Sleep Apnea: A Somewhat Different Viewpoint
Sleep Apnea: A Somewhat Different Viewpoint
Author: Raymond G. Russell, MD, FACP, CBSM, Kettering Sleep Disorders Center, Dayton, OH; Assistant Clinical Professor, Wright State University Boonshoft School of Medicine, Dayton, OH.
Peer Reviewer: Barbara Phillips, MD, MSPH, Director, UK HealthCare Good Samaritan Sleep Center, Professor, Division of Pulmonary, Critical Care and Sleep Medicine, University of Kentucky College of Medicine, Lexington.
The need for participation of the primary care physician in the treatment of sleep apnea has become much more apparent in recent years.1 Compliance with CPAP use has been found to be unacceptably low (on the order of only 46%).2 By intervening proactively, the PCP can increase compliance substantially and prevent the discontinuation of treatment in a majority of non-motivated patients.3,4
The Editor
Introduction
As I was going through the TSA security check on a recent flight to Florida, I heard the agent announce that we should put our computers and our CPAP machines in a separate bin. I was used to hearing the admonition about computers, but did not realize that CPAP machines were becoming so commonplace.
To start off our discussion, take as an example the case of 49-year-old Mr. Smith who is 5'10" and weighs 255 lbs. His neck is short and thick, and when you look into his throat, you have to push down hard on the tongue blade to see the posterior pharynx. You would like to visualize the tonsils, but you are afraid that any extra downward pressure on the wooden tongue blade might cause it to break. The patient did not come into the office of his own volition, but rather because his wife insisted that he do so due to long pauses in his breathing. Both the pauses and his very loud snoring have increased over the past year since he put on an extra 30 lbs. of weight. An easy clinical diagnosis. Or is it?
A large number of individuals snore, and a lot of overweight patients have hypopneas and not overt obstructive apneas. And what about central apneas? Not all patients who snore and appear to stop breathing have obstructive sleep apnea. How do we know whether Mr. Smith has obstructive sleep apnea? The standard to confirm our suspicion is to ask the patient to have a nocturnal polysomnogram, the most common of sleep study. However, the diagnosis of sleep apnea is not made by polysomnography alone; clinical evaluation also is required.
Before proceeding with our discussion, we should look at some of the terminology that might be confusing for some (and often for us). Many of these terms might be found by the primary care physician in a sleep study report or a sleep consultation letter.
Sleep apnea is what we would expect from the term: a cessation of breathing during sleep. When the term is used in conversations both inside and outside of the medical profession, people are usually talking about obstructive sleep apnea. Central apnea is indeed a cession of breathing, but in a different context. A corollary would be the use of the term "diabetes," which would generally refer to diabetes mellitus and usually not to diabetes insipidus.
Obstructive Sleep Apnea (OSA). An obstructive sleep apnea is defined as cession of airflow for at least 10 seconds and due to obstruction of the upper airway.
Hypopnea is an abnormal respiratory event lasting at least 10 seconds and associated with at least a 30% reduction in airflow, along with at least a 4% drop in oxygen saturation. An alternative definition is a 50% reduction in airflow with either a 3% or greater fall in oxygen saturation or an arousal from sleep.
Respiratory-Effort Related Arousal (RERA) refers to a respiratory event during sleep that does not meet criteria for either an apnea or a hypopnea but that results in a disturbance in sleep or oxygenation. The American Academy of Sleep Medicine Manual for the Scoring of Sleep and Associated Events defines an RERA as a sequence of breaths lasting at least 10 seconds characterized by increasing respiratory effort or flattening of the nasal pressure waveform leading to an arousal from sleep.
Obstructive Sleep Apnea-Hypopnea Syndrome (OSAHS) refers to a cluster of clinical features caused by repetitive episodes of obstructive apneas and hypopneas (what we are usually talking about when we say a patient "has sleep apnea"). These clinical features result from sleep fragmentation or hypoxemia, or both, and can include consequences such as sleepiness and hypertension.
Sleep Disordered Breathing. This term is somewhat general and refers to upper airways resistance syndrome and/or obstructive sleep apnea-hypopnea syndrome (see above). The term probably would not be seen on an official sleep study interpretation but often is used in the consultation letter from a sleep specialist. Its primary use is to distinguish respiratory-related sleep problems from other sleep disorders such as narcolepsy or leg movement disorders.
Apnea plus Hypopnea Index (AHI). The sum of the obstructive apneas and hypopneas divided by the hours of sleep. Insurance companies and naïve clinicians often base the diagnosis or assign severity based solely on the AHI.
Respiratory Disturbance Index (RDI). The RDI is the sum of apneas, hypopneas, and RERAs divided by the total sleep time, in hours.
Upper Airways Resistance Syndrome (UARS) is a term that has started to fall into disuse recently and is not recognized by many insurance companies as a diagnostic entity. It has been used to describe sleep-disordered breathing that consists mostly of RERAs. It basically refers to some degree of inspiratory obstruction that is not enough to qualify as hypopnea and with no apnea. The term has been used primarily to describe loud snorers who do not have apnea but who have sleep fragmentation.
