The Missed Acute Myocardial Infarction in the ED: Strategies to Reduce the Risk for Both the Patient and the Physician
The Missed Acute Myocardial Infarction in the ED: Strategies to Reduce the Risk for Both the Patient and the Physician
We have all heard a story that starts with, "Remember that patient you saw and discharged yesterday? Well, ...." This next series of EM Reports tackles one of the most common subjects of such a story: missed heart attacks.
It seems to me that in the diagnosis and treatment of heart attacks, we physicians have entered into a Faustian bargain. We have been given an impressive array of assessment, diagnostic, and therapeutic modalities to use. But with them, we are expected to be perfectan obvious impossibility, but one that continues to be played out in medical liability proceedings.
Like many physicians, I have developed techniques to deal with the potential for missing an MI. I am more attuned to atypical patients and atypical presentations. I commonly obtain serial ECG and cardiac biomarkers measurements. I readily observe patients. And I never discharge a patient with the phrase, "There is nothing wrong, everything is fine." That phrase has gotten me into more trouble than any other.
I hope you find this series useful in your practice.
J. Stephan Stapczynski, MD, FACEP, FAAEM, Editor
Introduction
The following patients are seen in the emergency department (ED) on a busy shift: an 84-year-old woman with altered mental status; a 32-year-old male diabetic with shortness of breath; a 25-year-old man with chest pain after using cocaine; a 52-year-old woman with weakness and fatigue. What do they have in common? They are all suffering from acute myocardial infarction (MI) – or are they? Each of them could have multiple other diagnoses besides acute MI, but missing this diagnosis will have significant consequences both for the patient and the physician. Missed MI lawsuits consistently pay out the largest awards in medical malpractice cases, accounting for nearly one-third of all dollars paid. The fact that their short-term mortality is as high as 25% when the acute MI is inadvertently discharged from the ED1 certainly contributes to these awards.
Thus, the task of the emergency physician is to accurately identify all patients who present to the ED with acute MI. While this is the goal, it has not been possible to reach 100% accuracy. Anywhere from 2% to 6% of ED patients who present with acute MI are mistakenly discharged without accurate diagnosis.1,2,3 This number has remained relatively constant for several decades, despite advances in enzyme testing and heightened awareness of the problem. Conversely, only 30% of patients admitted for suspected acute coronary syndrome (ACS) are ultimately diagnosed with MI or unstable angina. The 70% of patients not found to have ACS after admission cost more than $5 billion annually in the United States alone, and pressure is mounting to reduce the number of these admissions.4
Patients who present in classic fashion, with chest heaviness, shortness of breath, diaphoresis, and dramatic ST segment elevation are not among the missed MIs. Several studies of missed MI cases consistently have identified similar reasons for these patients not being diagnosed in the ED. Missed MI patients tend to be younger, have fewer risk factors, present atypically, and have subtle or non-diagnostic electrocardiogram (ECG) changes.2,5 Yet, in as many as 25% of cases, overt misreading of the ECG by emergency physicians also is cited as a contributing factor.3,6
This article will explore these identified reasons why MIs are missed in the ED, review current data on the limits of cardiac enzyme testing, discuss atypical presentations, as well as review the ECG findings of acute MI. ECG mimics of acute MI also will be discussed to review useful tools that identify these ECGs. New diagnostic advances, such as coronary computed tomography (CT) scans and use of recent nuclear stress test/catheterization results also will be discussed. Given that more than 1 million patients are diagnosed with acute MI each year in the United States,7 the odds are high that practicing emergency physicians will encounter these difficult-to-diagnose patients on multiple occasions during their careers. Only through combined understanding of the issues, appropriate use of new diagnostic modalities, and vigilance can one hope to reduce the risk of discharging the acute MI patient from the ED.
Defining Terms
Acute Coronary Syndrome. Several terms describe the process of myocardial ischemia and necrosis. The terms accurately describe different clinical entities in the continuum of what is now referred to as acute coronary syndrome (ACS). The continuum of ACS includes unstable angina and acute MI (both STEMI and non-STEMI).
Unstable Angina. Unstable angina refers to a change in the typical pattern of angina, or anginal-equivalent symptom (e.g., shortness of breath). Any one of the following three features defines the change in angina: occurs at rest or minimal exertion and often lasts > 20 minutes without treatment by nitroglycerin; is of new onset (< 1 month); or occurs with a crescendo pattern of increasing severity and frequency than previously experienced.8 Notably, as many as 50% of patients admitted with unstable angina will show evidence of mild myocardial necrosis by rising cardiac biomarkers and ultimately be diagnosed with non-ST elevation MI (non-STEMI).9 Before sensitive cardiac troponin testing became widely used, these patients were all considered to have unstable angina. However, elevated troponin levels now are considered diagnostic criteria for acute MI. Unstable angina can be produced by a variety of pathological conditions. The most common is by atherosclerotic plaque rupture or erosion causing non-occluding thrombus formation in a coronary artery. Slow erosion leading to thrombosis of a coronary artery may be more significant than first thought. Up to 35% of sudden deaths from MI are caused by plaque erosion rather that rupture.10 Unstable angina also can result from coronary artery spasm (Prinzmetal angina), progressive mechanical narrowing of a coronary artery (for example, from muscle hypertrophy), or by increased myocardial oxygen demand in the setting of limited oxygen supply (for example in symptomatic anemia). Lastly, the unstable angina patients are roughly six times more common than acute MI patients in ED populations complaining of chest pain,11 and the majority of these patients will have negative cardiac biomarkers and non-diagnostic ECGs.
Acute MI. Acute MI is defined as myocardial cell necrosis in the setting of ischemic symptoms and ECG changes indicative of ischemia (ST segment elevation or depression, development of Q waves) combined with typical rise and fall of specific cardiac biochemical markers.7 This definition recently was updated in a consensus statement from the European and U.S. cardiology organizations, by selecting rising cardiac troponins as the most specific markers of myocardial necrosis.12
STEMI and Non-STEMI. ST elevation MI (STEMI) results when one of the primary coronary arteries (left anterior descending, circumflex, or right) is suddenly and completely occluded, typically as the result of atherosclerotic plaque rupture/erosion and thrombus formation. Abrupt occlusion of a coronary artery results in acute ischemia of larger areas of the heart and produces characteristic injury patterns on the ECG (ST segment elevation), and results in development of Q-waves if blood flow is not restored before significant cell death occurs. Thus, the older term for STEMI was Q-wave or transmural infarction. Non-STEMI occurs when rising cardiac biomarkers are detected without ST segment elevation on the ECG, and replaced the term non-Q wave infarction.
 |
Missed MI. The term "missed MI" is not clearly defined in the literature. For the purposes of this article, missed MI refers to the patient discharged from the emergency department and subsequently found to have sustained an acute myocardial infarction during the next 48 to 72 hours. Because myocardial ischemia is a dynamic and unpredictable process, it is problematic to ascribe infarctions that occur more than a few days after the ED visit as having been "missed." Such later-occurring infarctions are more likely the natural progression of a chronic disease process. The acute MI can be either a STEMI or non-STEMI, and is defined as above with symptoms of ischemia, characteristic ECG changes, and rising cardiac biomarkers. This article will not include discussion of patients who develop an MI during their stay secondary to a different primary diagnosis, such as MI complicating sepsis. Patients with silent or unrecognized MIs will not be discussed.
Silent and Unrecognized MI. Silent MI strictly refers to patients who undergo myocardial infarction without any clinical symptoms recalled by the patient. Unrecognized MI occurs when the patient does not realize he or she is having an MI and does not present for care. This can be either because it is truly silent or because the patient has some symptoms associated with the MI but does not recognize their significance. Thus, the MI is unrecognized by the patient, not by a physician. Therefore, neither unrecognized nor silent MIs are "missed MIs" as defined in this paper. In both cases, these events are detected incidentally at a later date by discovery of new Q waves, echocardiographic evidence of wall motion defects, or on autopsy. Confusion can arise, as the term unrecognized MI is used by some authors interchangeably with silent MI,13 while most others consider silent MIs to be a subcategory of unrecognized MIs.7
The concept of truly silent ischemia has been challenged recently. Perhaps silent events are not truly silent. Even though the patient may not recall any symptoms does not necessarily mean that subtle or atypical symptoms were not present during the episode. One study of patients undergoing exercise stress testing with myocardial perfusion imaging found that 12% of women and 36% of men had evidence of ischemia without pain.14 Instead of pain, these patients reported vague symptoms such as diaphoresis, headache, tense muscles, or hot flashes.14 Unfortunately, but perhaps not surprisingly, the authors concluded that while symptoms were present with the patient's ischemia, they were too vague to be clinically useful alone.
The percentage of MIs that are thought to be unrecognized is much higher than one might first expect. Approximately 35% of all MIs are thought to be clinically unrecognized.15 Although they are undetected when acute intervention could be used, they are no less deadly. Overall, unrecognized MIs carry the same prognosis as those that are clinically evident.16
Scope of the Problem
Every emergency physician learns early in training that missing the diagnosis of MI tops the list of highest dollar awards in malpractice cases every year in the United States, accounting for nearly 30% of all dollars paid out.17 While missed fracture and infection cases are more common malpractice lawsuits overall,18 missed MI awards are so high because the cases often end in death or severe disability of relatively young and seemingly "healthy" patients. It is not through lack of effort that these patients slip through the cracks. Emergency physicians do their best every day to avoid such outcomes, yet they still occur. Some surveys have shown that the looming specter of malpractice lawsuits prompts some physicians to order tests or procedures for defensive reasons when they are not necessarily indicated.18 One recent study attempted to determine the effect that fear of a malpractice lawsuit has on a group of emergency physicians in the context of evaluation of chest pain patients.17 They found that physicians who had the highest level of fear of malpractice lawsuit as estimated by a questionnaire ordered more chest radiographs and troponin levels and admitted their patients more frequently to telemetry or ICU beds.17
These awards are against primary care physicians as well as emergency physicians, but emergency physicians are at particularly high risk due to the number of patients who come to the ED for evaluation of chest pain. The numbers are intimidating. According to the National Center for Health Statistics, there were 119.2 million ED visits in 2006, or 40.5 visits per 100 people in the United States.20 Chest pain was the most common chief complaint for all patients older than 15 years of age, resulting in more than 6 million visits in 2006 and nearly 2 million admissions.20 Another 2.6 million patients presented for dyspnea or abdominal pain, both of which can be the chief complaint in acute MI. From these millions of patients, the emergency physician is charged with correctly identifying those whose symptoms are caused by acute MI from those with other causes (i.e., GERD, pneumonia, emphysema, CHF, etc).
