Fibromyalgia
Fibromyalgia
Authors: Vani Velkuru, MD, Fellow, Rheumatology, Loma Linda University, Loma Linda, CA ; and Keith Colburn, MD, Professor of Medicine, Chief of Rheumatology, Loma Linda University and Loma Linda VA Medical Center, Loma Linda, CA.
Peer Reviewer: John C. Jump, MD, Medical Director, Kettering Medical Center, Department of Physical Medicine and Rehabilitation, Electrodiagnostic Medicine, Kettering, OH.
This issue deals with a common but often frustrating and unrewarding condition that confronts primary care physicians: fibromyalgia. As scientists and clinicians, we always search for the pathophysiologic mechanisms of disease, but fibromyalgia has, in many aspects, defied that search. We all feel more comfortable when we have a definitive laboratory test to confirm a diagnosis rather than the complexity of a syndrome with predominantly subjective complaints. Yet the condition is real and can be clearly characterized and effectively treated if accurately recognized.
The article highlights in a practical manner:
epidemiology;
etiology and pathophysiology;
clinical features;
diagnostic criteria;
laboratory findings;
physical examination; and
management, including pharmacological and non-pharmacological treatment options.
The Editor
Fibromyalgia, previously known as fibrositis or fibromyositis, is an idiopathic, multisystem, chronic condition characterized by fatigue, sleep disturbance, diffuse widespread musculoskeletal pain, paresthesias, and a variety of associated symptoms. Other synonyms of the fibromyalgia syndrome are non-articular rheumatism, neurasthenia, muscular rheumatism, and functional somatic syndrome.
In 1904, Sir William Gowers coined the term "fibrositis." In 1976, the term "fibrositis" was changed to "fibromyalgia" because it was discovered that the tissues associated with the diffuse tenderness of affected patients lacked inflammation.1 The concept of a non-inflammatory condition was further established by Smythe and Moldofsky. During the past 30 years, fibromyalgia gradually has been recognized as one of the most common causes of chronic widespread musculoskeletal pain, especially in women. In current medical practice, it is seen by most physicians as a distinct syndrome.
Epidemiology
The prevalence of fibromyalgia in the general population is 3-5%. Studies reveal that it is more common in women than men, and the incidence increases progressively with age. It is diagnosed most frequently in middle-aged women, but also is seen in older people, children, and possibly as many as 1-2% of all adolescents. According to a study in 2001, 15% of patients hospitalized in internal medicine wards had fibromyalgia, of which 91% were women.2
Certain studies show that fibromyalgia is more common in relatives of patients with fibromyalgia, suggesting the contribution of both genetic and environmental factors.3 There is also a high prevalence of pain syndromes and mood disorders in these families. Psychological factors associated with fibromyalgia include somatization disorder, anxiety, and personal or family history of depression.4
Etiology and Pathophysiology
There are multiple theories explaining the etiology of fibromyalgia, including muscle injury, non-refreshing sleep, neurohormonal abnormalities, psychophysiology, and abnormal central sensory processing of pain signals. However, both the etiology and pathophysiology of fibromyalgia remain elusive.5 The etiology appears to be polygenic and complex with the contribution of several overlapping syndromes such as anxiety and depression. The role of psychological factors in the pathogenesis is not very clear.
Genetic Factors. As previously mentioned, there appears to be a strong familial component or genetic basis6 contributing to the genesis of fibromyalgia. Family members of patients with fibromyalgia are many times more likely to be diagnosed with the same condition.7 They tend to have lowered thresholds for pressure-induced pain and are much more likely to have a host of regional pain syndromes and mood disturbances.
Specific genetic polymorphisms were found to be associated with an increased risk of developing a variety of pain syndromes in some patients with fibromyalgia. Genes affecting serotonin and catecholamine metabolic or signaling pathways may increase patients' susceptibility to develop variation in their sensitivity to pain and sensory processing. Serotonin 5-HT2A receptor polymorphism and inherited deletions in the gene for the human serotonin transporter,8 COMT (catecholamine o-methyl transferase) polymorphisms, and monoamine oxidase A gene polymorphism are examples of possible molecular explanations for the production of chronic pain.
Gender. Fibromyalgia affects women more frequently than men.9 In general, female patients are at a higher risk for pain syndromes because of increased pain sensitivity compared to males. They appear to lack certain pain-inhibitory mechanisms. Traumatic or stressful stimuli have a propensity to evoke greater sympathetic nervous system, hormonal, and psychosomatic responses in women than in men. Reproductive hormone levels can influence central pain-modulatory mechanisms.
Environmental Factors. Environmental stress factors play an important role in the development of fibromyalgia. The onset of symptoms may be triggered by emotional stress, war, catastrophic events, accidents, physical trauma, or illnesses including certain infections such as hepatitis B and C, human immunodeficiency virus (HIV) disease, parvovirus, Lyme disease, and Epstein-Barr virus.5 Other diseases associated with increased risk of acquiring fibromyalgia include hypermobility syndrome and chronic underlying autoimmune conditions such as rheumatoid arthritis, systemic lupus erythematosus (SLE), and inflammatory bowel disease (IBD).
Pain sensitivity may be modified by the psycho-social environment in which individuals develop. Adverse experiences during childhood, such as poverty or physical or sexual abuse, increases susceptibility to the development of chronic pain in adulthood. Several retrospective studies suggest that physical trauma may precipitate fibromyalgia.10 Patients with a history of psychological trauma associated with anxiety (for example, childhood trauma or sexual abuse) have an increased number of tender points.11 Hence, a pertinent history is important.
