Blunt Pelvic Trauma
November 1, 2014
Reprints
Related Articles
-
Is Artificial Intelligence Coming for Your Job?
-
Can Pulmonary Artery Pressure Help in the Decision to Operate in Chronic Aortic Regurgitation?
-
Combined TAVR and LAAO Studied in a Randomized Controlled Trial
-
Another Hemodynamic Variant of Aortic Stenosis
-
Clinical Features of Tachyarrhythmia-Induced Cardiomyopathy
Executive Summary
- Schulman et al found that patients presenting with pelvic ring fractures are at increased risk of death regardless of other associated injuries. Additionally, they were able to demonstrate that patients with pelvic fractures presenting with age greater than 65 years, injury severity index greater than 18, respiratory rate greater than 30 bpm, SBP less than 90 mmHg, and GCS score less than 8 had an even further increased risk of mortality.
- The structures at greatest risk for injury due to their location adjacent to the pelvic bones and ligaments are: the sciatic, femoral, and obturator nerves, as well as the bladder, urethra, and extraperitoneal rectum.
- Higher-energy forces are required to cause APC3 injuries, and the posterior sacroiliac ligaments are completely disrupted, resulting in a severely unstable pelvic ring in which the highest clinical suspicion for hemorrhage should be considered.
- The correct way to test for pelvic stability is to grab the iliac crests and push inward in a gentle motion. Also, the symphysis pubis can be palpated to assess for widening.
- In the setting of pelvic fracture and hematuria, the CT cystogram with distended bladder and post-emptying views has replaced the retrograde cystourethrogram for the diagnosis of bladder injury.
Blunt pelvic trauma can be devastating, and should be suspected in patients with a significant mechanism of injury or signs of shock. Controversies continue, despite extensive research, regarding the optimal management, particularly for unstable pelvic fractures. A high degree of suspicion and prompt diagnosis and management are critical to optimize patient outcome.
— Ann M. Dietrich, MD, Editor
Introduction
Trauma to the pelvis is a great example of how an entity in trauma has undergone major evolutionary changes. Despite advances, the treatment of pelvic trauma continues to pose difficult challenges, and, thus, it continues to be a widely studied topic. Controversies remain regarding the correct sequence in the treatment algorithm of the unstable trauma patient when pelvic injury is considered to be the primary source of bleeding. It has been proven that a multidisciplinary approach in the treatment of these patients improves outcomes, and has become the standard of care.1 A team of specialists in emergency medicine, interventional radiology, trauma, and orthopedic surgery should be coordinated for rapid evaluation and treatment.2 This article will focus on the recognition and management of the trauma patient with pelvic injury.
Epidemiology
The overall mortality rate for patients with pelvic ring fractures is approximately 6%.2 This rate increases to 18-40% in patients with uncontrolled pelvic-related hemorrhage. Death that occurs within the first 24 hours of injury is most often a result of acute blood loss, while concomitant head trauma accounts for 31% of deaths.2-6 The incidence of abdominal injury in the presence of pelvic fracture can range from 16% to 55%.7-8 Schulman et al found that patients presenting with pelvic ring fractures are at increased risk of death regardless of other associated injuries.9 Additionally, the researchers were able to demonstrate that patients with pelvic fractures presenting with age greater than 65 years, injury severity index greater than 18, respiratory rate greater than 30 breaths per minute, systolic blood pressure (SBP) less than 90 mmHg, and Glasgow Coma Scale (GCS) score less than 8 had an even further increased risk of mortality. Demetriades et al studied the differences between pelvic fractures among the pediatric population and the adult population.10 They found that adults were twice as likely to suffer pelvic fractures compared to patients in the pediatric group. However, age group was not a significant risk factor for severe pelvic fractures. This difference was identified with all common mechanisms of injury (motor vehicle collisions, pedestrian struck, and falls greater than 15 feet). In falls from a height greater than 15 feet, the risk of pelvic fractures in adults was approximately seven times higher than that in children. The incidence of gastrointestinal and solid organ injury was high in both age groups. Aortic injuries were found exclusively in the adult group, and the researchers identified age older than 55 years as a significant risk factor for aortic rupture in patients with pelvic fracture. When pelvic fractures occur, the most common mechanism of injury is blunt force impact caused by motor vehicle collisions (MVC).11 Mortality has been demonstrated to decrease when algorithms for management of these injuries are implemented.12-13
Anatomy
The pelvis is a complex structure with a small and rigid cavity that contains important nervous, vascular, gastrointestinal, and genitourinary structures. It is composed of three bones that articulate to each other to form the pelvic ring; these are the two large innominate bones and the sacrum. The pubic bones are the most common pelvic bones at risk for fracture due to their thin structure.2 Posteriorly, the innominate bones articulate with the sacrum to form the sacroiliac (SI) joints, and anteriorly they attach to each other in the pubic symphysis. The SI joint, which receives its support from the anterior and posterior sacroiliac ligaments, is the strongest joint in the body. The sacrospinous and sacrotuberous ligaments provide the integrity for the pelvic floor; they join the sacrum to the ischial spine and the ischial tuberosity, respectively.2 The acetabulum articulates with the femoral head to form the hip joint like a ball and a socket. Even though the acetabular joints form an important anatomical part of the pelvis, these fractures will not be emphasized in this article.