Anatomy
If we look at the oral cavity of a patient with suspected sleep apnea, we would expect to see some sort of narrowing of the opening into the back of the throat. If we shine a light into the mouth and ask the patient to say the traditional "aah," a wide open airway with good visualization of the entire back of the throat and a small uvula hanging down would make significant OSA much less likely (although not impossible).
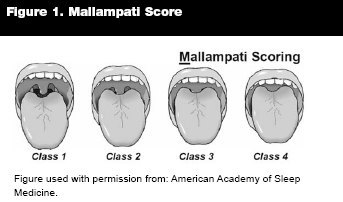
A classification by Mallampati has been helpful in describing the physical features of upper airways narrowing. (See Figure 1.) With a class 1 configuration, the entire uvula can be seen, along with any tonsillar tissue. A class 2 configuration demonstrates only part of the uvula, and class 3 shows only the base. With a Mallampati class 4 configuration to the posterior pharynx, none of the uvula is visualized and about all that can be seen is the tongue and the hard palate.
 |
Retropalatal and Retroglossal Regions (behind the hard palate and behind the tongue). These locations are where the obstruction generally takes place.5 Patients often believe their snoring results from nasal problems, but this is seldom the primary location. Nasal congestion can cause an increase in the negative intraluminal pressure of the upper airways and can aggravate snoring, but the main problem in OSA generally is found behind the tongue. Alleviating nasal congestion with medication, polyp removal, or septal surgery can sometimes help snoring but usually will do very little for apnea. When a bed partner complains that a patient stops breathing at night, the nose is not the best place to look for a cause.6
The two most common contributing factors to a narrowed opening at the back of the throat is an elongated soft palate and/or a high-riding base of the tongue. Redundancy of the tissue of the lateral pharyngeal walls also can be seen as an anatomic characteristic, as well as the presence of enlarged tonsils. A backward displacement of the mandible (retrognathia) also is described as an aggravating factor. However, in practical terms, this is not a common finding. Differentiating between an elevated tongue base and true macroglossia can be difficult, although both can contribute to symptoms of sleep apnea.
The pharyngeal airway is maintained open due to neuromuscular influences that can be suppressed and subsequently cause apnea. Substances that decrease the size of the upper airway include alcohol, sedatives, and opiates. It would not be unusual for a wife to elect to sleep in another bedroom on a football night when she knows that her husband is going to be drinking a generous amount of beer, with the snoring that follows. The same laxity of the soft tissues that causes the snoring also can contribute to OSA.
Another major contributing factor in OSA is weight gain. It is not uncommon for a patient's bed partner to describe an increase in the snoring and pauses in breathing with observed weight gain. Once a critical weight has been achieved, as little as a 10-pound gain can trigger the onset of snoring and pauses (both of which can disappear with a similar weight loss).7 As weight increases, the parapharyngeal fat pads also enlarge and narrow the pharyngeal opening. This is more of a problem in men, since more fat is deposited in the upper body and trunk in men, whereas women have more in the lower body and extremities. Obesity in older patients is especially problematic. The strongest risk factors for OSAHS are obesity and age over 65. One study showed a body mass index (BMI) of at least 25 kg/m2 had a sensitivity of 93% for the syndrome. Neck circumference greater than 18 inches is also a good predictor for OSA.8
Pathophysiology
It is important to realize how simple a process OSA can be to understand. One of the main activities of the upper airway muscles is to dilate and stiffen the airway. This counters the negative intraluminal pressure that develops when we inhale.9 Patients with obstructive sleep apnea often have more collapsible or more narrow upper pharyngeal airways. This adds to the pharyngeal narrowing caused by the normal sleep-related reduction in the upper airway dilating muscle activity. The sleep state itself is associated with a decrease in motor output to the pharyngeal muscles and can add to the upper airway anatomic abnormalities often seen in sleep apnea. The pharyngeal airway (and not the nose) is the site of upper airway closure during sleep.
Sleep fragmentation and nocturnal oxygen desaturation are the ways by which OSA impacts daytime functioning. People normally awaken periodically during the night. This is usually not a problem because most people are able to promptly fall back asleep and have no recollection of these few, brief awakenings. A problem arises, however, when patients arouse from sleep frequently, as occurs with sleep apnea. Sleep is divided into two very distinct forms: rapid-eye movement sleep (or REM sleep), and nonrapid-eye movement sleep (or NREM sleep). Approximately 75% of each night's sleep is spent in NREM; the remaining 25% of sleep is spent in REM. NREM sleep has three stages (1-3) that occur in cycles. REM sleep is interspersed with NREM stages, with increasing amounts of REM sleep toward the morning, when body temperature is lowest. One can imagine that if an individual goes from awake to stage one, then back to awake, then back to stage one, then back to awake frequently during the night, the quality of that night's sleep will be diminished. Such repeated awakenings are the hallmark of sleep fragmentation.