Neuroendocrine Factors. It is unclear if the hormonal, biochemical, and neurotransmitter abnormalities seen in fibromyalgia are due to a disease process or to pain itself. Derangement of the hypothalamic-pituitary-adrenal (HPA) axis and the autonomic nervous system along with hyperactivity of the stress response was observed in patients with fibromyalgia.12 The hormonal response to stimuli tends to be abnormal in these patients. Studies show decreased levels of baseline cortisol and a blunted adrenal cortical response to ACTH. On neuroendocrine provocative tests, the HPA response was low. All of these changes influence nociception and may alter the response to pain. Nociception is defined as the neural processes of encoding and processing noxious stimuli. It is the afferent activity produced in the peripheral and central nervous system by stimuli that have the potential to damage tissue.
Levels of somatomedin C (insulin-like growth factor I [IGF-I]), a modulator of growth hormone's actions and a neuromediator of chronic stress that is usually secreted during stage 4 sleep, are subnormal. Studies show elevated levels of Substance P (a neurotransmitter associated with enhanced pain perception) and nerve growth factor along with decreased levels of serotonin in the cerebral spinal fluid13 (CSF) of patients with fibromyalgia. Serum levels of serotonin, its precursor tryptophan, and its metabolite, 5-hydroxyindoleacetic acid (5-HIAA), are reported to be low. Serotonin mediates pain reactivity through changes in sleep patterns and by modulation of neuron excitability and pain signaling in the central nervous system (CNS). Hence medications that enhance the action of serotonin might play an important role in treating symptoms of fibromyalgia. There is a reported association between CSF levels of corticotropin-releasing factor (a key mediator in the HPA cascade), sensory pain, and autonomic dysfunction in fibromyalgia.
Altered HPA axis activity may be linked to childhood trauma, especially physical abuse.15 Exposure to stressors early in life or exposure to chronic stress can permanently affect the HPA axis, which, in turn, can lead to the development of fibromyalgia. Conditions like depression or post-traumatic stress disorder (PTSD) are associated with HPA and/or autonomic dysfunction.
To summarize, fibromyalgia is characterized by HPA axis hypo-activation. It is yet unclear if the characteristic hormonal profile of fibromyalgia is involved in the pathophysiology of the disease or if it is just a reflection of the patient's symptoms.
Autonomic Dysregulation. Autonomic derangement16 characterized by increased sympathetic and decreased parasympathetic tone may play a role in an altered visceral sensitivity encountered in fibromyalgia. It is manifested by orthostatic hypotension on postural change (tilt table testing) and baseline increased heart rate with poor variability. One of the proposed mechanisms for autonomic dysregulation is neurogenic switching, by which adverse stimuli can elicit a response at a distant area. Other conditions with autonomic dysfunction are depression and PTSD.
Dysfunction in Pain Processing. One of the earliest hypotheses in the etiopathogenesis of fibromyalgia is neuroplasticity and aberrant central pain mechanisms.17 According to this hypothesis, symptoms of chronic widespread pain are due to abnormalities involving the pain and sensory processing systems and involve the spinal cord, insula, and thalamus of the brain. One of the most reproducible features in fibromyalgia is pain and sensory amplification. Normal peripheral pain could become abnormally amplified at the level of the spine. Glial cells around the pain neurons can modify and augment the signaling and perception of pain. Generalized up-regulation of the CNS is enhanced in these patients, resulting in non-nociceptive pain. Non-nociceptive pain (neuropathic) results from abnormal function in the central or peripheral somatosensory system. This pain may occur when the normal physiologic process of nociception has been altered, producing different subjective and objective findings. The receptor thought to be involved is the N-methyl-D-aspartic acid (NMDA) receptor, present in dorsal horns of the spinal cord. NMDA receptors interact with excitatory amino acids, increasing pain.
Individuals with chronic pain syndromes display diffuse hyperalgesia (increased pain to normally painful stimuli) and/or allodynia (pain to normally non-painful stimuli). Compared to healthy individuals, these patients have generalized lower pain threshold to noxious physical stimuli like pressure, heat, cold, and electricity. This leads to extreme sensitivity at tender points as well as visceral pain as seen in IBD, non-cardiac chest pain, and headaches. One of the underlying mechanisms for hyperalgesia is central sensitization (heightened excitability of the spinal cord neurons that transmit nociceptive information to the brain). Repetitive stimulation of the peripheral nerve that carries pain signals can lead to central sensitization18 via abnormal, temporal summation or magnified "second pain" or wind-up.19 (Repeated, equal non-painful stimuli are perceived to be more intensive and painful than the initial stimulus.)
The presence of mood disorders can further modulate pain processing. In patients with fibromyalgia, studies show hypoperfusion in areas of brain such as the thalamus and caudate nucleus, increased levels of substance P in the CSF, gray matter loss, and down-regulation of brain opioid receptors. The decreased mu opioid receptor availability could be the reason for the ineffectiveness of opioids in treating patients with fibromyalgia.
Sleep Abnormalities. One of the most persistent universal symptoms of fibromyalgia patients is insomnia, although fibromyalgia is not primarily a sleep disorder. Chronic pain interferes with the quality of sleep, and insomnia, in turn, can aggravate pain. Both pain and non-restful sleep can result in fatigue.
An underlying CNS dysfunction can be a contributing factor to the stage 4 sleep disorder seen in patients with fibromyalgia. The distinctive feature of disturbed stage 4 sleep is phasic alpha sleep activity20 seen as intrusion of alpha waves (high-frequency, low-amplitude waves) into the normal stage 4 delta waves (low-frequency, high-amplitude) of non-rapid eye movement sleep. Frequent night awakening can result in a sensation of unrefreshing and non-restorative sleep. Irregular cyclic alternating sleep pattern contributes to the severity of fibromyalgia.
Decreased growth hormone levels along with low IGF1 might be due to the interruption of stage 4 sleep. Other hormones associated with sleep such as serotonin and endorphins are noted to be altered as well.