The internal iliac arteries provide perfusion to the pelvis. They traverse alongside to a large venous plexus that drains into the internal iliac veins. The largest and most commonly injured vessel is the superior gluteal artery.2 Pelvic fracture-related bleeding arises because the vascular structures of the internal iliac arterial and venous systems are located just anterior to the ligaments that form the SI joints. Therefore, they are exposed to damage from the trauma that causes ligamentous disruption.11 The structures at greatest risk for injury due to their location adjacent to the pelvic bones and ligaments are: the sciatic, femoral, and obturator nerves, as well as the bladder, urethra, and extraperitoneal rectum.11 When the ligamentous structures rupture, the pelvic ring loses its integrity and widens, and bleeding potentially ensues. While bony fractures may appear impressive on plain films, what really matters in terms of pelvic stability is the degree of ligamentous disruption.
Classification
The most widely used classification for pelvic fractures is the Young and Burgess classification system, which is illustrated in a schematic adaptation in Figure 1. This classification is based on the fracture patterns resulting from vectors of force applied on the pelvic ring.14 These patterns are divided into lateral compression (LC), antero-posterior compression (APC), and vertical shear (VS). LC fractures are the most common pelvic ring fractures, occurring in approximately 60% of the cases. LC1 fractures are the most common, and are associated with a lower-energy mechanism.15 There is lateral impact in a horizontal plane. In this mechanism of injury, the force is applied to the side of the pelvis, as occurs in motor vehicle collisions in which the car is "T-boned," or when a pedestrian is struck on his side by the vehicle. As the pelvis "implodes," the structures migrate and rotate medially, causing the structure to collapse inward.
Manson et al conducted a retrospective case control study in which patients with LC1 fractures were analyzed based on outcome. They found that fracture pattern was not a good indicator of risk for death.15 In this series, mortality was related more to injuries to the chest, abdomen, and head than to the characteristics of the pelvic fracture pattern. They concluded that fracture pattern is a poor marker of mortality for cases of LC1 fractures.
In LC2 injuries, higher-energy forces are required to cause disruption of the posterior sacroiliac ligament and displacement of the sacroiliac joint. The stability of the joint will be dependent upon the degree of the force applied. LC3 injuries are unstable fractures. The force applied causes the hemipelvis to rotate inward to a point at which there is a complete disruption of the integrity of the SI joint. There is a close association between LC fractures and lethal torso injuries, such as aortic dissection.10,15
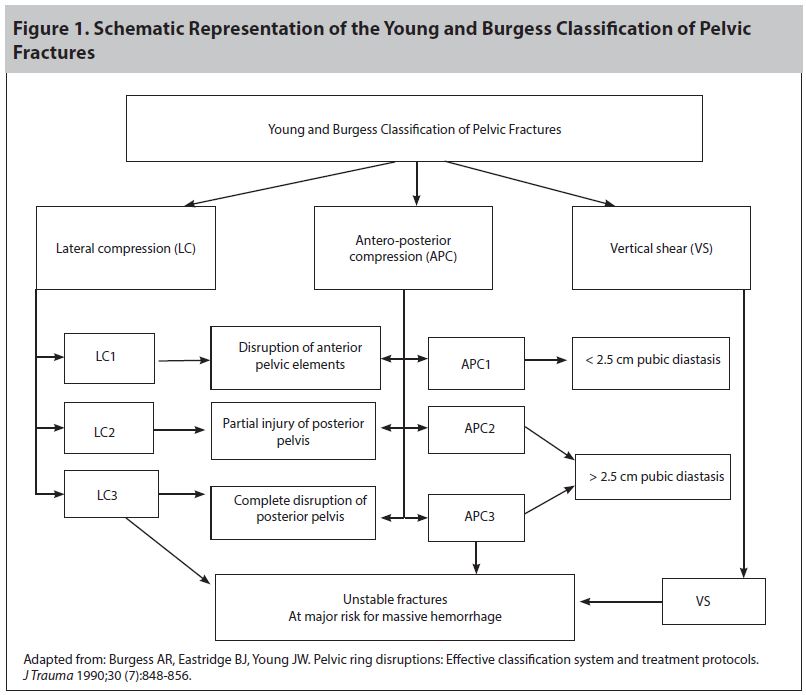
Adapted from: Burgess AR, Eastridge BJ, Young JW. Pelvic ring disruptions: Effective classification system and treatment protocols.