Fragmentation Triggers. The list of potential disturbances that can disrupt sleep is long and can include noise, a restless bed partner, an uncomfortable mattress, or respiratory distress because of apneas, hypopneas, or RERAs. As far as sleep apnea is concerned, there are several mechanisms involved in the arousal process. The most obvious to an observer is the simple struggling to breathe that occurs as the patient tries to breathe against a closed upper airway. The obstruction (and therefore the term "obstructive" sleep apnea) is an inspiratory problem.
Another major cause of arousals is hypoxia, which is why arousals are more common at the termination of apneic events.10 The patient usually falls back to sleep as the breathing pattern normalizes. Arousals (which generally are considered to be at least 3 seconds or greater) often are not remembered by the patient. Short periods of arousal frequently are associated with amnesia for the event, and patients can have arousals numbering in the hundreds and without being aware that their sleep has been fragmented. As the number of arousals increases, daytime sleepiness increases. Of special interest is the fact that excess daytime sleepiness occurs even when the total sleep time during the night is unchanged. It is the sleep fragmentation itself that is considered to be the cause of the excessive daytime sleepiness. A patient who goes in and out of sleep frequently during the night is also less likely to progress into the deeper stage-three sleep that generally is felt to be more restorative.
Epidemiology
The current estimate of the prevalence of sleep apnea in the United States is 5%, but this is likely a very conservative underestimate, as studies of several populations, notably commercial truck drivers, have indicated a much higher rate of symptomatic, clinically significant sleep-disordered breathing.11 In youth and early middle age, the male:female ratio for OSAHS is about 2:1, but after menopause, the incidence rises in women, and the prevalence difference between the sexes is small.12 Being Asian or Hispanic is probably an independent risk factor for sleep apnea. Data about African Americans also suggests that African ancestry may be a risk factor, but the evidence is not as strong as it is for Asians.13 Because our population is becoming heavier, older, and more diverse, the prevalence of sleep apnea is clearly increasing.
Genetics
Patients often ask whether sleep apnea can be inherited. The short answer is yes, but not in the strict Mendelian sense of the word as one sees in the trisomy 21 associated with Down's syndrome. A positive family history increases the risk by two to four times.14 A better term might be family clustering or familial aggregation. All of us inherit physical characteristics from our parents. If both of our parents have large Corinthian noses, there is a higher likelihood that we or one of our siblings will have a similar proboscis. OSA has strong anatomical characteristics, and family members often have shared craniofacial uniqueness (both soft tissue and bony structures). The contributing factors for OSA, however, are usually multifactorial. Obesity has a marked effect on apnea, and obesity is strongly heritable (obesity increases risk for OSA 10-14 fold). In summary, a positive family history of OSA is definitely a risk factor.
Diagnosis
Obstructive sleep apnea typically is diagnosed with a nocturnal polysomnogram. The patient arrives at the sleep center a little before bedtime, and a technician attaches electrodes and sensing devices to record oxygen saturation, electroencephalogram (EEG), electrocardiogram (ECG), electromyogram (EMG), eye movements, snoring, and occasionally other physiologic signals. Data then are retrieved from the computer readouts, and an analysis is made and coordinated with the sleep history and physical examination done previously. The study can detect exactly when the patient goes to sleep, the stages of sleep, what the breathing pattern is like, oxygen saturation, abnormal ECG and electroencephalographic patterns (alpha delta sleep), leg movement disorders, as well as many other sleep abnormalities.
The diagnosis needs to be suspected first, however, by the primary care physician before a polysomnogram can be considered.
Nocturnal Symptoms. Snoring is the most common complaint, although spousal concern about pauses in the patient's breathing is often the case. Snoring generally is more prevalent in the supine position, and the patient often complains of being awakened at night and told to roll over. The snoring is seldom a problem for the patient, although patients sometimes complain of waking themselves. Of more concern are nocturnal awakenings due to snorts or gasps that follow pauses in the breathing pattern. Although snoring (and sleepiness) are the stereotypical symptoms, it is becoming increasingly clear that women who have sleep apnea present differently from men. Women are much less likely to have been told that they snore or stop breathing during sleep and much more likely to present with insomnia, rather than sleepiness, than men.15
Daytime Consequences. Here is where it gets more difficult. The most frequent daytime complaint is often feeling "tired." However, the cardinal daytime symptom for OSA is supposed to be "excess daytime sleepiness." The problem is that patients have a hard time distinguishing between "tired," "sleepy," and "fatigued." It is difficult to go through a day without hearing someone complain of feeling tired. This complaint has almost become part of the American way of life, with people finding creative methods of wearing themselves out through work and other activities while often sleeping less.