Serological Abnormalities. There is no evidence at present to indicate that fibromyalgia is an immune-mediated condition.21 The importance and prevalence of anti-serotonin, anti-ganglioside, and anti-phospholipid antibodies in some patients with fibromyalgia are as yet not clear. Cytokines, including IL-8 and IL-6, are being investigated in the etiopathogenesis.
Behavioral and Psychological Variables. Studies have debated the role and contribution of psychological factors22 in the development of fibromyalgia. There is an association of this illness with an increased incidence of psychiatric conditions such as anxiety, depression, PTSD, somatization, and personality disorders. The presence of a coexisting psychiatric disease may increase the severity of fibromyalgia symptoms by lowering pain threshold and tolerance. On the other hand, chronic pain syndromes may result in worsening of psychiatric symptoms. The relationship between these conditions might be because of the presence and use of similar neuropathways and neurotransmitters. It is not clear if a co-morbid psychiatric condition contributes to the development of fibromyalgia or vice versa. Chronic psychological distress can result in symptoms such as fatigue, anxiety, and pain by altering stress response systems like the HPA axis and autonomic nervous system.
Clinical Features
The cardinal feature of fibromyalgia is diffuse pain that is widespread in the musculoskeletal arena. Other clinical hallmarks include fatigue, generalized hyperalgesia, allodynia, insomnia, and paresthesias. The three key features of pain, fatigue, and sleep disturbance are present in virtually every patient with fibromyalgia.23
The onset of this illness can be gradual or acute, especially if there is exposure to stress. Presenting symptoms often are chronic, persistent, multifocal pain with fatigue interfering with daily activities. Pain often is described as widespread, migratory, exhausting, unbearable, deep, gnawing, aching, or burning in nature. Pain and stiffness initially may be localized to the neck and shoulders, with eventual involvement of the axial skeleton, chest wall, hips, and extremities. Variation of symptoms with waxing and waning is common. The majority of these patients describe fatigue as severe, worse in the mornings, and persisting all day. Another universal complaint is poor sleep patterns with difficulty falling asleep, frequent awakening, or early awakening. Symptoms of fibromyalgia are aggravated by cold and humid weather, poor sleep, and physical or mental stress. They are improved by warm and dry weather, moderate physical activity, adequate sleep, and relaxation.24 Probably because of amplified pain pathways, patients with fibromyalgia may experience a wide variety of other symptoms ranging from lightheadedness, fluid retention, migratory arthralgias, postexertional pain, palpitations, night sweats, dysphagia, dyspnea, vulvodynia, dysmenorrhea, sexual dysfunction, Raynaud's phenomenon, and allergic symptoms. Many also complain of pain in the chest, abdomen, or pelvic area.
Paresthesias (sensations of numbness, burning, tingling, or crawling) without objective neurologic findings are common. Mood disturbances (particularly depression, anxiety, and personality disorders), cognitive dysfunction (such as short-term memory loss and difficulties with concentration) and headaches (muscular or migraine-type) are present in a majority of patients.21,26
The coexistence of regional pain syndromes with fibromyalgia is frequent, including temporomandibular disorders, irritable bowel syndrome, myofascial pain syndrome, irritable bladder syndrome, interstitial cystitis, and restless leg syndrome. Other conditions such as chronic fatigue syndrome are very closely associated with fibromyalgia in about 50% of cases. It is important to differentiate fibromyalgia from other concurrent rheumatological conditions like rheumatoid arthritis, osteoarthritis, and SLE because often the doctor and patient alike confuse the symptoms of fibromyalgia with a worsening of the primary disease, when in fact there may be no relationship between the symptoms of each condition.
Diagnostic Criteria
In spite of the high prevalence of fibromyalgia, it often remains under-diagnosed and under-treated. The onset of symptoms frequently precedes the clinical diagnosis by years. Fibromyalgia often is debilitating to patients because of the chronic nature of the symptoms. Patients may have pain in any domain of musculoskeletal and non-musculoskeletal regions of the body. The syndrome complex of fibromyalgia is defined by the presence of widespread pain in particular areas of increased sensitivity called tender points.
In 1990, the American College of Rheumatology (ACR) developed criteria for the diagnosis and classification of fibromyalgia based on a multicenter study done by Wolfe, et al.24 The criteria are defined in Table 2.
 |
 |
The presence of a second clinical disorder does not exclude the diagnosis of fibromyalgia. These criteria have a sensitivity of 88.4% and a specificity of 81.1%. Failure to meet the ACR criteria does not completely rule out fibromyalgia.
Control points (areas like the thumbnail, forehead, and anterior mid-tibia) are regions that are expected to be non-tender and are supposed to increase the specificity of the tender points. On the other hand, tenderness at these control points does not rule out the clinical diagnosis of fibromyalgia.
Physical Examination
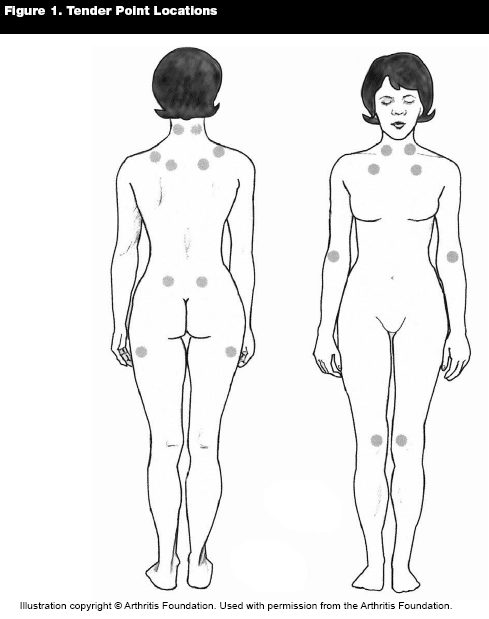
A thorough history and physical examination remain the most essential diagnostic tests for fibromyalgia. The most crucial part of physical examination is to palpate the 18 specific points to elicit tenderness. (See Figure 1.) These tender points initially were meant for classification purposes rather than diagnostic criteria; however, they are relied upon for diagnosis by most practitioners. The key feature to identify tender points is to apply consistent pressure equivalent to 4 kg/cm either by digital palpation or by using a dolorimeter (algometer). Applying enough pressure to whiten the examiner's fingernail bed generates approximately 4 kg/cm of pressure.27 Patients can feel pain, not just tenderness. The response at these tender points may be compared to control points (thumbnail, forehead, mid-forearm, etc.). Many patients with fibromyalgia complain of pain to pressure almost everywhere, including control areas.
