Procedural Sedation: General Principles and Specific Pharmacotherapeutic Strategies — Part I
Authors: Jonathan Glauser, MD, FACEP, Department of Emergency Medicine, Cleveland Clinic Foundation, Cleveland, OH; Faculty, Emergency Medicine Residency, MetroHealth Medical Center, Cleveland, OH; Brian Cullison, MD, Resident, Department of Emergency Medicine, MetroHealth Medical Center, Cleveland, OH.
Peer Reviewers: Sandra Schneider, MD, FACEP, Professor and Chair, Department of Emergency Medicine, Strong Memorial Hospital, University of Rochester, NY.
Procedural sedation and analgesia entail the proper administration of drugs to obtund, dull, or reduce the intensity of pain or awareness. The administration of those drugs by any route is intended to allow the patient to maintain airway control independently and continuously, but carries the risk of loss of protective reflexes.1 There is a continuum from anxiolysis to conscious sedation, deep sedation, and general anesthesia. Many procedures performed by emergency physicians are time-sensitive, such as reduction of a dislocated extremity. As well, it is important to be able to discharge patients from the emergency department (ED) in a timely manner when the procedure is completed.
Patients undergoing deep sedation require more vigilant monitoring and airway management, including possible intubation. As a rule, those undergoing conscious sedation require pulse oximetry, cardiac monitoring, blood pressure monitoring, and close observation, since these patients are more easily arousable throughout the whole procedure. It is essential that the ED physician be prepared to manage the airway in the event there is an unforeseen response to induction of sedation. Emergency intervention may require reversal agents (i.e., naloxone and flumazenil), oxygen, and availability of intubation equipment (i.e., suction, bag-valve-mask device, laryngoscopes, endotracheal tubes, stylet and oral and nasal airway adjuncts). Monitoring devices, including a cardiac monitor, pulse oximeter, blood pressure monitor, and possibly capnography should be in place before sedation is begun.
Sedation and analgesia also are being used with greater frequency in children than they have been in the past. Broad policy statements by the American Academy of Pediatrics (AAP) and the American College of Emergency Physicians (ACEP) offer guidelines for drug administration and monitoring in this patient population.2 There are multiple routes that are available for administration of these drugs, including inhalation, intravenous (IV), intramuscular (IM), rectal, oral, and nasal.
The preferred route of administration for agents used in procedural sedation is IV. The IV route enables repeated doses and titration to clinical effect. Onset of drug effect is more rapid and predictable via this route. As well, IV access enables prompt administration of reversal agents. Exceptions to this rule may occur, especially in children, when alternative route of drug administration may preclude needle sticks or when IV access is impractical.
It cannot be stressed enough that sedation optimally is accomplished using at least two clinicians, one of whom must be a physician. The other must be a trained clinical provider who is dedicated to monitoring the patient while the physician performs the procedure for which sedation is required. It is critical to appreciate that, when inducing sedation, host response may vary the dose of medication administered. As a rule, dosages are just recommended guidelines that reflect the patient’s expected response to the drug in question.
Because patient response will vary, a dedicated observer and a qualified physician must be available to respond to any untoward or exaggerated host response. For purposes of emergency physicians, the term procedural sedation will be employed, as level of consciousness desired may vary by procedure. Sedation in the ED, in general, serves the purpose of accomplishing a specific task, whether it be computed tomography (CT) in a child, fracture reduction, cardioversion, or laceration repair, after which it may be desirable for the patient to be discharged in the care of a responsible adult. Patient evaluation is essential. (See Table 1.)
 |
All deep sedation must be carried out in an area fully equipped for resuscitation, and managed by providers knowledgeable in the medications used and skilled in advanced life support. The sedation must be monitored by a provider whose sole function is to directly monitor the patient’s cardiopulmonary status.
This two-part series outlines general issues and specific approaches to optimize results and minimize risks for patients undergoing procedural sedation.—The Editor
Classification of Agents
Definitions of levels of procedural sedation are presented in Table 2. Many types of medications are available for achieving analgesia, amnesia, anxiety alleviation, and sedation. Inadequate sedation analgesia, which may be due either to physician misperception or severity of the pain produced by the procedure, can cause great discomfort to the patient. It is helpful to define some of these terms.
 |
Analgesia is an altered state of mind in which pain perception is relieved but without necessarily achieving sedation.
Anxiolytic drugs reduce apprehension without a reduction in awareness.
Hypnotic drugs promote sleep. Their intent is to diminish level of consciousness.
Sedatives, in appropriate doses and depending on host response, decrease the awareness of pain and of a patient’s surroundings. They decrease activity and calm patients while protective airway reflexes are maintained.
Conscious sedation, dissociative sedation, deep sedation, and unconscious sedation define progressive degrees of sedation; the last two states require airway management because of the potential loss of protective airway reflexes.
Some anxiolytic drugs may require the addition of analgesics to alleviate pain, although they can, in appropriate doses, cause sedation. Patients in whom sedation is accomplished electively in the ED must be chosen carefully.
The American Society of Anesthesiology’s physical status classification may be a useful tool for selecting patients suited for elective conscious sedation. The classification includes the following categories: healthy patients, patients with mild systemic disease, patients with severe systemic disease, patients with severe systemic disease with constant life threat, and surgical candidates whose survival depends upon an operation and who are moribund. (See Table 3.)
 |
As a rule, only patients from the first two categories should undergo procedural sedation electively in the ED. Patients in the other categories either should be taken to the operating room if they need elective procedures, or they should have their airways managed either with or without deep sedation in the ED before being sent to the operating room for non-elective procedures.
