Pediatric Rapid Sequence Intubation
Pediatric Rapid Sequence Intubation
Authors: Jennifer E. Tucker, MD, FAAP, Assistant Professor of Emergency Medicine and Pediatrics, Medical College of Georgia, Augusta; Michael Coussa, MD, Resident Physician, Emergency Medicine Residency, Medical College of Georgia, Augusta.
Peer Reviewer: Robert A. Felter, MD, FAAP, PE, FACPE, Medical Director, Pediatric Emergency Medicine and Inpatient Services, Inova Loudon Hospital; Professor of Clinical Pediatrics, Georgetown University School of Medicine, Washington, DC.
Emergency physicians perform many lifesaving procedures every day; however, none is more important than effective airway management. The loss of an airway is one of the most common causes of morbidity and mortality in severely ill or injured children. It may occur suddenly, without any warning, and requires immediate action; therefore, all emergency medicine physicians must possess knowledge of the management of an airway using rapid sequence intubation (RSI) and alternative techniques. One must know the differences between the adult and pediatric airways, the indications for RSI, the characteristics of a difficult airway, the different options for sedation and muscle relaxation, the mechanisms for confirming proper airway placement, and the alternatives available when RSI is unsuccessful.
The Editor
Introduction
The purpose of RSI is to effect a state of unconsciousness and neuromuscular blockade, allowing for endotracheal intubation without positive pressure ventilation (PPV).1 The avoidance of interval PPV is of particular importance in an emergency situation, since most patients are presumed to have full stomachs, thus increasing the risk of aspiration of gastric contents. Additionally, a goal of RSI is to blunt the body's physiologic responses to laryngoscopy; therefore, adjunctive pharmacologic agents may be indicated for the prevention of bradycardia and intracranial pressure (ICP) elevation. About half of RSIs are performed for traumatic events, while the other half are performed for medical emergencies such as seizures, depressed mental status (ingestions), and asthma.2 Twenty-five percent of all RSIs are secondary to head injury.3 Older children and trauma patients are more likely to be intubated with RSI compared to younger children and patients presenting with medical illnesses.4 In one study, physicians used paralytics less than 40% of the time in children under the age of 1 year.2
According to the American Heart Association's Pediatric Advanced Life Support (PALS) provider manual, the indications for RSI include inadequate central nervous system control of ventilation resulting in hypoventilation; functional or anatomic airway obstruction; the loss of protective airway reflexes like the cough or gag reflex; and excessive work of breathing with concern for fatigue and subsequent respiratory failure. RSI is also indicated when a stable airway with controlled ventilation is required in the setting of closed head injuries, sedation for diagnostic studies, and in preparation for patient transport.5 RSI has been found to be faster and safer than other forms of intubation in adults and children which do not include pre-intubation paralysis.6 Conversely, RSI is not indicated in the setting of cardiac arrest, as patients undergoing cardiopulmonary resuscitation (CPR) do not require sedation or muscle relaxation. Additionally, RSI may not be the best option in those patients who are breathing spontaneously with adequate ventilation; those with major facial or laryngeal trauma; patients with upper airway obstruction; those with distorted facial or airway anatomy; patients in whom IV access is contraindicated (e.g., patients with epiglottitis); and in those patients in whom there is concern that mask ventilation or intubation may be unsuccessful.7
RSI involves six distinct steps for successful airway management and endotracheal tube placement ("the 6 Ps"). Each step is of equal importance and none can be omitted. The steps are:
Preparation
Preoxygenation
Pretreatment
Paralysis with induction
Placement of the tube, and
Post-intubation management.
Preparation
The integral first step in preparation involves understanding the anatomic and physiologic differences between the pediatric and adult patient. Anatomically, the pediatric patient has a more prominent occiput which flexes the neck when the patient is supine, resulting in mechanical airway obstruction. This obstruction may be relieved by placing a towel roll underneath the child at the level of the shoulders or by having a second person gently lift the child by the shoulders, causing gentle neck extension. The chin-lift or jaw-thrust maneuver can also be utilized in relieving airway obstruction. The chin lift involves placing one hand on the forehead to tilt the head back and the other hand's fingers on the bony part of the chin, forcing the mandible upward and outward, thereby lifting the tongue from the back of the throat. The jaw thrust involves using one's thumbs to push the posterior aspects of the mandible upward, pulling the tongue forward and preventing it from occluding the airway. The jaw thrust is the maneuver of choice in the patient with suspected cervical spine (c-spine) injury.
Other anatomic differences between children and adults include the relatively larger pediatric tongue and smaller nares, both of which increase airflow resistance. To decrease this resistance, the emergency medicine provider must always remember to bag the patient by maintaining contact with the mask and the bony structures of the face, while avoiding contact with the soft tissues. Yet another significant anatomical difference is that the pediatric larynx is higher and more anterior than that in an adult, causing the visualization of the cords to be more difficult. Other anatomic discrepancies that deserve mention exist in the trachea; the pediatric trachea is short, increasing the risk of bronchial intubation. Also, the narrowest part of the pediatric trachea is the cricoid cartilage, whereas the vocal cords represent the narrowest portion in an adult. When all of the aforementioned anatomic differences are combined with the fact that the pediatric epiglottis is longer, softer, and higher than that in adults, it becomes clear that RSI can be substantially more technically difficult in children compared with adults.8
In terms of physiologic differences, the child experiences arterial oxygen desaturation more rapidly than the adult.8 At rest, infants and children may consume two to three times more oxygen per kilogram of body weight than adults as a result of higher basal metabolic rate.9 They also have a diminished total lung capacity when anesthetized or paralyzed, increased respiratory rate, and a decreased lung elastic coil.10,11 The infant or child's increased consumption of oxygen and decreased oxygen reservoir cause a more rapid rate of oxygen desaturation in the setting of hypoxemia, a phenomenon which is exacerbated by pre-existing lung disease.8
After mastering the anatomic and physiologic differences between children and adults, the next step in preparation includes a good history and physical examination, to help determine medication choices and to assess for a potential difficult airway. The mnemonic "AMPLE" can be used to elicit a history:
Allergies,
Medications,
Past medical history,
Last meal, and
Events leading up to the intubation.