The APC injuries, also known as the "open book" fractures, are caused when the force is applied directly to the pubis, which may occur when a car strikes a pedestrian frontally. In APC1 fractures, the pubic symphysis widens no more than 2.5 cm and there is no injury to the SI joint or to the posterior pelvic elements. In APC2 injuries, the pubic symphysis widens more than 2.5 cm. In this subclassification, there is potential disruption of the anterior sacroiliac, sacrospinous, or sacrotuberous ligaments. The posterior sacroiliac ligaments remain uninjured. Because there is progressive widening of the pelvic ring, bleeding is more likely. Higher-energy forces are required to cause APC3 injuries. The posterior sacroiliac ligaments are completely disrupted, resulting in a severely unstable pelvic ring in which the highest clinical suspicion for hemorrhage should be considered. APC3 injuries are associated with the largest amount of blood loss.2,15
In the VS injury pattern, also known as the Malgaigne fractures, the vector of force is applied cephalad. These injuries are typically caused by falls from a height or motor vehicle or motorcycle collisions. The integrity of the ring is compromised because the hemipelvis is injured anteriorly to the pubic rami and/or pubic symphysis and posteriorly with disruption of the SI joint. As a result, the physical exam may reveal shortening of the leg ipsilateral to the injury, which occurs because there is unopposed force applied by the psoas muscle, pulling the hemipelvis upward. Combination injury patterns can also occur, most commonly with the LC and VS.2 Of note, there can be a significant amount of patients who have high-grade pelvic fractures without pelvic hemorrhage. Conversely, a great number of patients who have arterial sources of hemorrhage have only minimally displaced fractures in the pelvic plain film.1
Initial Assessment
The evaluation of the trauma patient has been standardized and should be guided by the Advanced Trauma Life Support (ATLS) guidelines in which a systematic approach is followed for a thorough assessment. There are several compartments into which a patient can bleed: the thoracic, abdominal, muscular, and pelvic/retroperitoneal. In the primary survey, airway, breathing, control of life-threatening hemorrhage, and the establishment of intravenous access are essential. As an adjunct of the primary survey, a Focused Assessment with Sonography in Trauma (FAST) should be performed. Also, focused abdominal and pelvic exams should be conducted to evaluate for tenderness, tympany, distention, or bruising.
The awake patient with a pelvic injury may complain of pain around the hip, lower abdomen, or lower back. When evaluating the stability of the pelvic ring, it is essential to avoid "rocking" of the pelvis. A disadvantage of performing this maneuver is eliciting intense pain. Every time the pelvis undergoes rotational forces applied by the physical examiner, there is potential for further widening of the ring, disruption of a potential clot, aggravation of hemorrhage, and further injury to adjacent organs. The correct way to test for pelvic stability is to grab the iliac crests and push inward in a gentle motion. The symphysis pubis also can be palpated to assess for widening.
Shlamovitz et al conducted a retrospective study in which 115 patients with blunt pelvic fractures, including 34 with mechanically unstable pelvic fractures, were analyzed.16 The study demonstrated that the presence of either a pelvic deformity or an unstable pelvic ring on physical examination has poor sensitivity for detection of mechanically unstable pelvic fractures in blunt trauma patients. The study suggested that blunt trauma patients with GCS greater than 13 and without pelvic pain or tenderness are unlikely to suffer an unstable pelvic fracture.
The management algorithm for a patient with a pelvic injury depends on the hemodynamic status at the time of arrival. The Western Trauma Association has defined hemodynamic instability in an adult blunt trauma victim with a pelvic fracture as SBP less than 90 mmHg, significant transfusion requirement (4-6 units of packed red blood cells), or a base deficit of -6.1 It is essential to recognize the institutional limitations and the necessity for a higher level of care early in the evaluation and resuscitation phase for critically injured patients. Transfer arrangements should be made as soon as possible to minimize delays in definitive treatment.
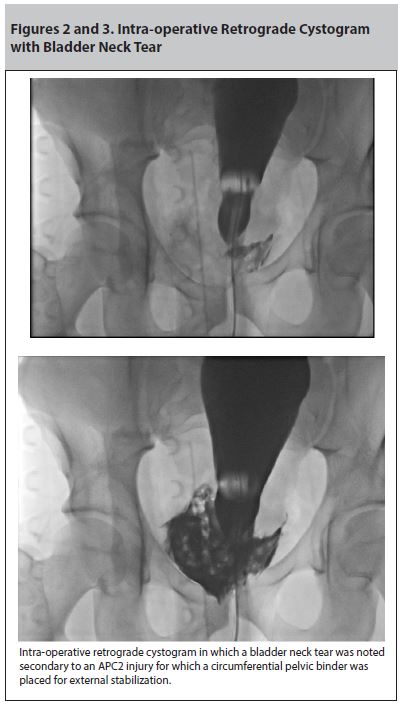
Intra-operative retrograde cystogram in which a bladder neck tear was noted secondary to an APC2 injury for which a circumferential pelvic binder was placed for external stabilization.
Genitourinary Injuries
Pubic bone fractures can be closely associated with genitourinary injuries (GUI). Attention should be focused on identifying findings that can suggest GUI, such as blood in the urethral meatus, perineal or scrotal hematoma, suprapubic tenderness, lacerations surrounding the perineum, vagina, or rectum, or blood or a periprostatic hematoma with rectal examination. Bjurlin et al analyzed 1,400 patients with pelvic fractures using the national trauma data bank and found that males have twice the incidence of GUI as females.18 Urethral injuries are fairly uncommon, occurring in approximately 1% of blunt pelvic injury patients. More than 80% of patients who present with bladder injury will have an associated pelvic fracture.18 Importantly, a negative physical examination for GUI does not exclude it.