Other Diagnostic Approaches to OSAHS. The combination of ambulatory monitoring and oximetry (a.k.a., portable sleep studies) has been studied recently as an alternative to facility-based polysomnography. Although in-lab polysomnography has historically been the prime diagnostic tool to confirm the presence of obstructive sleep apnea, home testing has been evaluated as an alternative diagnostic approach.16 In general, patients with significant sleep apnea have greater fluctuation in oxygen saturation and heart rate than those without sleep apnea. However, thinner, younger patients without lung disease can have significant breathing and sleep disturbance without remarkable oxygen desaturation. In other words, oximetry and portable testing can be used to help rule sleep apnea in, but they fall short in ruling it out. This form of testing may be especially helpful in areas not served by facility-based sleep diagnostic centers such as in rural settings. Even though the Centers for Medicare & Medicaid Services (CMS) has begun to reimburse for CPAP treatment in patients who are diagnosed through home testing, it is still too early to see how this form of testing is going to hold up over time in a real world environment.
Clinical Prediction Formulas. Several investigators have developed prediction formulas based on findings in the history or physical examination. Among the most useful of such findings are history of witnessed apneas, male gender, BMI, and neck circumference. In general, these formulas are sensitive but not as specific as those for PSG. Prediction formulas probably do have a place in the expedited diagnosis and/or triage of patients with suspected sleep-disordered breathing. One of the most reliable screening tools for sleep apnea is the Berlin Questionnaire. The information from such studies, however, needs to be correlated with other aspects of the clinical history and physical findings.17
Treatment
Once the sleep study is done, the first decision to be considered is whether there is enough pathology to justify therapeutic intervention. As with many treatment options in today's managed care environment, the physician often does not make all of the decisions in this regard. Studies have suggested that apnea with a frequency greater than 5 episodes per hour (apnea plus hypopnea index) can have significant detrimental effects. Many insurance companies and Medicare used to pay for treatment only if the index was over 15 per hour. After dialogues with sleep organizations such as the American Academy of Sleep Medicine, a compromise was reached, and most insurance carriers now allow treatment if the episodes are 5 or greater and if the OSA is accompanied by a history of excessive daytime sleepiness, ischemic heart disease, stroke, hypertension, insomnia, mood disorders, or impaired cognition. From a practical standpoint, it is usually not difficult to find one of these symptoms in the typical OSA patient. This remains a somewhat murky area, however, in that the question often remains, how much hypertension, insomnia, etc. justifies treatment?
Other factors, such as whether nocturnal oxygen desaturation is present on the polysomnogram, usually can help in the decision making. Medicare uses a saturation of less than 89% for at least five minutes as a cutoff for home oxygen use. This number also is helpful in deciding if specific intervention for sleep apnea is justified. Would the patient with mild sleep apnea accompanied by oxygen desaturation do just as well with supplemental oxygen via nasal cannula as with CPAP? If we take an overweight, hypertensive patient with nocturnal oxygen desaturation and an apnea plus hypopnea index of only 5 episodes per minute, insurance carriers would most likely agree to reimburse for treatment. However, if the patient is opposed to using either CPAP, surgery, or a dental device, it may be appropriate to simply use nasal cannula oxygen while the patient works on weight loss.
Supplemental oxygen by itself, however, should not be used as a primary treatment for OSA. Correcting any hypoxia increases the length of apneas (and thus CO2 increases) but does not correct the cardiovascular side effects of the apneas and hypopneas. More importantly, it does not improve sleepiness,18 so the patient remains at risk for the important complication of motor vehicle accidents.
Behavior. Clearly, behavioral change has a major role both in causing and treating this condition. Weight loss can be curative. Even modest weight loss (10%) can relieve mild sleep-disordered breathing. Weight, appetite, and sleep are complex, interrelated factors that are probably mediated in part by leptin, cortisol, insulin, and metabolic rate. Patients newly diagnosed with obstructive sleep apnea have a greater increase in weight in the year prior to diagnosis than their weight-matched controls. All obese patients with SDB should be counseled about weight loss. Cigarette smoking is an independent risk factor for snoring and for sleep apnea. In addition, nicotine disrupts sleep, and smokers generally have more sleep disturbances than nonsmokers. Smoking cessation advice should be routine in treating all patients who smoke. Muscle relaxants, including alcohol and sleeping pills, can make apneas longer by reducing airway tone and by increasing the arousal threshold. Moderate drinking clearly has been shown to exacerbate OSA and should be discouraged.
CPAP (continuous positive airway pressure) is the mainstay for treatment of OSA. Before the first CPAP device became available in 1981, the only viable treatment option was to perform a tracheostomy. Multiple improvements have been made to the technology, especially in the past decade. The treatment basically consists of a mask that either fits over the nose or both the nose and mouth (full face mask). The mask is connected by a tube to a pressure generator that introduces ambient air under pressure into the upper airways and acts as an internal splint to keep the airway open and thus eliminate the obstructive events as well as the snoring. If this sounds uncomfortable, it usually is. Just like the first time a person puts on a pair of glasses, it takes a certain amount of effort to get used to having something strapped to the face with air blowing into the nose and/or mouth. I often tell patients not to use the CPAP the first night they have it, but simply put on the mask without hooking it up to the machine. They can watch television, help with the dishes, etc., and generally get used to wearing the device and hopefully "desensitize" themselves somewhat. After a day or two, they can connect the air-generating machine and get used to breathing against the flow of air while they are awake. Once they feel comfortable using the CPAP, they can then take it to bed and usually tolerate it better than if they had started it the first night.