Important information from the past medical history includes personal or family history of malignant hyperthermia, muscular dystrophy, or renal failure. Knowledge of the approximate time of the patient's last meal may predict risk of aspiration during RSI. The events preceding the intubation will help determine if head trauma protection and/or c-spine immobilization will be required.
After the AMPLE history is obtained, the physical exam should first focus on an overall inspection of the child, looking for any obvious signs of a difficult airway for example, facial trauma. The examination of the mouth and oropharynx provides further information about the difficulty of the intubation. A small oral opening with a large tongue may predict a difficult airway due to insufficient space for accommodating all intubation equipment and obstruction from the tongue during bag mask ventilation. A short mandible and neck, large front teeth, obesity, laryngeal edema, foreign body, and c-spine immobility are also predictors of a difficult airway. Improper alignment of the oral, pharyngeal, and laryngeal axes secondary to a short mandible and neck causes the larynx to be anterior and high-riding, yielding a very difficult intubation.
The Mallampati classification may be used to predict the difficulty of airway access by visualization of the oropharynx. The degree of difficulty progresses through four classes ranging from Class I, with complete visualization of the uvula, tonsils, and soft palate, to class IV, with visualization of the hard palate only. (See Figure 1.) Other predictors of the difficult airway include short thyromental distance (distance from the thyroid cartilage to the chin) and limited neck mobility (evaluation of maximal atlanto-occipital extension). Levitan, et al, however, showed in their retrospective study that Mallampati scores, thyromental distance, and neck mobility are difficult to assess in pediatric patients because many children are unable to follow simple commands, and frequent c-spine immobilization precludes assessment of neck mobility.12
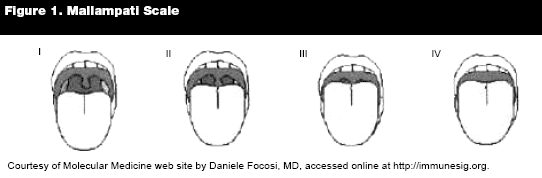 |
The final step in preparation involves assembling the supplies that may be needed for RSI. Necessary equipment includes a suction device, oxygen via a non-rebreather mask, IV access, a functioning laryngoscope with blades, a bag-valve-mask with a self-inflating bag, a secondary confirmation device such as an end tidal-CO2 detector, endotracheal tubes (ETT) with stylet, a 10 mL syringe, intubation medications, and an alternate airway device. In ideal circumstances, personnel should include a minimum of three persons with each having an assigned role. The first person is at the head of the bed, responsible for managing the airway. The second person administers medications, and a third person performs the Sellick maneuver (applying backwards pressure to the cricoid cartilage, compressing the esophagus against the underlying vertebral body) and monitors vital signs. The intubation team should obtain IV access and place the patient on a monitor recording heart rate, blood pressure, respiratory rate, and oxygen saturation.
When selecting a suction catheter, a good rule of thumb is that the diameter of the catheter is two times that of the ETT size. (More on selecting ETTs below.) It is also prudent to have the larger Yankauer suction available for clearing blood and secretions from the mouth. Laryngoscopes can be assembled with either one of two different types of blades. The Miller, or straight blade, is used by placing the tip of the blade to lift the epiglottis and then view the glottic opening. The Macintosh, or curved blade, is used by placing the tip of the blade into the vallecula to displace the tongue anteriorly; then traction is exerted upward to move the base of the tongue and epiglottis anteriorly, exposing the glottic opening. In younger children, because of the anatomical differences mentioned above and the increased angle of the epiglottis with the trachea, the Macintosh is often used like the Miller, for lifting the epiglottis up; placing it in the vallecula of young children may not allow adequate visualization of the cords. Always make sure to check the blade for a properly functioning light bulb prior to attempting any intubation. There is no rule for determining blade size; however, there are some recommendations for blade selection provided by the PALS manual.5 It is always important to have two separate blade sizes available in the event that the child is small or large for age or that the initial blade choice does not allow for maximum visualization.
The internal diameter of the ETT in millimeters can be estimated in children under the age of 12 years using one of two different equations:
[16 + (age in years)] / 4, or
(age in years /4) + 4.
A rough estimate may also be the approximate diameter of the patient's pinkie finger; however, this rule may be difficult and unreliable. In premature babies, gestational age divided by 10 roughly estimates ETT size (so 25 weeks equals a 2.5mm tube). Historically, physicians opted for uncuffed tubes in children younger than of 8 years of age, because the narrowest portion of the airway in this age group is the subglottic cricoid cartilage. This anatomic narrowing forms an adequate seal with the uncuffed endotracheal tube; therefore, there was concern that cuffed tubes would cause subglottic injury with inflation. New PALS guidelines (2007), however, recommend a cuffed tube in all age groups except neonates. The formula to estimate the size of a cuffed tube is (age in years/4) + 3. A cuffed tube is particularly useful in patients with poor lung compliance, increased airway resistance, or a large glottic air leak.
A good estimate for determining ETT depth after intubation in patients greater than 44 weeks gestational age is 3 times the endotracheal tube size. For those under 44 weeks gestational age, ETT depth should be approximately 6 cm plus the patient's weight in kilograms. These determinations are only estimates, however, and all tube placements must be verified by secondary confirmation devices, imaging, and the physical exam.
Preoxygenation
When the decision to intubate is made, 100% oxygen should be administered immediately via either a snug-fitting non-rebreather mask or a bag-valve-mask. There should be no need to bag the patient who is spontaneously breathing, as bagging will introduce air into the stomach, causing gastric distention. After two minutes, 95% of the lung's nitrogen will be replaced by oxygen, increasing the oxygen reservoir for continued oxygenation of the blood. This "nitrogen washout" provides just 3–4 additional minutes of apnea before a pediatric patient with normal functional residual capacity becomes hypoxemic; adults, however, may remain well saturated after up to 5–6 minutes of apnea.1,13,14 This discrepancy may be attributed primarily to the higher basal oxygen use per kilogram of weight in the pediatric patient. It is important to remember that preoxygenation helps prevent hypoxemia, but it has no impact on the level of carbon dioxide in the blood. Once apnea begins, hypercarbia begins to develop immediately, a fact which needs to be kept in mind especially in the head-injured patient in whom hypercarbia can be profoundly detrimental.
