The Current Status of Pediatric Meningitis
Author: Catherine A. Marco, MD, FACEP, Attending Physician, St. Vincent Mercy Medical Center, Toledo, Ohio; Associate Professor, Medical College of Ohio, Toledo.
Peer Reviewer: Ronald M. Perkin, MD, MA, Professor and Chairman, Department of Pediatrics, The Brody School of Medicine, East Carolina University, Greenville, NC.
Pediatric meningitis is a diagnosis feared by parents and practitioners. Although vaccination practices have decreased the incidence of meningitis, it remains a significant cause of pediatric morbidity and mortality. The decreasing occurrence of meningitis has resulted in less familiarity with the disease process and a need for heightened awareness, especially in unvaccinated and immunocompromised children. Maintaining a high index of clinical suspicion, completing a focused historical and physical assessment, rapidly completing diagnostic modalities, and timely institution of definitive therapy can reduce the mortality rate and rate of significant disability. Meningitis continues to be a leading cause of malpractice litigation, and emergency department (ED) physicians need to review carefully the possible pitfalls presented in this article and utilize them as a method to reduce risk in their practice.—The Editor
Definition of the Problem
Meningitis results in significant morbidity and mortality among children each year. It has been estimated that bacterial meningitis strikes 1.3-10 cases/100,000 population/year.1,2 There were an estimated 2800 pediatric cases of bacterial meningitis in the United States in 1995,3 and there are an estimated 75,000 cases of enteroviral meningitis annually in the United States.4 Bacterial meningitis carries a 5-40% mortality rate5-8 and a 25-58% rate of significant disability.9-11
Epidemiology
The incidence of bacterial meningitis has changed dramatically since the widespread use of Haemophilus influenzae type B (HiB) vaccine. During a period of approximately 10 years, the incidence of bacterial meningitis in children younger than 5 years decreased by 87%, primarily due to a 94% reduction in H. influenzae (type B) infections.12 During the same period, a slight increase in Streptococcus pneumoniae meningitis was seen (4%), and decreases were seen in meningitis caused by Neisseria meningitidis (33%), group B streptococci, (25%), and Listeria monocytogenes (5%). In the past, most bacterial meningitis occured in children younger than age 5,13 but the median age of meningitis is now 25 years.14 Invasive meningococcal disease primarily affects children younger than age 4 (approximately 8 cases/100,000/year), although clusters occur in adolescents and young adults, primarily caused by serotype C.15,16
Among patients with meningitis following blunt head trauma, S. pneumoniae is the most likely etiologic agent.17
Patients predisposed to bacterial meningitis include: males; American Indians; African Americans; premature infants; patients with sickle cell anemia, anatomic asplenia, HIV infection or other immunodeficiencies; those with nephrotic syndrome, renal failure, or neoplasms (especially hematologic); malnourished patients; indigent urban dwellers; and immunocompromised patients. Other predisposing factors include neurosurgical procedures, recent lumbar puncture, or skull fracture.18 Potential portals of bacterial entry include sinuses, mastoids, and, most commonly, hematogenous seeding.
The incidence of viral meningitis is unknown, due to difficulties in accurate diagnosis and reporting. Many cases are benign, and likely underdiagnosed.19
Enterovirus causes a significant number of meningitis cases in patients younger than 1 year. Eighty-five percent of cases occur in infants younger than 4 months.20 In contrast, most herpes simplex virus (HSV) meningitis cases are diagnosed in adolescents and adults.21
Certain agents produce infection during identifiable times of year. Enterovirus and arbovirus infections occur most commonly in the summer and early fall, probably due to association with the mosquito season. One study demonstrated that 70% of positive enterovirus cultures from 1975 to 1994 occurred between the months of July and December.22 The typical incubation period is 4-6 days. Cases caused by N. meningitidis and S. pneumoniae often peak in the winter months, possibly occurring in association with respiratory tract infections.23 Listeria meningitis typically occurs in the spring and fall.24 Mumps virus infections often occur during late winter and early spring. Lymphycytic choriomeningitis infections typically occur during winter months.
Etiologies
Table 1 lists some causative agents for pediatric meningitis and some epidemiologic features. Meningitis may be caused by bacteria, viruses, fungi, or, rarely, other organisms.
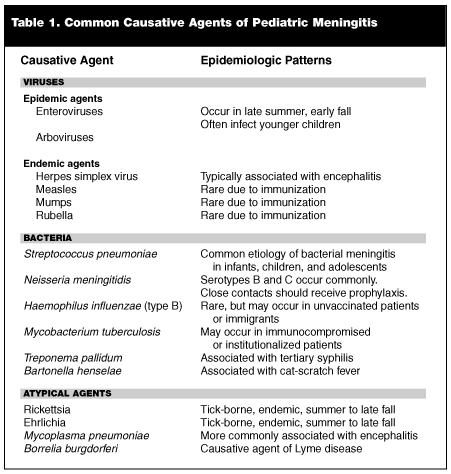
Bacteria. Organisms that cause bacterial meningitis vary by the age of the patient. (See Table 2.) Among the neonatal population, the predominant organisms include group B streptococcus (more than 50% of bacterial meningitis in this age group); Gram-negative enteric bacilli (30-40% of neonatal meningitis, and includes Escherichia coli, Klebsiella species, Enterobacteria species, Serratia species, and Citrobacter diversus); L. monocytogenes (approximately 5-20% of cases); Salmonella species, Enterococcus, Staphylococcus epidermidis, Staphylococcus aureus, S. pneumoniae, N. meningitidis, group D streptococci, Ureaplasma urealyticum, and H. influenza type B.25-28

Among older infants and children, 90% of bacterial meningitis is caused by S. pneumoniae and N. meningitidis. Although H. influenzae type B previously was a leading cause of bacterial meningitis, the incidence of this etiology has dropped dramatically since the institution of the HiB vaccine.29 However, it should be noted that the vaccine is less efficacious among Native Americans, who have a high incidence of this disease.30 Other etiologies may include Mycobacterium, Rickettsia, Ehrlichia, Salmonella species, Campylobacter species, L. monocytogenes, group G streptococcus, Francisella tularensis, and group B b-hemolytic streptococcus.31
Viruses. There are numerous viral agents that cause meningitis. In most clinical cases, the specific viral agent is not identified. Causative viruses may be classified in two groups, epidemic agents (which consist of enteroviruses and arboviruses), and endemic agents (including herpes viruses, rabies virus, etc.). Although measles, mumps, and rubella once caused significant numbers of cases of meningitis and encephalitis, vaccination has virtually eradicated these agents.