Although comprehensive in many aspects, this review excludes a number of issues. (See Table 4.)
 |
Agents for Procedural Sedation
From 1958 until the 1980s, one of the mainstays for pediatric analgesia and sedation was the combination of meperidine (Demerol), chlorpromazine (Phenergan) and promethazine (Thorazine), commonly called DPT or MPC, with papoosing. This "cocktail" has a slow duration of action and its intramuscular use makes it impossible to titrate accurately for effect. One report noted that 29% of children were judged insufficiently sedated with MPC. In that study, the total average ED time was 4.7 hours; time until the child was eating was 11 hours; and total time until the child appeared "normal" to the parents averaged 19 hours.4 With its long duration of action and limited efficacy, it no longer is recommended for sedation. The Agency for Health Care Policy and Research has discouraged its use.5 The DPT combination has been associated with respiratory depression, hypoxemia, prolonged sedation,6 hypotension, dystonic reactions, and reduction in seizure threshold.7 Procedural sedation now favors the use of shorter-acting, high-potency benzodiazepines and narcotics alone or in combination,8 as well as agents that depress level of consciousness. Further disadvantages of intramuscular injection include: lack of vascular access, if it becomes necessary for resuscitation or agent reversal; pain on injection; delayed action; and the possibility of erratic absorption.
Nitrous oxide offers safe sedation and analgesia.9 It has some amnestic, hypnotic, and analgesic properties, and can be self-delivered by the patient. Nitrous oxide has an onset of action of 3-5 minutes, with a duration of action of 3-5 minutes. It is used in dentistry and many other physician settings, such as orthopedics. During transport to hospitals, paramedics trained in the use of this gas can administer it for painful orthopedic injuries. The mechanism of action is not known, but the agent appears to blunt the reaction to pain. Nitrous oxide is colorless and diffuses across membranes extremely rapidly. It is self-administered at a fixed ratio of 1:1 with 02 (50% concentration), although lower percentages are used in some pre-hospital protocols and dental offices.10 The patient holds a facemask in place and, as sedation occurs, relaxation begins, leading to a release on the facemask’s grip. As the mask loosens or falls aside, the flow of nitrous oxide stops. It has been shown to be effective in decreasing anxiety for laceration repair in children.11 At higher altitudes (greater than 5280 ft), its effectiveness decreases unless 70% nitrous oxide concentrations are used, which may increase the risk of hypoxia. The drug is eliminated unchanged by the lungs.12 As the nitrous oxide is washed out of the system through the lungs, it displaces oxygen in the alveoli and can cause "diffusion hypoxia." Therefore, supplemental oxygen should be provided throughout the recovery phase.
Because nitrous oxide is much more soluble than nitrogen, it will exchange for this gas in body tissues. Its solubility coefficient is 32 times that of air, so nitrous oxide preferentially enters areas of the body such as the gut and middle ear, possibly leading to overdistension. Contraindications to its usage include bowel obstruction, pregnancy, pneumothorax, procedures using balloon-tipped catheters, patients with facial trauma, and patients with middle ear effusions.12
Some of the undesirable effects of nitrous oxide include vomiting13 and hypoxia, which usually is a problem only with concentrations in excess of 60%. It is prudent to avoid use of nitrous oxide in patients with significant coronary artery disease, as there is evidence of diminished cardiac output. Patients with significant pulmonary disease also should not be sedated with this agent. Patients with blocked nasal passages or eustachian tubes are not suitable for conscious sedation with nitrous oxide. Scavenging devices are necessary to avoid exposure of the surrounding health workers to gas, inasmuch as deleterious effects of long-term exposure to nitrous oxide has been reported. There is a synergistic effect with recent opioids and sedative-hypnotic agents.
Chloral Hydrate. Chloral hydrate is used in children only in selected situations to achieve sedation for procedures such as a CT scan. The typical oral or rectal dose is 25-50 mg/kg, though higher doses of 50-75 mg/kg have been recommended for pediatric sedation.14 It is a hypnotic sedative with a delayed onset of action varying from 30 minutes to 1 hour following administration, and, therefore, it may be better suited for procedures requiring extended duration that are elective in nature. Time to recovery averaged 42 minutes in one report.15 Sedation is unreliable, and its lack of reversibility makes it a second-line agent for sedation. The metabolite is the active ingredient, and safe use of chloral hydrate requires intact liver function. Arrhythmias, including a 10% incidence of ventricular ectopic beats,16 and paradoxical effects are common.
Ketamine. Ketamine, a sedative analgesic, induces a dissociation between the cortical and limbic systems, accounting for its dissociative properties. This single agent produces sedation, analgesia, and amnesia. It exhibits minimal toxicity and has only a slight effect on blood pressure, even in patients with depleted volume states. The fact that cardiorespiratory reflexes remain intact after its use make this agent desirable in the pediatric setting. At lower doses, ketamine produces solely analgesia and sedation. At threshold doses of approximately 1-1.5 mg/kg IV or 3-5 mg/kg IM, a characteristic trance-like cataleptic state appears.17,18 Muscle tone is preserved, even accentuated, with a glassy stare in a trance-like state. The patient’s pupils become dilated, there is some degree of bronchodilation, and the airway is preserved. No cases of aspiration were reported in 1022 cases in one report, although there was a 6.7% incidence of emesis.19 Increased salivation and bronchospasm, which sometimes are associated with this agent, can be minimized with the use of atropine as a pretreatment agent.
Ketamine best is used in children between the ages of 1 and 11 years. Ketamine doses of 4-5 mg/kg IM produced sedation in 93%, with doses of greater than 4.5 mg/kg adequate for sedation in 100% of children, in one review of 1022 patients. Time to discharge averaged 110 minutes.20 Emergence reactions are more common in adults than children and present as hallucinations, recovery agitation, extremely bizarre dystonic actions, and nightmares. Approximately 0-10% of children experience emergence reactions.21 Hallucinatory phenomena while emerging from the dissociative state have been reported in up to 50% of adults.12 A small dose of a benzodiazepine may be given, but does not necessarily attenuate these reactions in children.22 One significant advantage of ketamine is its multiple routes of administration, including IV and IM routes. The latter is effective within five minutes of administration. Return of coherence generally occurs within 15-20 minutes.23 Oral ketamine, in doses up to 10 mg/kg, also is effective for minor procedures. Nasal ketamine, 6 mg/kg for children 1-5 years of age, also seems to provide adequate sedation. Atropine 0.01-0.02 mg/kg or glycopyrrolate 0.005 mg/kg up to a total dose of 0.25 mg may be administered to decrease secretions. Ketamine is effective for sedation in asthmatic/chronic obstructive pulmonary disease (COPD) patients needing intubation, with or without paralyzing agents. The beneficial effects of ketamine on airway resistance permit its use in status asthmaticus even in the presence of acute myocardial infarction. It usually is administered in a IV drip form, 20 mg/h, after an initial bolus of 10 mg IV in adults. Ataxia, unsteady gait, or vomiting may occur for several hours. (See Table 5 for a list of contraindications to the use of ketamine.)
