Uncommon but Important Infectious Diseases
Uncommon but Important Infectious Diseases
Recently, we published a series of articles on the international traveler in the ED, emphasizing the risk factors and geography. This article provides a more standard review of some of the diseases seen in travelers to tropical areas and also some, like West Nile, that have migrated into temperate climates. This article will complete our series.
Sandra M. Schneider, MD, FACEP, Editor
Malaria
Worldwide, malaria is clearly the most deadly vector-borne illness. Each year, 300-500 million new cases of malaria occur, with approximately 1.5-2.7 million deaths annually,1 most in young children in sub-Saharan Africa. Roughly 1300 cases and 10 deaths per year are diagnosed in returning U.S. travelers.2 The majority of these cases occur in patients who have not taken chemoprophylaxis.3
Without proper treatment, severe malaria can progress from a few flu-like symptoms to coma and death in as little as 12-48 hours.4 It is suggested that "any patient who has potentially been exposed to malaria, however briefly, and presents with fever or unexplained systemic illness should be assumed to have life-threatening malaria until proven otherwise."5
History. The Centers for Disease Control (CDC) was established in 1946 and was responsible for efforts that eradicated malaria in the United States. However, localized outbreaks of mosquito-borne malaria have been documented as recently as 2003.6 Currently, the vast majority of cases of malaria currently diagnosed in the United States are in returning travelers.
Pathology. Malaria is caused by the protozoan Plasmodium, of which there are four species: P. falciparum, P. vivax, P. ovale, and P. malaria. Falciparum malaria accounts for up to 95% of the deaths worldwide, even though it represents only 50% of the infections.5 Both P. vivax and P. ovale produce dormant forms that can persist in the liver of infected people and resurface in several months or up to years after initial infection (longest recorded relapse time was 30 years).7 P. falciparum is most common in Africa, East Asia, Haiti, and the Amazon, while P. vivax is the dominant species in Central America, the Middle East, India, and Southeast Asia.8
Infection begins when a human is bitten by an infected mosquito of the Anopholes species, which are most often active at dusk/night. The incubation period typically is 1-3 weeks, but can be highly variable, ranging from several days or up to a year. Patients taking chemoprophylaxis may have delayed onset of illness and milder symptoms. Once inside the body, the malaria protozoan feeds on hemoglobin and other red blood cell (RBC) proteins, which results in lysis of the RBCs, causing anemia. P. falciparum infection produces a higher level of parasitemia than the others, and also changes the RBC surface so that it adheres to capillary beds and slows blood supply to the vital organs. Thus the brain, kidneys, liver, gut, placenta, etc., are at risk of focal ischemia, thrombosis, and infarction. This effect explains the widely varied clinical symptoms observed (cerebral malaria, acute renal failure, etc.). Patients slowly develop immunity living in an endemic area, which is why most fatal cases of malaria occur in children (or travelers) who have not yet attained protective immunity. After leaving the endemic area, immunity that took years to develop wanes in only about six months.
Signs and Symptoms. Depending on the species of Plasmodium, fever cycles begin and repeat at regular intervals (72 hours [quartan] for P. malaria and 48 hours [tertian] for the other three). However, the regular cycle of fevers does not fully develop until the infection is well established. More commonly, the patient presents with vague flu-like symptoms (low-grade fever, headache, malaise, abdominal pain without tenderness, vomiting, diarrhea, muscle aches) with several fever spikes throughout a 24-hour period. Less commonly, patients complain of shortness of breath or chest pain. On physical examination, hepatomegaly and splenomegaly are seen in roughly 25-35%, with jaundice evident in 16%.9 Lymphadenopathy and rash are not common. Likewise, elevated white blood cell count (WBC) is unusual (< 5% of cases) in acute malaria.10
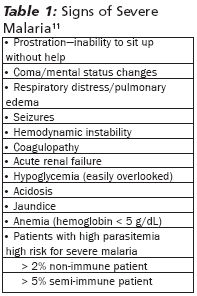
Progression to severe malaria is a life-threatening complication. Severe malaria is defined by the World Health Organization (WHO) as malaria infection and one or more of the signs listed in Table 1. Unfortunately, severe malaria is most common in travelers (non-immune patients) or in primigravida women. Once this stage is reached, even with modern intensive care unit (ICU) treatment, the mortality rate is greater than 30%.12 Cerebral malaria, a type of malaria-induced encephalitis, is the most common cause of death in severe malaria.13 Non-cardiogenic pulmonary edema occurs less often than cerebral malaria, but has an 80% mortality rate.14 Acute renal failure is rare in children, but is seen in 30% of non-immune adults with P. falciparum.9
 |
Diagnosis. One must maintain a high degree of suspicion in patients with a history of travel to an endemic area and any history of fever. Cases of "runway malaria" exist where it is suspected that an infected mosquito has been transported by airplane and may cause disease when released at the destination airport.15 Thus, any international traveler ill enough to require admission may require screening as well. Table 2 summarizes some of the pitfalls in making the diagnosis. While most patients become ill in the first month, some may take months (even years) to present. Table 3 summarizes high-risk travel that is associated with malaria.
 |
 |
Classically, the gold-standard for diagnosis of malaria is peripheral blood smear and identification of the parasites with a microscope (thick or thin smear with Giemsa stain). CDC recommends obtaining smears every 12-24 hours for three consecutive days before considering malaria excluded.16 Rapid detection tests (RDTs) using antibody detection techniques exist, but only one of these tests is currently approved for use in the United States (BinaxNOW Malaria). In cases of high suspicion, one should consider empiric therapy while waiting for test results.17
Treatment. Treatment of malaria depends on the species of Plasmodium with which the patient is infected and the severity of the illness. Resistance to anti-malarial drugs varies geographically. Obviously any acutely ill patient with suspected malaria should be admitted. Further, admission is advised for all children, pregnant women, and non-immune adults (most travelers). Anyone with malaria complications (see Table 1) or high parasite loads (> 2% in non-immune or > 5% in semi-immune patients) should be admitted to an ICU.5 Volume resuscitation, fever control, seizure control (phenobarbital prophylactically, benzodiazepines for treatment), serum glucose supplementation, and early hemodialysis for renal failure are the mainstays of treatment.