Premedication
The premedication aspect of RSI involves administering drugs that may blunt the negative physiologic responses and autonomic reflexes that result from various components of RSI. Premedication is of particular importance in children, as they exhibit exaggerated vagal responses to hypoxia, succinylcholine administration, laryngoscope introduction, and passage of the endotracheal tube through the cords.2,7,15 The use of anticholinergics, opioids, lidocaine and non-depolarizing neuromuscular blocking agents can help to dampen these responses. An anticholinergic agent like atropine may offset the bradycardia that young children can experience in response to the aforementioned stimuli. According to the American College of Emergency Physicians, the American Academy of Pediatrics, and the American Heart Association, atropine is a recommended premedication agent in RSI in all children under 1 year of age; children between 1 and 5 years of age who receive succinylcholine; adolescents and adults who receive more than one dose of succinylcholine; and anyone with bradycardia immediately preceding the intubation.16 The recommended dose administered 1–2 minutes prior to intubation is 0.01–0.02 milligrams per kilogram IV or IO with a minimum dose of 0.1mg to prevent reflex bradycardia, and a maximum dose of 1 mg. A retrospective cohort study performed by Fastle et al, however, determined that some patients will experience bradycardia despite atropine administration and that hypoxia is a stronger trigger for bradycardia than intubation or succinylcholine administration.16
Historically, laryngeal stimulation and endotracheal intubation have been thought to cause a transient but significant increase in ICP.17 Patients who have a head injury requiring airway management may suffer harmful effects from RSI because of this increase in ICP and consequent decrease in cerebral perfusion pressure (CPP = MAP – ICP). Lidocaine is a class IB antiarrythmic agent that has been used to thwart the possible increase in ICP from RSI. The exact mechanism for lidocaine's impact on ICP is unclear; however, some proposed theories are that it suppresses the cough reflex, depresses the brainstem, decreases cerebral metabolic rate, and stabilizes cell membranes.2 Lidocaine may also have a topical anesthetic effect that can attenuate the ICP rise in response to RSI; however, direct tracheal placement of lidocaine has been shown to have variable effects on ICP.7,8,18,19 Despite its widespread use, there has been no study examining the impact of lidocaine pre-treatment on neurologic outcome after RSI for acute head injury; however the National Emergency Airway Course continues to recommend its use for pretreatment in the setting of RSI of the head-injured patient.17,18
Opioids are used in conjunction with other induction drugs for amnesia and analgesia. They produce sedation and decrease the response to noxious airway stimuli by binding to receptors in key regulatory centers throughout the body, blunting increases in heart rate and blood pressure.7,20 The most common opioid used is fentanyl because of its rapid inhibitory effect on the sympathetic system, with rapid of action in 1–2 minutes. Fentanyl, like lidocaine, is most commonly administered during the premedication phase to prevent an increase in ICP from RSI. The impact of fentanyl administration on ICP was thought to be secondary to its sympatholytic action, but recent studies have rendered conflicting data on fentanyl's impact on ICP.21-22 The typical fentanyl sedation dose is 1–3 micrograms per kilogram given 1–3 minutes before laryngoscopy; however, sympathetic blockade requires higher doses, 5–7 micrograms per kilogram, a dose that may result in hypotension.7
The last drug that can be given during the premedication stage is a small dose (1/10th of the paralyzing dose) of a non-depolarizing neuromuscular blocking agent (discussed in detail later), which serves to decrease the fasciculations seen with succinylcholine administration. It is thought that muscle fasciculations may potentiate increases in ICP; however, it is unclear if it is the fasciculations or the drug itself that causes the increased ICP. Defasciculation is controversial in pediatrics and is only recommended in children older than age 5 years, because younger children have insufficient muscle mass to generate detrimental fasciculations.7 In the younger child, the combination of a non-depolarizing agent with succinylcholine may also induce severe bradycardia or asystole.23
Paralysis with Induction
After the administration of premedication drugs, the clinician should wait three minutes before administering an induction agent. Induction medication prevents the patient from having any memory of the event and assists in neuromuscular blockade, thus increasing the chance of a successful intubation. Ideally, induction agents should both induce unconsciousness rapidly and render return to consciousness quickly; if possible, an induction agent should be administered 2–3 minutes prior to paralytic administration to ensure adequate sedation but should be given no later than just prior to the neuromuscular blocking agent of choice.7 Certain situations may call for a specific agent, so a physician must be familiar with the indications and side effect profiles of all the available medication. (Table 1.)
 |
Etomidate is the most commonly used induction agent today, employed in greater than 40% of all RSIs.2,7 It is a nonbarbiturate, substituted imidazole hypnotic agent similar to the antifungal agent ketoconazole.1,25,26 It has been widely accepted by emergency medicine physicians for adult RSI; however, the manufacturer does not recommend its use in children under age10 years because of insufficient data for this age group.24 Recent studies have shown that etomidate is a highly safe and favorable induction agent for RSI in children, especially for the critically ill patient with potential circulatory or central nervous system disease.8,25,26,28 The dose of etomidate is 0.2–0.3 mg/kg IV given over 30–60 seconds, resulting in hypnosis in 5–15 seconds with a short duration of action of just 5–14 minutes.25 Etomidate exerts minimal cardiovascular and respiratory effects and actually decreases cerebral metabolic rate, making it the ideal drug in the setting of trauma with or without concurrent head injury.
Potential pitfalls of etomidate include myoclonus, vomiting, and pain with administration; etomidate also provides no analgesic effects.26 In patients with pre-existing seizure disorders, etomidate may precipitate seizure activity. Prior studies have shown that clinically significant adrenocortical suppression occurs after multiple doses of or a continuous infusion of etomidate, but there is not a clinically significant release of cortisol after a single dose.27 Etomidate provides effective intubating conditions with minimal side effects, but it should be used with caution in patients at risk for adrenocortical suppression or seizure.25-28
Thiopental, a short-acting barbiturate, is the second most common induction agent used in RSI.2 After a standard dose of 2–5 mg/kg IV, the onset of action is 30 seconds and the duration of action is 5–10 minutes.2 It is extremely lipid-soluble, enabling it to cross the blood-brain barrier easily, thus producing rapid central nervous system (CNS) depression, decreased ICP, and decreased cerebral metabolic and oxygen demands. Indications for thiopental, then, should include head-injured patients with normal blood pressure, patients with increased ICP secondary to CNS infection, and patients with status epilepticus. Thiopental causes cardiac depression and vasodilatation producing hypotension, especially in those with hypovolemia or cardiomyopathy; therefore, lower doses of thiopental should be administered in this subset of patients. Two percent of patients have an allergic reaction to thiopental, causing coughing, laryngospasm, and bronchospasm;29 therefore, asthma is a relative contraindication to its use. Other side effects include respiratory depression, hypersalivation, and a lack of analgesic properties.