 |
Etomidate. This drug is a nonbarbiturate carboxylated imidazole-containing compound with a rapid onset of action of 2-3 minutes. It lacks cardiovascular side effects, making it an excellent sedative drug for intubation. It has been used to assist in the reduction of major joint dislocations in the ED.24 It exerts depressant effects primarily on the ascending reticular activating system. Although it does not increase intracranial pressure, it may activate seizure foci. There is a similar incidence of pain on injection, as compared to propofol.25 It is not used for prolonged sedation in the intensive care setting, as it can produce transient depression of adrenal steroidogenesis.26 It has been associated with myoclonic jerks and should be avoided if a pre-existing seizure disorder is present. Cardiovascular and respiratory depression are limited with this agent, so that it may be used in patients with tenuous hemodynamic status.27 Dosage is 0.1-0.3 mg/kg. It is not approved for children under the age of 12.
Propofol. This is a nonopioid, nonbarbiturate sedative hypnotic, not related to other agents available for sedation. Analgesia should be administered in combination if propofol is used for a painful procedure. This agent also has amnesic properties and anticonvulsant activity, and can cause hypnosis when administered intravenously. Patients recover quickly and completely from its effects.28 In comparison studies, it has been shown to be better tolerated and to have a shorter recovery time than midazolam when used for endoscopy.29,30 It can produce cardiovascular depression and lower intracranial pressure. It is a highly lipophilic compound, most of which is bound to plasma proteins. It is available as a thick, white liquid containing glycerol, soy oil, and egg lechitin in a concentration of 10 mg/mL.31 Because of its distribution and high lipid solubility, lean body weight should be used when calculating doses. Rapid clearance from plasma permits rapid recovery, a major advantage to its use in the ED. It should be noted that propofol has limited analgesic properties at sedative does (1.0-1.3 mg/kg for sedation and 2.0-2.5 mg/kg for deep sedation). Propofol also has some antiemetic properties. Its first reported use in adult ED patients showed no significant complications utilizing 2 mcg/kg of fentanyl pretreatment and 0.21 mg/kg/min of propofol. The mean time to sedation was 7 minutes, with a mean recovery time of 6 minutes.32 Emergence from anesthesia is rapid, even following prolonged infusions.33,34 Typical induction doses range from 1-3 mg/kg, with sedative infusion rates from 25-120 mcg/kg/minute titrated to effect.
In children at least 3 years of age, it is used for induction of anesthesia. It has been used successfully for procedural sedation in children age 2-18 in the ED, with a mean recovery time of 15 minutes, as compared to 76 minutes for children sedated with midazolam.35 Due to pain at the injection site, 2% preservative-free lidocaine may be given over 1-2 minutes, in a dose of 0.5 mg/kg IV push. No significant hypotension was associated with its use in that study, although propofol has been associated with a significant incidence of hypotension when used in the pediatric intensive care unit setting.36 The recommended dosage in children is 1 mg/kg bolus over 2 minutes, followed by 67-100 mcg/kg/min infusion. Pediatric patients generally require maintenance infusion rates of 25-50% greater than those effective in adult patients.37
There is a similar incidence of apnea associated with thiopental, methohexital, and etomidate. Although it may cause hypotension, propofol can be used in patients with coronary disease without adverse effects on myocardial perfusion.38 Preliminary work has showed promise for patient-controlled propofol sedation.39,40
Barbiturates. Barbiturates are excellent short-acting hypnotics. It is important to remember that they do not have analgesic properties and, therefore, should be used in combination with pain-reducing drugs or local anesthetics when necessary. Barbiturates increase neuronal response to GABA (g-amino-butyric acid) and decrease neuronal excitability. Ultra-short and short-acting barbiturates are preferred for sedation in the ED. With thiopental and methohexital, emergence is rapid because of high lipid solubility. The initial dose of thiopental is 2-5 mg/kg of lean body mass. Its onset is 30-60 seconds after administration. Methohexital is administered in a dosage of 0.75-3 mg/kg IV and has a rapid onset of one minute, with a duration of action of 10 minutes. Barbiturates can decrease cardiac output and, on occasion, cause hypotension and respiratory depression.41 When titrated slowly, methohexital has minimal adverse effects.42 In addition to their primary role as inducing agents for surgery, methohexital and pentobarbital also can be used for short duration procedures. Both of these short-acting barbiturates are available in rectal suppository form, which makes them useful in children.43 However, via the rectal route, the onset of full sedation (30-60 minutes) is unpredictable and recovery time (sometimes more than 2 hours) is also variable. As a result, this method is more useful in radiology for imaging procedures than in the ED.44 IM routes are unpredictable and produce only slightly more rapid onset of action than the rectal route. Rectal methohexital for imaging in pediatric patients 1 month to 14 years of age has been used with excellent results. History of seizure disorder may be a contraindication to the use of methohexital.41 IV use of these barbiturates affords more controlled rapid onset and recovery end points and may be more useful in the ED. Since methohexital provides no analgesia, a short-acting opioid may be given for painful procedures.
Benzodiazepines. These are the drugs of choice for treating anxiety, sedating patients, and producing a significant degree of amnesia. These agents also have anticonvulsant properties, and cause no change in intracranial pressure. They, for the most part, are metabolized by the liver45 and excreted by the kidneys. Although respiratory depression is the primary major side effect of these sedatives, when administered slowly and incrementally, significant adverse effects may be prevented. Midazolam, a short-acting benzodiazepine first approved for use in the United States in 1986, has become popular for achieving conscious sedation in a wide range of clinical settings. Lorazepam and diazepam are longer-acting benzodiazepines, with the former having a slower onset of action. For immediate effects, IV bolus administration of these drugs is the preferred route in the ED. Compared to diazepam, midazolam is less painful at site of injection, possesses superior amnestic properties46 and produces more rapid onset of sedation. Despite clear advantages of midazolam for procedural sedation, many ED physicians continue to use diazepam or lorazepam sedation because they have more experience with those agents.