Despite being uncommon, injury to the urethra is not confined to males. In a female patient with pubic bone fractures, a vaginal examination is also necessary. The injury patterns that have been most associated with urethral and bladder injuries are straddle fractures (bilateral superior and inferior pubic rami fractures) and forces that cause abrupt thigh abduction.11 If there are no physical exam findings that suggest GUI, the initial approach should be to attempt to insert a urinary catheter, which should be done by an experienced provider. If any resistance is felt, the procedure should be aborted and the patient should have a retrograde urethrogram for evaluation of the urethra. Insertion of a urinary urethral catheter can potentially exacerbate or worsen a pre-existing injury.
If there is any concern for urethral injury, imaging should be obtained. As mentioned previously, gross hematuria noted after spontaneous voiding or after the insertion of a urinary catheter is the most common sign of bladder injury. In the setting of pelvic fracture and hematuria, the CT cystogram with distended bladder and post-emptying views has replaced the retrograde cystourethrogram for the diagnosis of bladder injury.11 Figures 2 and 3 are examples of an intra-operative retrograde cystogram with a bladder neck tear. Bladder rupture can occur intra- or extraperitoneally. Management depends upon the location of the tear. Intraperitoneal bladder rupture is an indication for operative repair. Extraperitoneal rupture can usually be managed with an indwelling bladder catheter.11
Laboratory Workup
Blood analysis should be sent for all patients with a suspected pelvic injury. The single most important blood sample to send is the blood type and cross. A chemistry and complete blood count should be sent, but waiting for these results should not dictate the need to start early transfusion of blood products in the setting of shock or hemodynamic instability. A coagulation profile can give information regarding possible coagulopathy, which can be quite common in the trauma patient. Serum lactate will define the severity of acidosis, which is translated into the degree of end-organ hypoperfusion, and can be drawn serially to determine the response to resuscitation and resolution of shock. Urinalysis should be ordered to evaluate for hematuria since pelvic fracture patients with ureteral injury do not exhibit gross hematuria 15-45% of the time. In the setting of bladder injury, the hallmark is gross hematuria, which can be seen in up to 95% of patients. Hence, approximately only 5% of patients with bladder injury present with microscopic hematuria.11,17
Plain Films
Routinely, the pelvic antero-posterior (AP) plain film has been utilized during the initial trauma evaluation to identify fracture patterns that can potentially predispose the patient to hemorrhage. The plain pelvic AP film is a very useful tool in the poly-trauma patient who presents with hemodynamic instability or a depressed level of consciousness in which pelvic injury needs to be assessed. In patients who are awake, alert, do not complain of pelvic pain, and have no physical exam findings suggestive of pelvic injury, the pelvic plain film has little utility. In the patient who presents with pelvic pain and has stable vital signs, the plain film is an appropriate initial imaging study. However, if the plain film is unremarkable or equivocal and the patient continues to complain of pelvic pain, a computed tomography (CT) scan of the pelvis should be obtained.
When evaluating plain films, acetabular and posterior pelvic fractures are commonly underestimated and overlooked. Barlebem et al conducted a retrospective study in 174 blunt trauma patients with pelvic fractures.19 They reported that 51% of blunt trauma patients with pelvic fractures were underdiagnosed by pelvic plain film in the ED, and 22% were completely misdiagnosed as having no pelvic fracture on pelvic plain films. A thorough physical exam and pelvic CT scan are more sensitive and accurate than the pelvic X-ray for the diagnosis of pelvic fracture in blunt trauma patients. The study also concluded that the pelvic X-ray in the ED is obsolete in the absence of hemodynamic instability and significant physical examination findings.
The pelvic plain film should be evaluated with a standardized approach to minimize missing subtle injuries. One systematic way to assess the film is to look away from the bones and focus on the soft tissues to evaluate for edema or foreign bodies. Then look at the three circles and evaluate the bony cortex, as it should be contiguous. Next, focus on the large iliac bones, the sacrum, and the SI joints. Even if bony displacement is not seen but there is loss of the continuance of the lines, that patient most likely has a pelvic fracture. Finally, look at the patient’s acetabula. Acetabula and anterior fractures can be hard to diagnose, as they can be quite subtle. It is important to remember that the pelvis is a circular structure and it is almost impossible to break a circle at only one point. If you see one fracture, look carefully, because it is highly likely that there is a second fracture. Inlet and outlet views of the pelvis better evaluate the SI joints, sacrum, and migration of a hemipelvis. Judet views for evaluation of the acetabulum have been replaced by CT scan.
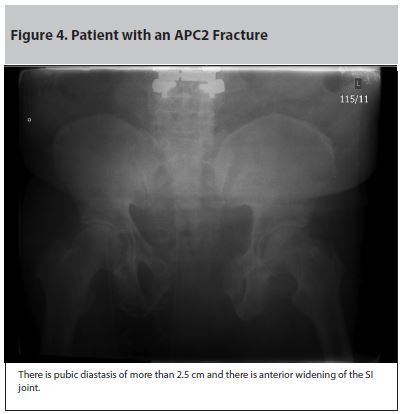
There is pubic diastasis of more than 2.5 cm and there is anterior widening of the SI joint.
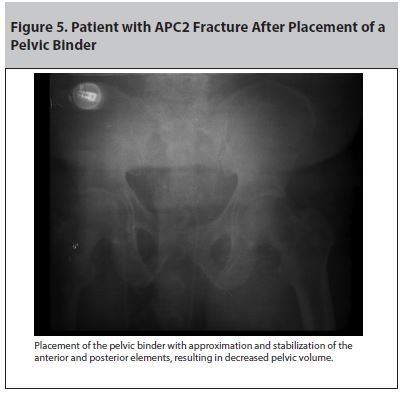
Placement of the pelvic binder with approximation and stabilization of the anterior and posterior elements, resulting in decreased pelvic volume.