Propofol is a highly lipophilic, rapidly-acting induction agent with onset of action in 20–30 seconds and a duration action of 10–15 minutes.2 The rapid onset and resolution of the sedation allow it to be titrated rapidly on and off, making the drug especially useful when serial exams are indicated.7 The dose is 1–3 mg/kg IV, with higher doses required for younger children, and continuous sedation may be achieved using 25–100 mcg/kg/min.7 Given propofol's highly lipophilic nature, it rapidly crosses the blood-brain barrier, decreasing ICP and cerebral metabolism, making it an excellent agent for head-injured patients. The potential adverse effects with propofol are that it causes hypotension and pain on injection; also, there are no analgesic properties, and it must be maintained in an emulsion to remain in solution. The hypotension observed with propofol use is caused by decreased systemic vascular resistance and can be tempered by administration of crystalloid IV fluids.29 Patients with allergies to lecithin (soy/eggs) may also exhibit hypersensitivity reactions to propofol, since propofol's formulation contains both egg lecithin and soybean oil; therefore, it should be used with caution in this subset of patients.
Ketamine is a phencyclidine derivative that produces analgesia and amnesia at a dose of 1–4 mg/kg IV, with an onset of action in less than two minutes and duration of action of 10–30 minutes.2,29 It is considered a dissociative induction agent rather than a pure sedative, because patients are unaware of their surroundings but are not actually asleep.7 The patient may keep his or her eyes open during this state but will continue to experience profound analgesia. Ketamine does not inhibit spontaneous respirations and does not suppress the body's protective airway reflexes.29 It causes catecholamine release, resulting in increases in blood pressure and heart rate via beta-1 receptors and dilatation of small airways through beta-2 receptors.1 This increase in heart rate helps prevent the bradycardia observed with vagal stimulation during laryngoscopy, and the elevation in blood pressure may offset hypotension in the patient with multiple trauma.1 The bronchodilatory properties of ketamine make it the drug of choice for status asthmaticus or chronic obstructive pulmonary disease.
Because of its sympathomimetic properties, ketamine may also increase ICP by cerebral vasodilatation and should be used with caution in closed-head injury.30 Some studies suggest, however, that ketamine increases the mean arterial pressure more than it increases ICP, thus improving the cerebral perfusion pressure (CPP = MAP – ICP) and rendering ketamine potentially cerebroprotective.31 Patients who receive ketamine may experience emergence phenomena (frightening hallucinations or dreams) for the first three hours after awakening from the dissociative state. These reactions are more common in adolescent females and adults after higher doses of ketamine, and the administration of a benzodiazepine may temper this potential side effect.1,29 Ketamine also produces excessive airway secretions and laryngospasm. The bronchorrhea and salivation can be ameliorated by pretreatment with an anticholinergic agent such as atropine or glycopyrrolate; the laryngospasm can be reversed with a dose of a short-acting paralytic. The indications for using ketamine include hypotension and reactive airway disease. Current contraindications include head trauma, penetrating eye trauma, glaucoma, and preexisting hypertension.1,2,7,29
Benzodiazepines are efficient sedatives and amnestics that serve as the sole induction agent in fewer than 20% of rapid sequence intubations, with midazolam as the benzodiazepine of choice in 90% of these cases.2 Midazolam exhibits the quickest onset of action and shortest duration compared to other benzodiazepines. When given in its correct dose of 0.2–0.3 mg/kg IV, midazolam's onset of action is 30 seconds and duration is 15–20 minutes. It can take up to 3–5 minutes before optimal sedation ensues, however, so midazolam should ideally be administered up to two minutes prior to paralysis.7 Benzodiazepines are popular sedatives because of their anticonvulsant effects; however, they do possess negative inotropic properties that can induce hypotension, especially in the elderly and the hemodynamically compromised. Other side effects or pitfalls of benzodiazepines include respiratory depression, slower progression to unconsciousness when compared with other induction agents, and a lack of analgesic properties.
Fentanyl is a short-acting and easily reversible synthetic opioid which is occasionally used as an agent for RSI induction. It provides analgesia as well as sedation; however, large doses are required to achieve sedation sufficient for intubation. The standard initial dose is 2 mcg/kg IV, but induction doses may be as high as 15 mcg/kg.7 Fentanyl may be combined with benzodiazepines to reduce the dose required for sedation, but this combination may cause a drop in blood pressure as both medications decrease systemic vascular resistance.1 Other side effects of fentanyl include chest wall rigidity, respiratory depression, and seizure-like activity,. Given that opioids alone are unreliable induction agents, they are usually not a preferable medication for RSI.1,7
Once the patient is adequately sedated, a paralytic should be administered to effect complete skeletal muscle relaxation, thus increasing the success of the intubation. There are two types of paralyzing agents: depolarizing and nondepolarizing paralytics. Both classes are highly water-soluble; therefore, they do not pass the blood-brain barrier, and both work at the motor end plate on the acetylcholine receptor to paralyze the muscle. (Table 2.)