Midazolam may be administered intravenously in doses of 0.02-0.03 mg/kg slowly over 2 minutes, and repeated as necessary. A total of more than 5 mg generally is not necessary for the majority of procedures.47 The suggested IM dose is 0.07-0.08 mg/kg. Time of onset after IV administration is approximately three minutes, and after IM injection, five minutes. The elimination half-life of midazolam is 1.5-3 hours, as compared to more than 20 hours for diazepam.48 Its pharmacologic duration of action is approximately 60-120 minutes.47
In children, intranasal, rectal, and oral routes for midazolam have been used. Intranasal dosing is 0.2-0.3 mg/kg administered slowly over two minutes. A tuberculin needleless syringe may be used, with half of the volume in each nostril.49 The dose may be repeated. The oral dose is 0.2-0.3 mg/kg, repeated as needed in 10 minutes. Dilution in juice may make the bitter taste more palatable.50 Rectal dosing is 0.3 mg/kg diluted in 5 cc normal saline. Oral, nasal, and rectal administration all have approximately a 15-minute onset.51 In addition to respiratory depression, hypoxia and emergency delirium have been reported with midazolam in children. For purposes of imaging, intranasal midazolam has been shown to be especially beneficial. It should be stressed that midazolam does not have analgesic properties and should be used in conjunction with an analgesic for painful procedures. Time from midazolam administration to ED discharge by either oral or nasal route was approximately 55 minutes in one report.50
Cytochrome P450 inhibitors such as omeprazole, macrolide antibiotics, cimetidine, and ranitidine can reduce the metabolism of midazolam.52 Hiccups, cough, nausea, and vomiting are the most commonly reported adverse effects.51 Paradoxical hostility, aggression, and rage have been reported. It is listed as pregnancy category D by the Food and Drug Administration.53 Most excretion of midazolam is via the kidneys; in elderly, chronically ill patients, and in those with renal impairment, the dose should be reduced by at least half.9
Opioids. Opioids are considered the drugs of choice for severe pain.54,55 They have analgesic as well as sedative properties. They do not reliably produce sedation or unconsciousness. Meperidine, morphine, fentanyl, alfentanyl, and sufentanyl are examples of this therapeutic class. IV, IM, and oral routes can be used, but the IV route is the most reliable for both early onset and monitoring the degree of sedation. Respiratory depression is the most important adverse effect of opioids. Effects on the gastrointestinal tract include decreased motility, constipation, and ileus. In the pediatric population, intranasal or oral transmucosal routes for sufentanyl and fentanyl citrate are available. In one study, intranasal sufentanyl and midazolam used in children ages 1-4 years was found to be as effective as the old standby—a combination of IM meperidine, chlorpromazine, and promethazine.
Morphine in doses of 0.1-0.2 mg/kg IV or IM has an onset of action of 20-60 minutes, with a duration of action of 3-5 hours.9 Meperidine is given in a usual dose of 1-2 mg/kg IV or IM. Its onset of action following injection is 15-30 minutes, and its duration of action is 3-4 hours. It is approximately one-eighth as potent as morphine. Due to their prolonged effects, these agents are not recommended for procedural sedation in the ED.
Fentanyl is the opioid most commonly used to provide analgesia during procedural sedation in the ED.2,56 It is very lipid soluble and 80-100 times more potent than morphine, leading to a much more rapid onset than morphine. Although its elimination half-life is three hours, its high degree of lipid solubility leads to a rapid redistribution into tissues from the central nervous system, so that its clinical effects in the acute setting are approximately 30 minutes.57 The respiratory depression seen with fentanyl is less than that with either morphine or meperidine.55 Histamine release is minimal compared with other narcotics, so that it should not induce significant hypotension. The recommended dose is 2-4 mcg/kg titrated in doses of 0.5-1.0 mcg/kg every 3-5 minutes.57 A rare complication is rigid chest syndrome, which may require paralysis and mechanical ventilation to treat. This complication has been reported when much higher doses of fentanyl than recommended for ED use have been given, and has not been reported in the emergency literature.12,56,58 Since metabolism of fentanyl in infants is prolonged, it has been recommended that one-third the usual dose be administered in this group.12
Oral transmucosal fentanyl citrate (commonly called the "lollipop," or OTFC) has been used with success in children between 2 and 8 years of age in the ED to achieve both analgesia and sedation for laceration repairs.59 It is administered as a fentanyl-impregnated, sweetened matrix in lozenge form on a holder, first utilized in children as premedication for surgery.60,61 If 10-15 mcg/kg doses are used, effects are noted within 12-30 minutes. In one report, only one child in 30 reported developed oxygen desaturation below 95%, requiring supplemental oxygen for five minutes.59 Pruritus was common, and vomiting occurred in 10%. Mean time to discharge from the ED was 93 minutes. Transdermal fentanyl is available as well, although it has found its greatest use in patients with chronic pain.
Other Short-Acting Opioids. Sufentanil and alfentanyl are synthetic structural analogs of fentanyl. Alfentanil is approximately one-third as potent as fentanyl. It has an onset of action of approximately 45-60 seconds,62 and a shorter duration of action of approximately 30-40 minutes. The total IV dose is 5-20 mcg/kg, titrated in 2.5-5 mcg/kg doses administered slowly over 5 minutes.9 As with fentanyl, there is a significant incidence of rigidity which can be decreased by slower administration. Its elimination half-life of 70-103 minutes is shorter than that of fentanyl.63 Hypotensive effects, even in cardiovascularly impaired patients, are minimal.64
Sufentanil is 5-10 times more potent that fentanyl.65 Low to moderate dosages are in the 0.7-4.6 mcg/kg range. It tends to not affect histamine levels even up to 15 mcg/kg.66 Its plasma elimination half-life of 140 minutes is shorter than that of fentanyl. Intranasal sufentanil has been used alone and as a premedicant prior to surgical procedures. Dosages of up to 4.5 mcg/kg have been associated with respiratory depression.67 The recommended dosage is 2 mcg/kg intranasally; when used with midazolam, the nasal dose should be lowered to 0.75 mcg/kg.68,69
Intranasal midazolam and sufentanyl was better tolerated than IM meperidine, promethazine, and chlorpromazine in children in one report, with average time to discharge of 54 minutes in the intranasal group vs 81 minutes in the IM group.68
Remifentanil, rapifentanil, and mirfentanil are piperidine derivative analogues of fentanyl with short half-lives,70 which may become useful in the ED due to their titratable effects and short duration of action of 8-15 minutes. Their use in the ED setting has not been reported.