Ultrasound
The FAST exam has become an indispensable tool during the evaluation of the blunt trauma patient in the acute setting. It is utilized to detect free intra-peritoneal fluid, which is presumed to be blood until proven otherwise. It can guide therapeutic interventions based on its findings and the patient’s hemodynamics. The FAST is most useful when the patient presents with hemodynamic instability. A positive FAST in an unstable patient mandates the need for an emergent operative intervention. A positive FAST in a stable patient allows for better characterizing the extent of the injuries, most likely with a CT scan.
Chiu et al studied 772 blunt trauma patients, some of whom had concomitant pelvic fractures that were initially evaluated with FAST upon admission.20 They found that there was a 29% incidence of intra-abdominal injury without hemoperitoneum; the FAST exam as a sole diagnostic tool is not reliable.
In another study, Tayal et al analyzed 87 patients with major blunt pelvic injuries.21 The authors found that the sensitivity and specificity of the FAST exam decreased when there was a concomitant major pelvic fracture. They theorized that this could be due to several factors, including an intra-peritoneal bladder rupture causing uroperitoneum or seepage of retroperitoneal hematoma, which can cause intra-peritoneal fluid collections secondary to a displaced fracture. The study argues that although intra-peritoneal bladder rupture mandates a therapeutic operative intervention, this should not supersede the need for emergent pelvic angiographic embolization therapy for life-threatening bleeding. They propose the use of an ultrasound-guided supra-umbilical diagnostic peritoneal aspirate (DPA) to differentiate urine from blood. In the DPA, the catheter is inserted above the umbilicus using the Seldinger technique, and intraperitoneal fluid is aspirated. A positive DPA is aspiration of greater than 10 mL of gross blood.1 Thus, DPA should be considered prior to laparotomy in a patient responding to resuscitation with a positive FAST and a concomitant major pelvic fracture.
Computed Tomography Scanning
The computed tomography (CT) scan has become the gold standard for imaging in the blunt trauma patient with multiple injures. It offers superior information that can be easily missed with plain films, ultrasound, DPL, or DPA. The CT scan also helps to better characterize the severity and displacement of the pelvic fracture. In addition, it can identify and quantify the presence and size of a pelvic hematoma. Three-dimensional reconstructions provide even more refined images for management planning.
Another important advantage of CT imaging is the administration of intravenous (IV) contrast, which can help identify the presence of "blush" or active contrast extravasation (ACE) in the presence of arterial injury. The identification of ACE or blush on CT scan has been reported to have an accuracy of 98% for identifying patients who require embolization.1,22 Data from an analysis of a European trauma registry propose that inclusion of a whole-body CT scan in the initial evaluation of severely injured patients is associated with improved survival.23 Due to the high incidence of concomitant intra-abdominal injury associated with pelvic fractures, it is recommended to order at least an abdominal and pelvic CT scan as part of the standard initial evaluation. The disadvantage of the CT scan is that the patient has to be hemodynamically stable to be transported into the radiology suite. It can also take time, and there is the potential risk for IV contrast-induced complications, such as allergic reactions and contrast-induced nephropathy.
External Pelvic Stabilization Techniques
Whenever there is a high suspicion for a pelvic fracture or confirmation of one in a hemodynamically unstable patient, external compression of the pelvis should be performed. The goal is to reduce the pelvic diameter and potentially stabilize the pelvic elements. Limiting the space available for bleeding provides a tamponade effect. There is evidence that external compression reduces the pelvic volume by 10%.24,25 If there is no widening of the pelvic ring or pubic symphysis diastasis (i.e., LC-type injuries, pubic rami fractures), external pelvic stabilization is not likely to be helpful and may exacerbate the injury.1
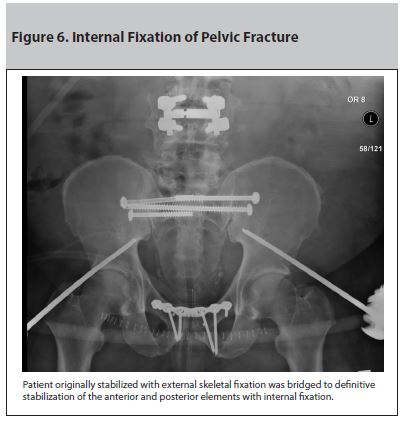
Patient originally stabilized with external skeletal fixation was bridged to definitive stabilization of the anterior and posterior elements with internal fixation.
Noninvasive external compression can be achieved with a bed sheet or with a commercial pelvic circumferential binder. The bed sheets, which should be readily available in any emergency department, are criss-crossed over the patient’s pelvis and tied down tightly. The bed sheet should be kept wide, around 20 cm, to distribute the force better. The circumferential pelvic binder can be adjusted with Velcro and pulleys. Figure 4 illustrates a patient with an APC2 fracture, and Figure 5 shows the same patient after placement of a pelvic binder. To confirm the correct placement of these devices, they should be centered over the greater trochanters of the femur and should cover the buttocks underneath in most patients.26 One common mistake is to place the binder over the lower part of the abdomen, which will not provide adequate pelvic compression. It can also precipitate abdominal compartment syndrome and further worsen hypotension in an already unstable patient due to a decrease in venous return to the heart. These devices are radiolucent and they can be cut if femoral access is needed.
