Hematuria: Implications and Management
Hematuria: Implications and Management
Author: Priya Jain, MD, Hematology/Oncology Specialist, Private Practice, Dayton, OH.
Peer Reviewer: Siegmund Teichman, MD, Professor of Medicine; Chief, Division of Nephrology, Loma Linda University, Loma Linda, CA.
Primary care physicians often are confronted with patients presenting with either gross or microscopic hematuria. Either situation demands a clinical investigation. Gross hematuria typically calls for a referral to a urologist. In today's cost-conscious mindset, it is important to work with the urologist collaboratively toward an effective and timely work-up that avoids unnecessarily expensive or unproductive tests. Microscopic hematuria typically begins with a careful history and thoughtful investigation, most often to exclude occult neoplastic causes, and may more often involve the expertise of a nephrologist. Commonly, however, the presence of microscopic hematuria can be intermittent and the work-up unrevealing, requiring regular follow-up.
This issue highlights: definition of hematuria; epidemiology; etiology; diagnostic work-up; and common causative conditions and therapeutic implications.
The Editor
Introduction
Evaluation of hematuria is a common problem in the primary practice setting. Hematuria may be distinguished between gross hematuria and microscopic hematuria. Differentiating between the two types is important in both evaluation and clinical management of either cause. Microscopic hematuria often is an incidental finding on urinalysis, whereas the patient often calls attention to manifestations of gross hematuria. In certain cases, the evaluation of the patient can yield no cause of the symptoms; however, a thorough evaluation is necessary given that the differential can extend from infection and nephrolithiasis to renal disease and malignancy.
Definition of Microscopic vs. Gross Hematuria
It is important to distinguish the difference in defining microscopic hematuria from macroscopic or gross hematuria. Microscopic hematuria is found on a urine dipstick or urinalysis often done for another purpose. The American Urological Association defines clinically significant microscopic hematuria as three or more red blood cells per high power field on microscopic evaluation on two of three carefully obtained specimens.1 Note that others may advocate two or more cells as abnormal but that lowering or increasing the cutoff can result in either increasing the number of false positives or missing manifestations of significant pathology.2 The urine sediment (or direct counting of red blood cells [RBC] per mL of uncentrifuged urine) is the gold standard for the detection of microscopic hematuria. Dipstick for heme can detect as low as 1-2 RBCs per high power field but may lead to detection of a higher number of false positives in the presence of myoglobinuria, alkaline urine with a pH greater than 9, or the presence of semen in the urine. Given the intermittent nature in which hematuria may present, some advocate for patients to have two of three specimens positive before having a formal evaluation.3
Therefore, if a dipstick is found to be positive, it should be confirmed with a microscopic analysis of the urine. If it is found to be negative, this virtually excludes the presence of RBC in the urine.4 However, false-negative urinalysis has been noted in patients who ingest large amounts of vitamin C.5
The primary care physician may be alerted to the presence of gross hematuria based on the patient's history of brownish or red urine. The presence of as little as 1 mL of blood per liter of urine can cause this discoloration, so a thorough evaluation is necessary to help differentiate the cause. The initial step is to centrifuge the urine specimen to see if the source of discoloration is from the supernatant or urine sediment. (See Figure 1.)
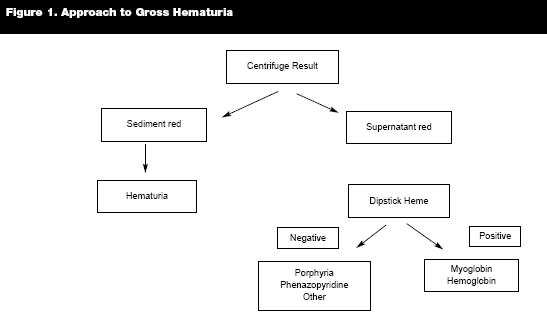 |
In menstruating females, to prevent contamination of the specimen from menstrual blood, the test should be repeated after bleeding has ceased. Gross hematuria will result in detection of heme in the urinary sediment. Detection of heme in the supernatant can be caused by myoglobinuria and hemoglobinuria, which should be distinguished from gross hematuria given the different causes associated with each pathology. Separating extraglomerular causes from glomerular causes of gross hematuria is important given the different evaluation process of each cause. The presence of blood clots in the setting of gross hematuria is typically associated with extraglomerular causes affecting the lower genitourinary tract. An exception may be IgA nephropathy and Alport's disease.
In defining gross versus microscopic hematuria, it is important to note that typically neither situation causes any immediate danger to the patient, with the exception of gross hematuria caused by extraglomerular causes that may lead to obstruction of the ureter or, in trauma, can lead to massive hemorrhage. In these cases, immediate referral to a urologist is necessary. Also, in defining the symptoms the patient presents with, it is important to note the age of the patient and the time frame of the symptoms.6 Unfortunately, even in older patients, often a source of the symptoms cannot be identified despite an extensive workup.7
Epidemiology
The prevalence of microscopic hematuria varies according to the age and gender of the population. The type of testing useddipstick testing alone or combined with microscopic analysisalso affects the number of positive screening tests. Studies have reported prevalence as low as less than 1% to as high as 22%, depending on the characteristics of the populations studied.8-9 In a large screening study of 1000 adult patients, with the screening cutoff of greater than three RBC per high power field in two of three samples, the prevalence was only 0.1%, with an increase with age older than 50 years.2 Note that microscopic hematuria, when found, can be persistent or transient. More commonly, transient hematuria may be precipitated by vigorous exercise, sexual intercourse, contamination, or trauma.