Enteroviruses currently cause 80-90% of viral meningitis.4 Enteroviruses are small, ribonucleic acid (RNA) viruses with approximately 70 serotypes, including echoviruses, coxsackie viruses, and polio viruses.32 Most viral meningitis is caused by enteroviruses 68-71, echoviruses, coxsackie viruses A and B, and polio viruses.33,34
Arboviruses are transmitted by an arthropod vector, typically a mosquito or tick. Arboviruses may cause diseases in a variety of vertebrates, including humans, horses, small mammals, and birds. The most common arboviruses that cause meningitis in humans are the St. Louis encephalitis virus and California encephalitis virus.
Endemic viruses that may cause meningoencephalitis include HSV, varicella-zoster virus, human herpes virus-6, measles, mumps, human parvovirus B-19, rubella, rubeola, influenza viruses, lymphocytic choriomeningitis virus, and HIV. HSV type 1 typically causes encephalitis, although the meninges may be involved to variable degrees. HSV type 2 may cause encephalitis, particularly in neonates who acquire the disease at the time of delivery. Varicella typically causes central nervous system (CNS) infection which manifests as cerebellar ataxia. It also may reactivate later in life and cause herpes zoster meningoencephalitis. Mumps meningoencephalitis typically is mild, but may result in deafness.
Fungi. Although fungi such as Cryptococcus neoformans or Candida species may cause meningitis in immunocompromised patients, they rarely are seen in immunocompetent patients.
Other Organisms. Rarely, other atypical pathogens may cause meningitis or encephalitis, including such agents as Rickettsia, Ehrlichia, Borrelia burgdorferi, Mycoplasma pneumoniae, and Bartonella henselae.
Pathophysiology. The precise pathophysiology of meningitis varies, dependent upon the etiologic agent and certain host characteristics.
Bacteria often spread by hematogenous routes in patients with bacteremia. Bacteria also may spread hematogenously from a distant focal infection, or by direct extension from an adjacent infection. Twenty-five percent of infants with bacteremia will develop meningitis.35 Bacteria also may enter the cerebrospinal fluid (CSF) following head trauma. In the neonatal population, organisms colonize the skin, nasopharynx, oropharynx, and umbilical cord at the time of birth, and may later spread to the CNS. Bacteria must cross the blood brain barrier, anatomically located at the cerebral microascular endothelium of the arachnoid membrane and the choroid plexus epithelium. Bacteria multiply easily in the CNS, in part due to poor immunologic defenses in normal cerebrospinal fluid.36,28 Bacteria replicate and release chemotactic factors and other virulence factors. Following the release of these factors, inflammation of the subarachnoid space occurs. Inflammation has both positive and negative effects; while necessary to eliminate bacteria, inflammation also causes CNS damage and potentially long-term neurologic deficits. Numerous inflammatory mediators have been implicated as contributors to the pathophysiology of meningitis, including tumor necrosis factor, interleukins, macrophage inflammatory proteins, platelet activating factor, prostaglandins, and interferon. Inflammation may lead to decreased CSF outflow, and increased intracranial pressure (ICP) may result. Other potential etiologies of cerebral injury include increased free radial production and excitatory amino acids, which may enter the CNS through altered blood brain barrier permeability.
Viruses typically enter the body via mucous membranes, or by hematogenous spread following an insect bite. It is suggested that mucosal barriers are affected by viral upper respiratory infections.37 Once in the body, viral replication occurs and the bloodstream may seed various organs. During this extraneural phase, systemic symptoms may occur, such as fever, malaise, and myalgias. Later, the CNS is seeded, causing meningitis. Other viruses such as HSV-1 may enter the CNS by direct spread along neuronal axons. Neurologic damage in viral meningitis, which may include cerebral edema, increased ICP, decreased cerebral blood flow, vascular thrombosis, demyelination, and perivascular destruction, is caused by either actively multiplying viruses or by the host reaction to viral antigens.
Clinical Features
Clinical features of meningitis vary according to several factors, including etiologic agent, host factors, duration of illness, and many others.38 Importantly, no single complaint or physical finding confirms or negates the diagnosis of meningitis. Additionally, partially treated meningitis may present with less impressive findings of fever and altered mental status, and may be more difficult to diagnose.39
Most patients with bacterial meningitis present with a sub-acute course, with progressive symptoms occurring over 2-5 days. Presenting symptoms may include fever, malaise, irritability, altered sleep patterns, anorexia, nausea, vomiting, or diarrhea. Interestingly, these nonspecific symptoms often are seen in children with other, more benign conditions. Table 3 illustrates the variety of clinical signs and symptoms that may be present.

Clinical features also vary according to the age of the patient. Among neonates, symptoms may include irritability, lethargy, hypotonia, feeding difficulty, tachypnea, diarrhea, vomiting, abdominal distension, hypo- or hyperthermia, seizures, altered tone, nuchal rigidity, bulging fontanelle, cyanosis, petechiae, purpura, or delayed capillary refill time. Approximately 50% of neonates with meningitis will be febrile, with half presenting with normal temperatures or with hypothermia. Specific findings, such as nuchal rigidity or bulging fontanelle, are found only sporadically and should not be relied upon as consistent clinical features.40 Infants younger than 4 weeks (some authors suggest 8 or 12 weeks) with fever should be evaluated for suspected sepsis and/or meningitis with blood cultures and lumbar puncture.41-44
Among older infants and children, clinical features may include fever, chills, malaise, myalgias, headache, photophobia, altered mental status, neck pain, backache, tachycardia, altered mental status, focal neurologic signs, hearing deficit, seizure, petechiae, purpura, delayed capillary refill, anorexia, nausea, and vomiting. One study demonstrated that the most common symptoms included neck stiffness (76%), focal neurologic findings (69%), fever (62%), headache (62%), and vomiting (54%).45
Clinical features of viral meningitis may be acute, but often are less fulminant.46 Fever is exceedingly common, and is found in nearly 100% of patients.33,47 Often, the illness begins with nonspecific symptoms such as fever, malaise, myalgias, and exanthem. CNS symptomatology may follow several days later, with symptoms such as headache, hyperesthesias, nausea, vomiting, photophobia, and neck pain. Infants typically present with nonspecific symptoms of fever and irritability, while older children often present with fever and headache. Symptoms of dehydration, including sunken fontanelle, tachycardia, or tachypnea, may be present. Headache and nuchal rigidity commonly are seen, but their absence does not rule out viral meningitis. Focal symptoms such as focal weakness, or loss of bowel and bladder control, may occur. Cutaneous vesicles may be suggestive of HSV meningoencephalitis, but typically are seen in only 30-50% of infants with neonatal HSV, and rarely are seen in older children.27 A maculopapular rash is present in about 25% of patients with enteroviral infection,48 and also may be seen in association with echovirus and coxsackie infections.