A pelvic AP plain film should be obtained after the placement of an external compression device to confirm the reduction of the pelvic elements. If there is inadequate reduction, it is recommended to readjust and repeat the film. The compression device should not be left in place longer than 24 hours, as it can cause skin necrosis over the injured areas and bony prominences.27
Placement of external skeletal fixation is another tool for pelvic stabilization in the unstable patient. In the emergent setting, it has been almost completely replaced by binders and bed sheets because of their rapid application and effectiveness. Many resources are needed for the placement of external fixators in the emergent setting, and the availability will be mostly institutionally dependent. Like the temporary pelvic binder, the external fixator helps control bleeding by reducing and stabilizing the fractured bony fragments and by decreasing the diameter of the pelvis.26 Additionally, early placement of external fixation has been shown to reduce mortality.26,28 Because the binder is not a permanent treatment option, external fixation can be used as a bridge to definitive internal fixation or as definitive care itself.25,26 A patient with hemodynamic instability and an unstable pelvic fracture who will be taken to the operating room for exploratory laparotomy may benefit from skeletal external fixation.1 Even though skeletal external fixation offers some advantages, a comparison between external fixation and temporary pelvic binder showed that the external fixator was found to have higher blood transfusion needs at 24 and 48 hours when compared to the binder.29 Figure 6 illustrates placement of definitive internal fixation after the patient was initially stabilized with external skeletal fixation.
Table 1. Indications for Emergent Pelvic Angiography
- Pelvic fractures with hemodynamic instability or signs of ongoing bleeding after non-pelvic sources of hemorrhage have been excluded
- Evidence of active contrast extravasation or "blush" in the pelvic CT scan
- Ongoing bleeding after pelvic angiography with or without embolization after non-pelvic sources have been excluded
- Age 60 years or more with major pelvic fracture
- Evidence of pelvic hematoma of > 500 mL in size on pelvic CT scan
Angiography and Embolization
The approach to pelvic bleeding should be selected based on the institutional resources and the instability of the patient. Pelvic angiography is a useful tool and can be diagnostic and therapeutic in the management of arterial pelvic hemorrhage. For this intervention to be an effective option, an interventional radiologist, experienced staff, and an angiography suite need to be available.
The majority of pelvic bleeding is of venous origin. The Eastern Association for the Surgery of Trauma performed a systematic review and published guidelines for the management of hemorrhage in pelvic fracture. Table 1 lists the indications for angiography in these patients.30 Fracture pattern alone is not a predictor of hemorrhage that will require embolization.31 Pelvic angiography with embolization seems to be 85% to 97% effective in controlling arterial hemorrhage.32 Despite angiography with embolization, there is a subset of patients that will require a second angiogram due to ongoing bleeding. It has been reported that 4.6% to 24% of patients with either no bleeding seen on the initial angiogram or initially successful pelvic embolization will require a repeat pelvic angiography with embolization. Independent risk factors for recurring pelvic bleeding include: transfusion requirement of more than two units of packed red blood cells per hour before angiography, more than two injured vessels that required embolization, recurring hypotension after initial angiography, absence of intra-abdominal injury, and a persistent base deficit.31-33
Miller et al performed a prospective study in which 1,171 patients admitted with pelvic ring fractures were analyzed based on hemodynamics on presentation.34 The authors concluded that if patients presented with hypotension from a pelvic fracture and had transient or no response to initial resuscitation, this indicated the presence of arterial bleeding in more than 70% of patients. In this series, in patients who responded to initial resuscitation, arterial bleeding was unlikely, with a negative predictive value of 100%.
Pre-peritoneal Pelvic Packing
Newer operative techniques are being integrated into the treatment of unstable patients with pelvic fractures when there is a delay for angiography or when there are concomitant injuries that warrant operative interventions. Pre-peritoneal pelvic packing is a technique in which laparotomy pads are inserted into the pelvic space to tamponade pelvic bleeding. The procedure consists of performing a horizontal Pfannenstiel incision just above the pubic symphysis, dissecting and identifying the pre-peritoneal fascia, evacuating the pelvic hematoma, and packing the pelvic space with surgical lap pads. The goal of this procedure is to apply direct pressure to the pelvic vasculature without violating the peritoneal space. If the patient warrants an exploratory laparotomy, a separate vertical midline incision can be made. There are conflicting data regarding whether this intervention should be performed before or after angiography/embolization. Eighty-five percent of the bleeding from pelvic fractures is venous and bony in origin, and by "packing" the pelvis first, there is a reduction in the transfusion requirement.35,36 If the patient is taken to angiography first and has a negative study, time has been lost attempting to control the hemorrhage.