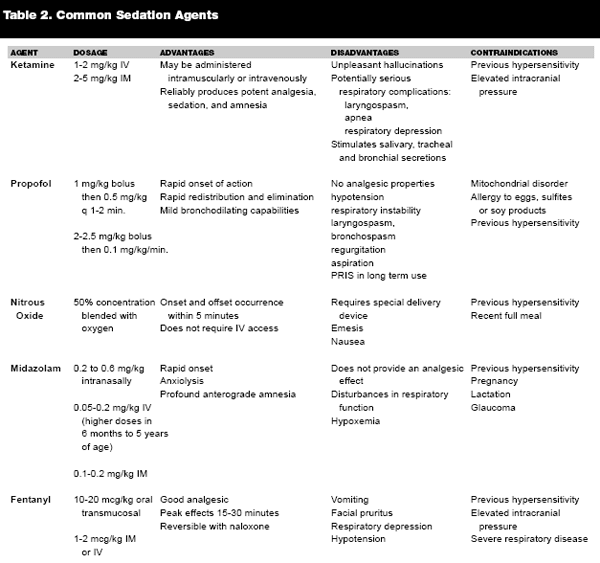 |
Succinylcholine is the only depolarizing agent used for RSI.7,29 The dose is 1–2 mg/kg IV and 4 mg/kg IM. It has an onset of action of 30–60 seconds IV and 4–6 minutes IM, duration of action of 5–7 minutes, and typical full return of function within 15 minutes. Succinylcholine is the preferred agent for use in RSI because of its rapid onset and short duration of action. In the event of an unsuccessful intubation, the patient will resume spontaneous breathing within several minutes, and there is a rapid return to baseline motor function, allowing for serial neurological exams.
Despite its positive attributes, succinylcholine has an extensive side-effect profile. The muscle fasciculations observed with succinylcholine may cause hyperkalemia and increased ICP, but the mechanism behind the increased pressure is unclear. The fasciculations can be prevented with a small dose of a nondepolarizing paralytic during the pretreatment phase, but the defasciculating dose does not necessarily abolish the associated side effects. Serum potassium increases by 0.5–1 mEq/L after a 1 mg/kg dose of succinylcholine.32 Normally the rise in potassium is clinically insignificant, but it becomes important in patients at risk for hyperkalemia. Succinylcholine should thus be avoided in patients who are bed-ridden or in those with renal failure, preexisting hyperkalemia, muscular dystrophy, acute denervation syndromes, acute crush injuries, adrenal crisis, significant burns greater than 48 hours old, or a personal history or family history of malignant hyperthermia.8 In the setting of burns, denervation injuries, and crush injuries, receptor proliferation does not occur until 48–72 hours after the event; therefore, succinylcholine is not contraindicated in acute burns or trauma but is contraindicated when these injuries are greater than 24 hours old.2,29,33 Succinylcholine should not be given in open-globe injuries or glaucoma, as it can increase intraocular pressure as well as ICP. Succinylcholine can also induce cardiac dysrhythmias, with bradycardias being the most common in children because of its effect upon the parasympathetic receptors on the heart. Although much less common, asystole and ventricular tachyarrhythmias have also been reported. Succinylcholine may cause masseter spasm, a phenomenon reported primarily in children that can be treated with a competitive neuromuscular blocking agent. If the spasm is severe and prolonged, malignant hyperthermia should be suspected.
Succinylcholine is broken down rapidly by plasma pseudocholinesterase prior to reaching the motor end plate; therefore, only small amounts of succinylcholine actually reach the acetylcholine receptor. In rare hereditary cases of defective or deficient enzyme, there will be a decreased amount of active plasma pseudocholinesterase available, resulting in prolonged paralysis for a period of up to 23 minutes.20 Succinylcholine will eventually diffuse away, however, restoring normal motor function.
Non-depolarizing agents, such as vecuronium and rocuronium, may also be used in RSI as an alternative to succinylcholine when contraindications are present. These agents work by binding competitively with the postsynaptic receptor on the motor end plate, thus preventing the binding of acetylcholine but without the depolarization observed with succinylcholine.1,7,29 Non-depolarizing agents do not cause fasciculations or hyperkalemia and can be reversed with acetylcholinesterase inhibitors like neostigmine or vagolytic doses of glycopyrolate or atropine. These agents have the distinct disadvantages of longer time to onset of action, longer duration of paralysis, and potential for bronchospasm secondary to histamine release. Given their longer duration of action, non-depolarizing agents should be reserved for patients who oxygenate easily and are not expected to be difficult intubations; a laryngeal mask airway (discussed in detail later) or other backup airway device should be available in case the intubation is unsuccessful.
Vecuronium is a dose-dependent aminosteriod which, when given at 0.1–0.2 mg/kg IV, has an onset of action of 1–4 minutes and a duration of 30–90 minutes. Larger doses of 0.25–0.3 mg/kg IV may hasten intubating conditions but will result in paralysis lasting up to two hours.7 Since vecuronium has a longer time to onset, it may be used in a time induction sequence or given as a split dose. In the time induction sequence, vecuronium is administered first, followed immediately by a rapid-acting induction agent, such that the vecuronium reaches peak effect concurrently with the induction agent, creating ideal intubation conditions. In the split dose, vecuronium is initially given at a small priming dose (0.01 mg/kg IV) which functions to shorten the time of onset. Three minutes later, the second dose of 0.15 mg/kg is administered, inducing paralysis in 35–90 seconds.7 Vecuronium is a safe drug for children with minimal cardiovascular effects.
Rocuronium is a metabolic derivative of the aminosteroid vecuronium and is age and dose-dependent. Its rapid onset of action of 30–90 seconds mirrors that of succinylcholine, making it the ideal non-depolarizing agent for RSI; the standard dose is 0.6–1.2 mg/kg IV, with higher doses taking effect at 30 seconds and lower doses at 90 seconds.34 A dose of 0.6 mg/kg lasts approximately 45 minutes in infants and 27 minutes in children.35 Rocuronium results in mild tachycardia, which can offset the bradycardia often associated with intubation. Since rocuronium has a better profile for RSI than vecuronium, it should be the drug of choice for children in whom succinylcholine is contraindicated.