The physical examination should be directed toward the identification of signs suggestive of meningitis. Vital signs and airway should be evaluated initially. Disease processes that predispose an individual to meningitis, (i.e., evidence of head trauma) should be sought. A complete neurologic examination should be performed to establish a baseline for comparison examinations during therapy. The physical examination should be performed rapidly and should not delay prompt initiation of antibiotic therapy.
A child with an altered mental status should significantly raise the suspicion of meningitis. One study found that 73-100% of patients with bacterial meningitis were lethargic or comatose, and 36-60% appeared toxic or moribund.49
The fontanelle should be assessed in infants, as a bulging fontanelle may suggest elevated ICP, and a depressed fontanelle may suggest dehydration.
Signs of nuchal rigidity should be assessed, including Kernig’s and Brudzinski’s signs, although signs of meningeal irritation rarely are identifiable in infants younger than 6 months.49 Although nuchal rigidity is predictive of acute bacterial meningitis, its absence is not predictive of a benign process. Two clinical maneuvers commonly are utilized to demonstrate nuchal rigidity. Kernig’s sign refers to pain on leg extension, following flexion of the hip to 90°. Brudzinski’s sign refers to involuntary flexion of the hips and knees, following passive flexion of the neck while supine. Both signs are considerably unreliable. A positive Kernig’s sign is seen in 43% of patients with bacterial meningitis, and a positive Brudzinski’s sign in 66% of patients.50
Patients with a history of head trauma should be examined for evidence of fractures with penetration into the sinuses, dural tears, and CSF otorrhea or rhinorrhea. These factors predispose a patient to the development of a CSF infection, but absence does not rule out the possibility of posttraumatic meningitis.
Hearing loss occurs commonly in association with bacterial meningitis. Up to one-third of patients may exhibit some degree of hearing loss, either during the acute phase, or during the recovery phase. Acute hearing loss, particularly in the setting of head trauma, should alert the clinician to the possibility of meningitis.
Seizures may be seen in 20-30% of patients with bacterial meningitis.51 Typically, altered level of consciousness persists long after the typical postictal period.52 The rate of disease progression also varies by etiologic agent. For example, N. meningitidis infection may rapidly progress to septic shock and fulminant multisystem failure within hours. Patients who present acutely with a fulminant course typically have a worse prognosis.53
Diagnostic Studies
Lumbar puncture is the gold standard for diagnosing meningitis. Lumbar puncture should be performed on all patients for whom there is a strong clinical suspicion of meningitis. This may include patients with altered mental status, irritability, fever, shock, neurologic deficit, headache, nuchal rigidity, photophobia, or seizures; or unexplained fever in infants younger than 3 months.
If a lumbar puncture feasibly cannot be performed, due to hemodynamic instability, respiratory insufficiency, or risk of increased ICP, antibiotic therapy should be administered without delay.27,54 Lumbar puncture may be performed semi-electively after stabilization, or the patient may be treated with a complete course of antibiotics, based on the presumptive, but not proven, diagnosis of meningitis.
Although many textbooks recommend performing lumbar puncture in the lateral decubitus position, with spine flexed and knees flexed, this procedure may be more successful (particularly in difficult cases) when performed in the upright sitting position. Particularly for obese patients, or patients with scoliosis, the midline may be deceptively difficult to identify in the lateral decubitus position. Fluoroscopic guidance may be required in certain cases, including patients with spinal deformities.
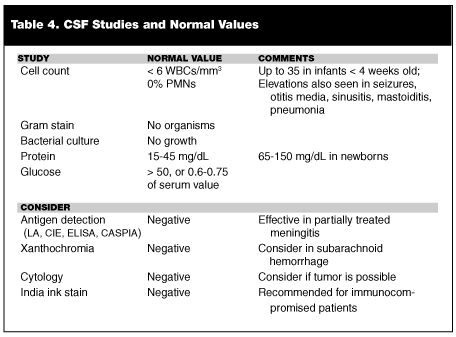
Studies that should be obtained are outlined in Table 4. Routine studies should include cell count, culture, Gram stain, protein and glucose. Types of culture may include bacterial, viral, fungal, and mycobacterial. Antigen detection may be useful, and may be done by a variety of techniques, including latex agglutination (LA), enzyme-linked immunoabsorbent assay (ELISA), countercurrent immunoelectrophoresis (CIE), and centrifugation-augmented solid-phase-immunoassy (CASPIA). Because antigens typically persist in the CSF for several days after the initiation of treatment, antigen detection may be particularly useful in diagnosing partially treated meningitis. Four tubes of CSF are routinely collected when meningitis is suspected. Some authors advocate certain tubes being sent for specific tests. For example, some authors recommend testing tubes one and four for cell count. Others believe this practice to be unnecessary, and advocate sending only tube four. Although some favor obtaining an opening pressure, we have found that this rarely influences clinical management.