Screening for microscopic hematuria is controversial and has not been recommended for the general population. Although bladder cancer is the most common malignancy associated with microscopic hematuria, the decision to screen lies with the physician and consideration of risk factors for underlying disease. (See Table 1.)
 |
Etiology
The cause of hematuria in the adult patient can vary in clinical significance. Causes can be secondary to benign sources, such as urinary tract infection and prostatic hyperplasia, to more severe or life-threatening causes such as bladder or renal cell malignancies. A thorough evaluation of both the upper urinary tract followed by cystoscopy can fail to identify the source of hematuria in a large number of patients. Based on several large studies, the percentage of nondiagnostic workups can range from 20-60%.10-13 In younger patients, the likelihood of a nondiagnostic workup is higher.14 (See Tables 2 and 3.)
 |
 |
The presence of additional abnormalities, such as proteinuria, red cell casts, or renal insufficiency, should prompt a referral to a nephrologist. These features may suggest the presence of renal parenchymal disease.
It is important to exclude benign causes such as menstruation, vigorous exercise, sexual activity, viral illness, trauma, and infection. These causes are commonly seen in a younger population presenting with hematuria. A thorough evaluation of the medication list can reveal some common medications that can cause hematuria. It should be noted that pathological gross hematuria can occur in younger individuals in a variety of conditions from bladder cancer, renal cystic diseases, and stones. (See Table 4.)
 |
Patients older than 40 years with hematuria are at greater risk for significant disease. Other risk factors for significant disease that warrant a thorough workup include smoking, exposure to chemicals or dyes, analgesic abuse, and history of pelvic irradiation.15
Diagnostic Studies and Evaluation
As noted, a thorough history and physical examination and assessment are essential to assess a patient's risk factors before proceeding with a radiographic evaluation of the upper urinary tract, urine cytology, cystoscopy, or a referral to a nephrologist. A diagnostic algorithm for the management and diagnosis of microscopic hematuria is shown. An evaluation of genitourinary (GU) pathology is also warranted in older individuals on anticoagulation therapy, since the bleeding could originate from a malignancy. (See Figures 2 and 3.)
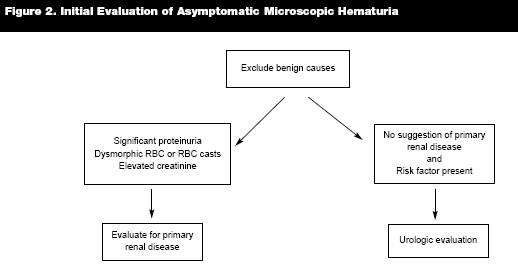 |
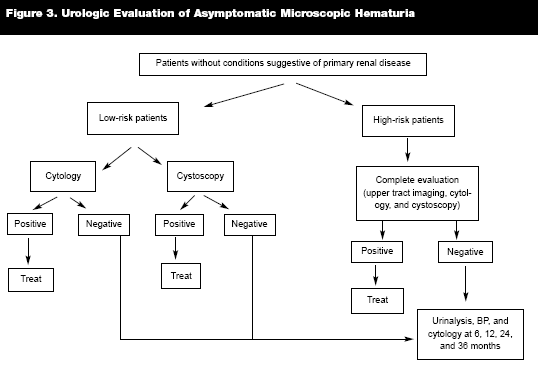 |
Radiographic Assessment of the Upper GU Tract. The upper urinary tract can be evaluated by the use of intravenous excretory urography, ultrasonography, and computed tomography. The goal of such imaging is to identify sources of hematuria, including renal carcinoma, urothelial carcinoma of the pelvicaliceal system and ureter, urolithiasis, renal infection, and benign conditions such as medullary sponge kidney disease, also known as papillary ectasia.
The intravenous excretory urogram (IVEU) has been the traditional initial approach for the evaluation of the upper urinary tract.16 This study defines the anatomy of the urologic tract from the kidney to the bladder. It is a relatively inexpensive and easily available diagnostic tool. However, it has not proved as sensitive in detection of smaller renal tumors. One study noted that intravenous urography identified 85% of lesions greater than 3 cm but only 21-52% of smaller lesions.17 Once a mass is detected using IVEU, further evaluation is needed using ultrasonography and computed tomography given that IVEU cannot distinguish solid masses from cystic ones.
Ultrasound is a less expensive and safer means for evaluation of microscopic hematuria because it does not expose the patient to intravenous contrast. It is limited in evaluating tumors less than 3 cm in diameter but is sensitive in detection of masses greater than 3 cm, cysts, and hydronephrosis.17
Computed tomography (CT) has a high sensitivity for identifying renal calculi.18 Unenhanced CT has replaced many other imaging techniques, including ultrasound and plain radiography, as the test of choice in patients with renal colic and microscopic hematuria. Although more expensive, contrast-enhanced CT has a higher sensitivity compared to ultrasonography and intravenous urography in detection of smaller renal masses. It also can be used to characterize a mass as cystic or solid and to allow for surgical staging in the setting of a malignancy.
Although not widely used and even more expensive, MRI can be used to assess the upper urinary tract. The sensitivity of CT, however, approximates that of MRI in detection of smaller masses.16
Iodinated contrast should be used cautiously in patients with impaired renal function, serum creatinine over 3 mg/dL (risk for developing AKI), and contrast MRI should be avoided if possible in patients with eGFR below 35 mL/min (risk of developing nephrogenic systemic fibrosis).
Radiographic Assessment of the Lower GU Tract. Assessment of the lower urinary tract can be necessary despite finding an abnormality in the upper tract, given the possibility of two separate lesions. Assessment of the lower urinary tract includes both urine cytology and cystoscopy. The cause of asymptomatic hematuria can be unknown in up to 70% of patients even after a thorough evaluation of the upper urinary tract.19
Cytology and Tumor Markers. Urothelial cancer of the collecting system, ureter, or bladder is the most common malignancy detected in patients with microscopic hematuria. Cytologic evaluation of cells within the urine is an easy, noninvasive method of detecting urothelial malignancy. However, cytology is very specific, but relatively insensitive (40-76%) for the detection of bladder cancers in comparison to cystoscopy and can be less useful in detection of low-grade tumors.20 The sensitivity is highest for high-grade lesions in the bladder and carcinoma in situ. Its primary advantage is its noninvasiveness in comparison to cystoscopy.