Not only is chest pain the most frequent chief complaint for ED visits, but cardiovascular disease is both common and deadly. Cardiovascular disease is the number one cause of death for adults in developed countries, with nearly 700,000 deaths each year in the United States. In 2008, roughly 1.2 million patients presented to EDs in the United States with diagnosis of acute MI.21 Given the published rates of missed MI (2-6%),1,2,3 that translates to 24,000–72,000 missed MIs each year in the United States. Another way to put this in perspective is to think of the number of MIs missed by the average emergency physician per year. If one sees roughly 2.5 patients per hour and works 2000 hours/year, 5000 ED patients will be seen each year. Around 250-400 patients (5-8%) will have chest pain, 50 patients (1%) will have MIs, and 1-3 patients (2-6% of the 50 MIs) each year inadvertently will be sent home having acute MIs. If one's career spans 25 years, then an individual ED physician will have missed roughly 25-75 MIs. For the average ED seeing 60,000 patients per year, this translates into 1-3 patients per month of acute MIs being discharged from just one ED.
Even with advances in diagnostics, such as use of cardiac biomarkers, the number of patients inadvertently discharged with undiagnosed MI has stayed relatively constant over the last few decadesbetween 2-6%.1-3 Interestingly, the numbers for other countries are similar: 3% for Canada22 and 6% for the U.K.23 Given the highly variable clinical presentation of MI patients (general fatigue, dyspnea without pain, indigestion, etc), it is not surprising that emergency physicians cannot reach 100% accuracy. Indeed, achieving up to a 98% success rate in this context could be considered a notable accomplishment. However, emergency physicians cannot be content with a 98% capture, as patients who are discharged from the ED with a missed MI have an increased risk of short-term mortality from 10-26%.1 Given the highly successful and time-sensitive treatments of cardiac catheterization and medical thrombolysis, it is essential to accurately detect as many patients presenting to the ED with acute MI as possible.
One recent study helped identify that MIs may be more likely to be missed in lower-volume EDs than in larger-volume ones.24 The percentage of missed MIs in this large study was consistent with lower number from most recent estimates, and was 2.1% of all MIs seen.24 However, significant variability was seen in the number of MIs missed compared to the overall number of MI patients seen annually. The study examined records of nearly 20,000 MIs seen in 171 EDs across Ontario, Canada, and found that the lower-volume EDs had roughly twice the number of missed MIs.24 Exact causes of the differences could not be determined, but this may correlate with similar lower complication rates for surgeons who perform higher numbers of a specific type of surgery. Regardless of the cause, it serves as a reminder for physicians who practice in lower-volume EDs that they may be practicing in higher-risk arenas. In addition, the patients in the study were identified when they returned to the ED for a second visit in which they were diagnosed correctly. This reinforces the concept of expanding the work-up when a patient returns to the ED just a few days after being seen.
Pathophysiology of Coronary Artery Disease
Recent advances have improved understanding of the nature of coronary artery disease from that of a simple cholesterol storage disease to a complex interaction between endothelial cells and multiple other factors. Coronary lesions that were once only thought of in terms of the degree of narrowing they produced are now considered to be more intricate as well. While emergency physicians are not the ones making decisions about which lesions require stenting, we often are using patient's previous catheterization reports in our judgment of whether to admit the patient. A brief overview of recent advances in the development of coronary lesions can help with understanding when patients are at higher risk.
Coronary lesions are defined in terms of size and generally are classed as non-obstructive (< 50% stenosis) or obstructive (> 50% stenosis). Obstructive lesions are further subdivided as moderate obstruction (50–70% stenosis), significant obstruction (70-90% stenosis), and high-grade lesions (90-99% stenosis). Interestingly, cardiology texts consider stenoses < 50% as "insignificant disease,"9 and some cardiology literature classifies those with < 50% stenosis as "normal" arteries.25
In general, the stable obstructive lesions are responsible for angina. It is thought that a lesion needs to be at least 60% to produce exercise-induced angina and > 95% to cause angina at rest.26 When the surface of lesion ruptures or erodes, it triggers a clot to form in and sometimes completely block the artery lumen, thus causing an MI. Although one may expect these high-grade lesions to be responsible for most acute MIs, in fact most acute MIs are caused by lesions < 50%.26 Only 15% of acute MIs arise from lesions > 60%.27 This is thought to occur because smaller lesions have a less stable covering and therefore lead to plaque rupture/erosion and vessel thrombosis more easily.27 It remains true that, individually, high-grade lesions are more likely to cause an MI; it's just that smaller lesions vastly outnumber the high-grade ones.28
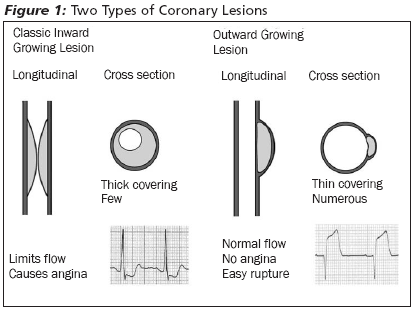
One major recent finding that can help the ED physician understand the variability seen with coronary lesions is that plaques do not just grow into the artery lumen; they grow outward as well.29 (See Figure 1.) Some authors believe that the lesion begins to grow outward at first, and only intrudes into the vessel lumen after maximum remodeling of the artery has occurred. Recent use of intravascular ultrasound and coronary CT has shown that patients can have extensive atherosclerosis but have a negative catheterization since the lesions are projecting outward rather than into the artery lumen.30 Thus, cardiac catheterization alone may be seeing only the tip of the iceberg of the true extent of coronary disease.27 The catheterization can be thought of as a "lumenogram," and disease not directly affecting the artery lumen can be overlooked.
Unfortunately, outward growing lesions still are at risk of triggering an acute MI, as these lesions still abut the vessel endothelium and can rupture into the artery producing STEMIs (see Figure 1).26 Unlike in patients with obstructing lesions, these patients do not have any preceding anginal symptoms and their MI comes "out of nowhere." Intravascular ultrasound studies also have confirmed autopsy data from young soldiers in the Korean and Vietnam wars by showing 17% of patients younger than age 20 years who were studied had evidence of coronary plaques.31 Further, all lesions were once thought to grow in a continuous fashion over time, but it is now believed that many grow in spurts that likely correspond to mild ruptures and thrombosis that do not lead to artery occlusion every time.28 Moreover, these minor ruptures are thought to be commonplace events in many patients with coronary disease.28 Thus a patient may have a "stable" lesion on serial catheterizations that suddenly becomes much larger, producing new symptoms or an acute MI.
 |
The location of the lesion in the vessel also is important. Ostial and bifurcation lesions are particularly high risk, both in terms of risk of rupture and risk of restenosis after intervention.32 With more proximal lesions, larger amounts of myocardium will be at risk when the plaque ruptures; hence the term "widow maker" for proximal left anterior descending (LAD) or left main lesions. High-grade lesions in both the proximal LAD and circumflex are referred to as left main equivalent, as they are the two branches of the left main. Lesions of smaller arteries can produce much smaller MIs in terms of heart muscle damage, but they can still trigger arrhythmias. Eccentric lesions and very long lesions also are more complicated to stent. The severity of longer, concentric lesions can be underestimated, as they are harder to visualize on catheterization. Likewise, other coronary branches can be superimposed on the vessel being studied and can hide lesions. In some patients, significant collateral flow can develop to compensate for high-grade lesions, or even complete large vessel occlusion. Thus, these lesions remain clinically silent longer than one would expect based on their appearance alone.
The bottom line for evaluating a patient whose previous catheterization results are available is for the ED physician to realize that coronary lesions are not simple static deposits of cholesterol. Size matters, in that high-grade lesions are dangerous, but the smaller or even lesions not visible with standard catheterization can more often be the source of an acute MI.