Complications of lumbar puncture include: infection, pain, bleeding, and rarely, herniation. Risk of infection may be reduced by the fastidious use of sterile technique. Pain of the procedure greatly may be reduced by the appropriate use of local anesthesia, and in certain cases, intravenous (IV) sedation. Postdural puncture headache is a known complication, and its incidence can be reduced by the use of small-gauge needles, restricting the fluid removal to 3 cc, and only moving the needle with the stylet in place. Although herniation of the temporal lobes or cerebellar tonsils has been described, this rare complication usually can be prevented by avoiding lumbar puncture in patients with evidence (physical or radiographic) of elevated ICP. The potential risk of hypoxia during the procedure may be reduced by preoxygenation and provision of supplemental oxygen during the procedure, particularly for infants.55
Abnormal CSF findings consistent with bacterial meningitis include: pleocytosis (typically 1000-20,000/mm3 with predominance of polymorphonuclear neutrophils (PMNs) , decreased glucose (< 0.4 of serum glucose), elevated protein, and elevated opening pressure. One study demonstrated that the finding of more than five white blood cells (WBC)/mm3 has a sensitivity of 98.4% and a specificity of 75.2% for predicting bacterial meningitis.56 Another recent study demonstrated that the finding of CSF WBC greater than 30/mL had a likelihood ratio of 10.3 for bacterial meningitis, and a positive predictive value of 22.3%, and that a significant fraction of children with bacterial meningitis did not demonstrate CSF pleocytosis.57 The Gram stain may demonstrate organisms, dependent upon the number of organisms in the sample. Although Gram stains have an overall sensitivity of 70-85%, 97% of Gram stains will be positive when the specimen contains 106 colony forming units/mL. The percentage of positive stains decreases with lower bacterial counts. Latex agglutination usually is positive. If bacterial meningitis is partially treated, CSF leukocytosis may be less pronounced, protein is likely to be elevated, and Gram stain, latex agglutination, and cultures may be positive, but may be sterile due to treatment.
Patients with tuberculous meningitis often have a CSF leukocytes up to 500/mm3, a dramatically elevated protein, and a glucose level less than 50 mg/dL. Acid fast smears often are falsely negative. Cultures may be positive if a large volume of CSF is cultured. The organism may be detected by polymerase chain reaction (PCR).
In viral meningitis, pleocytosis also may occur, but cell counts typically are not as high as is seen in bacterial meningitis. The CSF may contain between a few and several thousand cells/mm3. A mononuclear predominance often is noted, although predominance of PMNs may be seen early in the course. Opening pressure usually is normal or slightly elevated. The CSF protein may be slightly elevated, but often CSF glucose and protein are within normal ranges. Glucose may be decreased in 15-20% of mumps cases.30 The Gram stain is negative, but always should be performed to screen for bacterial meningitis. Isolation of the virus is more accurate when cultured early during the disease course, and false negative cultures may occur in up to 30% of viral meningitis.30 Enteroviruses, HSV, and CMV may be detected by PCR of the CSF. Since PCR results virtually are 100% specific,58 and often can be obtained within several hours, confirmation of viral meningitis may obviate the need for unnecessary hospitalization and antibiotic therapy.
In fungal meningitis, leukocytosis of up to 500/mm3 may occur. Protein may be between 25-500 mg/dL. Glucose often is below 50 mg/dL. Budding yeast forms may be seen on CSF examination and organisms may be recovered by fungal culture. If cryptococcal meningitis is suspected, cryptococcal antigen should be tested in the CSF and serum.
Repeat lumbar puncture occasionally may be required to establish a definitive diagnosis in a patient with an initially normal lumbar puncture. Approximately 1% of patients with bacterial meningitis may have a normal CSF profile.59 CSF composition may change drastically within a short time during acute meningitis.60
Other tests should be considered when evaluating a patient with possible meningitis to rule out other etiologies of symptoms or to determine the causative agent. Such studies may include: blood cultures (which identify the CSF organism in 80-92% of cases),31,61 nasopharyngeal cultures, serum glucose, complete blood count, arterial blood gas, chest radiograph, Gram stain of dermatologic lesions, urinalysis, and culture.
Radiographic tests that may be considered include computed tomography (CT) of the brain and magnetic resonance imaging (MRI). Head CT need not routinely be performed, but should be obtained whenever mass lesions or elevated incranial pressure are suspected, to rule out such entities as brain tumor, abscess, cerebral hemorrhage, intracranial hemorrhage, subdural empyema, cavernous sinus thrombosis, or lateral sinus thrombosis. Head CT should be performed prior to lumbar puncture if mass lesions are suspected, but need not be performed uniformly in all patients. Diffuse swelling of brain parenchyma, ventricular dilatation, or obliteration of the basal cisterns and sylvian fissures may be seen in acute meningitis.62 Importantly, radiographic tests should never delay the prompt initiation of antibiotic therapy for suspected meningitis.
Electroencephalograms (EEGs) may be performed. Although their diagnostic utility for meningitis is limited, they may be useful to rule out seizure activity. Typical EEG findings during acute meningitis include diffuse slow wave activity. Focal changes may be seen less commonly.
Differential Diagnosis
Because signs and symptoms of meningitis often are nonspecific, numerous other etiologies may be considered. Clinical diagnoses that may be entertained are depicted in Table 5. The differential diagnosis is extensive, depending on the clinical presentation, and includes such categories of illness as: infectious etiologies, neurologic disorders, neoplasms, metabolic disorders, toxic syndromes, vascular abnormalities, neuromuscular disorders, and other miscellaneous disorders.

Although making a definitive differentiation of bacterial vs. viral meningitis may not be possible, patients with viral meningitis typically have less fulminant clinical course, and symptoms often are accompanied by gastrointestinal complaints. Neck stiffness typically is milder than is seen with bacterial meningitis.
Management
Management for suspected meningitis includes airway assessment and intervention, if indicated; administration of supplemental oxygen, if indicated; IV fluids and circulatory support, if indicated; and prompt initiation of antibiotic therapy. Management strategies are outlined in Table 6.