Combining Sedative Agents. There may be advantages to combining different drugs to achieve the desired level of sedation. Some medications achieve sedation without producing analgesia, and therefore require a combination of drugs to achieve both effects. Moreover, a dose-related toxicity caused by a high dose of a single drug can sometimes be prevented with lower doses of two drugs. The major disadvantage of combining sedatives is additive toxicity and excessive sedation. Cost is another important factor in deciding whether to use sedative agents singly or in combination.
Reversal Agents. Reversing the effects of sedatives rarely should be indicated. Most agents used for reversal are competitive antagonists and can lull one into a false sense of security. Use of reversal agents should be restricted to obtaining information regarding the causative agent of sedation or for reversing life-threatening effects of narcotics or benzodiazepines. Patients should not be reversed for the sake of a more rapid discharge from the ED. Adequate time for natural reversal is mandatory for safe discharge.
Opioid Antagonists. Naloxone is a pure narcotic antagonist, a synthetic cogener of oxymorphone. It competitively blocks the sites used by narcotic agents and, therefore, may reverse narcotic effects. Doses of 0.4-10.0 mg have been used in adults without adverse effects. Recommended dosage by weight ranges from 5-10 mcg/kg titrated to effect. The patient who has satisfactorily responded to naloxone should be kept under continued surveillance.
Nalmefene is a pure opiate antagonist that is structurally similar to naloxone. It is available intravenously, in concentrations of 1 mg/mL, 2 cc vials, as well as 100 mcg/mL in 1 mL vials. It is long-acting; a 2 mg IV dose was able to prevent respiratory depression from fentanyl challenge in volunteers for eight hours after administration.71 It has been shown to be safe and effective in the reversal of opioid sedation in children.72
Naltrexone is a potent, long-acting synthetic opiate antagonist available in oral form in 50 mg tablets. It is well-absorbed orally, and has been shown to block the agonist effects of morphine and heroin for 24 hours after IV injection.70,73 As with benzodiazepine antagonism, it is unwise to discharge patients home after administration of opiate antagonists for respiratory depression.
Benzodiazepine Antagonists. Flumazenil is a pure benzodiazepine antagonist. Seizure activity can be induced by this drug, especially when reversing benzodiazepine administered for seizures. It has been reported to precipitate seizures in the setting of tricyclic antidepressant overdose, as well as in patients who suffer benzodiazepine dependence.74,75 Intracranial pressure could be raised in patients who have head injury and have been given this reversal agent. Doses ranging from 0.2 mg to 1.0 mg can be administered. Most patients will respond to this dose, although some may require up to 3 mg for reversal. Flumazenil reverses the effects of midazolam on sedation, psychomotor impairment, and memory loss within 5 minutes of its administration.76 Its efficacy in reversing benzodiazepine-induced respiratory depression is less clear. One report showed an improvement in minute ventilation and tidal volume at 3 minutes compared with placebo, but no difference at 30 minutes.77 Midazolam-induced respiratory depression is short-lived and tends to respond to stimulation and oxygen alone.78,79 The duration of action of flumazenil is generally 1-5 hours; since the half-life of benzodiazepines may be longer, particularly in the elderly and in patients with renal or hepatic insufficiency,45,80 it is inadvisable to discharge patients home because their mental status has improved after flumazenil administration.
Neuroleptic Agents. Haloperidol and droperidol are butyrophenones agents with sedative properties at higher doses. They primarily are used in critically ill patients to treat delirium and agitation.81 They have little effect on respiratory depression, nor do they potentiate respiratory depression caused by opioids. Hypotension is a side effect in rare instances.82
Haloperidol can be given IM, but the IV route is preferred in critical care settings. Although the butyrophenones have been used for agitation prior to minor surgical and diagnostic procedures,83 they should be reserved for acute psychotic episodes or the treatment of extreme agitation. Dystonia, extrapyramidal reactions, oculogyric crisis, and neuroleptic malignant syndrome are the principal adverse effects.84
Droperidol is a sedative antiemetic chemically similar to haloperidol. Since it does not possess significant analgesic effects, it is used primarily in combination with a narcotic for its antiemetic and anxiolytic properties.85 Typical doses range from 1.25-10.0 mg IV; antiemetic effects are seen at lower doses and anxiolysis at higher doses. When clinically indicated, droperidol can be used in combination with small doses of midazolam or lorazepam. (See Tables 6a-b for a summary of dosages, onset, and duration of agents discussed.)
Quality Improvement and Monitoring
Monitoring is a requirement for both conscious and deep sedation during the procedure and after until the level of consciousness is restored.86 Opportunities and equipment for monitoring patients with conscious sedation will vary among EDs. What is clear, however, is that vigilant, device-mediated monitoring of patients to whom sedating agents or narcotic analgesics have been administered is necessary. The use of pulse oximetry to detect hypoxemia, ECG for cardiac monitoring, and blood pressure monitoring are recommended. Pulse oximetry is noninvasive, gives continuous real-time estimates of arterial saturation in the range of 70-100%, and gives early warning of loss of pulse.87,88 One major shortfall is that pulse oximetry has failed to reflect significant carbon dioxide retention during procedural sedation, especially when supplemental oxygen is administered. Therefore, hypoventilation and hypercapnia may precede a decrease in oxygen saturation by many minutes.89 As well, oxygen saturation of less than 90% has been reported during sleep in 43% of randomly selected men90 and occur routinely in normal passengers on commercial aircraft.91
Capnography by nasal cannula for measuring CO2 concentration with the use of an end tidal CO2 monitor appears to be a useful tool in some cases. It has been demonstrated that obtained PaCO2 levels correlate well with partial pressure of expired CO2 (PETCO2) levels.92 In one report, the average nasal end-tidal CO2 in one report increased from 35.9-42.1 mmHg in heavily sedated ED patients; this clinical significance of this finding was uncertain. Apnea alarms (PETCO2 < 15 mmHg) and close clinical observation are mandatory.93 In general anesthesia settings, especially for intubated patients, this has already become standard of care.94 End tidal CO2 monitoring via calorimetric or solid state spectophotometric techniques, has become an adjunct for ED management of conscious sedation, intubated patients, and cardiopulmonary resuscitation.