Risk Factors for Coronary Artery Disease
Five major risk factors for the development of coronary artery disease were identified more than 40 years ago in the classic study of people in Framingham, MA: smoking, hypertension, diabetes, increased cholesterol, and family history.33 These risk factors have been verified as predictive of the patient's risk of developing coronary artery disease over his or her lifetime. Since then, additional risk factors of obesity, inactivity, male sex, and age older than 65 years have been identified.34 Others, such as chronic inflammation, have been suggested.35 The declining mortality rate from coronary artery disease, down since a peak in the 1950s, has also led some to suggest that a "prolonged infectious epidemic" (Chlamydia pneumoniae) may play a role in coronary disease as well.35
 |
Do Risk Factors Matter in the ED? Although these risk factors for coronary artery disease are well known, how much influence they should have on ED evaluations of patients with chest pain has been harder to define. In the past, it was purported that lack of any risk factors for cardiac disease was seen in nearly 50% of patients treated for acute MI.36 Some of these data originated from the Framingham studies as well.37 Indeed, multicenter studies done on patients presenting with chest pain have shown only existing diabetes and family history conferred increased risk of coronary disease, and even then the increased risk was minimal.38 This led to suggestions that risk factors, with the exception of age, male sex, and family history of premature disease, are not considered relevant in evaluation of ED patients with chest pain. Likewise, their absence could falsely reassure the physician that a particular patient is less likely to have active coronary disease.39 Thus traditional risk factors were thought of as items to target for reducing a patient's lifetime risk of coronary artery disease, and not as factors that correlate with a given chest pain patient's risk of MI during any one ED visit.39
This advice seems counter-intuitive to one's experience as a practicing ED physician. It still seems to be that patients with tobacco use, hypertension, obesity, etc., are at higher risk for ACS when they present to the ED with chest pain. It also seems hard to reconcile that if classic risk factors increase patients' lifetime risk of coronary disease, shouldn't the risk of presenting to the ED with an acute MI go up as they age as well? Further, past use of nitrates for chest pain, presumably for a diagnosis of coronary disease, does correlate with diagnosis of ACS in the ED.40 This should be distinguished from relief of chest pain with nitroglycerin in the ED, which does not.41 Fortunately a much larger study has shown that classic risk factors are indeed associated with increased risk of MI.42
This study, by Khot, et al, examined 76,716 STEMI cases and 35,527 non-STEMI/unstable angina cases and evaluated them for the presence of diabetes, hypertension, tobacco use, and elevated cholesterol. They found that overall, nearly 85% of women and 81% of men did indeed have at least one of these risk factors.42 In other words, only 15-20% of patients with acute MI did not have any risk factors; much less than the 50% level that had been reported in the past. For younger patients with MI (men < 55 years, women < 65 years), the results were even more dramatic; only 10-15% lacked any risk factors.42 Cigarette smoking in both men and women, as well as diabetes in women, correlated with the development of premature coronary artery disease. Tobacco use lowered the age of onset by 10 years for every group.42 The authors may have identified an explanation for the disparity of the results in this and in past studies. A significant number of patients who presented with acute MI had undiagnosed disease. Fully 32% of the patients had undiagnosed hypertension,43 and 30% had undiagnosed diabetes.44 Thus patients may not have presented with documented risk factors at the time of their acute MI, but the conditions were still present in many cases. They were simply undiagnosed and therefore undocumented. Even after this research, the relevance of risk factors when evaluating chest pain patients in the ED is still debated.45 At this point, the best assessment is to be careful and realize that just because a patient does not have risk factors for coronary artery disease does not mean one should disregard their chance of having an acute MI. Ultimately, their description of the symptoms and ECG findings are more important than the presence or absence of classic risk factors.46
Classic Risk Factors. Braunwald's text on cardiology identifies tobacco use as the single most important risk factor for development of coronary artery disease.34 Nearly 50% of the 450,000 annual deaths in the United States from tobacco use are due to ischemic heart disease.34,47 Women who smoke and use oral contraceptives or who also have diabetes are at especially high risk. Hypertension is a particular risk, as nearly 50 million people in the U.S. have hypertension, and it is often clinically silent. Up to 33% of patients with hypertension remain undiagnosed, and 25% are not effectively controlled.48 Further, as more becomes known about hypertension, even "normal" levels of 130-139 mmHg systolic and 85-89 mmHg diastolic have been shown to increase risk by a factor of two.49
 |
Increased cholesterol levels also have been shown to begin the process of atherosclerosis in people as young as teenagers.34 Thus elevated cholesterol and triglyceride levels increase risk of artery disease in general, not just in the coronary bed. Family history of premature onset coronary artery disease, defined as onset of disease < 50 years, and/or coronary artery disease in first-degree relatives also is associated with increased risk of coronary disease. Family history alone also has been shown to be an independent risk factor for development of disease.50 The effect seems to confer higher risk for women with first-degree relatives and nearly doubles their risk compared to men.51
The risk of diabetes has been equated with aging 15 years, an impact even higher than smoking.52 Lastly, obesity, defined as a BMI > 30, is now present in 30% of the U.S. population.34 Even mild obesity has been shown to correlate strongly with coronary artery disease, even in the absence of other risk factors.53
The History and Physical: Utility of "Classic" Symptoms and Signs
Angina was first described by Heberden in 1768,54 and physicians are very familiar with the current classic description of angina: crushing substernal pain that feels like an elephant on your chest. Angina often is described as pressure, tightness, heaviness, or burning instead of pain by the patient. The pain is made worse by exertion, relieved by rest; radiates to the left arm, shoulder, neck, or jaw; and is associated with shortness of breath, nausea, vomiting, and diaphoresis. It is unlikely that patients presenting with these classic symptoms will be among the missed MIs. However, it is important to take the time to elicit the proper history and ask about onset, location, severity, duration, radiation, precipitating/alleviating factors, and associated symptoms. Often classic symptoms can be present, but patients not used to giving medical histories may not provide them unless specifically prompted. Also be aware that "sharp" pain can mean different things to different cultures; some define it as knife-like, while in others it simply may mean severe.
This section will review data on the usefulness of symptom descriptions. It is important to remember that no single type of data, be it physical examination, ECG, or enzymes alone can rule out presence of an MI, and history alone is no exception.55 While history itself cannot rule out the presence of coronary disease, it is useful to classify patients into low-, medium-, and high-risk groups.56 Beware that atypical presentations are especially common in certain groups (elderly, women, younger patients), and these will be reviewed in the next section.
Chest Pain. Several studies have shown that the description of pressure-type pain only weakly correlates with presence of MI.57 Sharp, pleuritic, or positional pain is less likely to be cardiac but is still seen in 5% of MIs.58 Only a few studies examined usefulness of the actual location of the chest pain and found none.59 Right-sided pain can be just as dangerous as left-sided or central pain.46 Likewise, the area of the heart infarcting (anterior, posterior, inferior) in general does not correlate with a certain location on the chest, but inferior MIs are seen with upper abdominal pain more often then anterior ones.60 In addition, nausea and/or vomiting is seen more often with inferior compared to anterior MIs.7 Radiation of pain to the shoulders or arms has been shown to correlate with increased risk for MI in multiple studies.55,61
Pain precipitated by exertion is associated with increased risk of MI.61 Besides exertional pain, pain that wakes the patient in the morning does correlate with acute MI.46 Chest pain that reaches maximum intensity at onset is more suggestive of aortic dissection, whereas pain that follows a crescendo pattern is more indicative of angina. Pain that lasts for hours or days with each episode or pain that lasts only seconds is considered unlikely to be ischemic, but this has not been formally studied.62 Angina typically resolves in 15-20 minutes, whereas MI pain persists after 30 minutes. Pain reproduced by palpation also is associated with non-cardiac pain,61 but remember it is still seen in 5% of MIs.58 However, one older study found that risk was even less in patients with a combination of atypical features. No patients had an MI and only 3% had unstable angina who presented with sharp/stabbing chest pain that was pleuritic, positional, or reproduced by palpation.63
Relief of chest pain with nitroglycerin or a GI cocktail has be widely used in the past to confirm or exclude a patient's chest pain as likely cardiac or non-cardiac, respectively.55 While nitroglycerin does relax coronary vessel smooth muscle, it also relaxes esophageal smooth muscle as well and can provide relief from esophageal-based chest pain. At least two recent studies have shown a lack of association between relief from nitroglycerin and MI.41,64 Likewise, studies have shown that relief of chest pain with GI cocktail does not predict absence of MI.65 Lastly, the Levine sign (closed fist over chest) also has been classically associated with angina, but when tested is less clinically significant. One study found that it had reasonable specificity (78-86%), but the positive predictive value was only 55%.66
Associated Symptoms. Few studies have looked specifically at associated symptoms for acute MI, which typically include nausea, vomiting, shortness of breath, or diaphoresis. These features often are highlighted in certain patient populations, such as the elderly patients with shortness of breath, and will be discussed in a later section on atypical presentations. Results available on this topic are mixed. One study of clinical features in patients with chest pain showed no correlation with nausea, vomiting, or diaphoresis and acute MI,61 while two meta-analyses did show a correlation with nausea and diaphoresis with MI.57,67 However, the data were varied; nausea was 24-56% sensitive and 70-84% specific, while diaphoresis was 24-61% sensitive and 73-84% specific.67 Another study evaluated these symptoms as presenting complaints in patients with MI but no pain.68 In these patients, dyspnea was twice as common as other associated symptoms (seen in 49%); diaphoresis was seen in 25%, and nausea and vomiting in 24%.68
Physical Examination. Physical examination findings generally are absent in stable patients with acute MI. One study found no correlation with physical findings in this patient population.67 One thing that can be helpful is to examine the patient's skin in the area of pain, especially when pain has been present for more than 1 day. In a few cases, early signs of shingles will be seen, thus solving the dilemma of the chest pain's origin. When physical signs are present in acute MI, more often they identify severely ill patients. Signs of acute CHF (such as jugular venous distension, rales, peripheral edema, and a third heart sound) resulting from acute MI indicate abrupt pump failure. Likewise, hypotension or shock with pallor, cool skin, and mottled extremities may be signs of impending arrest and should be treated aggressively. As mentioned above, reproducibility of chest pain by palpation does lessen the risk of MI but is still seen in 5% of acute MIs.58
In practice, one often encounters a patient complaining of chest pain whose blood pressure is elevated. It is not uncommon for patients who can detect when their blood pressure is elevated to notice chest pain, shortness of breath, headache, or blurred vision with elevated blood pressure. Several authors have stressed the importance of not over-treating blood pressure numbers alone due to the risks of rapidly lowering the patient's blood pressure.69 Realize that hypertensive emergency requires both elevated blood pressure, typically greater than 180/110 mmHg, and evidence of end-organ damage. In the context of this article, acute MI, unstable angina, and pulmonary edema from left ventricular failure are recognized as evidence of end-organ damage not simply the presence of chest pain.70 While pulmonary edema may be easy to rule out in the ED, the presence of MI or unstable angina may require admission to the hospital or a chest pain unit. While not every patient will require admission of any kind, correlation with the history for the event and patient's past medical history will be needed to determine safety and timing of discharge. It is useful to note that the most recent study of missed MI patients did not find any correlation with blood pressure elevation between patients who were missed and those diagnosed.1
Atypical Presentations
It is not surprising that in studies of missed MI patients, atypical presentation is one of the common reasons cited as an explanation for why the MI was not detected. Atypical presentations can include a lack of chest pain with emphasis on associated symptoms (dyspnea, diaphoresis, nausea/vomiting), new-onset heart failure, apparent stable angina, odd locations of pain (back, abdomen, isolated jaw pain, etc.), or vague symptoms such as general weakness.7 Besides atypical presentation, studies of missed MI patients also found that these patients lack typical risk factors, are relatively young or elderly, more often are women, and have non-diagnostic or subtle ECG findings.2,5,68 It is important to note that more than one of these "risk factors" for missed MI often occurs in the same patients, probably increasing their risk of being missed. One recent study of MI patients with normal or non-diagnostic ECGs found they presented without chest pain (11-14%) more often than those with diagnostic ECGs (24%).71 These patients are nearly 8 times more likely to be discharged from the ED with acute MI than those with diagnostic ECGs.1
The prevalence of MI patients with atypical presentations is higher than one might suspect. Several studies have found that atypical presentations are seen in 52-67% of all patients with acute MI.72,73 In a large study of 10,689 patients with suspected acute coronary syndrome, 18% presented without chest pain.74 Certain patient groups (diabetics, elderly, young, etc.) appear to be at higher risk for atypical presentations and will be discussed individually. The results of one recent report suggest that patients with chronic kidney disease also may be added to the list of those at higher risk for presenting with dyspnea instead of chest pain.75 One must maintain suspicion for these presentations because, as expected, these patients are at much higher risk for less aggressive care and experience increased mortality. One study found the mortality rate for patients presenting without chest pain was 23% compared to 9% for MIs with chest pain.76
Diabetics. Diabetes is an epidemic problem both in the United States and globally. According to the CDC, 24 million people, or 8% of the U.S. population, have diabetes. Nearly 95% of these are type II diabetics, and 25% of them are undiagnosed.77 While these are large numbers, they are likely to grow in the near future. Obesity and inactivity are increasingly common in young people, and it is currently estimated that another 57 million people in the United States are pre-diabetic.