Patients diagnosed with malaria should be started on appropriate anti-malarial drugs (usually chloroquine, quinine, or artesunate combined with doxycycline, tetracycline, or clindamycin). However, the exact regimen to use is best decided in consultation with an infectious disease specialist. Patients with severe malaria should be given anti-malaria drugs intravenously, with quinidine and artesunate being the only choices. Parenteral quinidine is cardiotoxic, and its use is recommended only in an ICU setting with cardiology consultation. The Malaria Hotline is available from the CDC 8 AM - 4:30 PM Eastern time at 770-488-7788, and on-call malaria specialists are available after hours as well at 770-488-7100.
Prevention. More than 50% of the cases of imported malaria between 1992 and 2001 (2,616 cases) involved travelers who took no prophylaxis,18 but more recent reviews show even higher numbers of non-compliance (96% of 1579 cases).3
Prophylaxis is taken for 1-2 weeks prior to travel, once weekly on the same day of the week during travel, and for 4 weeks after completion of travel. Taking prophylaxis does not ensure protection. More than 70% of the cases (351 people) of imported malaria in the United States. between 1995 and 2001 were due to malaria prophylaxis failure.18 Most "failures" are from not taking the proper doses or due to drug-resistance issues. In addition to medication, one should employ netting, screens, and repellent as well. The CDC maintains a good resource web site for information of malaria prophylaxis (www.cdc.gov/malaria/travel/index.htm).
Dengue Fever
Like malaria, dengue is mosquito-borne with a similar global distribution. Historically, it was only found in Africa, but it has spread to all tropical regions of the world as a result of international trade. There are an estimated 50-100 million cases of dengue fever annually.19 Of these cases, about 250,000 develop dengue hemorrhagic fever, resulting in about 24,000 deaths. Dengue fever tends to occur in outbreaks or epidemics, and the attack rates can be as high as 70-90% during an outbreak.20 In only 6 years (1995-2001), the number of cases reported in Central/South America doubled. The majority of recent major epidemics have occurred in Central/South America (Mexico, Guatemala, El Salvador, Nicaragua, Columbia, Brazil) and the Caribbean. As with malaria, many of the dengue fever cases occur in children.
Dengue fever is becoming more common in ill returning travelers. Small outbreaks of dengue were detected in Laredo, TX, in 2005 as a result of dengue spreading across the Rio Grande from Nuevo Laredo, Mexico.21
Pathology. Dengue fever is caused by an RNA virus of the Flavivirus genus. The virus is transmitted directly by mosquito from another infected human, and there are no natural animal reservoirs. Currently there are four different virus serotypes, and unfortunately infection with one serotype does not provide protective immunity from the others. Initial infection with the virus (classic dengue fever) typically is not lethal, but the second infection with another serotype can produce the lethal form of dengue: dengue hemorrhagic fever or dengue shock syndrome. For reasons that remain unclear, hemorrhagic fever complications appear to be more common in whites and Asians than in blacks, in females than in males, and in well-nourished than in malnourished children.22
The dengue virus is transmitted to humans from the Aedes species of mosquito, which are active in the daytime. They are found in urban areas, which puts all travelers to endemic areas at risk. The incubation period varies from 3-14 days, with an average of 4-7 days. The virus primarily attacks hepatocytes and endothelial cells.
Signs and Symptoms. Classic dengue fever produces a flu-like syndrome with high fever, headache, arthralgias, and a generalized macular blanching rash. The rash is a helpful clue to distinguish dengue from malaria, in which rashes are uncommon. Symptoms return with fever and rash beginning simultaneously. In the second phase, the rash is mobilliform, maculopapular, sparing the palms and soles, with occasional desquamation. Small (< 1 cm) islands of normal or "spared" skin often are noted in the midst of the rash.19 The fever in the second phase typically is not as high and lasts another 2-5 days.
Another hallmark of dengue fever is the severe arthralgias, leading to the term "breakbone fever."20 The pain begins after the first fever onset, increases in severity with time, and may last several weeks. The most common sites affected are the legs, large joints, and lumbar spine. Interestingly, the bony pain is not present in dengue hemorrhagic fever or dengue shock syndrome.
The more severe form of dengue is dengue hemorrhagic fever. On the third to seventh day of illness around the end of the first phase, an abrupt shock syndrome develops. Patients develop abdominal pain with restlessness, hypothermia, mental status changes, and a sudden drop in platelet count. Thrombocytopenia contributes to spontaneous hemorrhage, ranging from petechiae to life-threatening gastrointestinal bleeding. Epistaxis, bleeding from gums, and menorrhagia also are noted. Hypotension and narrowed pulse pressures are noted, indicating intravascular volume depletion from increased capillary permeability. Loss of fluid into extracellular spaces rather than frank hemorrhage is the usual cause of shock.
The mortality of untreated dengue hemorrhagic fever can be as high as 50%, but ICU support can lower the number to < 5%.23 Most deaths occur from cardiovascular collapse and/or adult respiratory distress syndrome (ARDS). Although children respond to aggressive fluid resuscitation, it recently has been suggested that more refractory cases may be a result of depressed cardiac output, the cause of which is yet to be determined.24 Serum troponin is not elevated in these patients, so it does not appear to result from direct myocyte damage by the virus.
Diagnosis. In most cases, fever that begins after 2 weeks following departure from endemic areas or lasts longer than 10 days is not likely to be dengue fever.
The WHO has listed clinical criteria that support the diagnosis of dengue fever. (See Table 4.) WHO dengue hemorrhagic fever criteria include: recent history of fever, hemorrhagic complications, thrombocytopenia and hematocrit > 20% average for patient's sex, age and population.
 |
Fortunately, laboratory tests can provide clues, as leukopenia (< 5000/mm3), neutropenia (< 3000/mm3), thrombocytopenia (seen in 50% of cases), or mildly elevated AST levels (> 60 U/mL) often are present.25 Hematocrit levels should be monitored, as levels rising > 20% often precede development of shock.20 Likewise, platelet counts < 100,000 cells/µL are seen before initial fever defervescence and onset of shock. Ultimately, proof of diagnosis depends on isolation and identification of the virus by PCR or ELISA tests.
Treatment. Uncomplicated dengue fever can be managed with fever control and fluid replacement. Patients presenting with signs of hemorrhagic fever or shock should be managed aggressively. Patients with a history of repeated travel to endemic areas should be observed for possible development of complications. In most cases, the patient will be admitted for diagnosis confirmation and inpatient observation. Patients who survive the first 48 hours of shock syndrome recover rapidly and do not experience relapse.