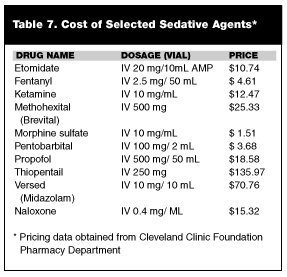
Administration of sedatives requires assessment and drug monitoring with impeccable protocols and quality improvement tools. Prices of agents vary. (See Table 7.) In hospital units where procedures are conducted (i.e., bronchoscopy, cardiac catheterization lab, endoscopy, and the ED) using conscious sedation and deep sedation, JCAHO requires leaders to develop and implement mechanisms designed to assure uniform performance of patient care processes throughout the organization.
 |
In this regard, many scales are available to measure the degree of sedation, including the Ramsay Sedation Scale, which ranks from 1 (anxious and agitated) to 6 (no response to loud noise). (See Table 8.) Sedation guidelines for monitoring infusion of drugs based on this sedation scale also are available. These are used primarily in the ICU and slowly are finding their way to the ED with appropriate modifications. Policy development and review in pediatric and adult population should address issues of criteria for the use of sedative agents, staff awareness of the actions and contradictions of each agent, familiarity with all aspects of airway management and monitoring parameters, and institution of continuous quality improvement tools for each patient that undergoes sedation. Although fasting history in the ED of necessity will vary due to the unscheduled nature of the practice, broad guidelines may be recommended. (See Table 9.)
 |
 |
These rules are not only applicable to ED physicians but to medical support personnel. Should help be required at any time during the management of the patient, it is prudent to consult an anesthesiologist to help avoid serious injury. In this context, JCAHO guidelines suggest approval of credentialing, education, and skill protocols by the department of anesthesia. A joint departmental cooperative effort to help formulate protocols, educate, and credential emergency medicine physicians in the use of conscious sedation and deep sedation in the ED seems reasonable. An ongoing monitoring and continuing medical education program for these important skills could be conducted by taking a written examination periodically or by placing requirements for a certain number of sedations to be performed over a given period. Many residency programs have pain control and sedation protocols established for their ED.
References
1. Holzman R, Cullen D, Eichhorn J, et al. Guidelines for sedation by nonanesthesiologists during diagnostic and therapeutic procedures. J Clin Anesth 1994;6:265-276.
2. Jagoda AS, Campbell M, Karas S, et al. Clinical policy for procedural sedation and analgesia in the emergency department. Ann Emerg Med 1998;31:663-677.
3. Ramsay MAE, Savage TM, Simpson BRJ, et al. Controlled sedation with alphaxalone-alphadolone. Br Med J 1974;2:656-659.
4. Terndrup TE, Dire DJ, Madden CM, et al. A prospective analysis of intramuscular meperidine, promethazine, and chlorpromazine in pediatric emergency department patients. Ann Emerg Med 1991;20: 31-35.
5. U.S. Department of Health and Human Services. Public Health Service; Agency for Heath Care Policy and Research; Acute Pain Management Guideline Panel: Clinical Practice Guideline—Acute Pain Management: Operative of Medical Procedures and Trauma. February 1992.
6. Green CJ. Options for sedating children. West J Med 1993;159: 596-597.
7. Terndrup TE, Dire DJ, Madden CM, et al. Comparison of intramuscular meperidine and promethazine with and without chlorpromazine: A randomized, prospective, double-blind trial. Ann Emerg Med 1993;22:206-211.
8. Baraff LJ. Conscious sedation of children. Ann Emerg Med 1994; 24:1170-1172.
9. Blackburn P, Vissers R. Pharmacology of emergency department pain management and conscious sedation. Emerg Med Clin North Am 2000;18:803-829.
10. Wilson S. A Survey of the American Academy of Pediatric Dentistry membership: Nitrous oxide and sedation. Pediatr Dent 1996; 18: 287-293.
11. Burton JH, Auble TE, Fuchs SM. Effectiveness of 50% nitrous oxide/50% oxygen during laceration repair in children. Acad Emerg Med 1998;5:112-117.
12. Sacchetti A, Schafermeyer R, Gerardi M, et al. Pediatric analgesia and sedation. Ann Emerg Med 1994;23:237-250.
13. Muir JJ, Warner M, Offord K. Role of nitrous oxide and other factors in postoperative nausea and vomiting. Anesthesiology 1987; 66:513.
14. Thompson JR, Schneider S, Ashwal S, et al. The choice of sedation for computed tomography in children: A prospective study. Radiology 1982;143:475-479.
15. Binder LS, Leake LA. Chloral hydrate for emergent pediatric procedural sedation: A new look at an old drug. Am J Emerg Med 1991;9: 530-534.
16. Saarnivaara L, Lindgren L, Klemola UM. Comparison of chloral hydrate and midazolam by mouth as premedicants in children undergoing otolaryngological surgery. Br J Anaesth 1992;69:65-69.
17. White PF, Way WL, Trevor AJ. Ketamine—Its pharmacology and therapeutic uses. Anesthesiology 1982;56:119-136.
18. Hurwitz GA, DeVore DT. Decreased doses of ketamine hydrochloride in children: A triple blind study. J Baltimore Coll Dent Surg 1975;30:28-34.
19. Green SM, Rothrock SG, Lynch EL, et al. Intramuscular ketamine for pediatric sedation in the emergency department: Safety profile in 1022 cases. Ann Emerg Med 1998;31:688-697.
20. Green SM, Hummel CB, Wittlake WA, et al. What is the optimal dose of intramuscular ketamine for pediatric sedation? Acad Emerg Med 1999;6:21-26.
21. Green SM, Johnson NE. Ketamine sedation for pediatric procedures 2. Review and implications. Ann Emerg Med 1990;19:1033-1046.