Diabetes is one of the most significant risk factors for development of coronary disease. It is estimated to increase the risk of coronary disease equivalent to aging 15 years, which is even more of an impact that smoking.52 One study found 57% of patients admitted for ACS were undiagnosed diabetics.78 By 55 years of age, 35% of people with type I diabetes have died of coronary disease, and coronary disease is the number one cause of death for type II diabetics regardless of how long they have had diabetes.79 Further, in-hospital mortality for diabetics with MI is nearly twice that of non-diabetics, which has remained constant despite the development of thrombolytics or use of stents.80
It is well established that diabetics also are at high risk for atypical presentations of acute MI, especially silent MI or painless MI.7 Diabetics with MI often have associated symptoms (dyspnea, nausea/vomiting) as well as vague complaints of weakness or fatigue as their primary complaint. One study found these associated symptoms were the presenting complaint in 40% of diabetics with MI.81 The reasons for this are unclear, but it is suspected that they may have a "defective anginal warning system" from autonomic neuropathy.82 Acute dyspnea is one of the most common presenting complaints in these patients and is seen in 15-30% of painless MI cases.83 Often, dyspnea in these cases is due to acute pulmonary edema resulting from depressed cardiac function.
Elderly Patients. A host of factors make an acute MI more difficult to detect and more deadly in elderly patients. To start, elderly patients are at higher risk in general because their increased age translates to an increased incidence of coronary disease. Autopsy studies have shown 50% of elderly women and up to 80% of elderly men have obstructive coronary disease.84 In addition, their lesions are more severe and diffuse compared to those of younger patients.84 As expected, then, they also are at higher risk of death compared to younger patients. Patients older than 65 years account for 65% of the MIs and 80% of the deaths from MI each year.85 Data show the in-hospital mortality for patients younger than 60 years with MI was only 2.8% compared to 19% for patients older than 70 years.86 Another study found a higher rate of 32% for patients older than 75 years with MI.87
Secondly, the elderly are more likely to present with atypical symptoms. A study of more than 4100 elderly patients with unstable angina found that 51% of those older than 65 years had atypical presentations.73 Atypical presentations were defined as those lacking chest tightness/pressure with arm pain worsened by exercise or relieved by nitroglycerin. In that study, dyspnea was by far the most common chief complaint and was seen in 69% of cases. Only 14% of patients even had any chest pain,73 but arm or shoulder pain was seen in 11% and 7%, respectively. Upper abdominal pain was found in 8%, and 6% complained of neck pain. Nausea was found in 37%, but vomiting was found in only 11%, and diaphoresis occurred in 25% of cases. The most elderly patients (older than 85 years) have the most atypical presentations of all. Syncope, stroke symptoms, weakness, confusion, and delirium increase as presenting symptoms in this group.88
The elderly often present later in their course of illness. In some cases this can be because older patients can be more stoic. This may be due in part to reduced pain sensation. One study found that in patients with documented angina, older patients (62-70 years) had a significant delay in their perception of angina compared to younger patients (53-63 years).89 Other factors also contribute to their delayed presentations. Communication can be impaired from baseline dementia, making reliable histories extremely difficult to obtain, and they can be dependent on others for transportation. Comorbid conditions are more frequent in elderly patients with acute MI as well. Besides dementia preventing a clear history, studies on elderly patients with angina or MI show risk of atypical presentation is increased in older patients with history of CHF and diabetes, and in women.90 Risk for other diagnoses being confused with acute MI, such as aortic dissection, also goes up with age. The classic patient with an acute thoracic dissection is a man in his seventh decade.
Lastly, when the elderly do have MIs, they are more often non-STEMIs with non-diagnostic ECG findings rather than classic STEMIs. Elderly patients' ECGs often are abnormal at baseline as well, with bundle branch blocks, changes from hypertension, and rhythm abnormalities such as atrial fibrillation that complicate ECG interpretation. Thus, elevated enzyme markers are more often the path to diagnosis of elderly patients with MI. However, using troponin primarily to diagnose MI can be problematic as they are not 100% sensitive or specific. Elevated troponin also can be seen in renal failure91 and sepsis.92
Younger Patients. Younger patients, defined as those younger than 45 years of age, account for about 10% of acute MIs annually.93 Classic cardiac risk factors of diabetes, obesity, metabolic syndrome, and tobacco use appear to play a significant role in development of coronary disease in these patients. One study of 800 patients 18-45 years old with MI found that these patients were more often male, nonwhite, had history of hypertension, were current smokers, obese, and dyslipidemic compared to older patients with MI.94 Family history of MI was not addressed in that study but was confirmed to be a significant factor in other studies.95 It is important to note that another study still found the majority of younger patients were males, but 25% were female compared to only 10% in other series.96 In this same study diabetes and hypertension were actually less likely to be present in younger patients with MI. The risk from smoking is impressive and is equivalent to the risk of previous MI in older patients.94 Likewise, obesity has been noted to double the prevalence of heart disease in both men and women < 50 years.97 Cocaine abuse is also a major risk factor for MI in younger patients but is discussed in a separate section. Younger patients are also less likely to have angina episodes prior to their MI.93
Younger patients tend to have a higher frequency of anterior MIs than older patients, are more likely to have a STEMI than non-STEMI, and more often had single vessel disease.94 Up to 20% of young MI patients have no coronary lesions, and coronary spasm can be a factor in producing infarction. However, it should be noted that in several previous reviews, "normal" arteries were defined as those with less than 50% stenosis; thus the presence of non-obstructing lesions was ignored.25 When they included all coronary lesions, they found that 60% of young patients had single vessel, but 29% had two-vessel disease, and 11% had triple-vessel disease.25 Even so, younger patients with MI continue to experience lower morbidity and mortality than older patients with MI. However, youth alone did not provide longer-term protection when patients suffered from large MIs. Risk of poor outcome (death, repeat MI, CHF) was as high as 64% over 5 years in patients with ejection fractions < 45% after their first MI.95
Anomalous Coronary Artery. Although rare, anomalous coronary arteries also can be a source of MI in young patients. In some cases the defect is severe enough to produce symptoms in infancy (i.e., left main arising from pulmonary artery), but otherwise, they can produce sudden and profound ischemia during intense exercise in adolescents and young adults. An example is the single coronary artery where only one artery arises from the aorta to supply the entire heart. While the incidence from autopsy studies is only 1% in the general population, they are thought to cause 12-19% of the sudden deaths seen in high school and college athletes in the United States.98 Obviously we can be of little help to the young patient who dies on the field, but this can be considered when young athletes suffer chest pain and syncope during exertion. These patients should be referred to a cardiologist before being cleared to return to sports.
Kawasaki Disease. At times patients in their twenties can present with acute MI as a complication of previous infection with Kawasaki disease. Kawasaki disease is a relatively rare autoimmune disease that produces a systemic vasculitis in young children. Children younger than age 5 years account for 80% of cases, and it remains a clinical diagnosis.99 Most recent data show that Kawasaki disease is most common in Asian and Pacific Island Americans, followed by African Americans and Hispanics; the incidence is lowest in Caucasians.100 Coronary artery aneurysms can result and are seen in 20-25% of untreated patients, but use of intravenous immune globulin reduces the incidence to less than 10%.99 In most situations, development of new aneurysms after 6 weeks is rare and risk of MI is highest in the first year after illness. Small or medium size aneurysms (< 4-8 mm) typically spontaneously regress within 5 years,101 whereas giant ones (> 8 mm) may require bypass surgery. Recent data show that even though the aneurysms have regressed, intimal thickening and abnormal dilation may persist at that site. Unfortunately, cases exist of acute MI in 20-year-old patients as a result of known or previously unknown Kawasaki disease,102,103 with some even requiring bypass for extensive lesions.104 These patients have no other risk factor for coronary disease, and in some cases previous angiograms had shown regression of known aneurysms.102 Thus even apparently healthy, drug-free young people can present with acute MI, although this is rare. A screening ECG should be sufficient to detect STEMI from Kawasaki disease, as most aneurysms develop in proximal locations.
Women. Heart disease is the number one cause of death for women in the United States, and women account for about 50% of all MIs.83,105 Women do seem to have a higher incidence of unrecognized MI than men (54% vs. 33%) in at least one study.13 They also tend to be about a decade older than men when presenting with MI and therefore have higher incidence of comorbid conditions. When they do seek medical care for an MI, women tend to wait longer before coming in, especially those > 65 years of age.l06 Once diagnosed, several studies have found that women also fare worse that men when admitted for MI. A study of 2800 MIs in the 1990s found that the 30-day mortality for women with MI was 17% compared to 9% for men, but the 30-day to 1-year mortality differed less dramatically (9% for women vs. 5% for men).107
Some of these differences may be explained by the fact that women tended to be older and had more comorbidity. Another more recent work that matched subjects closely found that women overall did survive as well as men, but that women with STEMIs did not.108 Women had an in-hospital mortality double that of men (10% vs. 5%). In this study, less aggressive care for women with MI was suggested to explain the differences in survival rates.108 Women were less likely to receive aspirin, early beta-blocker treatment, and had delayed reperfusion therapies more often than men. In one series, women with typical chest pain were 32% less likely to undergo catheterization than men.109 Other studies have confirmed but not explained why women tend to receive less aggressive and invasive care compared to men.110
Women also are at higher risk for missed MI in the ED, especially if younger than 55 years of age.1 Some studies suggest that when women do have chest pain, they may not associate it with cardiac causes as often as men.111 Women also present with atypical features of MI more often than men, such as dyspnea, nausea, abdominal pain instead of chest/ neck/back/jaw pain.112 This was confirmed in a meta-analysis that found that ACS and no chest pain occurred in 30-37% of women compared to 17-27% of men.113 These authors suggested, though, that many of these atypical features also are common in elderly patients, and since women tend to present 10 years later than men, increased age at presentation may be more important than sex to explain these results.