Rickettsial Diseases
Currently, rickettsial diseases consist of three main types of illnesses: spotted fevers, typhus, and Q fever. Spotted fevers (e.g., Rocky Mountain spotted fever) are transmitted via tick bites, while the typhus group of diseases is transmitted via fleas/lice, and Q fever is not vector-borne. Regardless of the type of rickettsial disease, treatment with doxycycline is usually the first-line therapy, with fluoroquinolones or macrolides as alternatives. Eschars seen at the site of the vector insect bites are common in all rickettsial diseases (except Q fever), and are caused by the high level of organisms present there.
Spotted Fevers. Rickettsial organisms cause a variety of spotted fevers, including Rocky Mountain spotted fever, Mediterranean spotted fever, African tick-bite fever, north Asian tick typhus, Queensland tick typhus, Japanese spotted fever, and flea-borne spotted fever. Spotted fevers are named for the rash that accompanies the other symptoms. These diseases are found around the globe except Antarctica. Cases are transmitted from infected animals (dogs, cows, other mammals) by tick bites. Often, the history of tick bite is absent, as transmission also can occur via the bite of immature forms whose bites go unnoticed. Most cases are seen in warmer months.
Typhus. Typhus is a group of illnesses (epidemic typhus, murine typhus, scrub typhus) caused by rickettsial organisms and transmitted to humans by lice or fleas. Epidemic typhus tends to occur during natural disasters or in areas of war/poverty.26 A few cases have been reported in the eastern United States in recent years in patients who had close contact with flying squirrels.27
Symptoms of epidemic typhus appear after an incubation period of 8-16 days. The organism invades endothelial cells and produces a multi-system vasculitis. Most patients experience abrupt onset of fever, headache, myalgias, conjunctival injection, and rash. The rash is maculopapular or petechial and involves the entire body except for the face, palms, and soles. Regional lymphadenopathy is seen near the louse bites. Unless antibiotics are initiated, the symptoms progress to delirium, cough, gangrene, coma, and death in a median 12.5 days after symptom onset.26 The mortality rate for untreated epidemic typhus varies from 20% (healthy patients) to 60% (elderly, pre-existing disease). With antibiotic treatment, the rate lowers to 3-4%. Patients who survive without antibiotic therapy recover about 14 days after onset of symptoms. Some patients, even with treatment, develop occult infection termed Brill-Zinsser disease. They harbor low levels of rickettsia in their bodies and may develop recurrent typhus months or even years later, although the course of recurrent disease is milder than epidemic typhus.
Most cases are diagnosed via rising antibody titers, but these are not present until the second week of illness. Doxycycline (100 mg BID for 7-10 days) is the treatment of choice, while chloramphenicol (60-75 mg/kg/day divided into 4 doses) is an effective alternative.26
Murine typhus is a similar infection caused by a different species of rickettsia and is transmitted to humans from infected rat flea bites. Isolated cases are seen in the United States (Texas, southern California) associated with cats and opossums. Murine typhus is seen primarily in port cities located in subtropical and temperate coastal areas. Cases recently have been reported in travelers returning from Hong Kong, China, Brazil, and Indonesia.28 Symptoms are similar to epidemic typhus, although the fatality rate is less (1-4%). Definitive diagnosis is via rising antibody titers, but distinction from epidemic typhus can be difficult due to shared antigens. Treatment is with doxycycline or chloramphenicol.
Scrub typhus is caused by another rickettsial species and transmitted by infected mites (chiggers). It is endemic in southern and eastern Asia (Japan, China, Korea), as well as Indonesia, India, and the Philippines. An estimated 1 million people are infected each year.29 Symptoms are similar to epidemic typhus, although the rash is less severe and often overlooked. Fatality rates can vary from 3% to 60% without treatment, but treatment with doxycycline or chloramphenicol greatly improves recovery.
Q Fever. Q fever is caused by Coxiella burnetii and the disease is endemic in many parts of the world. It is one of the diseases that has been mentioned as a bioterrorism agent. Of note, this organism recently was reclassified as a protobacteria (distantly related to Legionella).30 It is particularly common in southern Spain.31 It is also endemic in the United States, causing 60-70 cases per year in recent years. The disease primarily affects those in close contact with livestock (cattle, sheep, and goats) who breath the organisms into their lungs directly from close contact with animal body fluids (urine, feces, milk, amniotic fluid). No inoculation eschars are seen. The incubation time is variable, but the average time is 18-21 days (range of 9-40 days).32 Only about 50% of infections manifest with clinical symptoms, and mortality of acute infection is generally < 1%. Acute infection symptoms are those of pneumonitis with fever, headache, myalgias, cough, and pleuritic chest pain. For reasons that are unclear, some patients progress to chronic illness (lasting > 6 months) and are at risk for complications of hepatitis and endocarditis. Diagnosis is through rising antibody titers. Treatment is with doxycycline (100 mg BID for 14-21 days).
Schistosomiasis
Schistosomiasis is found in a total of 74 countries in Africa, South America, Asia, the Middle East, and the Caribbean. However, 80% of cases today are seen in sub-Saharan Africa.33 Prevalence in endemic areas can approach 50%, and even short-term travelers to these areas are at risk of infection. Dams and irrigation projects have introduced the disease into new areas, and transmission has been reported even in large urban areas in Africa and Brazil.34 Outbreaks of schistosomiasis also have been reported in adventure travelers on river trips in Africa and among Peace Corps volunteers.35 There are an estimated 400,000 people with schistosomiasis currently in the United States, and they occasionally present with complications of chronic infection.36 However, the disease cannot be passed on here, as the snail species required to complete the parasites life cycle is not found in the United States.
People become infected when they enter fresh water containing the free-swimming larval form that can penetrate normal skin. In people with previous infection, a self-limited inflammatory reaction produces a papular dermatitis (swimmer's itch) that can begin only a few hours after infection.33 Once in the body, larvae migrate into large veins of the liver, mesentery, or ureters depending on the species. After about 2-8 weeks, eggs are produced that pass from the intestines or urine into fresh water to infect specific snail hosts, which in turn release the free-swimming larvae. Once eggs begin to be released, many people develop an acute systemic reaction termed Katayama fever or acute schistosomiasis. The retention of the eggs produces the clinical disease of chronic schistosomiasis. Four major species migrate to produce hepatobiliary or gastrointestinal disease (S. mansoni, S. intercalatum, S. mekongi, and S. japonicum) and one to produce urinary disease (S. haematobium).
