VRSA Has Arrived
Abstracts & Commentary
Synopsis: The dialysis catheter used to manage a 40-year-old Michigan resident became infected with strains of Staphylococcus aureus resistant to 128 mg/L vancomycin and > 16 µg/mg/L oxacillin.
Sources: Quirk M. First VRSA isolate identified in USA. Lancet Infect Dis. 2002;2:510; Staphylococcus aureus resistant to vancomycin—United States, 2002. MMWR Morb Mortal Wkly Rep. 2002;51:565-567; Bartley J. First case of VRSA identified in Michigan. Infect Control Hosp Epidemiol. 2002;23:480.
A 40-year-old resident of Michigan, Ill, with diabetes and peripheral vascular disease had undergone dialysis for chronic renal failure. He had been treated for chronic foot ulcerations with repeated courses of antimicrobial therapy, some of which included vancomycin for over a year. In April of this year he developed bacteremia due to methicillin-resistant Staphylococcus aureus associated with amputation of a gangrenous toe. The arteriovenous hemodialysis graft was suspected to be the source and was duly removed and treatment was started with vancomycin and rifampin. In June, a catheter exit-site infection was apparent, the temporary dialysis catheter was removed and cultures of each yielded S aureus. The MIC of vancomycin, teicoplaninin, and oxacillin were > 128 mg/L, 32 mg/L, and > 16 mg/L, respectively. The strains contained both the mecA gene for methicillin/oxacillin-resistance and the vanA gene for vancomycin resistance. The infection responded to outpatient treatment comprising aggressive wound care and systemic antimicrobial therapy with trimethoprim/sulfamethoxazole to which the isolate was susceptible in vitro. The VRSA was also susceptible to chloramphenicol linezolid, minocycline, quinupristin/dalfopristin, and tetracycline.
The S aureus was also recovered from a chronic foot ulcer together with vancomycin-resistant Enterococcus faecalis (VRE), and Klebsiella oxytoca but not from the healed catheter exit site or the anterior nares.
So far, the VRSA has not been isolated from any of the health care workers, dialysis centre patients, familial contacts, or community contacts.
Comment by J. Peter Donnelly, PhD
It might have taken 8 years since the first vancomycin intermediately susceptible strain of S aureus was reported in Japan for the true VRSA to arrive—but it is here, and, likely here to stay. True, there was no evidence of spread and the strain seems to have been effectively eradicated from the patient, but S aureus is the emperor of nosocomial infections and knows only too well how to acquire new clothes. In fact, knowing the enemy is actually our best defense. The necessary ingredients for breeding multi-resistant S aureus have been known for a long time and were also present in this case (see Figure).
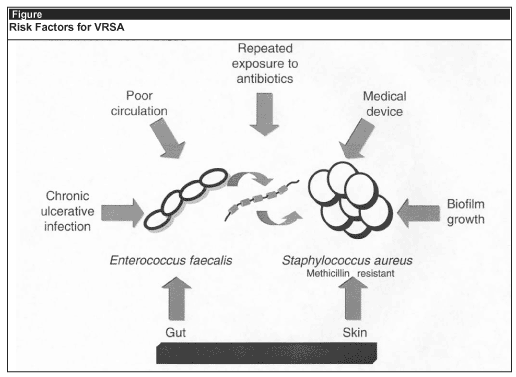 |
S aureus needs to have originated from somewhere and although technically the affected individual was an outpatient, the hospital is the most likely culprit since the patient was in and out for at least a year, had had a gangrenous toe amputated, suffered bacteriemia due to the methicillin-resistant S aureus emanating from an infected hemodialysis catheter and another clinically defined infection of a temporary dialysis catheter. Although the VRSA was not detected in the foot ulcer and the Enterococcus was not found around the catheter exit site, the 2 must have come into close contact with each other for the necessary exchange of DNA to take place. Chronic infection of dead or dying tissue such as the foot ulcer would seem the ideal place. Such lesions are easy prey to other opportunists migrating from their more usual residence and with the help of fingers and fomites, fecal bacteria like Enterococcus faecalis, and Klebsiella oxytoca could easily have spread from the gut and the MRSA from the catheter exit site. It was unlucky that the particular strain of E faecalis bore potentially transmissible genetic material that conferred resistance to vancomycin. Lastly, both enterococci and staphylococci dwell in a biofilm in their natural habitats and cohabitation would provide the intimate contact necessary to allow for the successful exchange of genes. In essence, the Petri dish was recreated. Having known it could happen in-vitro we now know it can also take place in-vivo but can only speculate on where and when it will occur next. My guess is the same places where its cousin MRSA resides and it is likely to happen sometime soon. Seems like a good time to check that the appropriate infection control precautions really are being taken.
Dr. Donnelly, Clinical Microbiologist, University Hospital, Nijmegen, The Netherlands, is Associate Editor of Infectious Disease Alert.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