Another study looked at prodromal symptoms experienced by women and found atypical symptoms were frequent then as well. Of the 515 women in the study, 95% noted prodromal symptoms, but the most common were unusual fatigue (70%) and sleep disturbance (48%).114 Only 29% reported any chest pain preceding their MI. Likewise, the most frequent symptoms associated with their MI were: dyspnea (58%), weakness (54%), and fatigue (43%). When women noted pain, it was most commonly in the back (37%). Left arm or shoulder pain (22%) was almost as common as chest pain (28%). These atypical presentations appear not to develop because women are having smaller MIs. At least one study found that ECG findings and peak enzyme levels were similar between women with atypical symptoms and men with classic presentations.83 Women also tend to have more frequent non-obstructing mechanisms for chest pain (mitral valve prolapse, syndrome X, or Prinzmetal angina), thus giving them a broader differential to work through. Lastly, detecting coronary disease in women with screening tests can be problematic, as women experience false positive stress tests more often than men.105 Ironically, when women do under angiography, they are much more likely than men to have normal coronary vessels. One study of 32,856 patients found 23% of women had normal vessels compared to 7% of the men, and the women were still more than 4 times more likely than the men to be re-admitted after their catheterization for continued chest pain.115
Not all authors think that coronary disease in women is that different, though. There also have been studies to suggest that women and men do not have significantly different presentations of MI. One study of chest pain in men and women during balloon inflation found no differences between the sexes.116 Further, chest pain in general was a poor indicator of ischemia, as 33% of men and 25% of women did not have pain during balloon inflation producing ST segment elevation.116 One also should be aware that the American College of Cardiology stated in the 2004 update on STEMI treatment that "symptom profiles for STEMI by sex generally appear more similar than different."117
Takotsubo Syndrome (Apical Ballooning Syndrome). The Takotsubo syndrome is a variation of STEMI presentation in women. This syndrome was first described in early 2000 in Japan118 and consists of patients with a characteristic "ballooning" of the apex of the heart. While first described in Japan, where it is believed to be responsible for about 1% of all admissions for suspected MI,119 it is being reported with increasing frequency in Caucasians in the United States.120 It is believed by some to be responsible for 1-2% of patients presenting with acute MI.121 The name comes from the Japanese word for a round-bottomed ceramic pot to catch octopus that is thought to resemble the shape of the left ventricle lumen seen on ventriculogram. These patients present with acute MI patterns on ECG but typically have no coronary lesions on angiography. In some cases they can develop elevation in their cardiac biomarkers, perhaps due to coronary spasm, but these elevations are mild in comparison to the large areas of akinetic regions seen on echocardiography.122 Most patients with this syndrome can become quite ill for a short time with complications from their apparent MI, and it can lead to cardiac arrest.123
This syndrome has several unique features. It is primarily seen in post-menopausal women (90% of cases), is reversible over a period of days to weeks, and seems to be triggered by emotional distress such as the loss of a loved one (hence the name "broken heart syndrome"). The majority of patients survive the episode, but many require aggressive care with vasopressor support and intra-aortic balloon pumps required in 40% of cases.119 The current theory is that the patient's stress condition triggers an excessive release of catecholamines, which leads to the cardiac effects.123 Although the mechanism behind these dramatic changes appears not to be due to coronary disease, the patients' ECGs undergo similar changes to standard STEMI. They present with chest pain and ST elevation in anatomic distributions, and the ECG changes evolve with return of ST elevation followed by deep T wave inversion similar to the natural evolution in acute MI.119 Even though these patients have dramatic ECG changes, their cardiac biomarkers elevate only modestly, and the wall motion defects seen on catheterization often cover the territory of all three coronary vessels in 90% of these women.119 The bottom line here is that these patients are clinically indistinguishable from standard acute MI. Although thought of as a mimic of acute MI, these patients present as a STEMI and must be treated as one, as it is only after catheterization that the true diagnosis is revealed.
Cocaine and Methamphetamine
Cocaine. Cocaine use, especially habitual use, adversely affects the heart in multiple ways. Besides MI, cardiovascular complications of cocaine include dilated cardiomyopathy, aortic dissection, and sudden death. On a cellular level, cocaine acts by releasing norepinephrine, dopamine, epinephrine, and serotonin. Norepinephrine induces ischemia by contraction of vascular smooth muscle, and epinephrine increases oxygen demand by increased contractility and heart rate. Cocaine also increases platelet activation and aggregation promoting thrombus formation. These changes can produce MIs in patients with normal coronary vessels, and those with pre-existing disease are at even higher risk. Vasospasm has been shown to be more severe in diseased vessels compared to normal vessels.124 Autopsy studies also suggest the rate of atherosclerosis is accelerated in habitual users.125 The frequency of coronary disease was much higher than in non-users and also much more advanced than expected for their age. One study of people who died with cocaine in their bloodstream found that 36% had at least 75% obstruction in one or more major coronary vessel; their average age was only 32 years.125 Cocaine also blocks sodium channels in cardiac myocytes, producing occasional wide complex tachycardias. Thus it is not surprising that cocaine use is the most frequent cause of drug-related deaths reported by medical examiners.126
Obtaining accurate history in cocaine-induced chest pain patients has unique problems. Many times, patients will not volunteer the fact that they have used cocaine, and may adamantly deny it when asked. They may be worried about legal issues such as parole violations, that their family may discover, or their job may be in jeopardy if use is confirmed. In most cases the urine drug screen is positive for 1-2 days after single use, but in cases of heavy abuse (> 5 g/day) urine tests may remain positive for 2-3 weeks after last use.127,128
Several large studies have confirmed that only about 6% of patients seen in the ED for cocaine chest pain will have an MI, and another 15% will have ACS.129,130 The typical patient with cocaine MI is a young man, non-white tobacco user, with repetitive cocaine use but few classic cardiac risk factors.129 The location, duration, and quality of chest pain ultimately are not useful alone to allow one to detect patients with higher risk of MI from those with only chest pain.131 Up to 33% of patients describe pleuritic pain.132
However, one study of 246 patients with cocaine chest pain found more classic angina symptoms were relatively common. Pain was substernal in 71% and pressure-like in 46%, while dyspnea was seen in 59% and diaphoresis in 38%.131
The risk of MI is independent of amount used, route of ingestion, and frequency of past use. Both casual users and habitual users are at risk,124 and MI has occurred in first-time users.133 Even in otherwise low-risk patients, the risk of MI is increased about 24-fold in the first 60 minutes after use.134 Infarction within the first hour is from direct cocaine toxicity, with events that occur after again several hours after use from the effects of cocaine metabolites. The incidence of MI is highest in the first 24 hours after use.129 In one study where cocaine users in drug treatment wore Holter monitors, silent episodic ST segment elevation was seen in 38% of patients up to two weeks after last use.135 Thus vasospasm can occur even when blood levels of cocaine are undetectable.133
Diagnosis of cocaine-induced MI can be problematic at best. First, one should remember several other diagnoses that can go along with cocaine chest pain. Pneumomediastinum occurs from forceful breath holding associated with smoking crack and can be a subtle fining on chest radiography. Acute aortic dissection also is associated with cocaine abuse. The incidence of cocaine-related dissection is thought to be rare, but one study of 38 cases of aortic dissection found that 37% were cocaine related.136 Most of these patients had other risk factors for dissection, such as hypertension and tobacco use. Cocaine users with aortic dissections tended to be younger (mean age 38 vs. 63 years for non-users), tobacco smokers, had higher blood pressures on presentation (210/130 mmHg on average), and had a higher incidence of type B (descending aorta) dissections (69% vs. 3%).137
ECG findings in cocaine MI are variable and may be misleading as often as helpful. The ECG can be non-diagnostic when the patient is having an MI and suggestive of ischemia when the patient is not.138 One study found patients having cocaine-related MI were just as likely to have normal or non-diagnostic ECGs as ones showing ischemia.132 In this study, 15% of patients with MI were missed in the ED based on their ECG readings, and patients were inadvertently discharged. Up to 84% of patients with cocaine chest pain can have abnormal ECGs.132 Early repolarization from left ventricular hypertrophy and J-point elevation can make the ECG more difficult to interpret and can obscure ischemic changes. Another review of cocaine-MIs found that 43% of patients had ECGs that met criteria for reperfusion but did not have MI.139 It is likely that transient vasospasm produced many of these findings. Ultimately the sensitivity of the ECG for MI with cocaine chest pain has been shown to be only 36%.132 Thus the ECG has a much higher false-negative rate in this population than in most chest pain patients.
Cardiac biomarkers are the ultimate tool for diagnosis of MI in the ED when ECG findings are nonconclusive, but they, too, can be tricky in setting of cocaine chest pain. General elevation of creatine kinase (CK) is common from skeletal muscle damage in cocaine abuse, and this can produce false positive CK-MB levels as well.140 Perhaps the most useful aspect of measuring CK-MB levels is to follow serial levels; those with elevated enzymes from skeletal muscle damage are likely to decline with time, whereas patients with actual MI should continue to rise over the next 6-12 hours.140 Troponin levels are more accurate than CK-MB for diagnosis of acute MI, and this is true in the setting of cocaine chest pain as well.141 Troponin-I and -T levels are specific for cardiac muscle and are less likely to be altered by rhabdomyolysis. In the end, most MIs will be diagnosed with enzymes rather than ECG findings.
Disposition of cocaine chest pain patients can be challenging as well. There are no clinical criteria alone to determine who is safe for discharge.131 Short-term observation, such as in a chest pain unit, is the best choice in most settings. Weber and colleagues studied 344 cocaine chest pain patients and found that most complications occurred within 12 hours of hospital presentation.142 Patients were observed for a 9-hour period with continuous ST segment monitoring and cardiac biomarker sampling every three hours. If no abnormal results were obtained, a cardiology consult followed with a Bruce protocol stress test. When not available in the ED, this could be arranged on a outpatient basis. The patients with negative stress tests were followed for 30 days afterward. No deaths were recorded, and there was only a 1.6% rate of subsequent MI, all of which occurred in those who continued to use and had additional coronary risk factors.142 On average 60% will continue to use cocaine after discharge from the ED, and 75% will have recurrent chest pain.129
Methamphetamine. Crystal methamphetamine is similar to crack cocaine in that both are smoked, producing a rush of euphoria. However effects of methamphetamine last for up to 12 hours, compared to only 15 minutes for crack cocaine. Those who binge with methamphetamine can stay awake for as long as 10 days with little food or drink.143 Cardiovascular complications of methamphetamine abuse also appear similar to cocaine but are less frequently documented in the literature.