Systemic symptoms begin about 2-8 weeks after initial exposure to contaminated water but can take up to 12 weeks to appear. The delay between fresh-water exposure and onset of symptoms can make the connection difficult to discern. Symptoms and physical findings include fever, myalgias, fatigue, abdominal pain, bloody diarrhea, lymphadenopathy, hepatomegaly, non-productive cough, and suspicious laboratory findings of patchy pulmonary infiltrates with eosinophilia are seen. Higher nocturnal fevers and eosinophilia suggest parasitic infection and may be the only specific clues to acute schistosomiasis.37 Most patients' symptoms resolve spontaneously in 2-10 weeks, but some develop persistent abdominal pain, diarrhea, dyspnea, and weight loss.
Symptoms of chronic schistosomiasis usually appear 2-10 years after initial infection and correlate with the area of the body infected. (See Table 5.) They result from inflammation and scarring produced by chronic egg retention.
 |
Diagnosis is via microscopic examination of stool or urine for eggs. Serologic antibody detection is available at the CDC, but it cannot distinguish between active or past infections. Early treatment is crucial to prevent complications of long-term infection; even a single pair of worms is thought to be able to produce severe complications (i.e., transverse myelitis) over time.33 Praziquantel (Biltricide 40-60 mg/kg/d PO divided BID to TID for 3-6 days) is the drug of choice, and just a single treatment can cure 65-90% of patients or reduce egg production by 90%.
Typhoid Fever
Typhoid fever is caused by infection with Salmonella typhi and paratyphoid fever by S. paratyphi. Both diseases are clinically similar, although paratyphoid symptoms generally are less severe and are collectively known as enteric fever. Typhoid fever now is seen mostly in developing countries with poor sanitation.38 There are an average of 400 cases per year in the United States, with 72% of these diagnosed in travelers, most of whom went to India or Latin America (Mexico).39 Travel to India carries the highest risk (18 fold higher).40 About 1-4% of untreated individuals can become chronic carriers of the bacteria. The most infamous carrier was "Typhoid Mary," who unknowingly spread typhoid fever while she worked as a cook in New York in the early 1900s.
After bacteria are ingested, they invade the enteric mucosa and can spread to the lymphatic tissue (Peyer's patches), spleen, liver, and bone marrow. Use of antacids, proton pump inhibitors, history of gastrectomy, and immune deficiency all increase risk of infection. The incubation period varies, with an average of 7-14 days (3-60 day range). Untreated, symptoms usually last around four weeks, though patients given antibiotics recover rapidly over just 2-3 days. Early clinical symptoms consist of headache, abdominal pain, anorexia, dry cough, and myalgias that often precede the fever. Ironically, constipation occurs with equal frequency as diarrhea and is thought to result from hypertrophy of Peyer's patches. Classic physical findings include fever, abdominal pain, and enlarged liver and/or spleen. Fever may be absent during the first week, but it rises to a sustained high fever in weeks 2-4. Abdominal distension appears in the second week and peaks in the third week. Classically, "rose spots" appear in about 30-50% of cases and are subtle salmon-colored blanching maculopapular lesions about 1-4 cm in size.38 Relative bradycardia with the fever is also suggestive of typhoid fever. Conjunctivitis and rales also are reported in about half of cases. Intestinal bleeding is the most common complication (12% in one series)41 and may precede perforation, which occurs at heavily infected Peyer's patches. If patients do not die from intestinal perforation, GI bleeding, myocarditis, or sepsis, they begin to improve in the fourth week, although the recovery is slow and relapses are seen in 10% of cases.38 Mortality can be as high as 90% without antibiotics, but their early use reduces this to < 1%.
Laboratory diagnosis is by culture of a Salmonella species. S. typhi can be cultured from blood (70% successful), stool (30% successful), urine, and the rose spots, but bone marrow aspirates have the highest sensitivity (80-95%) and remain a good source even after several days of antibiotic treatment.42 Presumptive treatment should be started as soon as the disease is suspected. Ciprofloxacin is no longer effective against species from Southeast Asia, and alternatives include ceftriaxone (Rocephin) for severe cases or trimethoprim/sulfamethoxazole (Bactrim) for mild cases.43
Leishmaniasis
Leishmania are intracellular parasites transmitted by the bite of sand flies. The three main types of disease caused are visceral (most dangerous), cutaneous, and mucocutaneous. Cutaneous leishmaniasis is more common than the visceral form. This topic was discussed in the Oct. 13, 2008 issue of Emergency Medicine Reports. The same species of Leishmania can produce more than a single clinical syndrome.
Leishmania are found in most of the world (also in the United States along the Mexican border), except for Australia, the Pacific Islands, and Antarctica. The vast majority of cases are clustered in several regions: 90% of visceral leishmaniasis is in eastern India, Bangladesh, Brazil, Nepal, and Sudan; 90% of mucocutaneous cases are from just Brazil, Bolivia, and Peru; and 90% of the cutaneous cases are from Iran, Iraq, Saudi Arabia, Syria, Afghanistan, Brazil, and Peru.44 Soldiers in Iraq and Afghanistan have frequent encounters, coining the term "Baghdad boil" for cutaneous disease. More than 500 cases in soldiers have been reported between 2002 and 2004. One estimate is that up to 10% of those deployed to the Persian Gulf may have leishmaniasis.45 Cases have been documented to occur via blood transfusion46 or needle sharing in IV drug users.47 Treatment is complicated due to the drugs used and frequent treatment failures; consultation with ID specialists is recommended.
Visceral Leishmaniasis. The visceral form of leishmaniasis is the most severe. It is also called Kala-azar or black fever. Travel to eastern India, Bangladesh, Brazil, Nepal, and Sudan are the main areas of risk. Travelers with HIV/AIDS, though, are at much higher risk (about 100-1000 times higher)48 The average incubation period is 3-8 months, but can be as short as 10 days or more than 1 year.45 Protozoa move to infect the spleen, liver, and bone marrow, and patients develop fevers, anorexia, weight loss, abdominal distension, hepatomegaly, and splenomegaly. Primarily in cases from India, hyperpigmentation develops on the hands, feet, abdomen, and face (hence the term black fever). Bone marrow involvement causes pancytopenia with severe anemia and leukopenia that can prompt opportunistic infections. Without treatment, the majority of cases prove fatal. Diagnosis is difficult and is typically through visualization of parasites (or culture) from bone marrow aspiration or liver or spleen biopsy. Serum antibody ELISA tests also are available. The first choice for treatment is amphotericin B (AmBisome) for 4-6 weeks. Miltefosine is a new oral agent being used with success in India, where resistance is 40%.44
Yellow Fever
In 1900, Dr. Walter Reed first confirmed Carlos Finley's idea that yellow fever was transmitted by mosquitoes. The last outbreak in the United States occurred in 1905 in New Orleans. Currently, yellow fever is still endemic to Africa and South America, with most of the cases in sub-Saharan Africa.