Determining how much a patient needs to use the CPAP for it to be effective has been a topic of contention for a number of years. Recent studies suggest that at least four hours of nightly use are needed to have an adequate therapeutic effect. Six hours are considered adequate presently, but these numbers may change as more research is done. Current wisdom suggests that the more hours the CPAP is used during the sleep period, the better. Nevertheless, CPAP appears to be extraordinarily effective in intention-to-treat studies.19 The newer CPAP machines make it easy to compute how much the patient actually uses the device thanks to a memory chip that can record the time used. If a primary care physician is notified by a medical supply company that memory chip information indicates suboptimal use of the CPAP, simply discussing this with the patient often is enough to improve compliance.20 If the problem continues, referral back to the sleep specialist may be needed. The emerging field of behavioral sleep medicine also has been helpful in improving compliance.
Surgery. The surgical approach to OSA was developed at about the same time CPAP was developed; however, this form of treatment never gained the popularity that the positive pressure therapy ultimately achieved. One reason for this was the poor response rate. About half of the patients had adequate resolution of their apnea, but this translated into a 50% failure rate in the short term, and long-term data are even more dismal. Indeed, the Cochrane database has concluded that initial surgical treatment of sleep apnea cannot be justified on the basis of currently available data.21 Another problem with this approach is the discomfort associated with the procedure. I have never had a patient describe the aftermath in kind words, and some female patients have even suggested that they would have preferred childbirth to this type of surgery.
The most common surgery is uvulopalatopharyngoplasty (UPPP). Basically, the posterior part of the soft palate is trimmed off, along with the uvula and with removal of any redundant tissue from the lateral pharyngeal walls. Long-term side effects can include voice changes (especially with some foreign languages) and liquids exiting through the nose if swallowed too quickly.22 Special circumstances such as exceptionally large tonsils with a lot of lateral pharyngeal wall tissue might merit having surgery offered as a first choice, but, generally speaking, the UPPP surgery is reserved for patients who do not tolerate CPAP or who resist the CPAP as a permanent fact of life. Some of the less invasive surgical techniques, such as laser surgery, can be considered in less severe cases, but, again, the failure rate is substantial. Surgical procedures that involve moving the lower jaw forward and placing bone grafts, such as in cases of retrognathia, are seldom needed and are generally limited to special referral centers.23
Dental Devices. In contrast to the low surgical success rates, oral appliances are proving to be an effective treatment in select cases of mild to moderate sleep apnea and snoring.24,25 Mandibular repositioners that pull the mandible forward (and with it, the tongue) are the most common appliances. There is now a board examination for Dental Sleep Medicine with a promising increase in new data coming out showing favorable results in treating OSA.
Nasal Dilators. Football players often wear the semi-rigid, stick-on patches across the top of the nose. The purpose of the patch is to externally splint the nasal passages open. It was hoped that these devices also would help snoring and possibly even OSA. The help with snoring is only mild and sporadic at best, and dilation of the anterior nasal valve using a nasal dilator has virtually no impact on OSA.26
Primary Care Treatment. An often under-utilized form of treatment is position therapy, which can be initiated by the patient's primary care physician. When a patient presents with a chief complaint of snoring but with no daytime consequences, simply getting the patient out of the supine position can be of significant help. It also can be of benefit in patients who have positional sleep apnea that is caused in great part by a falling back of the tongue, due simply to gravity when the patient sleeps in the supine position. There have been numerous creative ways to keep patients off their backs. The most popular solution is sewing a pocket into the back of a pajama top or heavy tee shirt. This usually includes a flap with a button so that a tennis ball or similar object can be inserted and later removed for washing. The discomfort of lying on the ball is usually enough to make the patients roll over to their side or abdomen. This generally reduces or stops the snoring, although I have had overweight patients who have required a softball or larger objects to be effective.
Why All of the Fuss About Sleep Apnea Recently?
Sleep medicine had a slow start in the 1970s, with the Stanford University group leading most of the early research. Initially the medical community was reluctant to accept sleep disorders as real nosologic entities, and it was not until the Department of Transportation became involved that physician awareness rose. Semi truck drivers were the first targets, due to the link between obesity and highway accidents related to falling asleep at the wheel secondary to sleep apnea.27 The previous attitude that sleep problems were more of a nuisance than real medical issues has dramatically changed. Multiple medical problems are now being linked to OSA.