After administration of an induction agent, the Sellick maneuver can be initiated until the airway is secure. Otherwise known as cricoid pressure, the Sellick maneuver involves applying light backwards pressure to the cricoid cartilage with the thumb and index finger, compressing the esophagus against the underlying vertebral body. This maneuver should serve to occlude the esophageal lumen, preventing regurgitated gastric contents from passing into the pharynx with subsequent aspiration into the pulmonary tree.36,37 The Sellick maneuver also improves visualization of the vocal cords, making the intubation easier; but it should only be performed upon a patient who is adequately sedated, as vomiting can occur in an awake patient. There are some pitfalls to the Sellick maneuver, however. It consistently reduces tidal volume and increases peak inspiratory pressures when applied during bag-valve-mask ventilation, and it can adversely affect ventilation and cause airway obstruction even when performed appropriately.37
Another mechanism to aid in the visualization of the vocal cords is bimanual laryngoscopy, a technique which involves intubator-directed manipulation of the vocal cords. Bimanual laryngoscopy coordinates maneuvering by the intubator's right hand with simultaneous direct intubator observation. Once the intubator has optimized his or her view, an assistant takes control of the manipulation of the larynx, freeing the intubator's right hand to place the tracheal tube 38
Placement of the tube
Once the patient is appropriately sedated and paralyzed, laryngoscopy and intubation can take place. Adequate sedation and paralysis can be assessed by manipulating the patient's mandible to evaluate for muscle tone. When performing laryngoscopy, one should avoid pressing the blade up against the teeth or the lips to prevent intubation trauma. In an uncomplicated airway, it should be easy to visualize the vocal cords and the ETT as it passes through them. The chest should be observed for equal rise and fall and should be auscultated on both sides, listening for equal breath sounds; auscultation of the epigastrum evaluates for breath sounds over the stomach, a finding which suggests esophageal intubation. Condensation in the ETT may support proper tracheal intubation; however, condensation alone is not sufficient for determining tube position. Decreased breath sounds in the left chest may suggest that the ETT is advanced too far into the right lung mainstem bronchus, an error which may be corrected by withdrawing the tube a centimeter or two while auscultating the chest.
A method to confirm endotracheal intubation and to detect inadvertent esophageal intubation is the end tidal CO2 detector.39 Disposable, colorimetric end tidal CO2 detection devices attach directly to the ETT and function by changing their color from purple to yellow on the detection of carbon dioxide during ventilation. The device comes in only two sizes: adult >15kg, and pediatric from 1–15kg, with each size equipped with the appropriate dead-space chamber. Detection of expired CO2 is the best method for the confirmation of tube placement in patients with a perfusing rhythm, because expired CO2 is both specific and sensitive for ETT placement in the trachea. In all circumstances, however, it is important to observe the color change for at least 6 breaths, because small amounts of CO2 in the esophagus may produce a brief purple to yellow color change in an esophageal intubation. The false-positive color change is more likely to occur if the patient has recently ingested a carbonated beverage or received bag-valve-mask ventilation resulting in air in the stomach. Washout of esophageal CO2 should be complete after six breaths, revealing the false-positive result. In children who are in cardiac arrest, the position of the tube must be confirmed by clinical evaluation and direct laryngeal examination, because expired CO2 may not be detected in the circumstances of low cardiac output with subsequent low pulmonary blood flow. Expired CO2 in the setting of cardiac arrest, however, is highly predictive of tube placement in the trachea.40 False negative results can occur in infants with diminished lung compliance, in near complete tracheal obstruction, and when using an adult-sized capnometer for a pediatric patient.39 Correct placement of the ETT should be confirmed primarily with physical exam findings and end-tidal CO2 detection, after which it should be carefully taped in place.
Secondary mechanisms for determining correct placement include the esophageal detector device, and fiber-optic bronchoscopy. The esophageal detector device is a bulb syringe that is used to aspirate air from the trachea. Since the esophagus is a muscular structure that collapses on itself, no air should be freely aspirated; therefore, air can easily be aspirated from a tube that has been placed correctly in the trachea. This device has been shown to be accurate in children of varying ages, but tracheal secretions may occlude the syringe resulting in no aspiration of air, suggesting an esophageal intubation.41,42 Direct laryngoscopy is not recommended to confirm tube placement, since airway trauma, secretions, and edema can make visualization difficult. Also misinterpretation of tube placement can easily occur, especially if the same clinician who attempted the intubation performs the confirmation laryngoscopy.
Post-intubation Management
The gold standard for verifying tube placement is the post-intubation chest x-ray, which should demonstrate the tip of the ETT just superior to the carina. Once proper placement of the tube has been confirmed and the patient is being ventilated adequately, protection of the ETT position is paramount. Patients should be sedated and paralyzed in order to prevent dislodgement of the tube and to blunt the bodies' physiologic response to the intubation. Versed or propofol drips are commonly used, as they can be easily titrated to achieve a suitable level of sedation, and vecuronium boluses can be administered repeatedly to induce paralysis. A blood gas should be performed one hour after intubation, and ventilator settings should be adjusted based upon the results.
The Failed or Difficult Airway
Some airways will be difficult or impossible to intubate because of their baseline anatomy or distortion resulting from trauma or illness, and clinicians should be prepared to handle the harrowing possibility of a failed intubation. The first step after an unsuccessful intubation attempt is to ensure that there is a tight seal between the mask and the patient's face and that the patient's oxygen saturations rise with bag-valve-mask ventilation. As long as the patient is oxygenating well, bag-valve-mask ventilation can continue indefinitely; however, the patient is unlikely to tolerate bag-valve-mask ventilation once the effects of the paralytics wane. After the patient's saturations improve with bagging, the clinician should reattempt the intubation with a maximum of three tries. After three unsuccessful attempts, a different person with more intubation experience should try to establish the airway; if that person is unsuccessful or the patient begins to deteriorate, alternative techniques should be attempted.
The laryngeal mask airway (LMA) is a well established airway device that can be used as a backup tool in a failed intubation. It consists of a semirigid tube with an inflatable silicone mask on the distal end and is inserted blindly into the pharynx. When resistance is met upon reaching the hypopharynx, the mask is inflated, forming a seal around the glottic opening that acts to ventilate the trachea with minimal gastric insufflation. The LMA comes in different sizes based upon the age of the patient, and appropriate size selection ensures proper placement and seal of the mask with consequent successful ventilation. (Table 3.) Most of the literature supporting the use of the LMA in pediatrics comes from case reports in pediatric anesthesia and not in pediatric resuscitation.43,44 Because its use has not been studied in emergency situations, the LMA is not considered a primary airway device for the resuscitation of infants and children; however, the Pediatric Emergency Medicine Committee of the American College of Emergency Physicians does advocate the LMA as the optimal alternative when RSI is unsuccessful.40,45 While the LMA is a good rescue device, it has flaws. It does not provide complete protection against aspiration, it can be dislodged easily by simple movements of the child, it can inadequately ventilate if placed improperly, and it may not generate sufficient inflation pressures for ventilation in asthmatic patients.
