Glucosamine Sulfate for Osteoarthritis
Glucosamine Sulfate for Osteoarthritis
By Dónal P. O'Mathúna, PhD. Dr. O'Mathúna is Senior Lecturer in Ethics, Decision-Making & Evidence, School of Nursing, Dublin City University, Ireland; he reports no financial relationship to this field of study.
Osteoarthritis is the most common form of arthritis and a leading cause of disability in the United States.1 As many as 40% of those older than 65 years may have symptomatic osteoarthritis of the hip or knee, the large joints most commonly affected by osteoarthritis.1 It is estimated that almost everyone 75 years of age or older is affected by osteoarthritis in at least one joint. However, only about half of the people who show evidence of the disease on X-ray report having any symptoms.2 The prevalence of osteoarthritis is expected to double in the next 20 years.3
Osteoarthritis was once viewed as a consequence of aging, but is now known to develop due to complex interactions involving genetics, age, gender, obesity, joint injury, and muscle weakness.3 Osteoarthritis tends to affect women at an earlier age than men, making it an important area of investigation for women's health.
Background
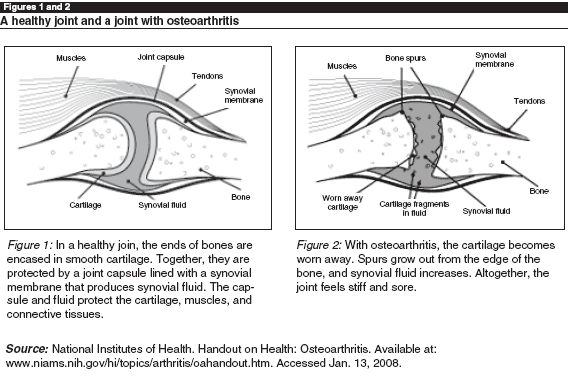
Medically speaking, osteoarthritis refers to a disease in which the cartilage inside joints gradually degenerates. This exposes the bone inside the joint and causes narrowing of the joint space leading to stiffness and pain. In the end, the joint may become so immobile and painful that joint replacement surgery is the best alternative. (See Figures 1 and 2.) Osteoarthritis is only one of a number of arthritic diseases, resulting in the term arthritis being used commonly to refer to any pain and stiffness in the muscles and joints. For this reason, some who have osteoarthritis refer to it as arthritis. Technically speaking, arthritis involves inflammation in the joints and osteoarthritis is a non-inflammatory degenerative disease. All of the studies reviewed here were carried out with older adults diagnosed with osteoarthritis.
 |
Currently, osteoarthritis does not have a cure.4 Management of symptoms includes pharmacological and non-pharmacological strategies.1 Weight control, pain relief, exercise to maintain and improve the range of motion and stability of the joints, and limiting functional disability are important strategies in treating osteoarthritis. Frequently, drug therapy, including analgesics like acetaminophen and nonsteroidal anti-inflammatory drugs (NSAIDs), is needed. The newer cyclooxygenase-2 (COX-2) inhibitors are recommended widely as perhaps being safer than older NSAIDs. Although all of these are important for managing symptoms, they do not treat the underlying pathology.
Concerns about the toxicity and side effects of pharmaceutical drugs have fueled interest in complementary approaches to managing osteoarthritis symptoms. Some dietary supplements are alleged to treat the underlying condition. Foremost among these are glucosamine sulfate and chondroitin sulfate, which are some of the most popular dietary supplements sold in the United States.5 Although the two supplements are usually taken together, most of the studies have tested them separately.3 This review will focus specifically on the use of glucosamine sulfate.
Glucosamine occurs naturally in the body as one component of the cartilage and synovial fluid found within joints.5 Glucosamine forms part of the structure of some compounds whose levels are reduced with osteoarthritis. A number of salts of glucosamine are available, with some debate over their relative effectiveness. While the exact mechanism of action of glucosamine remains unknown, it is presumed that glucosamine supplementation facilitates the production and regeneration of cartilage.
Clinical Studies
Clinical studies have been conducted on glucosamine sulfate for osteoarthritis for more than 20 years. Many of the early studies were conducted in Europe using a product available there by prescription as 1,500 mg sachets to be dissolved in water (manufactured by Rotta Pharmaceuticals of Italy). These studies were sponsored by its European manufacturer and generally had favorable results. Later independent studies tended to have negative results, raising concerns about bias. However, the differences among various trials are more complicated, involving different glucosamine formulations and study designs. Independent studies have used products available in the United States as dietary supplements, many of which are formulated as capsules and may not be absorbed well.2
An important publication in the history of glucosamine was a systematic review and meta-analysis published in 2000.6 This review identified six double-blind, randomized, controlled trials (RCTs) in which glucosamine sulfate was administered for at least four weeks. The trials were conducted between 1980 and 1997 and all evaluated knee osteoarthritis symptoms. The analysis revealed moderate-to-large benefits from taking glucosamine sulfate. However, all of the trials were of relatively short duration and had methodological limitations that are known to exaggerate beneficial effects. These included inadequate allocation concealment and lack of intention-to-treat analysis. There was also a question of publication bias.2
Another meta-analysis was published in 2003 which reviewed trials published between 1980 and 2002.7 Seven RCTs of glucosamine sulfate for osteoarthritis were included in the analysis. The treatment periods ranged from four weeks to three years, and only high-quality studies were included. Glucosamine was found to significantly improve symptoms as measured by the Western Ontario and McMaster Universities (WOMAC) osteoarthritis index, Lequesne Index (LI), visual analog scale (VAS) for pain, and mobility tests. The effect sizes were less than those found in the 2000 meta-analysis, which was interpreted as being due to the inclusion of only higher-quality studies. The 2003 review also included two studies in which joint space reduction was measured by X-ray. Both found significant improvements which suggested slowing of the joint cartilage degenerative process.