An effective live-attenuated vaccine was developed in 1937 and is still used today. It is effective with side effects of low-grade fevers, malaise, and injection-site reactions seen in fewer than 5% of individuals. The vaccine can be given to older people, but patients older than 70 years of age have a 13-fold increased risk of this complication.49 Yellow fever vaccination is required by many countries for entry by anyone traveling to or with stop-over in Africa and South America. Currently no vaccinations are required to return to the United States.50
Pathology and Symptoms. Yellow fever is caused by an RNA virus of the Flavivirus genus. It is transmitted to humans by mosquitoes. Unfortunately, these mosquitoes are found throughout urban areas in endemic areas, with the highest risk of transmission in the rainy seasons. Once a person is bitten, the virus infects many tissues, including the liver, kidneys, lymph nodes, spleen, and bone marrow.
After a short incubation period of 3-6 days, a variety of symptoms can result, ranging from mild illness to hemorrhagic fever and rapid death. Nearly 90% of infections are mild or asymptomatic, but more serious cases occur in about 15% of patients, in whom mortality can be as high as 50%.51 ICU care can lower this number in many cases to about 5%.52 The first or acute phase of yellow fever begins with fever, headaches, and myalgias. Conjunctival injection, facial flushing, and relative bradycardia with the fever (Faget's sign) are seen.20 Symptoms abate after about 3-4 days but will return in 24 hours to 2-3 days in the 15% who progress to the life-threatening stage or toxic phase of yellow fever.
The toxic phase is characterized by return of high fever, headache, low back pain, abdominal pain, vomiting, and somnolence (or period of intoxication). Jaundice appears as liver damage increases, giving the disease its name. Spontaneous hemorrhage is common with epistaxis, gum bleeding, GI bleeding, and petechiae/purpuric rashes. Renal and hepatic failure (hepatorenal syndrome) is a common cause of death and typically occurs within 7-10 days after symptom onset. Those who do survive take many weeks to fully recover from residual fatigue.20
Diagnosis and Treatment. While there are few clinical findings that suggest yellow fever, the relative bradycardia seen with the high fever (Faget's sign) can be a clue when present. Strict questioning of the patient about the vaccine is important because, when used, the vaccine is very effective and provides protective immunity in 95% of recipients within one week.53
Common laboratory abnormalities include mildly low WBCs or neutrophil counts, signs of coagulopathy (elevated prothrombin times, low fibrinogen levels, low hemoglobin, and thrombocytopenia), rising liver enzymes and bilirubin levels, and elevated serum creatinine. Albuinuria is a consistent finding that helps differentiate yellow fever from other causes of viral hepatitis.20 Ultimately, the diagnosis can be confirmed only by ELISA or PCR tests that detect the presence of the specific virus or by direct culture of the virus. Liver biopsy also can confirm the diagnosis but should be avoided in coagulopathic patients.
Treatment is supportive, as there are no specific treatments for yellow fever. All patients who do not progress to the toxic phase of the illness survive without sequelae. Of the 15% who progress to the toxic phase, 50% mortality is common without treatment. Aggressive fluid resuscitation, ventilation, dialysis, and management of disseminated intravascular coagulation can reduce this number to 5%.
West Nile Encephalitis
West Nile encephalitis was first recognized in 1937. The virus came to the forefront, though, in 1999 when it caused a series of deaths among wild crows and exotic birds.54 Later that year, the virus was isolated from 59 individuals in the eastern United States. By 2002, the disease had spread across the United States to the Pacific.20 Since 1999, there have been 19,525 cases reported in the United States, causing 771 deaths.55 In 2007, cases of West Nile disease were reported from 43 states, with the largest numbers in Colorado (576), California (379), and North Dakota (369). To date, only Washington, Alaska, and Hawaii have not reported cases.
West Nile disease is caused by another species of Flavivirus. It is transmitted to humans by the bite of infected mosquitoes, usually of the Culex species. The virus exists in the wild, primarily in bird populations (especially crows, jays, and sparrows), but also can infect other mammals (cats, dogs, horses, etc.). The virus also can be transmitted by infected blood products or by organ donation.
The majority of infections in humans are asymptomatic, with only 20-40% of people developing clinical symptoms.55 When symptoms do occur, they are seen after a 2- to 14-day incubation. Most people develop flu-like disease with fever, cough, sore throat, headache, myalgias, vomiting, and diarrhea that lasts 3-6 days. Fewer than 1% of infected people progress to severe neuroinvasive disease.55 Besides neurologic complications, myocarditis, hepatitis, and pancreatitis also have been reported.20
Those who progress to neurologic disease develop a flu-like prodrome lasting 1-7 days. They seem to improve before relapsing with more severe symptoms. When neurologic disease develops, it is clinically indistinguishable from other forms of viral encephalitis. Patients can have signs of meningeal irritation (headache, stiff neck, photophobia) or develop delirium and focal neurologic signs (dysarthria, tremor, seizures, ataxia). Encephalitis (63% of cases) is more common than meningitis (29%), and some patients develop only headache (8%).54 Case fatality rates range from 4-14%, with most deaths in the United States occurring in older patients.20
West Nile also is known to cause a flaccid paralysis that can follow 1-2 weeks after the flu-like illness.55 The onset is rapid, with peak weakness occurring in only a few hours. Minimal or no sensory changes are noted, and bowel/bladder function is affected only in a minority of patients.56 Unfortunately, recovery from flaccid paralysis is highly variable, with some studies reporting only 30% recovering the ability to walk after one year, while in other cases some patients recovered completely in a matter of weeks.55
Currently, no tests can identify West Nile infection while patients are still in the ED. ELISA tests for serum IgM antibodies are required.
Trypanosomiasis
Trypanosomiasis is caused by infection with protozoa of the Trypan-osoma genus, and there are two distinct disease forms in humans. American trypanosomiasis (Chagas' disease) is caused by Trypanosoma cruzi, and African trypanosomiasis (sleeping sickness) is caused by Trypanosoma brucei.