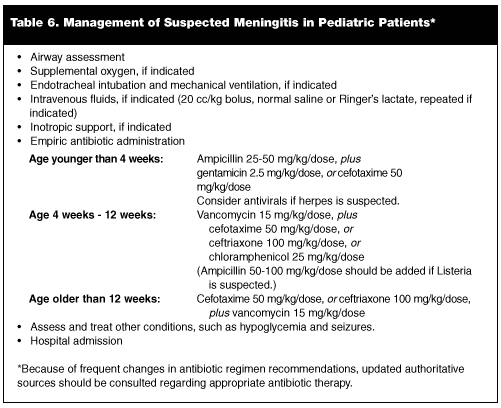
Some patients with meningitis may have shock and associated respiratory failure. Such patients should have aggressive airway intervention, which may include administration of supplemental oxygen, and/or endotracheal intubation and mechanical ventilation. Circulatory support may be indicated, with IV fluids and pressors, if necessary, to maintain adequate perfusion. Although correcting hypotension is a priority, excessive fluid administration should be avoided, as it may contribute to brain edema. However, fluid restriction, a previously advocated practice, should be avoided, as fluid restriction may contribute to poorer outcomes.63,64
Prompt initiation of antibiotics is crucial. Delayed antibiotic administration is associated with adverse outcomes.65 Although some practitioners delay antibiotics while diagnostic tests are performed, radiographic studies and even lumbar puncture results are not likely to be affected by the prompt administration of antibiotics.66 The choice of empiric antibiotics should be made with consideration of the age of the patient, likely organisms, and local antibiotic resistance trends. Table 6 lists some recommended antibiotic regimens that cover common pathogens. Of note, because of increasing rates of penicillin resistance among pneumococcal isolates (12-40%),67 vancomycin is considered the treatment of choice for penicillin-resistant S. pneumoniae.68
Administration of steroids is controversial. Although the administration of dexamethasone to patients with H. influenza type B meningitis has been associated with improved outcomes,69,70 this effect has not been demonstrated clearly for other types of meningitis.71,72 Additionally, adverse effects of steroids must be considered, such as reduced penetration of vancomycin,73,74 gastrointestinal bleeding, hyperglycemia, worsened neurologic outcome,75 and others. Although some authors still advocate the use of steroids for therapy for bacterial meningitis,76,77 others recommend that dexamethasone therapy be withheld, except for cases of suspected or proven H. influenza type B meningitis, when steroid therapy is appropriate.40,75 If given, dexamethasone should be administered in a dose of 0.15 mg/kg IV, 10-15 minutes prior to antibiotic administration.
ICP may be elevated in patients with CNS infections. If elevated ICP is suspected, an ICP monitor should be considered and measures undertaken to reduce ICP; measures may include raising the head of the bed;78 administration of IV mannitol (0.25-1.0 g/kg over 10 minutes); administration of diuretics and barbiturates; and maintenance of normothermia, normoglycemia, and adequate oxygenation.27,79,80
Treatment of viral meningitis primarily is supportive. Antibiotics usually are administered until CSF cultures are negative for a bacterial etiology or viral testing is positive. Dehydration should be managed with IV fluids. Antipyretics and analgesics may be indicated for symptomatic relief of fever and headache. Antiemetics may be indicated for treatment of nausea and vomiting, but care should be taken with their administration to differentiate medication effect from progressive alteration in mental status. Early reports suggest clinical efficacy of pleconaril for the treatment of enteroviral infections.81,82 If HSV meningoencephalitis is suspected, an antiviral such as acyclovir should be administered.
Treatment of Contacts. Contacts of patients with suspected bacterial meningitis should receive prophylactic antibiotics, particularly if meningococcal meningitis is suspected or confirmed. For known or suspected HIB, prophylaxis should be administered with rifampin (20 mg/kg once daily for 4 days) to household contacts, daycare staff, and enrollees. For known or suspected meningococcal disease, rifampin (10 mg/kg twice daily for 2 days) should be administered to household contacts, medical personnel, and day care contacts.54 Rifampin should be avoided in pregnant patients. Prophylaxis is not indicated for streptococcal infections.
Outcomes
Bacterial meningitis carries a 5-40% mortality rate4-6 and 25-58% rate of significant disability.7-9 The prognosis after meningitis is dependent on a variety of factors, including: age of the patient,83 severity of illness at presentation,84-85 extent of parenchymal involvement, hemodynamic status,65 presence of seizures,65,87 etiologic agent,28 abnormal EEG,88 and altered mental status.65 Delayed antibiotic administration also is associated with adverse outcomes. In the neonatal population, prolonged seizures, coma, need for inotropic support, and leukopenia are predictors of poor outcome.89 Long-term sequelae more commonly are reported among patients with gram-negative etiologies of neonatal bacterial meningitis than other etiologies.28
Neurologic complications, such as motor incoordination, seizure disorders, deafness, visual disturbances, and behavioral disturbances, may be seen.8-10,90 It is recommended that neurodevelopmental and audiologic evaluations routinely be performed in follow-up of patients with meningitis.
Additional Aspects
Complications. A wide variety of complications may be associated with meningitis. Respiratory insufficiency or aspiration may require intubation and mechanical ventilation. Seizures may be seen in 20-30% of patients with bacterial meningitis, and should be managed with benzodiazepines (for example, lorazepam 0.05-0.1 mg/kg IV) or other antiepileptic agents (for example, phenytoin 15 mg/kg IV, or phenobarbital 10-20 mg/kg IV). Hyperpyrexia should be treated with acetaminophen or nonsteroidal antiinflammatory agents. Fluid and electrolyte disturbances should be managed appropriately. If elevated ICP is suspected, an ICP monitor should be considered to guide ICP management. Mannitol administration may be considered in patients with significantly elevated ICP, but should not be instituted routinely.
Disseminated intravascular coagulation may be seen in patients with bacterial meningitis. The primary goal of therapy is to adequately and appropriately treat the underlying disease process. If necessary, active bleeding may be controlled by treating with platelet transfusions, vitamin K, and/or fresh frozen plasma.
Delayed complications may include intellectual impairment, deafness (partial or complete), motor deficits, psychiatric disorders, behavior problems, seizure disorders, hydrocephalus, visual disturbances, or cranial nerve damage.8-10,90
Complications of viral meningitis are relatively rare. CNS complications such as seizures, increased ICP, or coma may occur in 10% of patients younger than 2 years, but they rarely have long-term consequences.91 Diseases such as Guillain-Barré syndrome, acute transverse myelitis, hemiplegia, and cerebellar ataxia may occur.