 |
The Fibromyalgia Impact Questionnaire (FIQ)28 is one of the validated instruments to assess the functional abilities in daily life by measuring patient status and progress.
Other than the positive tender points, the physical examination usually is unremarkable for uncomplicated fibromyalgia. A detailed assessment of musculoskeletal and neurologic systems can help delineate this condition from other concomitant diseases including arthritis, connective tissue disorders, and neurological conditions. The presence of synovitis or signs such as hypermobility indicate the presence of concurrent disorders. It is important to identify other painful conditions such as bursitis, tendinitis, myofascial pain, and inflammatory soft-tissue conditions. The nociceptive pain from these conditions is amplified in fibromyalgia.
Laboratory Findings
Screening blood tests include a complete blood count, routine serum chemistries, erythrocyte sedimentation rate, C-reactive protein, liver function tests, thyroid function tests, and muscle enzymes. In uncomplicated fibromyalgia, probably the most important laboratory result is a normal sedimentation rate or C-reactive protein. There are no definitive laboratory tests to diagnose fibromyalgia. Laboratory testing is helpful mainly to screen and eliminate other commonly associated disorders. Extensive unnecessary workup should be avoided if possible.
Differential Diagnosis
A wide variety of diseases may overlap with or mimic fibromyalgia. It is important to differentiate fibromyalgia from conditions such as myofascial pain syndrome, chronic fatigue syndrome, hypothyroidism, and polymyalgia rheumatica. Infections such as infectious mononucleosis, HIV, HTLV, hepatitis, and Lyme disease should be considered in the differential diagnosis. Patients need to be evaluated for endocrine diseases such as hyperparathyroidism and Cushing's syndrome, entrapment syndromes (such as carpal tunnel syndrome), neurologic disorders (such as multiple sclerosis and myasthenia gravis), psychiatric disorders (such as depression), and sleep disturbances (such as sleep apnea). Other common conditions associated with fibromyalgia and previously mentioned in this article include depression, migraine, irritable bowel syndrome, interstitial cystitis, female urethral syndrome, metabolic and inflammatory myopathies, rheumatoid arthritis, SLE, and osteoarthritis. To provide optimal therapy and avoid over-treatment, it is important to recognize and treat patients for these overlapping conditions.
Management
In spite of improved understanding and recognition of fibromyalgia, treatment continues to be challenging. Management of chronic pain syndromes is frustrating for clinicians and patients alike. Because of a limited understanding of the etiology and the poor response to conventional pain treatments for fibromyalgia, a majority of the patients remain unsatisfied with the results of their therapy. Pain and fatigue often persist and limit daily functioning. There is no single effective therapy. Treatment strategies largely are empirical and are directed at controlling symptoms such as pain, sleep problems, depression, and anxiety. Treatment usually is long term since this condition is chronic, often with a waxing and waning pattern. Therapeutic strategies range from evidence-based approaches such as medications and exercise to less well studied alternative medical practices such as herbal substances and acupuncture. (See Table 4.)
 |
 |
The American Pain Society Fibromyalgia Panel has developed clinical practice guidelines29 and recommends a multidisciplinary or multimodal therapeutic approach that combines drug therapy with physical, psychological, and cognitive behavior strategies.30 The European League Against Rheumatism (EULAR) recently published evidence-based recommendations for the management of fibromyalgia.31
The response to treatment depends on the severity of the symptoms. Patients with mild symptoms need a simple regimen. Patients with severe symptoms will need a more sophisticated approach. A high pretreatment level of impairment was significantly associated with a better response to treatment.32 There is a lack of specific and sensitive tools to evaluate and assess the effectiveness of treatment.
Pharmacotherapy
In clinical trials, only limited numbers of medications were shown to be beneficial for the treatment of fibromyalgia. The drugs used with most success are antidepressants, muscle relaxants, anticonvulsants, and sedative-hypnotic drugs. The two FDA-approved drugs for the treatment of fibromyalgia are pregabalin (Lyrica) and duloxetine hydrochloride (Cymbalta). Because of aberrant central pain mechanisms in fibromyalgia, routine pain medications such as narcotics have been ineffective and their use in the treatment of fibromyalgia is not encouraged.
Tricyclic Antidepressants (TCAs). This group has been part of the traditionally used first-line medications in the treatment of fibromyalgia. Evidence shows that these medications improve pain, fatigue, and sleep quality. They are more beneficial in patients with coexisting depression. Low-dose sedating antidepressants (e.g. amitriptyline and trazadone) are used most frequently.
The TCAs have an analgesic effect that is independent of their antidepressant action.33 These agents increase the concentrations of both serotonin and norepinephrine in the central nervous system by inhibiting the reuptake at spinal dorsal horn synapses, with secondary activity at the sodium channels.34 Higher concentrations of these neurotransmitters can reduce pain signaling.