Positive cytology without evidence of an abnormality in the bladder should prompt an evaluation of the upper urinary tract and the prostatic urethra. If no clear lesion is found, washings of the ureters and renal pelvis can be performed.
Urine cytology specimens must be collected and stored properly to maintain a high level of diagnostic accuracy. Specimens obtained from a urinary catheter can lead to coalescing of cells in papillary groups, resulting in difficult interpretation and misdiagnosis of a low-grade TCC. Inflammation and chronic infection can cause degenerative cellular changes and atypica that can lead to misinterpretation. Care should be taken to avoid contamination with vaginal, cervical, or endometrial cells.
The role of urine flow cytometry in screening for urothelial malignancies is not clear. Flow cytometry is an automated procedure, requiring much less labor than cytology, but requires a catherized specimen. One of the drawbacks is the need for a sample with a large number of cells, making it less useful in the detection of CIS, where small populations of aneploid populations can exist.21
However, thus far, it appears that both cytology and flow cytometry used together do not appear to increase the diagnostic yield.22
The desire to find more noninvasive screening methods has led to the evaluation of newer tests such as urine histochemistry (ImmunoCyt test) and proteomics assays for the nuclear matrix protein NMP22 (NMP22 BladderChek test). These tests may hold more promise in the detection of lower-grade tumors.23
Urine histochemistry utilizes three monoclonal antibodies specific for two antigens expressed on the surface of TCC tumor cells. Urine histochemistry appears to be more sensitive than cytology in the detection of lower grade tumors, whereas cytology seems to have a higher sensitivity in detection of higher-grade tumors.24 However, the sensitivity of the histochemistry test does not seem high enough to replace cystoscopy.
Proteomics involves the analysis of protein expression in tissues, serum, and other biologic samples to identify or characterize malignant tumors on the basis of unique protein expression patterns. In a large study of more than 1300 patients with multiple risk factors for bladder carcinoma, all the patients underwent cystoscopy, with the comparison of urine cytology to the assay for NMP 22. Of the patients diagnosed with bladder carcinoma, the NMP assay was found to be more sensitive than cytology for lower- and intermediate-grade tumors.23
Other urinary biomarkers are being evaluated and have been found to have some utility in monitoring for recurrent carcinoma, but have not proven to be useful as a screening technique and have not eliminated the need for cystoscopy. These markers include qualitative fluorescence image analysis, molecular cytogenesis, telomerase expression, tumor associated intracellular or secreted products, oncogene mutations, microsatellite alterations, and markers of apoptosis.25
Cystoscopy. The disadvantages of cystoscopy are the discomfort of an invasive procedure and the limited ability to detect carcinoma in situ.26 However, it is the only accurate method of detecting a transitional cell carcinoma of the bladder and urethra.27
Cystoscopy has a lower yield in patients younger than 40 years with no risk factors for malignancy.28 In these select patients, cystoscopy may be deferred, but urine cytology, given its noninvasiveness, can be considered.
Bilateral ureteral brush biopsy, ultrasound, or endoscopy may be needed when transitional malignancy of the ureter is suspected, such as in a patient with analgesic-associated renal disease.
Glomerular vs. Urologic Hematuria
The differential diagnosis of hematuria is wide and can require management from different specialists, and this has led the attempt to find tests that could categorize asymptomatic microscopic hematuria as a marker for renal or urologic disease. As RBCs enter the urine as a result of glomerular bleeding, they pass through the renal tubules and can be subjected to high transmembrane osmotic gradients. This can lead to shrinking of cells, creating an irregular "crenated" shape in place of the normal biconcave disks of the RBC.29 The presence of such RBC has been evaluated by phase-contrast microscopy or automated size analysis in a Coulter counter. However, in a double-blind controlled comparison of the findings of two blinded observers using phase contrast microscopy, this technique was not successful.30 No larger-scale studies have been conducted to validate the use of these techniques in helping exclude causes of hematuria.
Management of Separate Diagnosis
The management of these patients is dictated by the diagnostic workup. For patients with an initial negative evaluation for asymptomatic microhematuria, most do not develop significant urologic disease. Yet, for a small percentage of patients, hematuria can precede the diagnosis of bladder cancer by many years, and some followup is necessary in higher-risk patientsespecially older patients with a history of smoking or occupational exposures.28 However, because the data regarding follow-up in such patients are sparse, recommendations regarding such follow-up are based on literature-based evidence and are not standardized. Based on AGU guidelines, consideration could be given to repeating urinalysis and urine cytology at six, 12, 24, and 36 months.2 After three years, the patient does not require further urologic monitoring. If a higher level of suspicion exists, consideration can be given to cystoscopy or further radiologic imaging.
Patients with hypertension, proteinuria, or evidence of glomerular bleeding in the form of red cell casts or dysmorphic red blood cells should be referred to a nephrologist for further monitoring and evaluation.
For patients found to have a suspicious malignancy, both the urologist and medical oncologist share joint management, with the help of radiation oncology in certain situations. Renal masses found to be suspicious for malignancy are removed surgically by urologists, without biopsies in most situations, given concern for tumor cell seeding with needle biopsy. In the last five years, with the availability of targeted receptors, the clinical outcome for patients with advanced renal cell malignancies has improved dramatically. Urothelial malignancies to the ureter, renal pelvis, urethra, and bladder are managed with surgery alone in the early stage, but require surgery, platinum-based chemotherapy, and radiation therapy for more advanced disease.