A subsequent review included all RCTs of glucosamine for osteoarthritis published up until 2005.8 Twenty studies were included in the analysis which showed much variability in their results. Overall, glucosamine showed a 28% improvement in pain and a 21% improvement in function as measured by LI. Sub-group analyses shed some light on the sources of variability. The eight studies of highest quality (which had adequate allocation concealment) showed no benefit in pain and WOMAC function scores. Ten of the studies were conducted with the Rotta product and their pooled results found glucosamine superior for pain and function. The 10 studies using non-Rotta products did not show statistical differences between glucosamine and placebo.
Since those reviews were published, the much-anticipated Glucosamine/Chondroitin Arthritis Intervention Trial (GAIT) published its results.9 This study randomized more than 1,500 patients with symptomatic knee osteoarthritis to daily doses of either 1,500 mg glucosamine hydrochloride, 1,200 mg chondroitin sulfate, both glucosamine and chondroitin, 200 mg celecoxib, or placebo. Two-thirds of the participants were women. The primary outcome measure was a 20% reduction in knee pain measured by the WOMAC scale after six months of treatment. No significant reduction in pain occurred for the groups taking glucosamine or chondroitin, alone or together, compared to placebo, while pain was significantly reduced in the celecoxib group. Subanalysis, however, revealed that those participants in the GAIT study with moderate-to-severe pain, had significantly better pain reduction after six months with combination therapy compared to placebo (P = 0.002).
Those patients with moderate-to-severe joint damage as revealed by X-ray were asked to continue in a follow-up study focused on joint space width.3 Patients continued to take the same treatment to which they had been assigned for a total of two years, with X-rays obtained at baseline, 12 months, and two years. No statistically significant differences in joint space width were observed between any of the groups.
Adverse Effects
The GAIT study reported no significant differences in adverse effects between the groups, with a similar number of participants withdrawing from each group because of adverse events.9 No differences in serious adverse events between groups were reported in the meta-analyses.7 Its safety profile is thus considered excellent.
Concerns have been raised that glucosamine might interfere with glycemic control in diabetic patients since animal models have demonstrated such effects. However, a double-blind RCT of Type 2 diabetes patients receiving either placebo or 1,500 mg glucosamine sulfate plus 1,200 mg chondroitin sulfate found no changes occurred in glycemic control after 90 days.10
Glucosamine sulfate is obtained from chitin extracted from marine exoskeletons, which has raised concerns about allergic reactions. Those with seafood allergies should probably avoid glucosamine, depending on the severity of their allergy, or be carefully monitored if they try it.4
Conclusion
The results of the GAIT study are consistent with previous research on glucosamine hydrochloride: It does not have the effectiveness found for glucosamine sulfate. Only the Rotta brand has consistently demonstrated effectiveness in reducing symptoms and slowing joint deterioration. The glucosamine may be more readily absorbed from this product as it is dissolved in water before consumption. Questions have been raised about the bioavailability of glucosamine in general.2 In addition, quality varies considerably among those products available in the United States as dietary supplements.11
For those with osteoarthritis, a trial of glucosamine sulfate may be warranted, especially for those who do not respond well to conventional treatments. However, even in those trials where glucosamine sulfate has been found effective, participants continued to use other analgesics when pain or discomfort were particularly problematic. Symptoms should be evaluated carefully over 2-3 months, at which time those who are likely to benefit will have responded.12 As with many chronic conditions, control of weight, exercise, and other symptom-relieving strategies will remain important in the overall management of osteoarthritis.
References
1. Zhang W, et al. OARSI recommendations for the management of hip and knee osteoarthritis, part I: Critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage 2007;15:981-1000.
2. McAlindon T. Why are clinical trials of glucosamine no longer uniformly positive? Rheum Dis Clin North Am 2003;29:789-801.
3. Sawitzke AD, et al. The effect of glucosamine and/or chondroitin sulfate on the progression of knee osteoarthritis: A report from the glucosamine/chondroitin arthritis intervention trial. Arthritis Rheum 2008;58:3183-3191.
4. Grainger R, Cicuttini FM. Medical management of osteoarthritis of the knee and hip joints. Med J Aust 2004;180:232-236.
5. Dahmer S, Schiller RM. Glucosamine. Am Fam Physician 2008;78:471-476.
6. McAlindon TE, et al. Glucosamine and chondroitin for treatment of osteoarthritis: A systematic quality assessment and meta-analysis. JAMA 2000;283: 1469-1475.
7. Richy F, et al. Structural and symptomatic efficacy of glucosamine and chondroitin in knee osteoarthritis: A comprehensive meta-analysis. Arch Intern Med 2003; 163:1514-1522.
8. Towheed TE, et al. Glucosamine therapy for treating osteoarthritis. Cochrane Database Syst Rev 2005(2): CD002946.
9. Clegg DO, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med 2006;354:795-808.
10. Scroggie DA, et al. The effect of glucosamine-chondroitin supplementation on glycosylated hemoglobin levels in patients with type 2 diabetes mellitus: A placebo-controlled, double-blinded, randomized clinical trial. Arch Intern Med 2003;163:1587-1590.
11. Russell AS, et al. Active ingredient consistency of commercially available glucosamine sulfate products. J Rheumatol 2002;29:2407-2409.
12. Hochberg MC. Nutritional supplements for knee osteoarthritisstill no resolution. N Engl J Med 2006;354:858-860.
Osteoarthritis is the most common form of arthritis and a leading cause of disability in the United States.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