Dosages of tricyclic drugs need to be individualized based upon efficacy and side effects. Due to the high incidence of anticholinergic side effects, they should be started at a low dose and then gradually increased. The usual starting dose of amitriptyline or desipramine is 5-10 mg at bedtime. In clinical trials, amitriptyline at doses of 25-50 mg is shown to be effective in providing analgesic effect, aiding sleep, and treating concomitant mood disorders.34 Adverse effects such as dry eyes, dry mouth, constipation, drowsiness, weight gain, grogginess, and difficulty concentrating are more common at higher doses. There is a possibility of cardiotoxicity in older patients, especially with amitriptyline. Desipramine has the advantage of fewer side effects. The effectiveness of tricyclic drugs may diminish gradually with time.37
Selective Serotonin Reuptake Inhibitors (SSRIs). SSRIs act on the CNS by decreasing the reuptake of serotonin and increasing its availability at the neuronal synapse. Studies show some benefit with SSRIs, but there are conflicting data in clinical trials regarding their effectiveness, especially as single agents.38-40 Drugs studied include fluoxetine, paroxetine, and citalopram. The combination of fluoxetine (SSRI) with amitriptyline was found to be more effective than using either drug in isolation.38 Similarly, a combination of fluoxetine with cyclobenzaprine was more beneficial than either alone over a 12-week period.41 A few clinical studies suggest that SSRIs are as effective as amitriptyline.42
Serotonin/Norepinephrine Reuptake Inhibitors (SNRIs). Based on the mechanism of action, SNRIs may have superior anti-nociceptive properties with fewer side effects when compared to SSRIs. These medications include the antidepressants venlafaxine (Effexor), duloxetine (Cymbalta), and milnacipran (not yet available in the United States). Newer medications such as duloxetine and milnacipran show improvement in pain, tenderness, and other outcome variables in randomized clinical trials of patients with fibromyalgia.
Duloxetine was superior to placebo in total FIQ scores and a number of outcome measures including the Brief Pain Inventory scores and global function scores in 207 patients with fibromyalgia in a 3-month clinical trial.43 Effective doses of duloxetine ranged from 30 mg daily to 60 mg twice per day. The adverse effects of duloxetine include insomnia, dry mouth, and constipation. Venlafaxine has been studied in smaller trials.44 Milnacipran, twice daily, reduced pain intensity and improved secondary variables in 125 patients with fibromyalgia over 12 weeks.34 Milnacipran 100 mg twice daily was more effective and better tolerated than at 200 mg once daily. Milnacipran also binds to N-methyl-D-aspartate receptors.
Other medications undergoing clinical trials are monoamine oxidase inhibitors and 5-HT3 receptor antagonists.
Analgesic Medications. Randomized clinical trials show that tramadol (Ultram), a centrally acting synthetic analgesic drug that has some opioid-like properties (with or without acetaminophen) to be beneficial in patients with fibromyalgia.46-48 Tramadol has numerous analgesic effects. It inhibits norepinephrine and serotonin reuptake, and its major metabolite binds weakly to mu opioid receptors. It is well-tolerated and efficacious in patients with mild to moderately severe pain. It should be started with a low dose and gradually titrated to avoid nausea and dizziness. Dosages range from 50 mg prn up to 100 mg four times per day. Clinical trials using a combination of tramadol and acetaminophen (37.5/325 mg) show that the discontinuation rates, pain scores, and Fibromyalgia Impact Questionnaire scores were better in the combination group than in the control group.48 The long-term abuse potential of tramadol is less compared to traditional narcotics but still is a concern.
When used alone, NSAIDs (including cyclooxygenase-2 selective agents) have, at best, a very modest benefit in treating fibromyalgia. Little objective evidence exists regarding their efficacy. There is no evidence of tissue inflammation in uncomplicated fibromyalgia. So it is the analgesic rather than anti-inflammatory properties of NSAIDs that might be beneficial. Overuse often can lead to rebound headaches, further worsening symptoms of fibromyalgia. NSAIDs were reported to be more effective when used in combination with medications such as the tricyclics and other CNS medications. There is very little data on the use of COX -2 inhibitors in treating fibromyalgia. However, our experience with NSAIDs is that not enough benefit is derived from their use in fibromyalgia to offset the potential side effects these drugs may cause.
Use of opioids is discouraged in treating patients with fibromyalgia. Almost invariably, they rapidly lose their effectiveness and require higher doses, leading to the need for stronger opioid agents. The opioids are associated with adverse effects such as addiction, constipation, sedation, and nausea.49 These medications should be reserved for short-term use in patients as a last resort after exhausting all other modalities.50
Muscle Relaxants. These medications are used both in the chronic management of fibromyalgia as well as for acute flares. There is very little evidence regarding their efficacy. The most studied medication of this group is cyclobenzaprine (Flexeril). It is structurally analogous to tricyclic antidepressants and was shown to be effective in improving sleep and decreasing pain.51 This medication reduces brainstem noradrenergic function and motor neuron efferent activity and induces skeletal muscle relaxation.52 Cyclobenzaprine should be started at a low dose of 10 mg at bedtime and titrated as tolerated to avoid excessive sedation. Chronic use seems to decrease its efficacy.
Other Central Nervous System-Acting Agents. Anticonvulsants/antiepileptic drugs (AEDs) have analgesic properties in addition to anticonvulsant properties and are used in the management of fibromyalgia and other chronic pain conditions. These medications include pregabalin (Lyrica), gabapentin (Neurontin), topiramate (Topamax), and tiagabine (Gabitril). Pregabalin, a second-generation anticonvulsant used in the action of neuropathic pain and epilepsy, was the first FDA approved drug to treat fibromyalgia. Studies show that it is effective in relieving pain, decreasing fatigue, and improving sleep.53 Two recent controlled trials report that pregabalin was as effective as monotherapy in improving symptoms of fibromyalgia.54,55 The analgesic mechanism of action is unclear, but it is known to disrupt neuronal signaling by binding to voltage-gated calcium channels in the central nervous system. It is recommended to start the drug at a low dose and gradually increase as tolerated to 300-450 mg per day. The diverse side effect profile seems to be dose-related and includes lightheadedness, sedation, weight gain, dry mouth, and peripheral edema.