Renal Malignancies
With approximately 38,000 cases diagnosed per year in the United States, renal cell carcinoma accounts for only about 2.5% of all malignancies.31 Approximately 25% of patients present with locally advanced invasive or metastatic disease. Approximately 30% of patients with resected disease have a recurrence.32 Given that the median survival for patients with advanced disease is slightly more than one year, there has been a focus on the development of new treatments.
As noted above, from the office of the primary care physician, hematuria can be the presenting symptom. Other suspicious symptoms include flank pain, fatigue, weight loss, and a palpable abdominal mass on physical examination. About 2% of diagnoses are associated with hereditary syndromes. The peak age of presentation is the 60s to 70s, and a there is an increased incidence in men, with a 1.6:1 ratio of men to women.31 Risk factors include obesity, smoking, and hypertension.33 Other adverse prognostic factors after diagnosis include poor functional status, high lactate dehydrogenase, a low hemoglobin level, and a high calcium level.
Surgical excision is the primary treatment for early stage renal-cell carcinoma. Radical nephrectomy, which includes removal of the kidney en bloc with Gerota's fascia, the ipsilateral adrenal gland, and regional lymph nodes, has been the standard therapy. The surgical approach is determined by the size and location of the tumor within the kidney, the stage of the tumor, and any other special anatomical considerations. Even in the setting of metastatic disease, there may be a role for nephrectomy. The combination of nephrectomy and interferon alfa has been shown to be superior to interferon alfa alone in advanced disease.34 In the setting of isolated metastasis, there is a role for resection of both the primary site of disease and of the isolated metastasis.
In the last few years, the treatment of advanced renal cell cancer has been revolutionized by new targeted therapies. Rates of response to chemotherapy alone are low (roughly 4-6%).35 Prior to this treatment, options were limited mainly to immunotherapy, with IL2 in selected patients with a good performance status with metastatic disease, and with interferon alpha in the adjuvant setting after nephrectomy. In a novel phase III trial, sunitinib, an orally administered inhibitor of tyrosine kinases, including vascular endothelial growth factor receptor (VEGF) and platelet-derived growth factor receptor (PDGFR), was compared to interferon alfa in previously untreated patients with metastatic disease. Progression-free survival, the primary endpoint, was found to be significantly longer with higher response rates in patients receiving sunitinib compared to patients who received interferon alfa.36 These receptor tyrosine kinases play an important role in the pathogenesis of clear cell carcinoma, the most common histologic diagnosis among renal cell carcinoma. This key role in pathogenesis occurs through involvement of the tumor suppressor gene, the von Hippel-Lindau gene, which is activated in a majority of sporadic cases by deletion, mutation, or methylation.
Following this study, another pivotal phase III study comparing sorafenib to placebo in patients with advanced renal cell carcinoma resistant to standard treatment showed that sorafinib prolonged progression-free survival compared to placebo. Sorafenib is also an orally active multikinase inhibitor, with effects on tumor cell proliferation and angiogenesis.37
With the results of these two phase III studies, the oral tyrosine kinase inhibitors have transformed the treatments of metastatic renal cell carcinoma. Currently, these targeted treatments are being studied in the adjuvant setting following surgery. The toxicities associated with these drugs commonly seen by both the oncologist and primary care physicians include diarrhea, hypertension, hand-foot-skin reactions, alopecia, nausea, bleeding, and cardiovascular events. Notably, patients receiving sunitinib can have a decline in their ventricular function. However, with discontinuing the medication, the cardiomyopathy is reversible.38
Another new agent, temsirolimus, was studied in poor prognosis patients with advanced disease. Temsirolimus is an inhibitor of the mammalian target of rapamycin (mTOR) kinase, a component of intracellular signaling pathways involved in the growth and proliferation of cells. A phase III trial compared temsirolimus to interferon alfa to the combination of both drugs in previously untreated, poor-prognosis metastatic renal-cell carcinoma. Overall survival and progression-free survival were improved in the temsirolimus group compared to interferon alfa alone. The addition of interferon alfa did not improve survival.39 The primary toxicities of temsirolimus included asthenia, rash, anemia, nausea, dyspnea, diarrhea, peripheral edema, hyperlipidemia, and hyperglycemia. This trial provided a new target, mTOR, in renal cell carcinoma. This drug has yet to be compared to the oral tyrosine kinase inhibitors in advanced carcinoma.
Clear cell histology is the most common histology in renal cell malignancies. These new medications were tested primarily in patients with clear cell histology. However, other histologies, such as papillary renal cell, chromophobe, collecting duct renal cell carcinoma, or oncocytoma, follow a different clinical course. Chromophobe renal cell carcinoma and oncocytoma follow a more benign course, whereas collecting duct carcinomas follow a more aggressive course. Chemotherapy may be more effective for collecting duct carcinoma. Papillary carcinoma follows a more favorable course when localized, but a more aggressive course if metastatic in contrast to clear cell histology. Therefore, the histology must be considered prior to starting the treatments available in renal cell carcinoma. Patients with impaired renal function and a small, well localized renal malignancy may be candidates for partial nephrectomy.
Urothelial Malignancies
Bladder cancer is the most common malignancy of the urinary system and the most common malignancy associated with microscopic hematuria. Transitional cell carcinoma (TCC), also known as urothelial carcinoma, is the predominant histology in the United States and accounts for 90% of bladder carcinoma. The most common risk factors for bladder carcinoma include smoking and occupational exposures. In regions outside the United States where schistosomiasis is more prevalent, nonurothelial histologies are more common. Transitional cell carcinoma also can involve the renal pelvis, ureter, and urethra.