Medicolegal Considerations
The failure to diagnose meningitis is one of the leading causes of malpractice litigation.92 Lumbar puncture or definitive treatment represents the standard of care for suspected bacterial meningitis. Even in cases in which bacterial meningitis is considered but deemed too unlikely to justify the risks of lumbar puncture, fastidious documentation of clinical findings and rationale may protect against future malpractice litigation. Specifically, instructions for appropriate and timely medical follow-up are good medical practice, and also may serve some medicolegal protection for the physician.
Pitfalls
Common errors that may result in medicolegal consequences are important to keep in mind when evaluating or managing a febrile child. These include:
- Failure to consider the diagnosis of meningitis in a febrile infant;
- Failure to consider the diagnosis of meningitis in a child with altered mental status, of any age;
- Failure to consider the diagnosis of meningitis in a child with persistent fever or positive blood cultures;
- Failure to consider the diagnosis of meningitis, following a lumbar puncture that revealed normal CSF studies;
- Failure to obtain blood cultures in a patient with suspected bacterial meningitis;
- Failure to document the history and physical examination thoroughly in patients with suspected bacterial meningitis; and
- Failure to provide detailed discharge instructions for patients with possible meningitis, including warning signs that would dictate return ED evaluation.
Disposition
Patients with suspected bacterial, fungal, or herpes meningitis should be admitted to an inpatient pediatric unit. Admission or transfer to a pediatric critical care unit should be considered for patients with hemodynamic instability, recurrent seizures, altered mental status, or evidence of shock.
Patients with suspected viral meningitis, who appear nontoxic, are able to ambulate, are tolerant of oral intake, with adequate home care, and with intact follow-up mechanisms, may be managed as outpatients. However, admission may be warranted until bacterial meningitis can be effectively ruled out.
Summary
Meningitis is one of the most devastating diagnoses in the pediatric emergency population. Both bacterial and viral meningitis are significant causes of pediatric morbidity and mortality. Maintaining a high index of clinical suspicion, completion of a rapid historical and physical assessment, rapid completion of diagnostic modalities, and timely institution of definitive therapy are the goals of treatment.
References
1. Segreti J, Harris AA. Acute bacterial meningitis. Infect Dis Clin N Amer 1996;10:797-809.
2. Dagbjartsson A, Ludvigsson P. Bacterial meningitis: Diagnosis and initial antibiotic therapy. Pediatr Clin North Am 1987;34:219-30.
3. Schuchat A, Robinson D, Wenger JD, et al. Bacterial meningitis in the United States in 1995. N Engl J Med 1997;337:970-976.
4. Sawyer MH. Enterovirus infections: Diagnosis and treatment. Current Opinion in Pediatrics 2001;13:65-69.
5. Arditi M, Mason EO, Bradley JS. Three-year multicenter surveillance of pneumococcal meningitis in children: Clinical characteristic and outcome related to penicillin susceptibility and dexamethasone use. Pediatrics 1999;201:1087.
6. Oliver LG, Harwood-Nuss AL. Bacterial meningitis in infants and children: A review. J Emerg Med 1993;11:555.
7. Durand ML, Calderwood SB, Weber DJ, et al. Acute bacterial meningitis in adults. A review of 493 episodes. N Engl J Med 1993; 328:21-28.
8. Aronin SI, Quagliarello VJ. New perspectives on pneumococcal meningitis. Hosp Pract 2001;36:43-51.
9. Grimwood K, Anderson VA, Bond L, et al. Adverse outcomes of bacterial meningitis in school-age survivors. Pediatrics 1995;95:646.
10. Pong A, Bradley JS. Bacterial meningitis and the newborn infant. Infect Dis Clin North Am 1999;13:711-733.
11. Klinger G, Chil CN, Beyene J, et al. Predicting the outome of neonatal bacterial meningitis. Pediatrics 2000;106:477-482.
12. Schuchat A, Robinson K, Wenger JD, et al. Bacterial meningitis in the U.S. in 1995. N Engl J Med 1997;337:970-976.
13. Bonadio WA, Mannenbach M, Krippendorf R. Bacterial meningitis in older children. Am J Dis Child 1990;144:463.
14. Kaplan SL, Mason EO, Barson WJ, et al. Three-year multicenter surveillance of systemic pneumococcal infections in children. Pediatrics 1998;102:538-545.
15. Centers for Disease Control and Prevention. Control and prevention of meningococcal disease and control and prevention of serogroup C meningococcal disease: Evaluation and management of suspected outbreaks. MMWR 1997;RR-5:1-21.
16. Zangwill KM, Schuchat A, Riedo FX, et al. School-based clusters of meningococcal disease in the U.S. JAMA 1997;277:389-395.
17. Bruyn GA, Kremer HP, de Marie S, et al. Clinical evaluation of pneumococcal meningitis in adults over a twelve-year period. Eur J Clin Microbiol Infect Dis 1989;8:695-700.
18. American Academy of Pediatrics Committee on Infectious Diseases. Peter G, ed. Red Book. 24th ed. Elk Grove, IL; AAP:1997:357-362, 410-419.
19. Rajnik M, Ottolini MG. Serious infections of the central nervous system: Encephalitis, meningitis, and brain abscess. Adol Med 2000;11:401-425.
20. Rotbart HA, McCracken GH Jr, Whitley RF, et al. Clinical significance of enteroviruses in serious summer febrile illnesses of children. Pediatr Infect Dis J 1999;18:869-874.
21. Koskiniemi M, Rautonen J, Lehtokoski-Lehtiniemi E, et al. Epidemiology of encephalitis in children: A 20-year survey. Ann Neurol 1991; 29:492-497.
22. Atkinson PJ, Sharland M, Maguire H. Predominant enteroviral serotypes causing meningitis. Arch Dis Child 1998;78:373-374.
23. Block C, Roitman M, Bogokowsky B, et al. Forty years of meningococcal disease in Israel: 1951-1990. Clin Infect Dis 1993;17:126-132.
24. Kessler SL, Dajani AS. Listeria meningitis in infants and children. Pediatr Infect Dis J 1990; 9:61-63.
25. Sadow KB, Durr R, Teach SJ. Bacterial infections in infants 60 days and younger: Epidemiology, resistance, and implications for treatment. Arch Pediatr Adolesc Med 1999;153:611.