African Trypanosomiasis (Sleeping Sickness). The West African version has a more delayed onset, with symptoms developing months to a year after the first bite. After symptoms develop in West African disease, it proceeds similarly to the East African version.
East African trypanosomiasis follows a more acute course. A painless inflammatory lesion at the bite site develops about a week after the bite. Even without treatment, this usually resolves spontaneously over a few weeks. About 3 weeks after being bitten, intermittent fevers, myalgias, general lymphadenopathy, rash, and headache occur. Like Chagas' disease, the heart can be involved at this stage with pericarditis and conduction disease.
Stage II develops when the parasites reach the central nervous system and may progress rapidly or may take months to years to develop. Persistent headaches, with behavioral changes, weight loss, and the characteristic disruption of sleep patterns (daytime somnolence, nighttime insomnia) the disease is named after are seen. Movement disorders, such as choreiform movements, fasciculations, ataxia, slurred speech, and tremors can prompt misdiagnosis with Parkinson's disease. Ultimately, the disease is fatal if untreated.
Definitive diagnosis relies on direct detection of the parasite in blood or tissue serologic antibody testing. Treatment is obtained through the CDC.
American Trypanosomiasis (Chagas' Disease). American trypanosomiasis, or Chagas' disease, is transmitted to humans from the bite of the kissing bug. People can become infected when the kissing bug bites, obtains a blood meal, and then defecates nearby. The protozoans are in high concentration in the bug's feces, and are then rubbed into the nearby, itching wound. Pregnant women can transmit the disease to their fetuses. Muscle tissue is most affected by T. cruzi infection, especially cardiac muscle, resulting in dilated cardiomyopathy as well as conduction system abnormalities. Apical aneurysms and mural thrombi are common. The esophagus and colon are the other tissues most commonly affected. Only 10-30% of people with chronic infection will become symptomatic.57
Two distinct clinical syndromes result from infection: the acute and chronic disease. Incubation periods are about 7-10 days and last between 3-8 weeks without treatment. Symptoms consist of localized swelling (chagoma) at the site of the insect bite, with localized lymphadenopathy. Systemic symptoms of fever, malaise, anorexia, and facial/extremity edema can be seen. Rarely, patients will develop meninngoencephalitis or congestive heart failure in the acute infection, and fatalities can result. Chronic Chagas' disease can take years to decades to produce symptoms, which consist of right heart failure, arrhythmias, or achlasia (megaesophagus).
Diagnosis of acute infection can be made by observing a fresh drop of anticoagulated blood at 400 x magnification for moving T. cruzi. When blood smears are not diagnostic, culture may be needed to prove infection, but this can take several weeks. PCR tests are useful to diagnose acute/chronic infections but are not sensitive enough for screening of donated blood.58 Treatment is only available through the CDC.57
Viral Hemorrhagic Fevers (Ebola, Lassa, Marburg)
At least 18 different species of viruses are known to cause hemorrhagic fever syndromes: arenaviruses (Lassa, Bolivian, Argentine, Brazilian hemorrhagic fevers), bunyaviruses (Hantavirus, Congo-Crimean hemorrhagic fever, Rift Valley fever), filoviruses (Ebola and Marburg fevers), and Flaviviruses (Dengue and yellow fever). All are RNA viruses in which humans are not the natural host, they cause human disease in sporadic outbreaks, and, with few exceptions, they have no specific treatment. Some of these viruses (Ebola, Lassa, Marburg) are so contagious they are classed as Category A bioterrorism agents comparable to anthrax and smallpox.59 This section will focus on the Ebola, Lassa, and Marburg fevers.
Ebola. Ebola fever is highly fatal in humans, with case fatality rates of 40-89%, and so far has been limited to infrequent outbreaks in sub-Saharan Africa (Congo, Gabon, Sudan, and Uganda). Very little is known about the virus, and the natural animal reservoir remains unidentified. Epidemics appear mysteriously, rapidly infect humans (and primates), and their spread is only limited by their deadly clinical course.
Ebola virus is highly infectious, and human-to-human aerosol spread through close contact with the infected patient or with their body fluids is well documented in those caring for these patients.60 Even persons preparing the dead for burial have become infected, presumably just by touching of the skin.60 Recent work shows that a few human Ebola infections remain asymptomatic, possibly due to a strong early immune response in these lucky individuals.61 After exposure to the virus, incubation periods range from only 5-10 days. Early symptoms include abrupt onset of fever, myalgias, headache, abdominal pain, diarrhea, and pharyngitis with herpetic lesions. Later, patients develop a maculopapular rash that evolves into petechiae with spontaneous bleeding from all mucosal sites and venipuncture sites. Severe conjunctival injection and bleeding also are seen.62 Most patients die from cardiovascular collapse and metabolic acidosis, but some improve markedly in the second week. Culture of the virus is diagnostic but must be performed in a level 4 biosafety facility. Otherwise, PCR detection of viral antigen or ELISA detection of antibodies is used. Currently there are no recommended antiviral drugs used to treat Ebola infections; treatment is strictly supportive.
Marburg Fever. This virus is closely related to the Ebola virus. Bats are the implicated natural reservoir, but so far no live virus from animals has been isolated.60 Clinically, Marburg fever is very similar to Ebola, although mortality in some outbreaks has been lower (25-30%).62 As with Ebola, there is no specific treatment, and the virus is highly contagious.
Lassa Fever. Lassa fever epidemics have been reported in West Africa (Nigeria, Liberia, Sierra Leone, and Guinea) with case fatality rates up to 50%. A case of Lassa fever was seen in New Jersey in 2004. It is estimated that between 100,000 and 300,000 cases (5,000 deaths) occur every year in West Africa. Only 1 in 20 infections leads to significant symptoms/hospitalization.63
The natural reservoir for Lassa virus is a small rodent. The exact mechanism of transfer to humans is unclear but likely occurs from contact, ingestion, or aerosol spread from the rat's urine or stool or by handling the rodents as they are prepared for food.64 Infected people are highly contagious, and contact with their body fluids, tissue, or even intact skin can transmit the virus.