In 2008, it was estimated that approximately 68,000 cases of bladder carcinoma were diagnosed in the United States.40 Bladder cancer typically is diagnosed in older individuals, with a median age of 69 in men and 71 in women.41 Although it is rare, bladder cancer can present in younger patients, but it usually is low grade, non-invasive disease.42 There is a higher risk of bladder cancer in white men, with roughly twice the incidence as African American and Hispanic men.43
Environmental exposures are a risk factor for urothelial carcinoma. The urothelium that lines the mucosal surfaces of the urinary tract is exposed to environmental carcinogens. This "field cancerization" effect is a hypothesis to explain the multifocal nature of carcinomas of the lower and upper urinary tract.44 Potential occupational carcinogens include benzene, polyaromatic hydrocarbons, and diesel exhausts.
Several agents have been evaluated as chemopreventive agents in the prevention of bladder cancer. Retinoids have been studied in both epidemiologic studies and in clinical trials. In a primary chemoprevention trial, 29,000 male smokers were randomized to beta-carotene, alpha-tocopherol (a vitamin E precursor), both, or placebo in the ATBC study. At a follow-up of 6.1 years, there was no statistical difference in the number of bladder carcinoma diagnoses in each treatment group.45 Other vitamin A derivatives, pyridoxine, vitamin E, and asorbic acid have been evaluated as chemopreventive agents, but no clear role has been established in either primary or secondary chemoprevention.
TCCs of the renal pelvis and ureter are thought to be due to the same etiologic factors as TCC of the bladder. Given the multifocal nature of these tumors, patients with a urothelial cancer of the upper urinary tract should have close surveillance for a tumor of the lower urothelial tract or the contralateral urinary tract. In a series of 82 patients who had complete resection of a TCC of the renal pelvis or ureter, urothelial carcinoma of the bladder was subsequently diagnosed in 44% of the patients at a median interval of 14 months.46
The clinical presentation of bladder cancer can range from superficial disease to invasive or metastatic disease. In the United States, the approach for high-grade, superficial bladder cancer is transurethral resection followed by intravesicular treatment with BCG. For patients with disease refractory to BCG, the treatment would then be radical cystectomy. Some patients with a projected longer survival proceed directly to immediate cystectomy, given the 50% risk of progression to muscle invasive disease.47
Approximately 10-20% of patients with invasive bladder cancer have locally advanced, inoperable tumors. These patients can be offered cisplatin-based chemotherapy to try to render patients unresectable.48 Chemoradiation is an option for patients who desire bladder preservation, with salvage cystectomy reserved for patients who do not respond. Patients with a poorer performance status can be treated with radiation alone or chemotherapy for palliative purposes.
Whereas transitional cell or urothelial carcinoma comprise 90% of bladder cancer in North America and Europe, in areas of the world where infection with Schistoma haematobium is prevalent, nonurothelial carcinoma of the bladder is more common. These patients often present with hematuria and irritative symptoms. Nonurothelial carcinomas are classified as epithelial or nonepithelial. The large majority are epithelial, which include squamous cell, adenocarcinoma, and small cell carcinoma. The nonepithelial include sarcomas, pheochromocytomas, melanomas, and lymphomas.
In these areas, squamous cell cancer accounts for approximately 75% of cases, transitional cell carcinoma for 20%, and adenocarcinoma for 5%.49 Although the pathophysiology behind the development of nonurothelial carcinoma is not understood, both bladder irritation and metaplasia are thought to be contributing factors. Areas with increased incidence of Schistoma haematobium have a significant increase of bladder cancer, including squamous cell cancer, adenocarcinoma, mixed histology, and transitional cell carcinoma, with squamous cell being most common. Squamous cell carcinoma without infection usually presents with bulky tumors and locally advanced disease, although metastatic disease is present in only about 10% of patients.50 Treatment is primarily surgery, given that chemotherapy and radiation have not been shown to be successful in affecting outcome, and these patients have frequent local recurrences. Schistosomal bladder carcinoma usually presents at a younger age, more commonly in men. These tumors are more low- to moderate-grade. The standard management includes radical cystectomy and lymph node dissection. Small studies have shown that chemotherapeutic agents have activity in advanced disease, although these studies have been underpowered to show a clinical significance.
Glomerular Disease and Hematuria
Although malignancy is a strong consideration in the workup of older patients with hematuria who have specific risk factors, hematuria in young adults is more likely to be caused by renal parenchymal disease than malignancy. Unfortunately, given the rare cases of urinary tract malignancies in individuals younger than 40 years, each young patient must be considered individually in determining whether a referral to a urologist, nephrologist, or both is necessary. Several studies have shown that renal biopsy in patients with no urologic explanation of hematuria has shown glomerular disease in a significant number of patients.51-52
In a series, 165 patients with microscopic or macroscopic hematuria were referred to nephrologists and underwent renal biopsies. All of these patients had no obvious signs of renal disease: normal serum creatinine, no evidence of significant proteinuria with protein excretion of < 300 mg/24 hours on urine collections. Of these patients, abnormal findings were noted on renal biopsies in 46% of patients.53 Approximately one-third of the patients had IgA nephropathy, seven patients had thin basement membrane nephropathy, and 20 had various other forms of glomerular disease.