22. Wathen JE, Roback MG, McKenzie T, et al. Does midazolam alter the clinical effects of intravenous ketamine sedation in children? A double-blind randomized controlled emergency department trial. Ann Emerg Med 2000;36:579-588.
23. Dachs RJ, Innes GM. Intravenous ketamine sedation of pediatric patients in the emergency department. Ann Emerg Med 1997;29: 146-150.
24. Dursteler BB, Wightman JN. Etomidate-facilitated hip reduction in the emergency department. Am J Emerg Med 2000;18:260-263.
25. Boysen K, Sanchez R, Krintel JJ, et al. Induction and recovery characteristics of propofol, thiopental and etomidate. Acta Anaesthesiol Scand 1989;33:689-692.
26. Wagner RL, White PF, Kan PB, et al. Inhibition of adrenal steroidogenesis by the anesthetic etomidate. N Engl J Med 1984;310: 1415-1421.
27. Soifer BE. Procedural anesthesia at the bedside. Crit Care Clinics 2000;16:7-28
28. Ruiz K, Coldwell SA, Hitchin N, et al. Propofol sedation in general dental practice. Dental Update 2000;27:16-24.
29. Jung M, Hofmann C, Kiesslich R, et al. Improved sedation in diagnostic and therapeutic ERCP: Propofol is an alternative to midazolam. Endoscopy 2000;32:233-238.
30. Krugliak P, Ziff B, Rusabrov Y, et al. Propofol versus midazolam for conscious sedation guided by processed EEG during ERCP: A prospective randomized double-blind study. Endoscopy 2000;32: 677-682.
31. Barr J. Propofol: A new drug for sedation in the intensive care unit. Int Anesth Clin 1995;33:1131-1154.
32. Swanson MD, Seaberg DC, Mathias S. The use of propofol for sedation in the emergency department. Acad Emerg Med 1996;3: 234-238.
33. Morgan DJ, Campbell GA, Crankshaw DP. Pharmacokinetics of propofol when given by intravenous infusion. Br J Pharmacol 1990;30:144-148.
34. Victory RA, Pace N, White PF. Propofol: An update. Anesthesiol Clin North Am 1993;11:831-844.
35. Havel CJ, Strait RT, Hennes H. A clinical trial of propofol vs midazolam for procedural sedation in a pediatric emergency department. Acad Emerg Med 1999;6:989-997.
36. Hertzog JH, Campbell JK, Dalton HJ, et al. Propofol anesthesia for invasive procedures in ambulatory and hospitalized children: Experience in the pediatric intensive care unit. Pediatrics 1999;103:E30.
37. Smith I, White PF, Nathanson M, et al. Propofol: An update on its clinical use. Anesthesiology 1994;81:1005-1043.
38. Hall RI, Murphy JT, Moffitt EA, et al. A comparison of the myocardial metabolic and hemodynamic changes produced by propofol-sufentanil and enflurane-sufentanil anesthesia for patients having coronary artery bypass surgery. Can J Anesth 1991;38:996-1004.
39. Ng JM, Kong CF, Nyam D. Patient-controlled sedation with propofol for colonoscopy. Gastrointest Endosc 2001;54:14-17.
40. Kulling D, Fantin AC, Biro P, et al. Safer colonoscopy with patient-controlled analgesia and sedation with propofol and alfentanil. Gastrointest Endosc 2001;54:122-126.
41. Zink BJ, Darfler K, Salluzo RF, et al. The safety and efficacy of methohexital in the emergency department. Ann Emerg Med 1991;20:1293-1298.
42. Dionne RA, Yagiela JA, Moore PA, et al. Comparing efficacy and safety for intravenous sedation regimens in dental out-patients. J Am Dent Assoc 2001;132;740-751.
43. Pomerantz ES, Chudnofsky CR, Deegan TJ, et al. Rectal methohexital sedation for computed tomography imaging of stable pediatric emergency department patients. Pediatrics 2000;105:1110-1114.
44. Sedik H. Use of intravenous methohexital as a sedative in pediatric emergency departments. Arch Ped Adolesc Med 2001;155:665-668.
45. Dundee JW, Halliday NJ, Harper KW, et al. Midazolam: A review of its pharmacological properties and therapeutic use. Drugs 1984;28:519-543.
46. Coughlin MW, Panuska HJ. Direct comparison of midazolam and diazepam for conscious sedation in outpatient oral surgery. Anesth Prog 1989;36:150-168.
47. Stanski DR, Hudson RJ. Midazolam pharmacology and pharmacokinetics. Anesth Rev 1985;12:21-23.
48. Wright SW, Chudnofsky CR, Dronen SC, et al. Comparison of midazolam and diazepam for conscious sedation in the emergency department. Ann Emerg Med 1993;22:201-205.
49. Hartgraves PM, Primosch RE. An effect of oral and nasal midazolam for pediatric dental sedation. J Dent Child 1994;61:175-181.
50. Connors K, Terndrup T. Nasal versus oral midazolam for sedation of anxious children undergoing laceration repair. Ann Emerg Med 1994;24:1074-1079.
51. Nordt SP, Clark RF. Midazolam: A review of therapeutic uses and toxicity. J Emerg Med 1997;15:357-365.
52. Fee JPH, Collier PS, Howard PJ, et al. Cimetidine and ranitidine increase midazolam bioavailability. Clin Pharmacol Ther 1987;41: 80-84.
53. Briggs GG, Freeman RK, Yaffe JJ eds. Drugs in Pregnancy and Lactation 4th ed. Baltimore: Williams and Wilkins:1994.
54. Levy MH. Pharmacologic treatment of cancer pain. N Engl J Med 1996;335:1124.
55. Drugs for pain. Med Lett Drugs Ther 1998;40:1033.
56. Proudfoot J. Analgesia, anesthesia, and conscious sedation. Emerg Med Clin North Am 1995;13:357-379.
57. Chudnofsky CR, Wright SW, Dronen SC. The safety of fentanyl use in the emergency department. Ann Emerg Med 1989;18:635.
58. Streisand JB, Bailey PL, LeMaire L, et al. Fentanyl-induced rigidity and unconsciousness in human volunteers. Anesthesiology 1993; 78:4.