 |
The intubating LMA (ILMA) is a modified version of the LMA that allows for blind tracheal intubation with either a special tapered ETT or a conventional ETT up to size 8. It is inserted in the same fashion as the basic LMA, but it has an epiglottic elevator on the mask and a flat metal handle that is repositioned after insertion to lift up the epiglottis and attempt a blind tracheal intubation. The ILMA has demonstrated high success rates for insertion, ventilation, and subsequent tracheal intubation in adults, but it can only be used in children who weigh more than 30 kg.46,47 Like the LMA, the ILMA does not provide complete protection against aspiration; however, it should be considered in situations in which a patient cannot be ventilated or intubated.
 |
Two devices which deserve mention but are rarely indicated in children include the tracheoesophageal Combitube and the fiber-optic bronchoscope. When blindly inserted into the hypopharynx, the Combitube lands in either the trachea or the esophagus. The clear lumen functions as an endotracheal tube in the event that the combitube lands in the trachea, and the blue lumen functions like an LMA if it lands in the esophagus, forcing air into the larynx through side perforations. It can only be used in patients older than 15 years and greater than 5 feet tall. Fiber-optic bronchoscopy can be a quick and efficient means of tube placement when in well-trained hands; however, it is expensive and operator dependent. It involves passing an intubating bronchoscope through the vocal cords under fiber-optic visualization. The bronchoscope can act as an introducer in which the endotracheal tube can pass over and through the cords. Fiber-optic intubation is useful in instances when airway assessment and intubation need to occur simultaneously or in the setting of airway injury or trismus. The fiber-optic bronchoscope is not standard equipment in most emergency departments and requires an extensive training period to become competent in its use.
A surgical airway may be required in instances in which the patient cannot be ventilated or intubated and the nonsurgical alternative techniques mentioned previously are unsuccessful. A surgical cricothyrotomy requires locating the cricothyroid membrane and making a vertical incision through the skin just superior to the membrane and extending the incision just inferior to it. The scalpel can then be used to make a lateral incision through the cricothyroid membrane, an incision which can be enlarged with a scalpel blade or handle to accommodate an ETT or a Shiley™ tracheostomy tube. Ventilation can then occur through the tube. Cricothyrotomy is difficult and dangerous in children younger than 8 years of age because of anatomic considerations, and these children should undergo needle cricothyrotomy with transtracheal ventilation instead.
 |
Needle cricothyrotomy with transtracheal jet ventilation involves inserting a 14-gauge IV catheter through the cricothyroid membrane. Aspiration for free flow of air through the catheter using a syringe indicates correct placement in the trachea. A 3.0 ETT adapter is then attached to the catheter, and a resuscitation bag supplying oxygen is connected to the catheter adapter. This form of ventilation is preferred in children younger than 5 years of age. An alternative method of ventilating the patient in children older than 5 years is to connect the catheter to a high-pressure tubing system with a pressure regulator and gauge. The system is connected to a wall oxygen source that is set to a pressure sufficient for ventilation, usually about 50 psi. A one-second burst of high-pressure oxygen is administered, followed by a three- to four-second period to allow for exhalation and to avoid barotrauma. This mode of ventilation is a temporary measure only, because it provides oxygenation but inadequate ventilation, resulting in CO2 retention. The catheter also can be easily kinked or dislodged. Complications from needle cricothyrotomy with jet insufflation include inadequate ventilation, pneumothorax, subcutaneous emphysema, mediastinal emphysema, esophageal injury, and bleeding.8
 |
Retrograde intubation is another life-saving alternative to RSI in difficult airways. It involves inserting a needle through the cricothyroid membrane and guiding a wire superiorly through the vocal cords and out of the mouth. The endotracheal tube can then be passed over the wire into the trachea with removal of the wire as the tube is advanced, but the success of retrograde intubation is absolutely contingent upon adequate anesthesia. This technique has been described in adults but should be considered in adolescents and children in the setting of a difficult intubation.
Conclusion
Pediatric RSI and airway management are critical skills that must be mastered in order to be a competent emergency physician. Physicians should be well-versed in indications for and techniques of RSI under normal conditions and when faced with a difficult airway. The recognition of the anatomical and physiologic differences between children and adults can help facilitate the procedure and prevent complications. There are many drugs available for premedication, induction, and paralysis, and all of these medications have advantages and disadvantages. With sufficient knowledge and training, emergency physicians should be able to pre-oxygenate, sedate, paralyze, and intubate any patient that presents to the emergency room; and in the event of an unsuccessful ETT placement, a physician should be skilled in alternate techniques of oxygenation and ventilation.
References
1. Bledsoe GH, Schexnayder SM. Pediatric rapid sequence intubation: A review. Ped Emerg Care 2004;20:339-344.
2. Zelicof-Paul A, Smith-Lockridge A, Schnadower D, et al. Controversies in rapid sequence intubation in children. Curr Opin Pediatr 2005;17:355-362.
3. Chng Y, Sagarin M, Chiang V, et al. Pediatric emergency airway management. Acad Emerg Med 2004;11:438-439.
4. Sagarin MJ, Chiang V, Sakles JC, et al. Rapid sequence intubation for pediatric emergency airway management. Ped Emerg Care 2002;18:417-423.
5. American Heart Association. Pediatric Advanced Life Support (PALS) Provider Manual. American Heart Association, 2002, 2007.
6. Gnauck K, Lungo JB, Scalzo A, et al. Emergency intubation of the pediatric medical patient: Use of anesthetic agents in the emergency department. Ann Emerg Med 1994;23:1242-1247.
7. Gerardi M, Sacchetti A, Cantor R, et al. Rapid sequence intubation of the pediatric patient. Ann Emerg Med 1996;28:55-74.
8. Sullivan K, Kissoon N. Securing the child's airway in the emergency department. Peds Emerg Care 2002;18:108-121.
9. Hill JR, Rahimtulla KA. Heat balance and the metabolic rate of newborn babies in relation to the environmental temperature; and the effect of age and of weight on the basal metabolic rate. J Physiol 1965;180:239-265.
10. Wetzell RC. Evaluation of children. In: Rogers MC, Tinker JH, Covino BG, et al, eds. Principle and practice of anesthesiology. St. Louis:Mosby;1993: 441-479.
11. Motoyama EK. Respiratory physiology in infants and children. In: Motoyama EK, Davis PJ, eds. Smith's Anesthesia for Infants and Children. St. Louis: Mosby;1996:11-67.