IgA Nephropathy
IgA nephropathy, also known as mesangial IgA disease or Berger's disease, is the most common primary glomerulonephritis in developed areas of the world.54 There is a peak incidence in the second and third decades of life, with a greater frequency in Asians and Caucasians.55 This disease can present with a wide spectrum of disease. Given that findings can be noted on renal biopsy, this diagnosis must be considered in otherwise relatively healthy patients with hematuria who have a relatively negative diagnostic workup. The more classic presentation is hematuriamore commonly gross hematuriaseveral days within the onset of a respiratory illness. Other presentations include nephrotic syndrome, hematuria and proteinuria, rapidly progressive glomerulonephritis, Henoch-Schoenlein purpura, chronic renal failure, or asymptomatic microscopic hematuria.56
The presence of IgA nephropathy is established only by renal biopsy. The pathognomonic finding is seen on immunofluorescence microscopy of the tissue, which demonstrates prominent, globular deposits of IgA (often accompanied by C3 and IgG) in the mesangium and, less often, along the glomerular capillary wall. IgA nephropathy is generally not associated with a marked cellular glomerular infiltration, which suggests that glomerular injury is mediated by resident glomerular cells. As IgG and complement components often are codeposited, IgA alone appears sufficient to provoke glomerular injury in the susceptible individual. This occurs predominantly through IgA-induced activation of mesangial cells and local complement activation.57
Some patients with IgA nephropathy have a coexisting diffuse thinning of the glomerular basement membrane that is indistinguishable from thin basement membrane nephropathy. It is not known whether these patients follow a different clinical course of patients with typical IgA nephropathy.58
Manifestations of IgA nephropathy are most often restricted to the kidney. However, mesangial IgA deposition can be seen with alcohol-related cirrhosis, celiac disease, and HIV infection.
Patients with IgA nephropathy who do not have evidence of proteinuria seem to have a lower risk of progression of their disease. Despite this, renal insufficiency and proteinuria can still develop in a percentage of patients over the long term, and these patients may progress to end-stage renal disease (ESRD).57 The possible role of persistent microscopic hematuria in predicting an adverse outcome in this group of patients is debated.54
The approach to the treatment of IgA nephropathy is not well standardized. Two approaches include symptomatic treatment of symptoms and manifestations as the disease progresses versus the use of corticosteroids. Attempts to slow progression include the use of angiotensin converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) to control blood pressures and monitoring of proteinuria. Corticosteroids can be used as treatment of underlying inflammatory disease.
Disease typically is monitored serially using the urine sediment, protein excretion (usually estimated from the protein-to-creatinine ratio), and the serum creatinine concentration. Hematuria is thought to mark the presence of persistent immunologic activity, but not necessarily of progressive disease. This finding may be a sign of a "smoldering" segmental necrotizing lesion, suggestive of "capillaritis."59 The presence of hematuria alone should not necessarily prompt the need for treatment, whereas the presence of moderately severe proteinuria may be an indicator of more severe disease and may mark the need for instituting treatment.
The two main nonimmusuppressive treatments include ACE inhibitors for blood pressure and proteinuria and statin therapy for lowering lipid levels. Small randomized studies have shown that both ACE inhibitors and ARBs are more effective than other antihypertensive drugs in slowing the progressive decline in glomerular filtration rate in IgA nephropathy.60 The addition of an ARB to an ACE inhibitor in patients with IgA nephropathy may produce a further antiproteinuric effect and has been evaluated in several trials.61 However, no randomized trials have shown that these combination regimens improve renal outcomes. Yet, given that a more antiproteinuric effect is thought to improve overall outcome, these treatments either alone or in combination continue to be used and studied.
Lipid-lowering with statins has been associated with a slower rate of loss of glomerular filtration rate in patients with mild to moderate chronic kidney disease (CKD).62 Both this and the association of chronic kidney disease and cardiovascular disease have led to the use of statin therapy as part of the management in patients with IgA nephropathy and manifestations of renal disease.
A variety of treatment regimens using corticosteroids, alone or in combination with other immmunotherapies, has been evaluated. Studies evaluating these treatments have not been conclusive, and often these studies have been small and have limited follow-up with conflicting results.63 In practice, most nephrologists will not treat mild or stable IgA nephropathy with glucocorticoids or other immunosuppressive therapies, given the limited evidence of benefit and toxicity from chronic use.64 Steroid therapy in such patients should only be administered when there is clinical and histologic evidence of active inflammation. Corticosteroid therapy for periods of 6 months to two years has been associated with a reduction in proteinuria and perhaps improved renal survival in certain studies, but this has not been a consistent finding.65 Immunosuppressive therapy should be considered only in patients with more severe disease as noted by a more rapidly progressive clinical course and histologic evidence of severe active inflammation.
Thin Glomerular Basement Membrane
Thin glomerular basement membrane disease, also previously known as benign familial hematuria, can be another common cause of asymptomatic glomerular bleeding. This disease can only be diagnosed by renal biopsy, with electron microscopy showing uniform thinning of the glomerular basement membrane (Lamina Densa). The most common manifestation of this disease is asymptomatic microscopic hematuria. The disease is inherited in an autosomal dominant pattern, and typically these patients have a good prognosis. Hypertension, proteinuria, and progressive renal failure are not common manifestations of this disease. However, progression in certain cases to end-stage renal failure has been reported.66 Typically these patients are observed and have an excellent prognosis for normal renal function.
Another glomerular disease reported in patients with asymptomatic microscopic hematuria is mesangial proliferative glomerulonephritis, similar to IgA disease, but without the presence of IgA deposits. The prognosis of this disease is similar to IgA nephropathy.
Summary
The implications of hematuria, either gross or microscopic, can be broad and range from renal, urologic, malignant, and benign causes. Given such a large differential diagnosis, the diagnostic workup can seem overwhelming. The patient's risk factors, history, and physical examination should dictate how to proceed. It is important to note that older patients, even if a renal cause may be suspected, deserve a full evaluation of the upper and lower urinary tract to exclude malignancy, given that these patients can be at increased risk. Also, an abnormality of the upper tract should not preclude an evaluation of the lower tract, given that patients can have anatomic abnormalities in both locations, which both warrant treatment. In patients who have a negative diagnostic evaluation for both renal and urologic disease, certain patients with asymptomatic hematuria warrant continued followup at future intervals to see if their symptoms resolve. This decision should include consideration of the risk factors for each patient and should be evaluated on an individualized basis.