When a patient presents to a primary care physician with cognition or performance deficits, especially if the patient is elderly, sleep apnea is usually not high on the list in the diagnostic differential. As with many medical issues, severe disease is much easier to identify. The subtle changes related to mild obstructive sleep apnea, however, can be elusive. Personality changes and inability to concentrate and maintain attention to detail can be seen even in the mildest of cases. The emotional lability seen in mild sleep apnea can be seen in so many other situations that the diagnosis usually needs additional historical data to be considered. In more severe cases, work performance can become a major problem, along with family discord. Memory is easily affected by the sleep deprivation associated with the fragmentation of sleep. Not only is the storage and processing of information hampered, but executive function such as judgment and decision making can be jeopardized.28
High on the list of complications of sleep apnea is a wide array of cardiovascular side effects. Any doubts a few years ago about the association between obstructive sleep apnea and hypertension have effectively been erased by numerous well controlled studies. There appears to be a "dose dependent" association between obstructive sleep apnea and hypertension, especially in the mild to moderate range of apnea. Since intermittent hypoxia has more of an adverse effect on vascular endothelial cells than sustained hypoxia, the hypoxia-reoxygenation cycles that occur with chronic OSA not only facilitate systemic hypertension but also pulmonary hypertension and coronary artery disease. It is now known that sleep apnea leads to increased sympathetic tone, which facilitates hypertension, and CPAP treatment can bring the sympathetic tone back down to normal.29-31 More patients are being referred to sleep disorders centers by cardiologists as they become aware of the association between OSA and a wide variety of cardiac conditions.
More recently, metabolic disturbances are being linked to the sleep fragmentation and nocturnal oxygen desaturation seen in sleep apnea.32 A bidirectional association between diabetes mellitus and OSA has been documented, with the autonomic dysfunction of diabetes contributing to sleep apnea.33 Even the earliest studies showed a causal relationship between OSA and glucose intolerance as well as insulin resistance. The intermittent hypoxemia and sleep fragmentation due to recurrent arousals related to apneas and hypopneas are being shown to contribute significantly to the now much-discussed metabolic syndrome.
Pre-operative clearance also merits mention. Patients sometimes fail to mention to their surgeon or anesthesiologist that they use a CPAP machine at night, so it is helpful to have the primary care office chart "flagged" as a reminder. General anesthesia can have serious deleterious effects in patients with OSA. If the anesthesiologist is unaware of the patient's condition, surprises in this regard can be substantial. If patients ask if they should take their CPAP machine with them for their surgery, tell them yes. This adds to the treatment options the anesthesia department has at its disposal. (Unlike mechanical ventilators, CPAP machines are individualized to a specific patient.)
Central Apnea
The other sleep apnea is one that we read about but do not hear patients and bed partners complain about very much. There may be little or no snoring associated with this form of pauses in breathing, and one would actually have to be looking at the patient to pick up any abnormal breathing pattern. Central apnea is characterized by pauses in breathing due to a lack of ventilatory effort. No signal is sent to the diaphragm, abdominal muscles, or accessory muscles of breathing for a breath to take place. Carbon dioxide control mechanisms can be defective in patients with chronic pulmonary disease, and Cheyne Stokes respiration can be observed in patients with congestive heart failure. Many neurologic diseases can cause central apneas, especially in the elderly. In addition, this form of apnea also is frequently seen in patients taking opioids for pain. From a sleep standpoint, physicians should remember that the central forms of apnea may cause hypoxia, and anything that causes hypoxia can cause arousals that ultimately fragment sleep and lead to daytime consequences. The diagnosis is made with a standard sleep study (polysomnogram), and treatment is usually aimed at the cause.
Growth Industry and Kickback
So far, the health insurance industry has been fairly lenient with diagnostic studies to rule out sleep apnea. Due to an initial scarcity of accredited sleep laboratories, Medicare allowed non-sleep trained physicians to buy into stand-alone centers and then refer their own patients into the centers for sleep studies. Normally this activity would constitute a violation of the Stark anti-kickback legislation, but due to the old regulations, non-sleep trained physicians can still refer their patients to their own sleep laboratories and receive their "kickback" in the form of stock value. The result has been a marked increase in free-standing sleep centers across the country, some even in rented motel rooms, and some with only marginal credentials. Among the difficulties that have been seen with some of the free-standing centers is that most are not set up to treat the broad scope of sleep disorders seen in the general population. The main reason for the existence of some of these independent laboratories is only to perform sleep studies. Once the studies are completed, the center may have little else to offer the patient. If a report of "sleep apnea" has been made, CPAP may be the knee-jerk reaction as far as treatment is concerned. If the center does not have an on-site sleep physician to evaluate treatment recommendations, other options such as surgery, dental devices, aggressive weight loss, position change in bed, etc., may not even be considered. Treatment of other sleep disorders found on the study as well as in the history may simply be ignored. Cognitive behavioral therapy and additional behavioral sleep medicine measures may not even be offered due to the lack of specialized physician training in these areas. The general lack of oversight in this regard may be coming to an end in the near future. The Office of the Inspector General, which is the investigative wing of the U.S. Department of Health and Human Services, issued a warning in October that it would be starting investigations into sleep-related services during its fiscal year 2009.