Methamphetamine can cause acute MI by coronary vasoconstriction with or without thrombus formation, and chronic users can develop a dilated cardiomyopathy.133 Methamphetamine also may accelerate development of atherosclerosis like cocaine, as one autopsy study of methamphetamine users found a 19% incidence of coronary disease compared to only 5% of controls.144 Another study found a relatively high rate of coronary disease (17%) in patients with positive drug screens observed in a CPU setting.145 In 2004, methamphetamine-related MIs were noticed in Hawaii and since have been reported in areas where methamphetamine abuse is epidemic.146,147 Methamphetamine abuse is also a risk factor for acute aortic dissection and may be more dangerous than cocaine. An autopsy series found that 20% of patients who died from acute dissection were positive for methamphetamine, which made it the second most important risk factor behind hypertension.148 Ultimately, methamphetamine users with chest pain likely can be cared for in a similar fashion to cocaine users.
Minorities. Minorities generally have a higher incidence of coronary disease risk factors and, therefore, also have a higher rate of MI compared to whites. Hypertension occurs in 30% of African-American men and 27% of women compared to 25% of white men and 24% of white women.149 Cholesterol levels tend to be higher in African Americans compared to whites as well. Likewise, diabetes is more common in Hispanic people compared to whites (20% compared to 11%, respectively). Thus, as expected, minorities tend to develop coronary disease at higher rates than whites. The prevalence of coronary disease in African Americans is 9% for women compared to 5% for white women, although the rates for men are both 7%.149 For Hispanic women, the rate is also higher than for white women (7% vs. 5%), and again men are similar at 7%. One study found these differences translated into black patients with MIs presenting an average of 8–10 years younger than whites.150
Since minorities develop coronary disease more often, it may not be surprising that they tend to be discharged with a missed MI more often. Pope, et al. found in 2000 that the missed MI rate for black patients was 5.8% compared to only 1.2% for whites in the same study.1 They suggested the fact that black patients were 8-10 years younger and that a high percentage were women as possible explanations for this. Another study of inner-city ED MI patients presenting without chest pain found that black patients presented this way 2.5 times more often than Hispanics and 4.6 times more often than whites.151 The typical non-chest pain MI patient in that study was black with a history of CHF and presented with elevated blood pressure/pulmonary edema.151
Other studies also have found that African Americans unfortunately have worse outcomes from MI compared to other racial groups. The mortality rate for African Americans is 1.8 times that of whites.149 Interestingly, the rate for whites is roughly 1.5 times that of Hispanics, but Hispanics die from diabetes complications twice as often as whites.149 As with women, studies have found that minorities tend to have less aggressive care for coronary disease. One study of Hispanics found that they received PTCA only 13% of the time compared to 23% for whites.152 Likewise, African Americans received thrombolytic treatment less often in the 1990s (19% compared to 29% for whites),152 and recent articles show the same for coronary catheterization rates for acute MI (38% for African Americans, 45% for whites).153 Multiple reasons for these findings have been suggested, including later presentation of African Americans, more frequent atypical presentations, and less willingness to undergo invasive procedures.152,153
A recent study found that Asians are another minority that may be at higher risk. In a study of 2189 South Asians with angina, the authors found that they presented with atypical symptoms more often than white patients (60% vs. 52%).154 Further, South Asians were 42% less likely to undergo catheterization than whites,154 which could not be explained completely by atypical presentations. Other studies have shown that Asians tend to have higher mortality rates from coronary disease.155 One also should be aware that language barriers can confound history taking and that people from different cultures can present in overly stoic or hypervigilant form. Extra time should be taken with these patients when they present with chest pain or possible ACS symptoms to ensure the history is as accurate as possible, as it can be the key to diagnosis in some cases.
References
1. Pope JH, Aufderheide TP, Ruthazer R, et al. Missed diagnoses of acute cardiac ischemia in the ED. N Engl J Med 2000;342:1163-1170.
2. Lee TH, Goldman L. Evaluation of the patient with acute chest pain. N Engl J Med 2000;342:1187-1195.
3. Schor S, Behar S, Modan B, et al. Disposition of presumed coronary patients from an emergency room. A follow-up study. JAMA 1976:236:941-943.
4. Mehta RH. Missed diagnosis of ACS in the ER continuing challenges. N Engl J Med 2000;342:1207-1210.
5. Rusnak RA, Stair TO, Hansen K, et al. Litigation against the emergency physician: Common features in cases of missed myocardial infarction. Ann Emerg Med 1989;18:1029-1034.
6. McCarthy BD, Beshansky JR, D'Agostino RB, et al. Missed diagnoses of acute myocardial infarction in the emergency department: Results from a multicenter study. Ann Emerg Med 1993;22:579-582.
7. Antman EM, Braunwald E. ST-elevation myocardial infarction: Pathology, pathophysiology and clinical features. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 1207-1230.
8. Braunwald E, Antman EM, Beasley JW, et al. ACC/AHA guideline update for the management of patients with unstable angina and non-ST segment elevation myocardial infarction-2002: Summary Article: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients with Unstable Angina). Circulation 2002;106:1893-1900.
9. Cannon CP, Braunwald E. Unstable angina and non-ST elevation myocardial infarction. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 1319-1340.
10. Virmani R, Burke AP, Farb A, et al. Pathology of the vulnerable plaque. J Am Coll Cardiol 2006;47:C13-C18.
11. Kohn MA, Kwan E, Gupta M, et al. Prevalence of acute myocardial infarction and other serious diagnoses in patients presenting to an urban emergency department with chest pain. J Emerg Med 2005;29:383-390.
12. Thygesen K, Alpert JS, White HD. Joint ESC/ACCF/AHA/WHF Task Force for the Redefinition of Myocardial Infarction. Universal definition of myocardial infarction. J Am Coll Cardiol 2007;50: 2173-2195.
13. de Torbal A, Boersma E, Kors JA, et al. Incidence of recognized and unrecognized MI in men and women aged 55 and older: The Rotterdam Study. Eur Heart J 2006;27:729-736.
14. D'Antono B, Dupris G, Arsenault A, et al. Silent ischemia: Silent after all? Can J Cardiol 2008;24:285-291.
15. Kim HW, Klem I, Shah DJ, et al. Unrecognized non-Q-wave myocardial infarction: Prevalence and prognostic significance in patients with suspected coronary disease. PLoS Med 2009 21;6:e1000057.
16. Sheifer SE, Manolio TA, Gersh BJ. Unrecognized myocardial infarction. Ann Intern Med 2001;135:801-811.
17. Katz DA, Williams GC, Brown RL, et al. Emergency physician's fear of malpractice in evaluation of patients with possible acute cardiac ischemia. Ann Emerg Med 2005:46:525-533.
18. Kachalla A, Gandhl TK, Puopolo AL, et al. Missed and delayed diagnoses in the ED: A study of closed malpractice claims from 4 liability insurers. Ann Emerg Med 2007:49:196-205.
19. Lawthers A, Localio A, Laird N, et al. Physicians' perceptions of the risk of being sued. J Health Polit Policy Law 1992;17:462-482.
20. Pitts SR, Niska RW, Xu J, et al. National hospital ambulatory medical care survey: 2006 Emergency Department summary. Natl Health Stat Report 2008:7:1-40.
21. Rosamond W, Fleagl K, Furie K, et al. Heart disease and stroke statistics2008 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2008;117:e25-146.
22. Christenson J, Innes G, McKnight D, et al. Safety and efficiency of emergency department assessment of chest discomfort. CMAJ 2004;170:1803-1807.
23. Collinson PO, Premachandram S, Hashemi K, et al. Prospective audit of incidence of prognostically important myocardial damage in patients discharged from emergency department: commentary: Time for improved diagnosis and management of patients presenting with acute chest pain. BMJ 2000;320:1702-1705.
24. Schull MJ, Vermeulen MJ, Stukel TA. The risk of missed diagnosis of acute myocardial infarction associated with emergency department volume. Ann Emerg Med 2006;48:647-655.
25. Valika AA, Pica M, Goldstein J. Normal coronary arteries are rare in young patients with acute myocardial infarction. Catheter Cardiovasc Interv 2007;70:683-690.
26. Brady WJ, Harrigan RA, Chan T. Acute coronary syndromes. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 1154-1195.
27. Libby P, Theroux P. Pathophysiology of coronary artery disease. Circulation 2005;111:3481-3488.
28. Libby P. The vascular biology of atherosclerosis. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 987-1001.
29. Clarkson TB, Prichard RW, Morgan TM, et al. Remodeling of coronary arteries in human and nonhuman primates. JAMA 1994;271:289-294.
30. Arnett EN, Isner JM, Redwood DR, et al. Coronary artery narrowing in coronary heart disease: Comparison of cineangiographic and necropsy findings. Ann Intern Med 1979;91:350-356.
31. Tuzcu EM, Kapadia SR, Tutar E, et al. High prevalence of coronary atherosclerosis in asymptomatic teenagers and young adults: Evidence from intravascular ultrasound. Circulation 2001;103:2705-2710.
32. Kini AS. Coronary angiography, lesion classification and severity assessment. Cardiol Clin 2006;24:153-162.
33. Kagan A, Dawber TR, Kannell WB, et al. The Framingham study: A prospective study of coronary heart disease. Fed Proc 1962:21:52-57.
34. Ridker PM, Libby P. Risk factors for atherothrombotic disease. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 1004-1022.
35. Muhlestein JB. Chronic infection and coronary artery disease. Med Clin North Am 2000;84:123-148.
36. Futterman LG, Lemberg L. Fifty percent of patients with coronary artery disease do not have any of the conventional risk factors. Am J Crit Care 1998;7:240-244.
37. Wilson PW, D'Agostino RB, Levy D, et al. Prediction of coronary heart disease using risk factor categories. Circulation 1998;97:1837-1847.
38. Jayes R, Beshansky J, D'Agostino R, et al. Do patient's coronary risk factors predict acute cardiac ischemia in the emergency department? A multicenter study. J Clin Epidemiol 1992;45:621-626.