Symptoms begin after 7-12 days, and a variety of symptoms can be seen. In order of decreasing frequency, they include retrosternal chest pain (perhaps from pericarditis), sore throat, back pain, cough, abdominal pain, vomiting, diarrhea, conjunctivitis, and facial edema.65 Mucosal bleeding is seen less often than with Ebola or Marburg. In some patients, encephalitis and/or meningitis complications occur. Diagnosis can be suggested by elevated serum aspartate transaminase (AST) levels, but serologic ELISA confirmation is required for accurate diagnosis. Patients who die from infection do so in the second week from cardiovascular collapse and pulmonary edema.
Ribavirin has been shown to be an effective treatment.66 Ribavirin now is recommended as effective for chemoprophylaxis in those exposed to infected patients.
Cholera
Cholera is a contagious diarrheal illness capable of causing epidemics and pandemics.67 Regional epidemics occur yearly. The overwhelming majority (66%) of recent cases are from Africa, followed by Southeast Asia (17%).68 Although cholera is primarily a disease of poor sanitation, developed countries such as the United States, Canada, and Australia have reported endemic cases.69 The U.S. cases were associated with raw seafood on the Gulf Coast, and cases were reported in association with hurricanes Katrina and Rita.70
Symptoms begin after a 24- to 48-hour incubation period, and the watery diarrhea ("rice water stools") can produce mild to life-threatening dehydration. Stool volume has been measured at > 250 mL/kg in the first 24 hours.67 Vomiting can be present, complicating oral rehydration attempts. Clinical symptoms will vary with degree of dehydration. If untreated, cholera can cause death in 1-3 days from dehydration with a reported mortality of 50%.67
Diagnosis is by visualization of mobile V. cholerae in the stool, by culture, or by PCR testing. Treatment centers on rehydration, with restoration of fluid losses in the first 2-4 hours followed by maintenance until diarrhea slows. Fluid requirements in the restoration phase can be extreme, up to 100 mL/kg/hr (7 liters for 70-kg person).67 Antibiotics are optional but do shorten the course of illness and limit fluid losses. Tetracycline resistance is spreading, and resistance to ciprofloxacin also has been reported in India. Erythromycin and azithromycin have been used with success.
References
1. Suh KN, Kain KC, Keystone JS. Malaria. CMAJ 2004;25:693-702.
2. Isturiz RE, Torres J, Besso J. Global distribution of infectious diseases requiring intensive care. Crit Care Clin 2006;22:469-488.
3. Millet JP, Garcia de Ollalla P, Carrillo-Santisteve P, et al. Imported malaria in a cosmopolitan European city: A mirror image of the world epidemiological situation. Malar J 2008;7:56.
4. Zucker JR, Campbell CC. Malaria: Principles of prevention and treatment. Infect Dis Clin North Am 1993;7:547-567.
5. Stanley J. Malaria. Emerg Med Clin North Am 1997;15:113-155.
6. Centers for Disease Control: Summary of Notifiable Diseases, U.S. MMWR Morb Mortal Wkly Rep 1997; Nov 1998;46:1-87.
7. Trampuz A, Jereb M, Muziovic I, et al. Clinical review: Severe malaria. Crit Care 2003;7:315-323.
8. Schwartz IK. Prevention of malaria. Infect Dis Clin North Am 1992;6:313-331.
9. White NJ, Ho M. The pathophysiology of malaria. Adv Parasitol 1992;31:34-173.
10. Kortepeter M, Brown JD. A review of 79 patients with malaria seen at a military hospital in Hawaii from 1979-1995. Mil Med 1998;163:84-89.
11. World Health Organization. Severe falciparum malaria. Trans R Soc Trop Med Hyg 2000;94:1-90.
12. White NJ. Malaria. In: Cook GC, ed. Manson's Tropical Diseases, 2nd ed. Philadelphia; WB Saunders; 1996:1087-1164.
13. Taylor TE, Wills B, Kazembre P, et al. Rapid coma resolution with artemether in Malawian children with cerebral malaria. Lancet 1993;341:661-662.
14. Warrell DA, Molyneaux ME, Beales PF. Severe and complicated malaria. Trans R Soc Trop Med Hyg 1990;84:1-65.
15. Connor MP, Green AD. Runway malaria in a British serviceman. J R Soc Med 1995;88:415P-416P.
16. Centers for Disease Control: Appendix Microscopic procedures for diagnosing malaria. MMWR Morb Mortal Wkly Rep 1997;46(SS-2):46-47.
17. McLellan SLF. Evaluation of fever in the returned traveler. Prim Care Clin Office Pract 2002;29:947-969.
18. Hexdall AH, Chiang WK. Malaria deaths following inappropriate malaria chemoprophylaxisUnited States, 2001. Ann Emerg Med 2002;39:86-88.
19. Pincus LB, Grossman ME, Fox LP. The examthem of dengue fever: Clinical features of two U.S. tourists traveling abroad. Am J Acad Dermatol 2008;58:308-316.
20. Tsai TF, Vaughn DW, Solomon T. Flaviviruses (Yellow Fever, Dengue, Dengue Hemorrhagic Fever, Japanese Encephalitis, West Nile Encephalitis, St. Louis Encephalitis, Tick-Borne Encephalitis). In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:1926-1945.
21. Centers for Disease Control: Dengue hemorrhagic feverU.S.-Mexico border, 2005. MMWR Morb Mortal Wkly Rep 2007;56:785-789.
22. Halstead SB, Streit TG, Lafontant JG, et al. Absence of dengue hemorrhagic fever despite hyperendemic dengue virus transmission. Am J Trop Med Hyg 2001;65:180-183.
23. Singhi S, Jayashree M. Dengue shock syndrome: At the heart of the issue. Pediatr Crit Care Med 2007;8:583-584.
24. Khongphatthanayothin A, Lertsapcharoen P, Supachokchaiwattana P, et al. Myocardial depression in dengue hemorrhagic fever: Prevalence and clinical description. Pediatr Crit Care Med 2007;8:524-529.
25. Kalayanarooj S, Vaughn DW, Nimmannitya S, et al. Early clinical and laboratory indicators of acute dengue illness. J Infect Dis 1997;176:313-321.
26. Raoult D, Walker DH. Rickettsia prowazekii (Epidemic or Louse-Borne Typhus). In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:2305-2309.
27. Duma RJ, Sonenshine DE, Bozeman FM, et al. Epidemic typhus in the United States associated with flying squirrels. JAMA 1981;245:2318-2323.
28. Ng CP, Lo CB, Wong KK, et al. A returned traveler with persistent fever due to murine typhus. Hong Kong Med J 2002;8:457-459.