References
1. Grossfeld GD, Wolf JS Jr, Litwin MS, et al. Asymptomatic microscopic hematuria in adults: Summary of the AUA best practice policy recommendations. Am Fam Physician 2001;63:1145-1154.
2. Sutton JM. Evaluation of hematuria in adults. JAMA 1990;263: 2475-2480.
3. Mariani AJ, Mariani MC, Macchioni C, et al. The significance of adult hematuria: 1000 hematuria evaluations including a risk-benefit and cost effective analysis. J Urol 1989;141:350-355.
4. Schroder FH. Microscopic hematuria. Requires investigation. BMJ 1994;309:70.
5. Brigden ML, Edgell D, McPherson M, et al. High incidence of significant urinary ascorbic acid concentrations in a west coast populationimplications for routine urinalysis. Clin Chem 1992;38:426.
6. Cohen RA, Brown RS. Clinical practice. Microscopic hematuria. N Engl J Med 2003;348:2330.
7. Khadra MH, Pickard RS, Charlton M, et al. A prospective analysis of 1,930 patients with hematuria to evaluate current diagnostic practice. J Urol 2000;163:524.
8. Woolhandler S, Pels RJ, Bor DH, et al. Dipstick urinalysis screening of asymptomatic adults for urinary tract disorders. JAMA 1989; 262:1214-1219.
9. Sutton JM. Evaluation of hematuria in adults. JAMA 1990;263: 2475-2480.
10. Khadra MH, Pickard RS, Charlton M, et al. A prospective analysis of 1,930 patients with hematuria to evaluate current diagnostic practice. J Urol 2000;163: 524-527.
11. Greene LF, O'Shaughnesy EJ Jr, Hendricks ED. Study of five hundred patients with asymptomatic microhematuria. J Am Med Assos 1956;161:610-613.
12. Ritchie CD, Bevan EA, Collier SJ. Importance of occult haematuria found at screening. Br Med J (Clin Res Ed) 1986;292: 681-683.
13. Tomson C, Porter T. Asymptomatic microscopic or dipstick haematuria in adults: Which investigations for which patients? A review of the evidence. BJU Int 2002;90:185-198.
14. Benson G, Brewer E. Hematuria algorithms for diagnosis. Hematuria in the adult and hematuria secondary to trauma. JAMA 1981; 246:993-995.
15. Jaffe JS, Ginsberg PC, Gill R, et al. A new diagnostic algorithm for the evaluation of microscopic hematuria. Urology 2001;57:889-894.
16. Gray Sears CL, Ward JF, Sears ST, et al. Prospective comparison of computerized tomography and excretory urography in the evaluation of asymptomatic microhematuria. J Urol 2002:168:2457-460.
17. Jamis-Dow CA, Choyke PL, Jennings SB, et al. Small (< or = 3-cm) renal masses: Detection with CT versus US and pathologic correlation. Radiology 1996;198:785-788.
18. Luchs JS, Katz DS, Lane MJ, et al. Utility of hematuria testing in patients with suspected renal colic: Correlation with unenhanced helical CT results. Urology 2002;59:839-842.
19. Mazhari R, Kimmel PL. Hematuria: An algorithmic approach to finding the cause. Cleve Clin J Med 2002;69:870, 872-874, 876.
20. Bakhos R, Shankey TV, Flanigan RC, et al. Comparative analysis of DNA flow cytometry and cytology of bladder washings: Review of discordant cases. Diagn Cytopathol 2000;22:65-69.
21. Gourlay W, Chan V, Gilks CB, et al. Screening for urothelial malignancies by cytologic analysis and flow cytometry in a community urology practice: A prospective study. Mod Pathol 1995;8:394-397.
22. Badalament RA, Kimmel M, Gay I, et al. The sensitivity of flow cytometry compared with conventional cytology in the detection of superficial bladder cancer. Cancer 1987;59:2078.
23. Grossman HB, Messing E, Soloway M, et al. Detection of bladder cancer using a point-of-care proteonomic assay. JAMA 2005;293: 810-816.
24. Pfister, C, Chautard D, Devonec M, et al. Immunocyt test improves the diagnostic accuracy of urinary cytology: Results of a French multicenter study. J Urol 2003;169:921-924.
25. Konety BR, Getzenberg RH. Urine based markers of urologic malignancy. J Urol 2001;165:600-611.
26. Koss LG, Deitch D, Ramanathan R, et al. Diagnostic value of cytology of voided urine. Acta Cytol 1985;29:810-816.
27. Harper M, Arya M, Hamid R, et al. Haematuria: A streamlined approach to management. Hosp Med 2001;62:696-698.
28. Sparwasser C, Cimniak HU, Treiber U, et al. Significance of the evaluation of asymptomatic hematuria in young men. Br J Urol 1994;74:723-729.
29. Schichiri M. Differentiating glomerular and non-glomerular hematuria. Lancet 1988;2:446-447.
30. Venkat Raman G, Pead L, Lee HA, et al. A blind controlled trial of phase-contrast microscopy by two observers for evaluating the source of haematuria. Nephron 1986;44:304-308.
31. Jemal A, Murray T, Ward E, et al. Cancer Statistics, 2005. CA Cancer J Clin 2005;55:10-30.
32. Cohen H. Renal cell carcinoma. N Engl J Med 2005;353: 2477-2490.
33. Chow WH, Gridley G, Fraumeni JF Jr, et al. Obesity, hypertension, and the risk of kidney cancer in men. N Engl J Med 2000;343: 1305-1311.
