Special Series: Assessing Risks/Benefits - ASSERT statement offers guidance in trial design
ASSERT statement offers guidance in trial design
Checklist aids in ethical evaluation
IRBs and investigators who would like an organized checklist of steps to take to ensure human subject protection and the highest ethical standards in designing clinical trials could look no further than the ASSERT statement. Based on existing ethical requirements and guidelines, including the CONSORT (Consolidated Standards of Reporting Trials) statement, published in The Lancet in April 2001, the ASSERT statement, which stands for A Standard for the Scientific and Ethical Review of Trials, was created by Howard Mann, MD, program associate of the division of medical ethics at the University of Utah School of Medicine in Salt Lake City. Mann also is a radiologist at the University Hospital and Clinics in Salt Lake City.
"I was prompted to develop the ASSERT statement and the associated checklist after becoming aware of the CONSORT statement that addresses the reporting of clinical trials," Mann says. "Important items in the CONSORT checklist are included in the ASSERT’s checklist." The ASSERT statement addresses important, but neglected, aspects of randomized clinical trials, he adds. Mann suggests that IRBs include ASSERT’s requirements in their applications for research, but to keep in mind that ASSERT alone is insufficient guidance for IRBs in evaluating clinical research proposals. Here’s a brief look at the ASSERT statement and the issues it raises:
• Checklist table: The on-line checklist provides links to longer explanations of an application section.
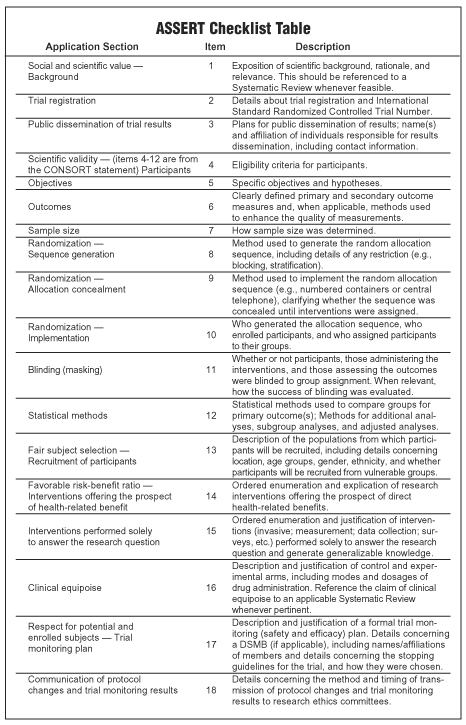 |
• Systemic reviews: Referencing the Declaration of Helsinki, the statement supports systemic reviews in order to determine a proposed clinical trial’s relevance, avoidance of duplication, and prevention of exposing human subjects to interventions that lack therapeutic efficacy. The four main parts of a systematic review, according to the statement, are:
- assessment of clinical value and relevance;
- assurance of clinical equipoise;
- assessment of significance of interim results analysis;
- contextual reporting of results.
Also, systemic reviews may reference existing reviews found in the Cochrane Library or a review performed by an investigator involved in the trial, the ASSERT statement adds.
• Outcomes and social value: All members of an IRB are responsible for assessing a potential trial’s social value, including relevant outcomes, according to ASSERT. "Investigators may not evaluate pertinent and scientifically-relevant outcomes," Mann says. "An example is the inappropriate choice of surrogate outcome measures." Important insights into social value may be gleaned from the input of consumers, patients, and laypersons, he says. Also, a study’s specified outcomes may not represent those that matter to patients, which is why it’s important to have patient/consumer involvement in planning trials, Mann adds. "Thus, IRBs should request investigators to justify their choice of outcome measures," he says. "A formal trial monitoring plan should indicate the manner in which outcomes will be assessed during the trial." Often, trial protocols do not contain an assessable monitoring plan, Mann notes.
• Disseminating trial results: The public release of a study’s results is a final step in the research process, and when investigators fail to disseminate scientific research results, this can have an adverse effect on published results of other trials, and investigators who fail to report their results are considered to have engaged in misconduct, the ASSERT statement says. The statement identifies these precise ethical imperatives for the public dissemination of research:
- a judgment that the research meets the requirement of scientific and social value presupposes the public dissemination of results;
- justification for the inclusion of nontherapeutic interventions designed to answer the research question;
- avoidance of publication bias, which adversely affects clinical decision making and the utility of systemic reviews and meta-analyses of the published literature;
- honoring the altruistic motivations of patient-subjects who agree to participate in a clinical trial;
- entitling participants to know the results of the research their enrollment made possible;
- consistent dissemination with the duty to share new knowledge with colleagues, commonly found in professional codes of conduct.
• Risk-benefit ratio: ASSERT states that the design of a research protocol should maximize benefits and minimize risks to participants, and to do so requires IRBs and research ethics committees to conduct a systematic, nonarbitrary assessment of risks and benefits associated with the protocol. In doing so, an IRB should make a distinction between therapeutic and nontherapeutic interventions, Mann says. "The regulatory criterion of minimal risk, when applicable, should be applied to the non-therapeutic interventions," Mann says. "An IRB should be rigorous in ascertaining that clinical equipoise is present."
Quoting the Declaration of Helsinki, principle 29, ASSERT states: The benefits, risks, burdens, and effectiveness of a new method should be tested against those of the best current prophylactic, diagnostic, and therapeutic methods. "Clinicians should consider the nature, magnitude, likelihood, reversibility, and timing of potential harms, when designing and evaluating research proposals," he explains.
(Editor’s note: To see the entire ASSERT statement and checklist, visit the following web site: www.assert-statement.org/.)
IRBs and investigators who would like an organized checklist of steps to take to ensure human subject protection and the highest ethical standards in designing clinical trials could look no further than the ASSERT statement.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