Cothren et al reported data advocating for the integration of external skeletal pelvic fixation in combination with pre-peritoneal pelvic packing as an initial approach before angiography.36 The authors propose that this technique can be advantageous in rural hospitals. It seems to be a safe procedure, and the packs are usually left in place for 48 hours. This novel approach potentially addresses the primary source of bleeding, which, the majority of the time, is likely to be venous and bony in origin. It can also be done concurrently with other operative procedures such as laparotomy, thoracotomy, fasciotomy, etc. In their study, fewer than 15% of patients required angiography/embolization for ongoing arterial bleeding after being packed. The study concluded that angiography/embolization should be seen as a complementary procedure for life-threatening hemorrhage control after pre-peritoneal pelvic packing. Dora et al demonstrated that pelvic packing performed as a first-line therapy with subsequent angiography, if needed, may be just as effective, if not better, than angiography alone for controlling bleeding.37
Endovascular Technology for Hemorrhage Control
Emerging endovascular technology is being applied in the setting of trauma. The resuscitative endovascular balloon occlusion of the aorta (REBOA) is a catheter device with a balloon in the distal end. It is inserted into the femoral artery through an introducer over a wire and is inflated once at the desired level, creating an internal occlusion of the aorta. The catheter position can be confirmed using a plain film or with ultrasound. When used for hemorrhage control in the setting of pelvic fractures, the balloon is inflated at the level of the infra-renal aorta before the bifurcation. It can serve as a temporizing measure for hemorrhage control. When caring for a patient with hemodynamic instability, it can serve as a temporizing measure and as a bridge for resuscitative efforts, induction of anesthesia, or transfer to the operating room or to the angiography suite in order to achieve definitive hemorrhage control.
Brenner et al published a case series in which the use of REBOA proved to be a feasible and effective method of internal aortic control for patients in end-stage shock from blunt and penetrating trauma mechanisms.38 Martinelli et al reported the use of intra-aortic balloon occlusion in patients with critically uncontrollable hemorrhagic shock secondary to pelvic fractures.39 Their study suggests that this procedure may be safe and effective to decrease mortality in this setting. However, further investigation is needed to establish the usefulness of this device.
Conclusion
Blunt pelvic trauma can be a devastating injury that can be accompanied by trauma to other structures, and which places the patient at risk for life-threatening hemorrhage. It is crucial to have a high index of suspicion for this diagnosis when faced with a patient who has unstable vital signs or who has a mechanism of severe trauma. The initial evaluation should always be standardized to all trauma patients. A thorough physical examination is essential. Early transfer arrangements should be made by institutions with limited resources. For patients with an unstable pelvic fracture as the source of hemorrhage, external stabilization of the pelvis should be accomplished as soon as possible. The pelvic plain film is a great tool as an initial imaging study in a patient with hemodynamic instability or depressed level of consciousness. The CT scan of the pelvis is the gold standard imaging study and, when performed with IV contrast, it can provide valuable information regarding ACE in the setting of arterial vascular injury. However, in the setting of hemodynamic instability, a FAST exam should be performed. If positive, the patient should be taken for an operative intervention in which pre-peritoneal packing and skeletal external fixation can be considered as reasonable treatment approaches. In the setting of a negative FAST and an unstable pelvic fracture, the decision of whether the patient goes for an angiogram or to the operating room for pre-peritoneal packing and external fixation depends on the institution. As further clinical research evolves with the REBOA, more patients with pelvic-related bleeding will potentially benefit from its use.
References
-
Davis J, Moore F, McIntyre R, et al. Western trauma association critical decisions in trauma: Management of pelvic fracture with hemodynamic instability.
J Trauma 2008; 65: 1012-1015. - Mattox K, Moore E, Feliciano D. Pelvis. Trauma, 7th ed. McGraw Hill; 2013: |655-668.
-
Moreno C, Moore EE, Rosenberg A, et al. Hemorrhage associated with major pelvic fracture: A multispecialty challenge.
J Trauma 1986;26:987-994. -
Smith W, Williams A, Agudelo J, et al. Early predictors of mortality in hemodynamically unstable pelvic fractures.
J Orthop Trauma 2007;21:31-37. -
Cothren CC, Osborn PM, Moore EE, et al. Preperitoneal pelvic packing for hemodynamically unstable pelvic fractures: A paradigm shift. J Trauma 2007;62:
834-839. - Gililand MD, Ward RE, Barton RM, et al. Factors affecting mortality in pelvic fractures. J Trauma 1982;22:691-693.
- Ben-Menachem Y, Coldwell DM, Young JW, et al. Hemorrhage associated with pelvic fractures: Causes, diagnosis, and emergent management. AJR Am J Roentgenol 1991;157:1005-1014.
-
Cryer HM, Miller FB, Evers BM, et al. Pelvic fracture classification: Correlation with hemorrhage. J Trauma 1988;28:
973-980. -
Shulman J, O’Toole R, Castillo R, et al. Pelvic ring fractures are an independent risk factor for death after blunt trauma.
J Trauma 2010;68:930-934. - Demetriades D, Karaiskakis M, Velmahos G, et al. Pelvic fractures in pediatric and adult trauma patients: Are they different injuries? J Trauma 2003;54:1146-1151.
- Flint L, Cryer H. Pelvic fracture: The last 50 years. J Trauma 2010;69:483-488.
- Balogh Z, Caldwell E, Heetveld M, et al. Institutional practice guidelines on management of pelvic fracture related hemodynamic instability: Do they make a difference? J Trauma 2005;58:778-782.