59. Schutzman SA, Burg J, Liebelt E, et al. Oral transmucosal fentanyl citrate for premedication of children undergoing laceration repair. Ann Emerg Med 1994;24:1059-1064.
60. Streisand JB, Stanley TH, Hague B, et al. Oral transmucosal fentanyl citrate premedication in children. Anesth Analg 1989;69: 28-34.
61. Feld LH, Chanpeau MW, Van Steenis CA, et al. Preanesthetic medication in children: A comparison of oral transmucosal fentanyl citrate versus placebo. Anesthesiology 1989;71:374-377.
62. Nauta J, deLange S, Koopman D, et al. Anesthetic induction with alfentanyl: A new short-acting narcotic analgesic. Anesth Analg 1982;61:267-272.
63. Camu F, Gepts E, Rucquoi M, et al. Pharmacokinetics of alfentanil in man. Anesth Analg 1982;61:657-661.
64. Murphy MR. Clinical pharmacology of alfentanyl and sufentanil. Anesthesiol Rev 1984;11:17.
65. Sebel PS, Bovill JG, Fiolet J, et al. Hormonal effects of sufentanil analgesia. Anesth Analg 1982;61:214-215.
66. Philbin DM, Rosow CE, Moss J, et al. Histamine release during induction with high dose sufentanil. Abstracts of the Sixth European Congress of Anesthesiologists. Paper 611:1982.
67. Henderson JM, Brodsky DR, Fisher DM, et al. Preinduction of anesthesia in pediatric patients with nasally administered sufentanil. Anesthesiology 1988;68:671-675.
68. Bates BA, Schutzman SA, Fleisher GR. A comparison of intranasal sufentanil and midazolam to intramuscular meperidine, promethazine, and chlorpromazine for conscious sedation in children. Ann Emerg Med 1994;24:646-651.
69. Zedie N, Amory DW, Wagner BK, et al. Comparison of intranasal midazolam and sufentanil premedication in pediatric outpatients. Clin Pharmacol Ther 1996;59:341-348.
70. Lineberger CK, Ginsberg B, Franiak R, et al. Narcotic agonists and antagonists. Anesth Clin N Am 1994;12:65-89.
71. Gal TJ, DiFazio CA. Prolonged blockade of opioid effect with oral nalmefene. Clin Pharmacol Ther 1986;40:537-542.
72. Chumpa A, Kaplan RL, Burns MM, et al. Nalmephene for elective reversal of procedural sedation in children. Am J Emerg Med 2001; 19:545-548.
73. Resnick RB, Volavka J, Freedman AM, et al. Studies of EN-1639A (naltrexone): A new narcotic antagonist. Am J Psychiat 1974;131: 646-650.
74. Mordel A, Winkler E, Almog S, et al. Seizures after flumazenil administration in a case of combined benzodiazepine and tricyclic antidepressant overdose. Crit Care Med 1992;20:1733-1734.
75. Lheureux P, Vranckx M, Leduc D, et al. Flumazenil in mixed benzodiazepine/tricyclic overdose: A placebo controlled study in the dog. Am J Emerg Med 1992;10:184-188.
76. Glass PS, Jhaveri RM, Ginsberg B, et al. Evaluation of flumazenil for reversing the effects of midazolam-induced conscious sedation or general anesthesia. South Med J 1993;86:1238-1247.
77. Gross JB, Weller RS, Conard P. Flumazenil antagonism of midazolam-induced ventilatory depression. Anesthesiology 1991:75: 179-185.
78. Sievers TD, Yee JD, Foley ME, et al. Midazolam for conscious sedation during pediatric oncology procedures: Safety and recovery parameters. Pediatrics 1991;88:1172-1179.
79. Bailey PL, Pace NL, Ashburn MA, et al. Frequent hypoxemia and apnea after sedation with midazolam and fentanyl. Anesthesiology 1990;73:826-830.
80. Baurer TM, Ritz R, Haberthur C, et al. Prolonged sedation due to accumulation of conjugated metabolites of midazolam. Lancet 1995;346:145-147.
81. Durbin CG Jr. Sedation of the agitated, critically ill patient without an artificial airway. Crit Care Clin 1995;11:913-936.
82. Berger I, Waldhorn R. Analgesia and paralysis in the intensive care unit. Am Fam Phys 1995;51:166-172.
83. Knight ME, Roberts RJ. Phenothiazine and butyrophenone intoxication in children. Pediatr Clin North Am 1986;33:299-309.
84. Riker RR, Fraser GL, Cox PM. Continuous infusion of haloperidol controls agitation in critically ill patients. Crit Care Med 1994;22: 433-440.
85. Caldwell JE. New skeletal muscle relaxants. Int Clin 1995;33: 39-60.
86. Lazzaroni M, Porro GB. Preparation, premedication, and surveillance. Endoscopy 2001;33:103-108.
87. Council on Scientific Affairs, American Medical Association. The use of pulse oximetry during conscious sedation. JAMA 1993;270: 1463-1468.
88. Standards for Basic Intra-Operative Monitoring. Park Ridge, IL:American Society of Anesthesiologists;1991.
89. Freeman ML, Hennessy JT, Cass OW. Carbon dioxide retention and oxygen desaturation during conscious sedation for ERCP, colon-oscopy, and upper GI endoscopy. Gastrointest Endosc 1991;37:233.
90. Block AJ, Boysen PG, Wynne JW, et al. Sleep apnea, hypopnea and oxygen desaturation in normal subjects. N Engl J Med 1979;300: 513-517.
91. AMA Commission on Emergency Medical Services. Medical aspects of transportation aboard commercial aircraft. JAMA 1982; 247:1007-1011.
92. McNulty SE, Roy J, Torjman M, et al. Relationship between arterial carbon dioxide and end-tidal carbon dioxide when a nasal sampling port is used. J Clin Monit 1990;6:93-98.
93. Wright S. Procedural sedation and analgesia in the emergency department. The value of capnography and pulse oximetry. Ann Emerg Med 1992;21:551-555.
94. Eichhorn JH, Cooper JB, Cullen DJ, et al. Standards for patient monitoring during analgesia at Harvard Medical School. JAMA 1986;256:1017-1020.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