34. Flanigan RC, Salmon SE, Blumenstein BA, et al. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med 2001;345:1655-1659.
35. Yagoda A, Abi-Rached B, Petrylak D. Chemotherapy for advanced renal-cell carcinoma: 1983-1993. Semin Oncol 1995;22:42-60.
36. Motzer RJ, Hutson, Thomas, et al. Sunitinb versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 2007;356:115-124.
37. Escudier, Bernard, Eisen T, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Eng J Med 2007;356:125-134.
38. Schmelzle T, Hall MN. TOR, a central controller of cell growth. Cell 2000;103:253-262.
39. Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon alfa, or both for advanced renal cell carcinoma. N Eng J Med 2007;356:2271-2281.
40. Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin 2008;58:71-96.
41. Lynch CF, Cohen MB. Urinary system. Cancer 1995;75:316-329.
42. Lynn JF, Sesterhenn I. The molecular characteristics of bladder cancer in young patients. J Urol 1998;159:1493-1496.
43. Howe HL, Wu X, Ries LA, et al. Annual report to the nation on the status of cancer, 1975-2003, featuring cancer among U.S. Hispanic/Latino populations. Cancer 2006;15:1711-1742.
44. Kang CH, Yu TJ, Hsieh HH, et al. The development of bladder tumors and contralateral upper urinary tract tumors after primary transitional cell carcinoma of the upper urinary tract. Cancer 2003;19:1620-1626.
45. Virtamo J, Edward BK, Virtanen M, et al. Effects of supplemental alpha-tocopherol and beta-carotene on urinary tract cancer: incidence and mortality in a controlled trial (Finland). Cancer Causes Control 2000;11:933-939.
46. Raman JD, Sosa RE, Vaugh ED Jr, et al. Pathologic features of bladder tumors after nephroureterectomy or segmental ureterectomy for upper urinary tract transitional cell carcinoma. Urology 2007;69: 251-254.
47. Cookson MS, Herr HW, Zhang ZF, et al. The treated natural history of high risk superficial bladder cancer: 15-year outcome. J Urol 1997;158:62-67.
48. Dodd PM, McCaffrey JA, Herr H, et al. Outcome of postchemotherapy surgery after treatment with methotrexate, vinblastine, doxorubicin, and cisplatin in patients with unresectable or metastatic transitional cell carcinoma. J Clin Oncol 1999;17:2546-2552.
49. Ghoneim MA, el-Mekresh MM, el-Baz MA, et al. Radical cystectomy for carcinoma of the bladder: Critical evaluation of the results in 1,026 cases. J Urol 1997;158:393-399.
50. Rundle JS, Hart AJ, McGeorge A, et al. Squamous cell carcinoma of bladder. A review of 114 patients. Br J Ur 1982;54:522-526.
51. Arm JP, Peile EB, Rainford DJ. Significance of dipstick haematuria. Correlation with pathology. Br J Urol 1986;58:218-223.
52. Burkholder GV, Dotin LN, Thomason WB, et al. Unexplained heamturia. How extensive should the evaluation be? JAMA 1969; 210:1729-1733.
53. Topham PS, Harper SJ, Furness PM, et al. Glomerular disease as a cause of isolated microscopic hematuria. Q J Med 1994;87: 329-335.
54. D'Amico G. Natural history of idiopathic IgA nephropathy and factors predictive of disease outcome. Semin Nephrol 2004:24; 179-196.
55. Galla JH. IgA Nephropathy. Kidney Int 1995;47:377-387.
56. Donadio JV Jr, Grande JP. Immunoglobulin A nephropathy: A clinical perspective. J Am Soc Nephrol 1997;8:1324-1332.
57. Dysart NK Jr, Sisson S, Vernier RL. Immunoelectron microscopy of IgA nephropathy. Clin Immunol Immunopathol 1983;29:254-270.
58. Geddes CC, Rauta V, Gronhagen-Riska C, et al. A tricontinental view of IgA nephropathy. Nephrol Dial Transplant 2003;18: 1541-1548.
59. Donadio JV, Bergstralh EJ. Proteinuria patterns and their association with subsequent end-stage renal disease in IgA nephropathy. Nephrol Dial Transplant 2002;17:1197-1203.
60. Maschio G, Cagnoli L, Claroni F, et al. ACE inhibition reduces proteinuria in normotensive patients with IgA nephropathy: A multicentre, randomized, placebo-controlled study. Nephrol Dial Transplant 1994;9:265-269.
61. Russo D, Pisani A. Additive antiproteinuric effect of converting enzyme inhibitor and losartan in normotensive patients with IgA nephropathy. Am J Kidney Dis 1999;33:851-856.
62. Ozsoy RC, Koopman MG. The acute effect of atorvastatin on proteinuria in patients with chronic glomerulonephritis. Clin Nephrol 2005;63:245-249.
63. Dillon JJ. Treating IgA nephropathy. J Am Soc Nephrol 2001; 12:846.
64. Locatelli F, Vecchio LD, Pozzi C. The patient with IgA glomerulonephritisWhat is the role of steroid treatment? Nephrol Dial Transplant 1999;14:1057.
65. Kobayashi Y, Hiki Y, Kokubo T, et al. Steroid therapy during the early stage of progressive IgA nephropathy. A 10-year follow-up study. Nephron 1996;72:237-242.
66. Dische FE, Weston MJ, Parsons V. Abnormally thin glomerular basement membranes associated with hematuria, proteinuria, or renal failure in adults. Am J Nephrol 1985;5:103-109.
Primary care physicians often are confronted with patients presenting with either gross or microscopic hematuria. Either situation demands a clinical investigation.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