39. Jones ID, Slovis CM. Emergency department evaluation of the chest pain patient. Emerg Med Clin North Am 2001;19: 269-282.
40. Pozen M, D'Agostino R, Selker H, et al. A predictive instrument to improve coronary care unit admission practices in acute ischemic heart disease: A prospective multicenter clinical trial. N Engl J Med 1984;310:1273-1278.
41. Henrikson CA, Howell, EE, Bush DE, et al. Chest pain relief with nitroglycerin does not predict active coronary artery disease. Ann Intern Med 2003;139:979-986.
42. Khot, UN, Khot MB, Bajzer CT, et al. Prevalence of conventional risk factors in patients with coronary heart disease. JAMA 2003;290:898-904.
43. Hyman DJ, Pavlik VN. Characteristics of patients with uncontrolled hypertension in the United States. N Engl J Med 2001;345:479-486.
44. Norhammar A, Tenerz A, Nilsson G, et al. Glucose metabolism in patients with acute myocardial infarction and no previous diagnosis of diabetes mellitus: A prospective study. Lancet 2002;359:2140-2144.
45. Haro LH, Decker WW, Boie ET, et al. Initial approach to the patient who has chest pain. Cardiol Clin 2006;24:1-17.
46. Boie ET. Initial evaluation of chest pain. Emerg Med Clin North Am 2005;23: 937-957.
47. Centers for Disease Control and Prevention (CDC). Annual smoking-attributable mortality, years of potential life lost, and economic costsUnited States, 1995-1999. MMWR Morb Mortal Wkly Rep 2002;51:300-303.
48. Chobanian AV, Bakris GL, Black HR, et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: The JNC 7 report. JAMA 2003;289:2560-2572.
49. Vasan RS, Larson MG, Leip EP, et al. Impact of high-normal blood pressure on the risk of cardiovascular disease. N Engl J Med 2001;345:1291-1297.
50. Shea S, Ottman R, Gabrieli C, et al. Family history as an independent risk factor for coronary artery disease. J Am Coll Cardiol 1984;4:793-801.
51. Pohjola-Sintonen S, Rissanen A, Liskola P, et al. Family history as a risk factor of coronary heart disease in patients under 60 years of age. Eur Heart J 1998;19: 235-239.
52. Booth GL, Kapral MK, Fung K, et al. Relation between age and cardiovascular disease in men and women with diabetes compared with non-diabetic people: A population-based retrospective cohort study. Lancet 2006;368:29-36.
53. Yan LL, Daviglus ML, Liu K, et al. Midlife body mass index and hospitalization and mortality in older age. JAMA 2006;295:190-198.
54. Heberden N. Some account of a disorder of the breast. Med Transactions 1772;2: 59-67.
55. Swap CJ, Nagurney JT. Value and limitations of chest pain history in the evaluation of patients with suspected acute coronary syndromes. JAMA 2005;294:2623-2629.
56. Kontos MC. Evaluation of the emergency department chest pain patient. Cardiol Rev 2001;9:266-275.
57. Panju AA. The rational clinical examination. Is this patient having a myocardial infarction? JAMA 1998;280:1256-1263.
58. Lee TH, Cook EF, Weisberg M, et al. Acute chest pain in the emergency room: Identification and examination of low risk patients. Arch Intern Med 1985;145:65-69.
59. Everts B, Karlson BW, Wahrborg P, et al. Localization of pain in suspected acute myocardial infarction in relation to final diagnosis, age and sex, and site and type of infarction. Heart Lung 1996;25:430-437.
60. Pasceri V, Cianflone D, Finocchiaro ML, et al. Relation between myocardial infarction site and pain location in Q-wave acute myocardial infarction. Am J Cardiol 1995;75:224-227.
61. Goodacre S; Locker T; Morris F, et al. How useful are clinical features in the diagnosis of acute, undifferentiated chest pain? Acad Emerg Med 2002;9:203-208.
62. Constant J. The diagnosis of nonanginal chest pain. Keio J Med 1990;39:187-192.
63. Kinlen L. Incidence and presentation of myocardial infarction in an English community. Br Heart J 1973;35:616-622.
64. Diercks DB, Boghos E, Guzman H, et al. Changes in the numeric descriptive scale for pain after sublingual nitroglycerin do not predict cardiac etiology of chest pain. Ann Emerg Med 2005;45:581-585.
65. Servi RJ, Skiendzielewski JJ. Relief of myocardial ischemia pain with a gastrointestinal cocktail. Am J Emerg Med 1985;3:208-209.
66. Marcus GM, Cohen J, Varosy PD, et al The utility of gestures in patients with chest discomfort. Am J Med 2007;120:83-89.
67. Chun AA, McGee SR. Bedside diagnosis of coronary artery disease: A systematic review. Am J Med 2004;117:334-343.
68. Brieger D, Eagle KA, Goodman SG, et al. Acute coronary syndromes without chest pain, and underdiagnosed and undertreated high-risk group. Chest 2004;126:461-469.
69. Flanigan JS, Vitberg D. Hypertensive emergency and severe hypertension: What to treat, who to treat, and how to treat. Med Clin North Am 2006;90:439-451.
70. Stewart DL, Feinstein SE, Colgan R. Hypertensive urgencies and emergencies. Prim Care Clin Office Pract 2006;33: 613-623.
71. Welch RD, Zalenski RJ, Frederick PD, et al. Prognostic value of a normal or nonspecific initial EKG in acute myocardial infarction. JAMA 2001;286:1977-1984.
72. Uretsky BF, Farquhar DS, Berezin AF, et al. Symptomatic myocardial infarction without chest pain: Prevalence and clinical course. Am J Cardiol 1977;40:498-503.
73. Canto J, Fincher C, Kiefe C, et al. Atypical presentations among Medicare beneficiaries with unstable angina pectoris. Am J Cardiol 2003;91:118-119.
74. Pope J, Ruthazer R, Beshansky J, et al. Clinical features of emergency department patients presenting with symptoms of acute cardiac ischemia: A multicenter study. J Thromb Thrombolysis 1998;6:63-74.
75. Sosnov J, Lessard D, Goldberg RJ, et al. Differential symptoms of acute myocardial infarction in patients with kidney disease: A community-wide perspective. Am J Kidney Dis 2006;47:378-384.
76. Canto JG, Shilpack MG, Rogers WJ, et al. Prevalence, clinical characteristics, and mortality among patients with myocardial infarction presenting without chest pain. JAMA 2000;283:3223-3229.
77. McKinlay J, Marceau L. US public health and the 21st century: Diabetes mellitus. Lancet 2000;356:757-761.
78. Conaway DG, O'Keefe JH, Reid KJ, et al. Frequency of undiagnosed diabetes in patients with acute coronary syndrome. Am J Cardiol 2005;96:363-365.
79. Krolewski AS, Kosinski EJ, Warram JH, et al. Magnitude and determinants of coronary artery disease in juvenile-onset, insulin-dependent diabetes mellitus. Am J Cardiol 1987;59:750-755.
80. Schoenhagen P, Nissen SE. Coronary atherosclerosis in diabetic subjects: Clinical significance, anatomic characteristics, and identification with in vivo imaging. Cardiol Clin 2004;22:527-540.
81. Jacoby RM, Nesto RW. Acute myocardial infarction in the diabetic patient: Pathophysiology, clinical course, and prognosis. J Am Coll Cardiol 1992;20:736-744.
82. Morrow DA, Gershe BJ. Chronic coronary artery disease. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008:1353-1405.
83. Lusiani L, Perrone A, Pesavento R, et al. Prevalence, clinical features, and acute course of atypical myocardial infarction. Angiology 1994;45:49-55.
84. Williams MA, Fleg JL, Ades PA, et al. Secondary prevention of coronary heart disease in the elderly (with emphasis on patientsR75 years of age): An American Heart Association scientific statement from the Council on Clinical Cardiology Subcommittee on Exercise, Cardiac Rehabilitation, and Prevention. Circulation 2002;105:1735-1743.
85. Goldberg RJ, McCormick D, Gurwitz JH, et al. Age-related trends in short- and long-term survival after acute myocardial infarction: A 20-year population-based perspective (1975-1995). Am J Cardiol 1998;82:1311-1317.
86. Maggioni AP, Maseri A, Fresco C, et al. Age-related increase in mortality among patients with first myocardial infarctions treated with thrombolysis. N Engl J Med 1993;329:1442-1448.
87. Goldberg RJ, Gore JM, Gurwitz JH, et al. The impact of age on the incidence and prognosis of initial acute myocardial infarction: The Worcester Heart Attack Study. Am Heart J 1989;117:543-549.
88. Bayer AJ, Chadha JS; Farag RR, et al. Changing presentation of myocardial infarction with increasing old age. J Am Geriatr Soc 1986;34:263-266.
89. Ambepitiya G; Roberts M; Ranjadayalan K, et al. Silent exertional myocardial ischemia in the elderly: A quantitative analysis of anginal perceptual threshold and the influence of autonomic function. J Am Geriatr Soc 1994;42:732-737.
90. Gupta R, Kaufman S. Cardiovascular emergencies in the elderly. Emerg Med Clin North Am 2006;24:339-370.
91. Obialo CI, Sharda S, Goyal S, et al. Ability of troponin T to predict angiographic coronary artery disease in patients with chronic kidney disease. Am J Cardiol 2004;94: 834-836.
92. Ammann P, Maggiorini M, Bertel O, et al. Troponin as a risk factor for mortality in critically ill patients without acute coronary syndromes. J Am Coll Cardiol 2003;41: 2004-2009.
93. Choudhury L, Marsh JD. Myocardial infarction in young patients. Am J Med 1999;107:254-261.
94. Anderson RE, Pfeffer MA, Thune JJ, et al. High-risk myocardial infarction in the young: The VALsartan In Acute myocardial iNfarcTion (VALIANT) trial. Am Heart J 2008;155:706-711.
95. Fournier JA, Cabezon S, Cayuela A, et al. Long-term prognosis of patients having acute myocardial infarction when < 40 years of age. Am J Cardiol 2004;94:989-992.
96. Doughty M, Mehta R, Bruckman D, et al. Acute myocardial infarction in the young The University of Michigan experience. Am Heart J 2002;143;4-6.