29. Raoult D. Scrub typhus. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:2309-2310.
30. Williams JC, Thompson HA. Q fever: The biology of Coxiella burnetii. CRC Press; 1991: 2.
31. Botelho-Nevers E, Raoult D. Fever of unknown origin due to rickettsioses. Infect Dis Clin North Am 2007;21:997-1011.
32. Fouts-Flicek B. Rickettsial and other tick-borne infections. Crit Care Nurs Clin North Am 2007;19:27-38.
33. Maguire JH. Trematodes: Schistosomes and other flukes. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:3276-3284.
34. Firmo JO, Lima Coast MG, Guerra HL, et al. Urban schistosomiasis: Morbidity, sociodemographic characteristics and water contact patterns predictive of infection. Int J Epidemiol 1996;25:1292-1300.
35. Ross AG, Bartley PB, Sleigh AC, et al. Schistosomiasis. N Engl J Med 2002;346:1212-1220.
36. Neal PM. Schistosomiasisan unusual case of ureteral obstruction. Clin Med Res 2004;2:216-227.
37. Ross AG, Vickers D, Olds, GR, et al. Katayama syndrome. Lancet Infect Dis 2007;7:218-224.
38. Thielman NM, Guerrant RL. Enteric fever and other causes of abdominal symptoms with fever. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:1273-1279.
39. Mermin JH, Townes JM, Gerber M, et al: Typhoid fever in the United States, 1985-1994: Changing risks of international travel and increasing antibiotic resistance. Arch Intern Med 1998;158:633-638.
40. Connor BA, Schwartz E. Typhoid and paratyphoid fevers in travellers. Lancet Infect Dis 2005;5:623-628.
41. Zuckerman MJ, Meza AD, Ho H, et al. Lower gastrointestinal bleed in a patient with typhoid fever. Am J Gastroenterol 2000;95:843-845.
42. Szakacs TA, McCarthy AE. Teaching case report: An all-inclusive vacation. CMAJ 2007;177:29-31.
43. Rowe B, Ward LR, Threlfall EJ. Multidrug-resistant Salmonella typhi: A worldwide epidemic. Clin Infect Dis 1997;24:S106-S109.
44. Jeronimo SMB, Sousa AQ, Pearson RD. Leishmania species: Visceral, cutaneous and mucocutaneous leishmaniasis. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:3145-3154.
45. Pehoushek JF, Quinn DM, Crum WP. Cutaneous leishmaniasis in soldiers returning from deployment to Iraq. J Am Acad Dermatol 2004;51:S197-S200.
46. Cardo LJ. Leishmania: Risk to the blood supply. Transfusion 2006;46:1641-1645.
47. Cruz I, Morales MA, Noguer I, et al. Leishmania in discarded syringes from intravenous drug users. Lancet 2002;359:1124-1125.
48. Desieux P, Alvar J. Leishmania/HIV coinfection: Epidemiology in Europe. Ann Trop Med Parasitol 2002;97:S3-S15.
49. Monath TP, Cetron MS, McCarthy K, et al. Yellow fever 17D vaccine safety and immunogenicity in the elderly. Hum Vaccin 2005:1:207-214.
50. Freedman DO. Protection of travelers. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:3638-3646.
51. Vasconcelos PF. [Yellow fever.] Rev Soc Bras Med Trop 2003;36:275-293.
52. McFarland JM, Baddour LM, Nelson JE, et al. Imported yellow fever in a U.S. citizen. Clin Infect Dis 1997;95:1142-1147.
53. Freeman DO. Infections in returning travelers. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:3647-3655.
54. Saks MA, Karras D. Emergency medicine and the public's health: Emerging infectious diseases. Emerg Med Clin North Am 2006;24:1019-1033.
55. Kramer LD, Li J Shi PY. West Nile virus. Lancet Neurol 2007;6:171-181.
56. Leis AA, Stokic DS, Polk JL, et al. A poliomyelitis-like syndrome from West Nile virus infection. N Engl J Med 2002;347: 1279-1280.
57. Kirchhoff LV. American trypanosomiasis (Chagas' disease). In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005: 3157-3158.
58. Piron M, Risa R, Casamitjana N, et al. Development of a real-time PCR assay for T. cruzi detecting in blood samples. Acta Trop 2007;103:195-200.
59. Pigott DC. Hemorrhagic fever viruses. Crit Care Clin 2005;21:765-783.
60. Marburg and ebola virus hemorrhagic fevers. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:2056-2059.
61. Leroy FM, Baize S, Volchkov VE, et al. Human symptomatic Ebola infection and strong inflammatory response. Lancet 2000;355:2210-2215.
62. Cleri DJ, Ricketti AJ, Porwancher RB. Viral hemorrhagic fevers: Current status of endemic disease and strategies for control. Infect Dis Clin N Am 2006;20:359-393.
63. McCormick JB, Webb PA, Krebs JW, et al. A prospective study of the epidemiology and ecology of Lassa fever. J Infect Dis 1987;155:437-444.
64. Peters CJ. Lymphocytic chroiomeningitis virus, Lassa virus and the South American hemorrhagic fevers. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005: 2090-2096.
65. McCormick JB, King IJ, Webb PA, et al. A case-control study of the clinical diagnosis and course of Lassa fever. J Infect Dis 1987;155:445-455.
66. McCormick JB, King IB, Webb PA, et al. Lassa fever: Effective therapy with ribavirin. N Engl J Med 1986;314:20-26.
67. Seas C, Gotuzzo E. Vibrio cholerae. In: Mandell GM, Bennett JE, Dolin R, ed. Principles and Practice of Infectious Diseases, 6th ed. New York: Churchill Livingstone; 2005:2536-2542.
68. Griffith DC, Kelly-Hope LA, Miller MA. Review of reported cholera outbreaks worldwide: 1995-2005. Am J Trop Med Hyg 2006;75:973-977.
69. Weber JT, Levine WC, Hopkins DP, et al. Cholera in the United States, 1965-1991. Risks at home and abroad. Arch Intern Med 1994;154:551-556.
70. Centers for Disease Control: Two cases of toxigenic V. cholerae O1 infection after hurricanes Katrina and RitaLouisiana, Oct 2005; MMWR Morb Mort Wkly Rep 2006;55:31.
Recently, we published a series of articles on the international traveler in the ED, emphasizing the risk factors and geography. This article provides a more standard review of some of the diseases seen in travelers to tropical areas and also some, like West Nile, that have migrated into temperate climates. This article will complete our series.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