Gabapentin, a well known agent used in the treatment of epilepsy and neuropathic pain, is another commonly used agent in the management of fibromyalgia. Gabapentin was shown to have significant analgesic effects in diabetic neuropathy, post herpetic neuralgia, migraine prophylaxis, and other neuropathic pain conditions.56 Although it is structurally related to gamma-aminobutyric acid, it inhibits the voltage-gated calcium channels similar to pregabalin. In a recent randomized controlled trial designed to compare gabapentin (1200-2400 mg/day) with placebo for its efficacy and safety in treating fibromyalgia-associated pain,57 the gabapentin-treated patients showed clinically significant improvement and experienced less pain, improved sleep quality, and less fatigue. It was well tolerated when started at a low dose with gradual titration up to 3600 mg/day. Adverse effects include dizziness, sedation, lightheadedness, and weight gain.
Non-pharmacologic Therapy
Non-pharmacologic therapy for fibromyalgia includes patient education, aerobic exercise, cognitive-behavioral therapy, and other modalities such as acupuncture, meditation, yoga, counseling, and biofeedback.
Exercise. Exercise is the "most" important treatment for fibromyalgia. Thus, the responsibility for improvement from this condition rests squarely on patients with this problem. Patients with fibromyalgia usually do not maintain a regular exercise regimen due to pain, fatigue, or both. The importance of exercise in fibromyalgia was stressed since 1976, starting with Moldofsky. Cardiovascular exercise is widely recommended in the management of fibromyalgia and chronic pain syndromes. Exercise programs (aerobic and strength-training exercises) were shown to have positive effects in patients with fibromyalgia, improving psychological status, pain, and physical function.58,59 Exercise reduced the number of patients fulfilling the ACR criteria for fibromyalgia, decreased tender point counts, and improved FIQ scores.60 Graded aerobic exercise is more effective than other types of training, including relaxation and flexibility training. The main goal of exercise is to restore and maintain musculoskeletal function. Exercise prescriptions should be individualized.58 Physicians should encourage patients to start slowly and then to increase the duration and intensity by gradual increments. Once a moderate level of cardiovascular fitness is achieved, exercise can be advanced to other dimensions. High-intensity workouts should be avoided initially. Aerobic exercises with low impact such as brisk walking, cycling, swimming, or water aerobics have more success in improving function and symptoms. Warm water-based exercise may be beneficial, especially as an initial regimen and seems to be better tolerated by fibromyalgia patients.61,62
Cognitive Behavioral Therapy (CBT). Studies show that psychological therapy, especially CBT, is effective in treating patients with fibromyalgia.63,64 It was shown to improve symptoms of fibromyalgia including pain, mood disorders, fatigue, and physical function.65 The main goal of these education-based programs is to instruct and train the patients to use techniques that can improve their symptoms. The programs can be either individualized or used in a group therapy setting. CBT also has been useful in management of other chronic pain conditions.
Other Non-pharmacologic Therapies. The various complementary and alternative medicine modalities66 that are used in addition to traditional therapies in the management of fibromyalgia include meditation-based stress reduction, acupuncture, trigger point or tender point injections,EMG biofeedback, hypnotherapy, massage therapy, energy therapy, chiropractic therapy, support groups, tai chi, and yoga. Some of them seem beneficial to individual patients, but controlled studies are essential to further investigate their effectiveness.
Patient Education. Patient education plays a crucial role in the success of treatment. Patients need to understand that fibromyalgia is a chronic illness and there is no instant cure. Also it is essential for the patient to realize the need to play an active role in the treatment plan and to develop a self-management program. Physicians should make every effort to build confidence and trust in these patients and provide positive reinforcement for their continued efforts.
Multidisciplinary Therapy. A multidisciplinary approach is essential in the management of fibromyalgia. It requires involvement of a variety of motivated health professionals. Treatment needs to be individualized. Optimal treatment includes a multifaceted regimen including symptom-based pharmacotherapy, non-pharmacologic therapies like aerobic exercise, and cognitive behavioral therapy with an emphasis on patient education.
Prognosis
Although fibromyalgia is a chronic condition, it is not life-threatening. With the availability of various types of treatment modalities, strongly motivated patients with fibromyalgia can be expected to lead a reasonably normal life. Many patients, however, refuse to exercise and end up miserably inactive. Patients with fibromyalgia have to take responsibility for getting well because recovery depends heavily on daily aerobic exercise.
References
1. Inanici F, Yunus MB. History of fibromyalgia: past to present. Curr Pain Headache Rep 2004;8:369-378.
2. Buskila D, Neumann L, Odes LR. The prevalence of musculoskeletal pain and fibromyalgia in patients hospitalized on internal medicine wards. Semin Arthritis Rheum 2001;30:411-417.
3. Neumann L, Buskila D. Epidemiology of fibromyalgia. Curr Pain Headache Rep 2003;7:362-368.
4. White KP, Harth M. Classification, epidemiology, and natural history of fibromyalgia. Curr Pain Headache Rep 2001;5:320-329.
5. Abeles AM, Pillinger MH, Solitar BM, et al. Narrative review: The pathophysiology of fibromyalgia. Ann Intern Med 2007;146:726.
6. Buskila D, Sarzi-Puttini P. Biology and therapy of fibromyalgia. Genetic aspects of fibromyalgia syndrome. Arthritis Res Ther 2006;8:218.
7. Arnold LM, Hudson JI, Hess EV, et al. Family study of fibromyalgia. Arthritis Rheum 2004;50:944.
8. Offenbaecher M, Bondy B, de Jonge S, et al. Possible association of fibromyalgia with a polymorphism in the serotonin transporter gene regulatory region. Arthritis Rheum 1999;42:2482.
