Special Feature: Primer for Chemical Warfare: Nerve Agents
Primer for Chemical Warfare: Nerve Agents
By Jacob W. Ufberg, MD
|
Following the disaster of September 11th and recent cases of anthrax, the possibility of attack by weapons of mass destruction seems more likely than at any time in our nation’s history. One particularly devastating family of chemical warfare agents, the nerve agents, is closely related to many commonly used organophosphate insecticides. It is imperative that emergency medicine specialists become familiar with these agents and the treatment of victims exposed to them. This review will provide a brief history of the nerve agents, their mechanism of action, their clinical effects, and their treatment.
Background
The nerve agents are organophosphorus compounds first synthesized as weapons of war in 1936 by a German scientist who was searching for a more potent agricultural insecticide. This agent became known as tabun, or GA. In the following years, two additional agents known as sarin (GB) and soman (GD), were synthesized.1 The code names stand for German (G) and followed in sequence (skipping GC, which was the medical code for gonorrhea). In 1954, the British synthesized the fourth nerve agent of military significance, VX, again while searching for a superior pesticide.2 In 1968, VX leaked from a tank and drifted outside a military base in Utah, killing approximately 6000 sheep. One year later, President Richard Nixon issued an executive order halting production and further development of chemical weapons.
The German military never deployed nerve agents during World War II. The first documented use of mili-tary nerve agents did not occur until Iraq used sarin against the Kurds and during its war against Iran. In 1994 and 1995, terrorists used sarin gas in attacks in Japan, resulting in large numbers of patients flocking to nearby emergency departments.
Though described as nerve "gases," these four agents are actually colorless liquids at normal ambient temperatures.2 They become aerosolized when sprayed or coupled with an explosive device.3 They are similar in structure to typical organophosphate insecticides such as malathion. The G agents are moderately volatile (the less volatile an agent is, the more persistent it is on material and terrain), posing a significant inhalational danger but persisting only a limited time before evaporation and dispersion. VX is much less volatile, and can remain where delivered for weeks.4 However, it may be an inhalational threat at ambient temperatures greater than 100°F. Nerve agent vapors are denser than air, and thus, tend to accumulate in low areas or underground shelters.5
Nerve agents are soluble in fat and water, allowing absorption through the eyes, skin, and respiratory tract. Vapor tends to affect the eyes first, followed by the respiratory tract, and then progressing to generalized effects if the exposure is great enough. The effect of vapor exposure is a function of the toxicity of the agent, the concentration of the agent in the air, and the time of exposure. Dermal exposure to a liquid agent produces localized effects which then progress to generalized effects if the exposure is large enough.
Mechanism of Injury
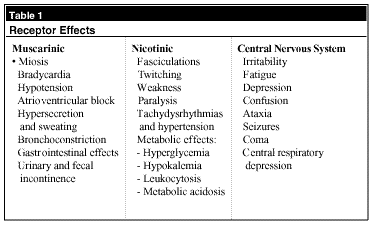
Acetylcholine (ACh) is a neurotransmitter found throughout the central nervous system (CNS), the autonomic nervous system, and at the skeletal muscle motor end plate. (See Table 1.) ACh binds and activates nicotinic and muscarinic receptors throughout the body. Nicotinic receptors are sodium channels at post-synaptic membranes in autonomic ganglia and at skeletal muscle motor end plates. Muscarinic receptors stimulate or inhibit cellular function through G proteins at cardiac muscle, secretory glands, and skeletal smooth muscle.6 The enzyme acetylcholinesterase (AChE) regulates ACh activity within the synapse by binding and hydrolyzing ACh. After hydrolysis, AChE dissociates itself and is free to act on another molecule of ACh.
 |
The toxicity of nerve agents is due to their ability to inactivate AChE, leading to an accumulation of toxic levels of ACh. This accumulation initially stimulates, then paralyzes, cholinergic nerve transmission. Nerve agents inhibit AChE by binding to a serine residue at the active site of the molecule, forming a phosphorylated protein that is incapable of breaking down ACh.2 Over time, AChE cleaves a portion of the bound nerve agent, producing a stable, irreversible bond between the nerve agent and AChE.6 This is called "aging," and the time required for this to occur varies from minutes (soman) to 48 hours (VX).3 For this process to be overcome, new enzyme must be produced.
There is evidence that nerve agents also affect CNS mechanisms that are non-cholinergic. Antagonistic effects on ¡-aminobutyric acid (GABA) may help explain seizure activity after poisoning. Additionally, the adenylcyclase and phosphodiesterase systems may be affected. Anti-dopaminergic activity also has been postulated to explain parkinsonian symptoms in insecticide toxicity cases, although none of these symptoms have been described in victims of nerve agent poisoning.2
Clinical Presentation
The nerve agents interfere with cholinergic function at muscarinic, nicotinic, and CNS receptors. This leads to dysfunction of multiple organ systems. (See Table 2.) The major determinants of clinical effects are the amount of nerve agent and the route of exposure. The two most important routes of exposure are dermal and inhalational, and progression of symptoms may range from gradual onset to death within minutes.