12. Levitan R, Everett W, Ochroch EA. Limitations of difficult airway prediction in patients intubated in the emergency department. Ann Emerg Med 2004;44: 307-313.
13. Strange G, ed. APLS: The pediatric Emergency Medicine Course. 3rd ed. Dallas,TX:American College of Emergency Physicians–American Academy of Pediatrics;1998:21.
14. Berthoud M, Read DH, Norman J. Pre-oxygenation-how long? Anaesthesia 1983;38:96-102.
15. Mazurek A, Rae B, Hann S, et al. Rocuronium versus succinylcholine: Are they equally effective during rapid sequence induction of anesthesia? Anesth Analg 1998;87:1259-1262.
16. Fastle R, Roback M. Pediatric rapid sequence intubation. Peds Emerg Care 2004;20:651-655.
17. Robinson N, Clancy M. In patients with head injury undergoing rapid sequence intubation, does pretreatment with intravenous lignocaine/lidocaine lead to an improved neurological outcome? Emerg Med J 2001;18:453-457.
18. Butler J, Jackson R, Mackway-Jones K. Lignocaine premedication before rapid sequence induction in head injuries. Emerg Med J 2002;19:554.
19. Lev R, Rosen P. Prophylactic lidocaine use per-intubation. A review. J Emerg Med 1994;12:499-506.
20. Walls, Ron M. Airway. In: Marx J, et al, edd. Rosen's Emergency Medicine Concepts and Clinical Practice, 6th Ed. Philadelphia:Elsevier Health Science;2005:2-23.
21. Lerman J, Kiskis AA. Lidocaine attenuates the intraocular pressure respone to rapid inutabation in children. Canada Anaesth Soc J 1985;32:339-345.
22. Wilson IG, Meiklejohn BH, Smith G. Intravenous lidocaine and sympathoadrenal responses to laryngoscopy and intubation. Anaesthesia 1991;46:177-189.
23. Schulte-Sasse U, Eberlein HJ, Schumacker I et al. Should the use of succinylcholine during pediatric anesthesia be reevaluated? Anesthesiol Reanim 1993; 18:13-19.
24. Amidate (etomidate). Package insert. North Chicago, IL:Abbott Laboratories, 1998.
25. Sokolove P, Price D, Okada P. The safety of etomidate for emergency rapid sequence intubation of pediatric patients. Ped Emerg Care 2000;16:18-21.
26. Zuckerman N, Pitetti R, Herr S, et al. Use of etomidate as an induction agent for rapid sequence intubation in a pediatric emergency department. Acad Emerg Med 2006;13:602-609.
27. Fellow IW, Bastow MD, ByrneAJ, et al. Adrenocortical suppression in multiple injured patients: A complication of etomidate treatment. BMJ 1983;287: 1835-1837.
28. Guldner G, Schultz J, Sexton P, et al. Etomidate for rapid-sequence intubation in young children: Hemodynamic effects and adverse events. Acad Emerg Med 2003;10:134-139.
29. Reynolds S, Heffner J. Airway management of the critically ill patient: Rapid-sequence intubation. Chest 2005;127:1397-1412.
30. Reves JG, Glass PSA, Lubarsky DA, et al. Intravenous nonopioid anesthetics. In: Miller RD, ed. Miller's Anesthesia. Philadelphia:Churchill Livingtone; 2005:317-378.
31. Sehev R, Symmons D, Kindl K. Ketamine for rapid sequence induction in patients with head injury in the emergency department. Emerg Med Australasia 2006;18:37-44.
32. Isenstein D, Venner D, Duggan J. Neuromuscular blockade in the intensive care unit. Chest 1992;102:1258-1266.
33. Rubin MA, Sadovnikoff N. Neuromuscular blocking agents in the emergency department. J Emerg Med 1996;14:193-199.
34. Doobinin KA, Nakagawa T. Emergency department use of neuromuscular blocking agents in children. Pediatr Emerg Care 2000;16:441-447.
35. Gronert BJ, Brandom BW. Neuromuscular blocking drugs in infants and children. Pediatr Clin North Am 1994;41:73-91.
36. Sellick BA. Cricoid pressure to control regurgitation of stomach contents during induction of anaesthesia. Lancet 1961;2:404-406.
37. Ellis D, Harris T. Cricoid pressure in emergency department rapid sequence tracheal intubations: A risk-benefit analysis. Ann Emerg Med 2007;50:653-665.
38. Levitan R, Kinkle W, Levin W, et al. Laryngeal view during laryngoscopy. Ann Emerg Med 2006;47:548-555.
39. Sullivan K, Kisson N, Goodwin S. End-tidal carbon dioxide monitoring in pediatric emergencies. Pediatr Emerg Care 2005;21:327-335.
40. Zideman D, Zaritsky A, Carlo W, et al. Airways in pediatric and newborn resuscitation. Ann Emerg Med 2001;37:126-136.
41. Sharieff GQ, Rodarte A, Wilton N, et al. The self-inflating bulb as an airway adjunct: Is it reliable in children weighing less than 20 kilograms? Acad Emerg Med 2003;10:303-308.
42. Sharieff GQ, Rodarte A, Wilton N, et al. The self-inflating bulb as an esophageal detector device in children weighing more than twenty kilograms: A comparison of two techniques. Ann Emerg Med 2003;41:623-629.
43. Johnston DG, Wrigley SR, Robb PJ, et al. Experience with the laryngeal mask airway in paediatric anesthesia. Anaesthesia 1990;45:924-927.
44. Mason DG, Bingham RM. The laryngeal mask airway in children. Anaesthesia 1990;45:700-763.
45. Butler K, Cyne B. Management of the difficult airway: Alternative airway techniques and adjunts. Emerg Med Clin North Am 2003;21:259-289.
46. Burgoyne L, Cyna A. Laryngeal mask vs intubating laryngeal mask: Insertion and ventilation by inexperienced resuscitators. Anaesth Intens Care 2001; 29:604-608.
47. Martel M, Reardon RF, Cochrane J. Initial experience of emergency physicians using the intubating laryngeal mask airway: A case series. Acad Emerg Med 2001;8:815-822.
Emergency physicians perform many lifesaving procedures every day; however, none is more important than effective airway management.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