9. Wolfe F, Ross K, Anderson J, et al. The prevalence and characteristics of fibromyalgia in the general population. Arthritis Rheum 1995;38:19-28.
10. Aaron LA, Bradley LA, Alarcón GS, et al. Perceived physical and emotional trauma as precipitating events in fibromyalgia. Associations with health care seeking and disability status but not pain severity. Arthritis Rheum 1997;40:453-460.
11. Katon W, Sullivan M, Walker E. Medical symptoms without identified pathology: relationship to psychiatric disorders, childhood and adult trauma, and personality traits. Ann Intern Med 2001;134: 917-925.
12. Crofford LJ, Pillemer SR, Kalogeras KT, et al. Hypothalamic-pituitary-adrenal axis perturbations in patients with fibromyalgia. Arthritis Rheum 1994;37:1583-1592.
13. Russell IJ, Vaeroy H, Javors M, et al. Cerebrospinal fluid biogenic amine metabolites in fibromyalgia/fibrositis syndrome and rheumatoid arthritis. Arthritis Rheum 1992;35:550-556.
14. Winfield J B, Dale DC, Federman DD, Antman K, et al. Section 15, Chapter 13. ACP Medicine, 2008 , Vols 1 & 2. American College of Physicians: New York, NY; 2008.
15. Weissbecker I, Floyd A, Dedert E, et al. Childhood trauma and diurnal cortisol disruption in fibromyalgia syndrome. Psychoneuroendocrinology 2006;31:312.
16. Cohen H, Neumann L, Shore M, et al. Autonomic dysfunction in patients with fibromyalgia: Application of power spectral analysis of heart rate variability. Semin Arthritis Rheum 2000;29:217.
17. Yunus MB. Towards a model of pathophysiology of fibromyalgia: Aberrant central pain mechanisms with peripheral modulation [Editorial]. J Rheumatol 1992;19:846-850.
18. Desmeules JA, Cedraschi C, Rapiti E, et al. Neurophysiologic evidence for a central sensitization in patients with fibromyalgia. Arthritis Rheum 2003;48:1420.
19. Li J, Simone DA, Larson AA. Windup leads to characteristics of central sensitization. Pain 1999;79:75-82.
20. Roizenblatt S, Modlofsky H, Benedito-Silva AA, et al. Alpha sleep characteristics in fibromyalgia. Arthritis Rheum 2001;44:222.
21. Yunus MB. Fibromyalgia and overlapping disorders: The unifying concept of central sensitivity syndromes. Semin Arthritis Rheum 2007;36:339.
22. McBeth J, Silman AJ. The role of psychiatric disorders in fibromyalgia. Curr Rheumatol Rep 2001;3:157-164.
23. Aaron LA, Buchwald D. A review of the evidence for overlap among unexplained clinical conditions. Ann Intern Med 2001;134:868-881.
24. Wolfe F, Smythe HA, Yunus MB, et al. The American College of Rheumatology 1990 criteria for the classification of fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum 1990;33:160-172.
25. Burkham J, Harris ED Jr, et al. Fibromyalgia: A chronic pain syndrome. In: Kelley's Textbook of Rheumatology, 7th ed. Philadelphia: Elsevier Saunders; 2005:522-536.
26. Marcus DA, Bernstein C, Rudy TE. Fibromyalgia and headache: An epidemiological study supporting migraine as part of the fibromyalgia syndrome. Clin Rheumatol 2005;24:595.
27. Okifuji A, Turk DC, Sinclair JD, et al. A standardized manual tender point survey. I. Development and determination of a threshold point for the identification of positive tender points in fibromyalgia syndrome. J Rheumatol 1997;24:377.
28. Burckhardt CS, Clark SR, Bennett RM. The fibromyalgia impact questionnaire: Development and validation. J Rheumatol 1991;18:728-733.
29. Burckhardt CS, Goldenberg D, Crofford L, et al. Guideline for the management of fibromyalgia pain in adults and children. National Guideline Clearinghouse. (Accessed August 2007.)
30. Goldenberg DL, Burckhardt C, Crofford L. Management of fibromyalgia syndrome. JAMA 2004;292:2388-2395.
31. Carville SF, Arendt-Nielsen S, Bliddal H, et al. EULAR evidence-based recommendations for the management of fibromyalgia syndrome. Ann Rheum Dis July 20, 2007
32. Burckhardt CS. Nonpharmacologic management strategies in fibromyalgia. Rheum Dis Clin North Am 2002;28:291-304.
33. Sawynok J, Esser MJ, Reid AR. Antidepressants as analgesics: An overview of central and peripheral mechanisms of action. J Psych Neurosci 2001;26:21-29.
34. O'Malley PG, Balden E, Tomkins G, et al. Treatment of fibromyalgia with antidepressants: a meta-analysis. J Gen Intern Med 2000;15:659-666.
35. Clauw DJ. Pharmacotherapy for patients with fibromyalgia. J Clin Psychiatry 2008;69[suppl 2]:25-29.
36. Goldenberg DL, Burckhardt C, Crofford L. Management of fibromyalgia syndrome. JAMA 2004;292:2388-2395.
37. Tofferi JK, Jackson JL, O'Malley PG. Treatment of fibromyalgia with cyclobenzaprine: A meta-analysis. Arthritis Rheum 2004;51:9.
38. Goldenberg DL, Mayskiy M, Mossey CJ, et al. A randomized, double-blind crossover trial of fluoxetine and amitriptyline in the treatment of fibromyalgia. Arthritis Rheum 1996;39:1852.
