:1471-1477.
11. Hurwitz ES, et al. Public Health Service study of Reye’s syndrome and medications: Report of the main study. JAMA. 1987;257:1905-1911.
12. Forsyth BW, et al. New epidemiologic evidence confirming that bias does not explain the aspirin/ Reye’s syndrome association. JAMA. 1989;261: 2517-2524.
13. Yeager MP, et al. Epidural anesthesia and analgesia in high-risk surgical patients. Anesthesiology. 1987; 66:729-736.
14. Ackerman WE 3d, et al. Beneficial effect of epidural anesthesia on oxygen consumption in a parturient with adult respiratory distress syndrome. Southern Med J. 1993;86(3):361-64.
15. Kirsch JR, et al. Preoperative lumbar epidural morphine improves postoperative analgesia and ventilatory function after transsternal thymectomy in patients with myasthenia gravis. Crit Care Med. 1991;19(12): 1474-1479.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