Can a primary care physician simply write a prescription for a polysomnogram? Certainly, just as he or she can write for an echocardiogram. The difference between these types of studies is who reads the data. Technicians are not allowed to render an official report on an echocardiogram, and diagnostic considerations are left to the expertise of a trained cardiologist. This is not the case with all sleep studies. Technicians "score" the sleep studies by looking at up to seven or eight hours of sleep data to verify the various stages of sleep and document observed abnormalities. Where the study goes from there is quite variable. Some of the free-standing centers simply have a "condensed" summary sheet that is compiled by the technician and sent to a sleep specialist, sometimes in another city. The specialist then looks at the summary sheet and signs the bottom, often without having access to the raw data and also no contact with the patient. Hospital-based sleep centers usually have stricter guidelines for the evaluation of their sleep studies, and somewhere in the process a sleep-trained specialist has access to the original tracings and the opportunity to actually over-read the information. In our hospital, a sleep-trained physician actually has to see the patient and get a very detailed face-to-face sleep history, including co-morbid medical and psychiatric problems along with a review of systems, physical examination, family history, occupational history, etc. A board-certified PhD sleep specialist then reviews the sleep studies before the data even reach the hands of the clinical sleep physician.
When patients are sent to major, hospital-based sleep centers for sleep evaluation, they may not get a standard sleep study. The examining physician may decide that a multiple sleep latency test or a maintenance of wakefulness study would be more appropriate; or maybe a sleep log; or maybe no study at all.
Legal Aspects of Cognitive and Performance Deficits
As public awareness increases concerning the deleterious effects of sleep apnea, so does the recognition by attorneys that this area is fertile ground for potential lawsuits. Civil liability claims are becoming popular for accidents related to decreased alertness.
Falling asleep at the wheel in New Jersey now counts as recklessness under the existing vehicular homicide law. Canadian courts have become even more punitive in their opinions, and physicians are high on the list of targets. Statutory reporting by physicians of impaired drivers in Canada with sleep apnea has reached the point that this duty supersedes the physician's obligation of confidentiality.34,35 The impetus behind this trend is that sleep apnea patients are up to four times more likely to have a motor vehicle accident, and effective treatment of sleep apnea can reduce collision risk to normal.36 Patients do not have to completely fall asleep at the wheel to be at risk. Sleepiness increases periods of inattention and has been linked not only to accidents on the road but also in occupational settings. From a primary care physician liability standpoint, at a very minimum, there should be documentation in the chart that the OSA patient understands that he or she is not to drive or engage in dangerous occupational activity if he or she is not actively using a CPAP machine or other treatment modality.
Future
As public awareness of OSA becomes even more pronounced, expect to see an increasing number of patients reporting snoring as a concern. The increase in obesity in our society also will be a challenge, and an overall reversal in this obesity trend could resolve a large percentage of OSA problems. A large number of undiagnosed patients remain who have significant OSA and who are functioning poorly in their personal and professional lives. Many also have co-morbid issues, such as the above-mentioned cardiovascular and metabolic consequences. What happens to these patients depends in large part on the primary care physicians in the country and their ability to think outside the box regarding the possible association between OSA and other medical conditions.
References
1. Aloia MS, Arnedt JT, Riggs RL, et al. Clinical management of poor adherence to CPAP: Motivational enhancement. Behav Sleep Med 2004;2: 205-222.
2. Raucher H, Popp W, Wanke T, et al. Acceptance of CPAP therapy for sleep apnea. Chest 1991;100:1019-1023.
3. Meurice JC, Dore P, Paquereau J, et al. Predictive factors for long term compliance with nasal continuous positive airways pressure treatment in sleep apnea syndrome. Chest 1994;105:429-433.
4. Aloia MS, Arnedt J, Zimmerman M, et al. Combined therapy to improve adherence to CPAP. Sleep 2005;28(suppl.):A192.
5. Issa FG, Sullivan CE. Upper airway closing pressures in obstructive sleep apnea. J Appl Physiol 1984;57:520-527.
6. Friedman M , Tanyeri H, Lim JW, et al. Effect of improved nasal breathing on obstructive sleep apnea. Otolaryngol Head Neck Surg 2000;122:71-74.
7. Peppard PE, Young T, et al. Longitudinal study of moderate weight change and sleep-disordered breathing. JAMA 2000;284:3015-3021.
8. Davies RJO, Strandling JR. The relationship between neck circumference, radiographic pharyngeal anatomy, and the obstructive sleep apnea syndrome. Eur Resp J 1990;3:509-514.
9. Smith PL, Wise RA, Gold AR, et al. Upper airway pressure-flow relationships in obstructive sleep apnea. J Appl Physiol 1988;64:789-795.