39. Arnold LM, Hess EV, Hudson JI, et al. A randomized, placebo-controlled, double-blind, flexible-dose study of fluoxetine in the treatment of women with fibromyalgia. Am J Med 2002;112:191.
40. Wolfe F, Cathey MA, Hawley DJ. Double-blind placebo controlled trial of flluoxetine in fibromyalgia. Scand J Rheumatol 1994;23:255.
41. Cantini F, Bellandi F, Niccolo L, et al. Fluoxetine combined with cyclobenzaprine in the treatment of fibromyalgia. Minerva Med 1994;85:97-100.
42. Celiker R, Cagavi Z. Comparison of amitriptyline and sertraline in the treatment of fibromyalgia syndrome [abstract]. Arthritis Rheum 2000;43:S332.
43. Arnold LM, Lu Y, Crofford LJ, et al. A double-blind multicenter trial comparing duloxetine to placebo in the treatment of fibromyalgia with or without major depressive disorder. Arthritis Rheum 2004;50:2974-2984.
44. Zijsltra TR, Barendregt PJ, van de Laar MA. Venlafaxine in fibromyalgia: results of a randomized placebo-controlled, double-blind trial [abstract]. Arthritis Rheum 2002;46:S105.
45. Gendreau RM, Mease PJ, Rao SR, et al. Minacipran: A potential new treatment of fibromyalgia [abstract]. Arthritis Rheum 2003; 48:S616.
46. Russell IJ, Kamin M, Bennett RM, et al. Efficacy of tramadol in treatment of pain in fibromyalgia. J Clin Rheumatol 2000;6: 250-257.
47. Biasi G, Manca S, Manganelli S, et al. Tramadol in the fibromyalgia syndrome: a controlled clinical trial versus placebo. Int J Clin Pharmacol Res 1998;18:13-19.
48. Bennett RM, Kamin M, Karim R, et al. Tramadol and acetaminophen combination tablets in the treatment of fibromyalgia pain: a double-blind, randomized, placebo-controlled study. Am J Med 2003;114:537-545.
49. Rao SG, Bennett RM. Pharmacological therapies in fibromyalgia. Best Pract Res Clin Rheumatol 2003;17:611-627.
50. Dworkin RH, Backonja M, Rowbotham MC, et al. Advances in neuropathic pain: diagnosis, mechanisms, and treatment recommendations. Arch Neurol 2003;60:1524-1534.
51. Tofferi JK, Jackson JL, O'Malley PG. Treatment of fibromyalgia with cyclobenzaprine: A meta-analysis. Arthritis Rheum 2004;51: 9-13.
52. Share NN, McFarlane CS. Cyclobenzaprine: A novel centrally acting skeletal muscle relaxant. Neuropharmacology 1975;14:675-684.
53. Crofford LJ, Rowbotham MC, Mease PJ, et al. Pregabalin for the treatment of fibromyalgia syndrome: Results of a randomized, double-blind, placebo-controlled trial. Arthritis Rheum 2005;52: 1264-1273.
54. Arnold LM, Russell J, Duan R, et al. Pregabalin for management of fibromyalgia syndrome (FDS): a 14-week randomized double-blind, placebo-controlled, monotherapy trial (abstr P03.075). Annual Meeting of American Academy of Neurology, Boston, April 28-May 5, 2007.
55. Crofford LJ, Simpson SI, Young JP, et al. A 6-month, double-blind, placebo-controlled trial of pregabalin as treatment of fibromyalgia syndrome: The Fibromyalgia Relapse Evaluation and Efficacy for Durability of Meaningful Relief (FREEDOM) trial (abstr OP0035). Annual Meeting of American Psychiatric Association, San Diego, May 19-24, 2007.
56. Backonja M, Glanzman RL. Gabapentin dosing for neuropathic pain: Evidence from randomized, placebo-controlled clinical trials [review]. Clin Ther 2003;25:81-104.
57. Arnold LM, Goldenberg DL, Stanford SB, et al. Gabapentin in the treatment of fibromyalgia: A randomized, double-blind, placebo-controlled, multicenter trial. Arthritis Rheum 2007;56:1336-1344.
58. Richards SC, Scott DL. Prescribed exercise in people with fibromyalgia: Parallel group randomised controlled trial. BMJ 2002;325:185.
59. Busch A, Schachter CL, Peloso PM, et al. Exercise for treating fibromyalgia syndrome. Cochrane Database Syst Rev CD003786, 2002.
60. Sprott H. What can rehabilitation interventions achieve in patients with primary fibromyalgia? Curr Opin Rheumatol 2003;15: 145-150.
61. Jentoft ES, Kvalvik AG, Mengshoel AM. Effects of pool-based and land-based aerobic exercise on women with fibromyalgia/chronic widespread muscle pain. Arthritis Rheum 2001;45:42-47.
62. Mannerkorpi K, Nyberg B, Ekdahl C. Pool exercise combined with an education program for patients with fibromyalgia syndrome: A prospective, randomized study. J Rheumatol 2000;27:2473-2481.
63. Nielson WR, Walker C, Mccain GA. Cognitive behavioral treatment of fibromyalgia syndrome: preliminary findings. J Rheumatol 1992; 19:98-103.
64. White KP, Nielson WR. Cognitive behavioral treatment of fibromyalgia syndrome: A follow-up assessment. J Rheumatol 1995;22:717-721.
65. Williams DA. Psychological and behavioral therapies in fibromyalgia and related syndromes. Best Pract Res Clin Rheumatol 2003;17:649-665.
66. Ernst E. Complementary and alternative medicine in fibromyalgia. In: Wallace DJ, Clauw DJ, editors. Fibromyalgia and Other Central Pain Syndromes. Philadelphia: Lippincott, Williams and Wilkins; 2005:389-394.
This issue deals with a common but often frustrating and unrewarding condition that confronts primary care physicians: fibromyalgia.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