26. DeLanois J. Acute bacterial meningitis in a newborn. J Antimicrob Chemother 1994;34:61.
27. Wilde JA: Meningitis and encephalitis. In: Harwood-Nuss A, ed. The Clinical Practice of Emergency Medicine. 3rd edition. Philadelphia: Lippincott Williams & Wilkins, 2001.
28. Polin RA, Harris MC. Neonatal bacterial meningitis. Semin Neonatol 2001;6:157-172.
29. Centers for Disease Control. Progress toward the elimination of Haemophilus influenza type b disease among infants and children. United States 1987-1995. MMWR 1996;45:901.
30. Behrman RE, Kliegman RM, Jenson HB. Nelson’s Textbook of Pediatrics. 16th edition. Philadelphia: WB Saunders Co; 2000:757.
31. Rubin DH, Conway EE, Caplen SM, et al. Neurologic disorders. In: Rosen P, Barkin R, Danzl D, et al, eds. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 5th edition. St. Louis, MO: Mosby, Inc; 2001: 2344-2350.
32. Rotbart HA and Hayden FG. Picornavirus infections: A primer for the practitioner. Arch Fam Med 2000;9:913-920.
33. Sawyer MH. Enterovirus infections: Diagnosis and treatment. Pediatr Infect Dis J. 1999;18:1033-1039.
34. Rotbart HA, McCracken GH Jr, Whitley RF, et al. Clinical significance of enteroviruses in serious summer febrile illnesses of children. Pediatr Infect Dis J 1999;18:869-874.
35. Klein JO. Bacterial sepsis and meningitis. In: Remington JS, Klein JO, eds. Infectious Diseases of the Fetus and Newborn Infants. Philadelphia: WB Saunders; 2000:943-998.
36. Lipton JD, Schafermeyer RW. Evolving concepts in pediatric bacterial meningitis. Part I: Pathyphysiology and diagnosis. Ann Emerg Med 1994;24:118.
37. Michaels RH, Myerowitz RL. Viral enhancement of nasal colonization with Haemophilus influenzae type b in the infant rat. Pediatr Res 1983;17:472-473.
38. Feigin RD, McCracken GH, Klein JO. Diagnosis and management of meningitis. Pediatr Infect Dis J 1992;11:785-814.
39. Rothrock SG, Green SM, Wren J, et al. Pediatric bacterial meningitis: Is prior antibiotic therapy associated with an altered clinical presentation? Ann Emerg Med 1992;21:146.
40. Rubin DH, Conway EE, Caplen SM, et al. Neurologic Disorders. Neurologic disorders. In: Rosen P, Barkin R, Danzl D, et al, eds. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 5th edition. St. Louis, MO: Mosby, Inc; 2001.
41. Kellogg JA, Ferrentino FL, Goodstein MH, et al. Frequency of low level bacteremia in infants from birth to 2 months of age. Pediatr Infect Dis J 1997; 16:381.
42. Jaskiewica JA, McCarthy CA, Richardson AC, et al. Febrile infants at low risk for serious bacterial infection: An appraisal of the Rochester criteria and implications for management. Pediatrics 1994;94:390.
43. Baker MD, Avner JR, Bell LM. Failure of infant observation scales in detecting serious illness in febrile, 4- to 8-week-old infants. Pediatrics 1990;85:1040.
44. Baker MD, Bell LM. Unpredictability of serious bacterial infection in febrile infants from birth to one month of age. Arch Pediatr Adolesc Med 1999; 153:508.
45. Kirkpatrick B, Reeves DS, MacGowan AP. A review of the clinical presentation, laboratory features, antimicrobial therapy and outcome of 77 episodes of pneumococcal meningitis occurring in children and adults. J Infect 1994;29: 171-182.
46. Cherry JD. Enteroviruses. In: Feigin RD, Cherry JD, eds. Textbook of Pediatric Infectious Diseases. Philadelphia: WB Saunders; 1992:1705-1753.
47. Rotbart HA. Viral meningitis and the aseptic meningitis syndrome. In: Scheld WM, Whitley RJ, Durack DT, eds. Infections of the Central Nervous System. Philadelphia: Lippincott-Raven; 1997:23-46.
48. Dagan R. Nonpolio enteroviruses and the febrile young infant: Epidemiologic, clinical and diagnostic aspects. Pediatr Infect Dis J 1996; 15:67-71.
49. Walsh-Kelly C, Nelson DB, Smith DS, et al. Clinical predictors of bacterial versus aseptic meningitis in childhood. Ann Emerg Med 1992;21:910-914.
50. Lipton JD, Schafermeyer RW. Central nervous system infections: The usual and the unusual. Emerg Med Clin North Am 1995;13:417.
51. Rosman NP, Peterson DB, Kaye EM, et al. Seizures in bacterial meningitis: Prevalence, patterns, pathogenesis, and prognosis. Pediatr Neurol 1985;1: 278-285.
52. Green SM, Rothrock SG, Clem KJ, et al. Can seizures be the sole manifestation of meningitis in febrile children? Pediatrics 1993;92:527-534.
53. Kilpi T, Anttila M, Kallio M, et al. Severity of childhood bacterial meningitis and duration of illness before diagnosis. Lancet 1991;338:406-409.
54. Mellis P. Bacteremia, sepsis and meningitis in children. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide. 5th edition. New York:McGraw-Hill, 2000.
55. Fiser DH, Gober GA, Smith CE, et al. Prevention of hypoxemia during lumbar puncture in infancy with preoxygenation. Pediatr Emerg Care 1993;9:81.
56. Rodewalk LE, Woodin KA, Szilagyi PG, et al. Relevance of common tests of cerebrospinal fluid in screening for bacterial meningitis. J Pediatr 1991;119: 363-369.
57. Freedman SB, Marrocco A, Pirie J, et al. Predictors of bacterial meningitis in the era after Haemophilus influenzae. Arch Pediatr Adolesc Med 2001;155: 1301-1306.
58. Rotbart HA, Ahmed A, Hickey S, et al. Diagnosis of enterovirus infection by polymerase chain reactino of multiple specimen types. Pediatr Infect Dis J 1997;16:409-411.
59. Sivakmaran M. Meningococcal meningitis revisited: Normocellular CSF. Clin Pediatr 1997;36:258-262.
60. Korones DN, Shapiro ED. Occult pneumococcal bacteremia: What happens to the child who appears well at reevaluation. Pediatr Infect Dis J 1994;13:382.