10. Berthon-Jones M, Sullivan CE. Ventilatory and arousal responses to hypoxia in sleeping humans. Am Rev Respir Dis 1982;125:632-639.
11. Young T, Peppard PE, Gotlieb DJ. Epidemiology of obstructive sleep apnea: A population health perspective. Am J Respir Crit Care Med 2002;165: 1217-1239.
12. Tishler PV, Larkin EK, Schluchter MD, et al. Incidence of sleep-disordered breathing in an urban adult population. JAMA 2003;289:2230-2237.
13. Young T, Peppard PE, Gotlieb DJet al. Epidemiology of obstructive sleep apnea: A population health perspective. Am J Respir Crit Care Med 2002; 165:1217-1239.
14. Palmer LJ, Buxbaum SG, Larkin E, et al. A whole-genome scan for obstructive sleep apnea and obesity. Am J Hum Genet 2003;72:340-350.
15. Valipour A, Lothaller H, Rauscher H, et al. Gender-related differences in symptoms of patients with suspected breathing disorders in sleep: A clinical population study using the sleep disorders questionnaire. Sleep 2007;30: 312-319.
16. Trikalinos TA, Ip S, Gowri R, et al. Home Diagnosis Of Obstructive Sleep Apnea-Hypopnea Syndrome. Rockville MD: Agency for Health Care Research and Quality. April 12, 2007.
17. Netzer NC, Stoohs RA, Netzer CM, et al. Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Ann Intern Med 1999;131: 485-491.
18. Phillips BA, Schmitt FA, Berry DTR, et al. Treatment of obstructive sleep apnea: A preliminary report comparing CPAP to nasal oxygen in patients with mild obstructive sleep apnea. Chest 1990;90:325-330.
19. McArdle N, Devereau G, Heidarnejad H, et al. Long-term use of CPAP therapy for sleep apnea/hypopnea syndrome. Am J Respir Crit Care Med 1999;159:1108-1114.
20. Kribbs NB, Pack AI, Kline LR, et al. Objective measurement of patterns of nasal CPAP use by patients with obstructive sleep apnea. Am Rev Respir Dis 1993;147:887-895.
21. Sundaram S, Bridgman S, Lim J, et al. Surgery for obstructive sleep apnoea. Cochrane Database Syst Rev 2005;(4):CD001004.
22. Fairbanks D. Uvulopalatopharyngoplasty complications and avoidance strategies. Otolaryngol Head Neck Surg 1990;102: 239-245.
23. Riley R, Powell N, Guilleminault C. Maxilofacial surgery and obstructive sleep apnea: A review of 80 patients. Otolaryngol Head Neck Surg 1989; 101:353-361.
24. Marklund M, Stenlund H, Franklin KA. Mandibulal advancement devices in 630 men and women with obstructive apnea and snoring. Chest 2004;125: 1270-1278.
25. Ferguson KA, Cartwright R, Rogers R, et al. Oral appliances for snoring and obstructive sleep apnea: A review. Sleep 2005:29:244-262.
26. Hoiger U, Ejnell H, Hedner J, et al. The effects of nasal dilators on snoring and obstructive sleep apnea. Arch Otolaryngol Head Neck Surgery 1992; 118:281-284.
27. Masa JF, Rubio M, Findley LJ, et al. Habitually sleepy drivers have a high frequency of automobile crashes associated with respiratory disorders during sleep. Am J Resp Crit Care Med 2000;162:1407-1412.
28. Naegele B, Thouvard V, Pepin JL, et al. Deficits of cognitive executive functions in patients with sleep apnea syndrome. Sleep 1995;18:43-52.
29. Marchant B, Stevenson R, Vaishnav S, et al. Influence of the autonomic nervous system on circadian patterns of myocardial ischaemia: Comparison of stable angina with the early post infarction period. Br Heart J 1994;71: 329-333.
30. Mancia G. Autonomic modulation of the cardiovascular system during sleep. N Engl J Med 1993;328:347-349.
31. Hung J, Whitford EG, Parsons RW, et al. Association of sleep apnoea with myocardial infarction in men. Lancet 1990;336:261-264.
32. Harsch IA, Schahin SP, Radespiel-Troqer M, et al. Continuous positive airway treatment rapidly improves insulin sensitivity in patients with obstructive sleep apnea syndrome. Am J Respir Crit Care Med 2003;169:156-162.
33. Resnick HE, Redline S, Shahar E, et al. Diabetes and sleep disturbances: Findings from the Sleep Heart Health Study. Diabetes Care 2003;26: 702-709.
34. Spillane v. Wasserman 13 CCIT (2D) 267 (GD), OJ No 2470 (CA).
35. Toms v. Foster OJ No. 1413 (CA).
36. Findley L, Smith C, Hooper J, et al. Treatment with nasal CPAP decreases automobile accidents in patients with sleep apnea. Am J Resp Critical Care Med 2000;161:857-859.
The need for participation of the primary care physician in the treatment of sleep apnea has become much more apparent in recent years.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