- Biffl WL, Smith WR, Moore EE, et al. Evolution of a multidisciplinary clinical pathway for the management of unstable patients with pelvic fractures. Ann Surg 2001;233:843-850.
-
Burgess AR, Eastridge BJ, Young JW. Pelvic ring disruptions: Effective classification system and treatment protocols.
J Trauma 1990;30 (7):848-856. - Manson T, Nascone J, Sciardini M, et al. Does fracture pattern predict death with lateral compression type 1 pelvic fractures? J Trauma 2010;69:876-879.
- Shlamovitz G, Mower W, Bergman J, et al. How (un)useful is the pelvic ring stability examination in diagnosing mechanically unstable pelvic fractures in blunt trauma patients? J Trauma 2009;66:815-820.
- Mattox K, Moore E, Feliciano D. Genitourinary trauma. Trauma, 7th edition. McGraw Hill; 2013: 669-708.
-
Bjurlin M, Fantus R, Mellet M, et al. Genitourinary injuries in pelvic fracture morbidity and mortality using the national trauma data bank. J Trauma 2009;67:
1033-1039. - Barleben A, Jafari F, Rose J, et al. Implementation of a cost-saving algorithm for pelvic radiographs in blunt trauma patients. J Trauma 2011;71:582-584.
- Chiu WC, Cushing BM, Rodriguez A, et al. Abdominal injuries without hemoperitoneum: A potential limitation of focused abdominal sonography for trauma (FAST). J Trauma 1997;42:617-623.
- Tayal V, Nielsen A, Jones A, et al. Accuracy of trauma ultrasound in major pelvic injury. J Trauma 2006;61:1453-1457.
- Pereira SJ, O’Brien DP, Luchette FA, et al. Dymanic helical computed tomography scan accurately detects hemorrhage in patients with pelvic fracture. Surgery 2000;128:678-685.
- Huber-Wagner S, Lefering R, Qvick LM, et al. Working group on polytrauma of the German trauma society. Effect of whole-body CT during trauma resuscitation on survival: A retrospective, multicenter study. Lancet 2009;53:1455-1461.
-
Bottlang M, Simpson T, Sigg J, Krieg JC, et al. Noninvasive reduction of open book pelvic fractures by circumferential compression. J Orthop Trauma 2002;16:
367-373. - Krieg JC, Mohr M, Ellis TJ, et al. Emergent stabilization of pelvic ring injuries by controlled circumferential compression: A clinical trial. J Trauma 2005;9:659-664.
- Feliciano D, Mattox K, Moore E. Pelvic Fractures. Trauma, 6th edition. McGraw Hill; 2008: 669-788.
- Jowett AJ, Bowyer GW. Pressure characteristics of pelvic binders. Injury 2007;38:118-121.
-
Burgess AR. The management of hemorrhage associated witg pelvic fractures.
Int J Orth Trauma 1992;2:101. - Krieg JC, Mohr M, Ellis TJ, et al. Emergent stabilization of pelvic ring injuries by controlled circumferential compression: A clinical trial. J Trauma 2005;59:659-664.
- Culliane DC, Schiller HJ, Ziellinski MD, et al. Eastern association for the surgery of trauma practice management guidelines for hemorrhage in pelvic fracture — update and systematic review. J Trauma 2011;71:1850-1868.
-
Starr AJ, Griffin DR, Reinert CM, et al. Pelvic ring disruptions: Prediction of associated injuries, transfusion requirement, pelvic arteriography, complications, and mortality. J Orthop Trauma 2002;16:
553-561. - Gorlay D, Hoffer E, Routt M, et al. Pelvic angiography for recurrent traumatic pelvic arterial hemorrhage. J Trauma 2005;59:1168-1173.
- Shapiro M, McDonald AA, Knight D, et al. The role of repeat angiography in the management of pelvic fractures. J Trauma 2005;58:227-231.
-
Miller P, Moore P, Mansell E, et al. External fixation or arteriogram in bleeding pelvic fracture: Initial therapy guided by markers or arterial hemorrhage.
J Trauma 2003;54:437-443. -
Cothren C, Osborn P, Moore E, et al. Preperitoneal pelvic packing for hemodynamically unstable pelvic fractures: A paradigm shift. J Trauma 2007;62:
834-842. - Cothren C, Moore E, Smith W, et al. Preperitoneal pelvic packing/external fixation with secondary angioembolization: Optimal care for life-threatening hemorrhage from unstable pelvic fractures. J Am Coll Surg 2011;212:628-635.
- Dora K, Tai M, Wing-Hong L, et al. Retroperitoneal pelvic packing in the management of hemodynamically unstable pelvic fractures: A level 1 trauma center experience. J Trauma 2011;71:79-86.
- Brenner M, Moore, L, Dubose J, et al. A clinical series of resuscitative endovascular balloon occlusion of the aorta for hemorrhage control and resuscitation. J Trauma Acute Care Surg 2013;75:506-511.
- Martinelli T, Thony F, Declety P, et al. Intra-aortic balloon occlusion to salvage patients with life-threatening hemorrhagic shocks from pelvic fractures. J Trauma 2010;68:942-948.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