97. Hubert HB, Feinleib M, McNamara PM, et al. Obesity as an independent risk factor for cardiovascular disease: A 26-year follow-up of participants in the Framingham Heart Study. Circulation 1983;67:968-973.
98. Van Camp SP, Bloor CM, Mueller FO, et al. Nontraumatic sports death in high school and college athletes. Med Sci Sports Exerc 1995;27:641-647.
99. Barron KS. Kawasaki disease: Etiology, pathogenesis and treatment. Cleve Clin J Med 2002;69:SII69-SII78.
100. Taubert K. Epidemiology of Kawasaki disease in the United States and worldwide. Prog Pediatr Cardiol 1997;6:181-185.
101. Akagi T. Interventions in Kawasaki disease. Pediatr Cardiol 2005;26:206-212.
102. Tsuda E, Hanatani A, Kurosaki K, et al. Two young adults who had acute coronary syndrome after regression of coronary aneurysms caused by Kawasaki disease in infancy. Pediatr Cardiol 2006;27:372-375.
103. Shiraishi J, Sawada T, Tatsumi T, et al. Acute myocardial infarction due to a regressed giant coronary aneurysm as possible sequela of Kawasaki disease. J Invasive Cardiol 2001;13:569-572.
104. Sivalingam SK, Parthasarathy HK, Choong CK, et al. Severe triple vessel coronary artery disease and aneurysms in a young white man: Disease progression of childhood Kawasaki disease. J Cardiovasc Med (Hagerstown) 2009;10:170-173.
105. Brady WJ, Harrigan RA, Chan T. Acute coronary syndromes. In: Marx, JA ed. Rosen's Emergency Medicine: Concepts and Clinical Practice, 6th ed. St. Louis: Mosby; 2006:1154-1195.
106. Lefler LL, Bondy KN. Women's delay in seeking treatment with myocardial infarction: A meta-synthesis. J Cardiovasc Nurs 2004;19:251-268.
107. Gottleib S, Harpaz D, Shotan A. Sex differences in management and outcome after acute myocardial infarction in the 1990s. A prospective observational community-based study. Circulation 2000;102:2482-2490.
108. Jneid H, Fonarow GC, Cannon CP, et al. Sex differences in medical care and early death after acute myocardial infarction. Circulation 2008;118:2803-2810.
109. Zaman MJ, Junghans C, Sekhri N, et al. Presentation of stable angina pectoris among women and South Asian people. CMAJ 2008;179:659-667.
110. Vaccarino V, Rathore SS, Wenger NK, et al. Sex and racial differences in the management of acute myocardial infarction, 1994 through 2002. N Engl J Med 2005;353:671-682.
111. Granot M, Goldstein-Ferber S, Azzam ZS. Gender differences in the perception of chest pain. J Pain Symptom Manage 2004;27:149-155.
112. Goldberg RJ, O'Donnell C, Yarzebski J, et al. Sex differences in symptom presentation associated with acute myocardial infarction: A population-based perspective. Am Heart J 1998;136: 189-195.
113. Canto JG; Goldberg RJ; Hand MM, et al. Symptom presentation of women with acute coronary syndromes: Myth vs reality. Arch Intern Med 2007;167:2405-2413.
114. McSweeney JC, Cody M, O'Sullivan P, et al. Women's early warning symptoms of acute myocardial infarction. Circulation 2003;108:2619-2623.
115. Humphries KH, Pu A, Gao M, et al. Angina with "normal" coronary arteries: Sex differences in outcomes. Am Heart J 2008;155:375-381.
116. Caldwell MA, Pelter MM, Drew BJ. Chest pain is an unreliable measure of ischemia in men and women during PTCA. Heart Lung 1996;25:423-429.
117. Kalla K, Christ G, Karnik R, et al. Implementation of Guidelines Improves the Standard of Care: The Viennese Registry on Reperfusion Strategies in ST-Elevation Myocardial Infarction. Circulation 2006;113:2398-2405.
118. Nyui N, Yamanaka O, Nakayama R, et al. "Tako-Tsubo" transient ventricular dysfunction: A case report. Jpn Circ J 2000;64:715-719.
119. Sharkey SW, Lesser JR, Zenovich AG, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation 2005;111; 472-479.
120. Desmet WJ, Adriaenssens BF, Dens JA. Apical ballooning of the left ventricle: First series in white patients. Heart 2003;89:1027-1031.
121. Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): A mimic of acute myocardial infarction. Am Heart J 2008;155:408-417.
122. Kurisu S, Inoue I, Kawagoe T. Apical ballooning in Takotsubo cardiomyopathy. Can J Cardiol 2008;24:921.
123. Wittstein IS, Thiemann DR, Lima JA, et al. Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med 2005;352: 539-548.
124. Lange RA, Cigarroa RG, Yancy CW Jr, et al. Cocaine-induced coronary-artery vasoconstriction. N Engl J Med 1989;321:1557-1562.
125. Dressler FA, Malekzadeh S, Roberts WC. Quantitative analysis of amounts of coronary arterial narrowing in cocaine addicts. Am J Cardiol 1990;65:303-308.
126. Lange RA, Hillis LD. Cardiovascular complications of cocaine use. N Engl J Med 2001;345:351-358.
127. Goldstein RA, DesLauriers C, Burda AM. Cocaine: History, social implications, and toxicityA review. Dis Mon 2009;55:6-38.
128. Burke WM, Ravi NV, Dhopesh V, et al. Prolonged presence of metabolite in urine after compulsive cocaine use. J Clin Psychiatry 1990;51:145-148.
129. Hollander JE. The management of cocaine-associated myocardial ischemia. N Engl J Med 1995;333:1267-1272.
130. Weber JE, Chudnofsky CR, Boczar M, et al. Cocaine-associated chest pain: How common is myocardial infarction? Acad Emerg Med 2000;7:873-877.
131. Hollander JE, Hoffman RS, Gennis P, et al. Prospective multicenter evaluation of cocaine-associated chest pain. Cocaine Associated Chest Pain (COCHPA) Study Group. Acad Emerg Med 1994;1:330-339.
132. Hollander JE, Hoffman RS, Gennis P, et al. Prospective multicenter evaluation of cocaine associated chest pain. Acad Emerg Med 1994;1:330-339.
133. Lange RA, Hillis LD. Toxins and the heart. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 1805-1814.
134. Mittleman MA, Mintzer D, Maclure M, et al. Triggering of myocardial infarction by cocaine. Circulation 1999;99:2737-2741.
135. Nademanee K, Gorelick DA, Josephson MA, et al. Myocardial ischemia during cocaine withdrawal. Ann Intern Med 1989;111:876-880.
136. Hsue PY, Salinas CL, Bolger AF, et al. Acute aortic dissection related to crack cocaine. Circulation 2002;105;1592-1595.
137. Singh S, Trivedi A, Adhikari T, et al. Cocaine-related acute aortic dissection: Patient demographics and clinical outcomes. Can J Cardiol 2007;23:1131-1134.
138. Hollander JE, Henry TD. Evaluation and management of the patient who has cocaine-associated chest pain. Cardiol Clin 2006;24:103-114.
139. Gitter MJ, Goldsmith ER, Dunbar DN, et al. Cocaine and chest pain: Clinical features and outcome of patients hospitalized to rule out myocardial infarction. Ann Intern Med 1991;115:277-282.
140. Tokarski GF, Paganussi P, Urbanski R, et al. An evaluation of cocaine-induced chest pain. Ann Emerg Med 1990;19: 1088-1092.
141. Hollander JE M. Levitt A, Young GP, et al. Effect of recent cocaine use on the specificity of cardiac markers for diagnosis of acute myocardial infarction. Am Heart J 1998;135:245-252.
142. Weber JE, Shofer FS, Larkin GL, et al. Validation of a brief observation period for patients with cocaine-associated chest pain. N Engl J Med 2003;348:510-517.
143. Buxton JA. The burden and management of crystal meth use. CMAJ 2008;178: 1537-1539.
144. Karch SB, Stephens BG, Ho CH. Methamphetamine-related deaths in San Francisco: Demographic, pathologic, and toxicologic profiles. J Forensic Sci 1999;44:359-368.
145. Diercks DB; Kirk JD; Turnipseed SD, et al. Evaluation of patients with methamphetamine- and cocaine-related chest pain in a chest pain observation unit. Crit Pathw Cardiol 2007;6:161-164.
146. Wijetunga M, Bhan R, Lindsay J. Acute coronary syndrome and crystal methamphetamine use: A case series. Hawaii Med J 2004;63:8-13.
147. Westover AN, Nakonezny PA, Haley RW. Acute myocardial infarction in young adults who abuse amphetamines. Drug Alcohol Depend 2008;96:49-56.
148. Swalwell CI, Davis GG. Methamphetamine as a risk factor for acute aortic dissection. J Forensic Sci 1999;44: 23-26.
149. Yancy CW. Heart disease in varied populations. In: Libby P, Bonow RO, Mann DL, et al, eds. Braunwald's Heard Disease: A Textbook of Cardiovascular Medicine, 8th ed. Philadelphia, PA: Elsevier; 2008: 23-33.
150. Maynard C, Fisher LD, Passamani ER, et al. Blacks in the Coronary Artery Surgery Study: Risk factors and coronary artery disease. Circulation 1986;74:64-71.
151. Clark LT, Adams-Campbell LL, Maw M, et al. Clinical features of patients with acute myocardial infarction presenting with and without typical chest pain: An inner city experience. J Assoc Acad Minor Phys 1989;1:29-31.
152. Borzak S, Havstad S, Tilley B, et al. Lower thrombolytic use for African Americans with myocardial infarction: An influence of clinical presentation? Am Heart J 1999;137:338-345.
153. Chen J, Rathore SS, Radford MJ, et al. Racial differences in the use of cardiac catheterization after acute myocardial infarction. N Engl J Med 2001;344: 1443-1449.
154. Zaman MJ, Junghans C, Sekhri N, et al. Presentation of stable angina pectoris among women and South Asian people. CMAJ 2008;179:659-667.
155. Harding S, Rosato M, Teyhan A. Trends for coronary heart disease and stroke mortality among migrants in England and Wales, 1979-2003: Slow declines notable for some groups. Heart 2008;94:463-470.
We have all heard a story that starts with, "Remember that patient you saw and discharged yesterday? Well, ...." This next series of EM Reports tackles one of the most common subjects of such a story: missed heart attacks.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