61. Bohr V, Rasmussen N, Hansen B, et al. Eight hundred seventy-five cases of bacterial meningitis: Diagnostic procedures and the impact of preadmission antibiotic therapy. J Infect 1983;7:193-202.
62. Ressler JA, Nelson M. Central nervous system infections in the pediatric population. Neuroimag Clin North Amer 2001;10:427-443.
63. Singhi SC, Singhi PD, Srinivas B, et al. Fluid restriction does not improve the outcome of acute meningitis. Pediatr Infect Dis J 1995;14:495-503.
64. Wubbel L, McCracken GH. Management of bacterial meningitis 1998. Pediatr Rev 1998;19:78.
65. Aronin SI, Peduzzi P, Quagliariello VJ, et al. Community acquired bacterial meningitis: Risk stratification for adverse clinical outcome and effect of antibiotic timing. Ann Intern Med 1998;129:862.
66. Talan DA, Hoffman JR, Yoshikawa, TT, et al. Role of empiric parenteral antibiotics prior to lumbar puncture in suspected bacterial meningitis: State of the art. Rev Infect Dis 1988;10:365.
67. Dowell SF, Schwartz B. Resistant pneumococci: Protecting patients through judicious use of antibiotics. Am Fam Phys 1997;55:1647-1658.
68. Quagliarello VJ, Scheld WM. Treatment of bacterial meningitis. N Engl J Med 1997;336:708-716.
69. Report of the committee on infectious diseases of the American Academy of Pediatrics: Dexamethasone therapy for bacterial meningitis in infants and children. Elk Grove Village, Ill: American Academy of Pediatrics, 1994.
70. McIntyre PB, Berkey CS, King SM, et al. Dexamethasone as adjunctive therapy in bacterial meningitis: A meta-analysis of randomized clinical trials since 1988. JAMA 1997;278:925.
71. Wald ER, Kaplan SL, Mason EO, et al. Dexamethasone therapy for children with bacterial meningitis. Pediatrics 1995;95:21.
72. Daoud AS, Batieha A, Al-Sheyyab M, et al. Lack of effectiveness of dexamethasone in neonatal bacterial meningitis. Euro J Pediatr 1999;158:230-233.
73. Chandy JC. Treatment failure with the use of a third-generation cephalosporin for penicillin-resistant pneumococcal meningitis: Case report and review. Clin Infect Dis 1994;18:188.
74. Friedland IR, Paaris M, Ehrett S, et al. Evaluation of antimicrobial regimens for treatment of experimental penicillin- and cephalosporin-resistant pneumococcal meningitis. Antimicrob Agents Chemother 1993;37:1630-1636.
75. American Academy of Pediatrics, Committee on Infectious Diseases: Therapy for children with invasive pneumococcal infections. Pediatrics 1997;99:289.
76. McIntyre PB, Berkey CS, King SM, et al. Dexamethasone as adjunctive therapy in bacterial meningitis. JAMA 1997;278:925-931.
77. Bonadio WA. Adjunctive dexamethasone therapy for pediatric bacterial meningitis. J Emerg Med 1996;14:165.
78. Yatsiv I. Central nervous system support techniques. In: Holbrook PR, ed. Textbook of Pediatric Critical Care. Philadelphia: WB Saunders, 1993.
79. Dacey RD. Monitoring and treating increased ICP. Pediatr Infect Dis J 1987;6:1161.
80. Allen CH, Ward JD. An evidence-based approach to management of increased intracranial pressure. Crit Care Clin 1998;14:485-495.
81. Sawyer MH, Saez-Llorenz X, Aviles CL, et al. Oral pleconaril reduces the duration and severity of enteroviral meningitis in children. In: Proceedings of the 1999 American Pediatric Society/Society for Pediatric Research Meetings, May 1-4, 1999; San Francisco, CA.
82. Shafran SD, Halota W, Gilbert D, et al. Pleconaril is effective for enteroviral meningitis in adolescents and adults: a randomized placebo-controlled multicenter trial. In: Proceedings of the 39th Interscience Conference on Anitimicrobial Agents and Chemotherapy; Abstract 1904, presented September 26-29, 1999; San Francisco, CA.
83. Oliver LG, Harwood Nuss AL. Bacterial meningitis in infants and children: A review. J Emerg Med 1993;11:555.
84. Segreti J, Harris AA. Acute bacterial meningitis. Infect Dis Clin N Amer 1996;10:797-809.
85. Kornelisse RF, Westerbeek CM, Spoor AB, et al. Pneumococcal meningitis in children: Prognostic indicators and outcome. Clin Infect Dis. 1995;21: 1390-1397.
86. Grimwood K, Anderson VA, Bond L, et al. Adverse outcomes of bacterial meningitis in school-age survivors. Pediatrics 1995;95:646.
87. Kaaresen PI, Flaegstad T. Prognostic factors in childhood bacterial meningitis. Acta Paediatr 1995;84:873-878.
88. Chequivi RS, Tharp BR, Dreimane D, et al. Prognostic value of EEG in neonatal meningitis: Retrospective study of 29 infants. Pediatr Neurology 1992;8:417-422.
89. Klinger G, Chin CN, Beyene J, et al. Predicting the outcome of neonatal bacterial meningitis. Pediatrics 2000;106:477-482.
90. Bedford H, de Louvois J, Halket S, et al. Meningitis in infancy in England and Wales: Follow up at age 5 years. Brit Med J 2001;323:1-5.
91. Rorabaugh ML, Berlin LE, Heldrich F, et al. Aseptic meningitis in infants younger than 2 years of age: Acute illness and neurologic complications. Pediatrics 1993;92:206-211.
92. Committee on Medical Liability. Liability issues in diagnosing meningitis. In: Robertson WO, Lockhart JD, eds. Medical Liability for Pediatricians. Elk Grove Village, IL: American Academy of Pediatrics; 1995:99-103.
Maintaining a high index of clinical suspicion, completing a focused historical and physical assessment, rapidly completing diagnostic modalities, and timely institution of definitive therapy can reduce the mortality rate and rate of significant disability from pediatric meningitis.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