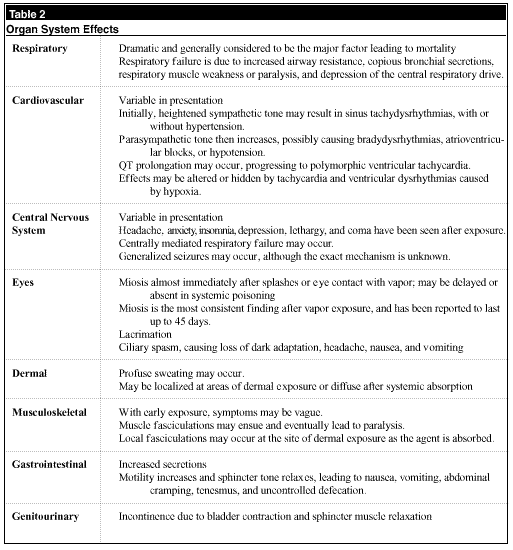 |
After exposure to nerve gas vapors, the patient will initially develop miosis and respiratory symptoms. With ongoing exposure, GI symptoms may develop and respiration becomes more difficult. The patient may develop muscular weakness leading to paralysis, and seizures may occur or be masked due to paralysis. Death ultimately results from respiratory arrest.
Dermal exposures may result in delayed onset of toxicity, with no effects for as long as 18 hours.7 This poses an increased risk of contamination for health care providers. When liquid nerve agent contacts the skin, local effects include sweating and fasciculations at the site of contamination. With progressive absorption, generalized symptoms may occur, progressing to full systemic toxicity. Of note, miosis tends to be absent in the early stages of dermal exposure.
Treatment
Initial Priorities and Decontamination. Emergency responders must take steps to ensure that they do not become casualties themselves. Rescuers must wear personal protective equipment—including a protective respirator (self-contained breathing apparatus [SCBA] device or a mask with a charcoal filter) and heavy butyl rubber gloves at minimum, and preferably a protective suit—when entering the scene of a chemical attack. To prevent further absorption of nerve agents and spread to others, decontamination should take place in a "hot zone" prior to transport, if possible. Decontamination of liquid nerve agent exposure starts with removal of all clothing. Copious irrigation with water physically will remove the nerve agent, but will not inactivate residual agent. Inactivation is accomplished by washing the skin with an alkaline solution, such as 0.5% hypochlorite solution (1:10 solution of household bleach) or an alkaline soap.8 Avoid hot water and vigorous scrubbing, which may increase absorption of the agent. No decontamination is necessary for pure vapor exposures.
Supportive Care. Severely intoxicated patients must receive the care necessary for any critically ill patient. The ABCs must be addressed. The patient should have supplemental oxygen or supportive ventilation as required. Intravenous (IV) access, cardiac monitoring, and pulse oximetry should be ensured. Suction is an important part of airway maintenance, as secretions will be copious. Rapid sequence intubation should be performed with a paralytic agent other than succinylcholine, as this agent is metabolized by plasma cholinesterase, leading to prolonged paralysis. Diazepam may be used to treat seizure activity in doses of 5-10 mg IV every 10 minutes, titrated to effect.
Antidotes. Two antidotes are used to treat nerve agent toxicity. Atropine is the initial drug used for the symptomatic patient. Atropine acts as a muscarinic antagonist and antagonizes some CNS effects of nerve agents, but does not reverse muscle weakness or paralysis. It alleviates bronchoconstriction and excessive bronchial secretions, vomiting, bradycardia, and diaphoresis, and may help stop seizures. Aggressive use of atropine will aid mechanical ventilation, which initially may be difficult secondary to bronchoconstriction and bronchorrhea. Tachycardia is not a contraindication to atropine use in this clinical circumstance.
The initial dose of atropine for moderately symptomatic patients is 2 mg (0.02 mg/kg for children, minimum 0.1 mg) IV every 2-5 minutes with a therapeutic endpoint of drying of the respiratory secretions and reversal of bronchoconstriction and bradycardia. In the severely intoxicated patient, doses may need to be increased to 5 mg IV every 2-5 minutes (0.05 mg/kg for children), with the same endpoints. The typical total dose is less than 20 mg in the first 24 hours, which is much less than the hundreds of milligrams that may be used in severe organophosphate pesticide poisonings.6
The other antidote used is pralidoxime chloride (2-PAM). This agent is indicated in all symptomatic exposures, and may be administered concurrently with atropine. 2-PAM reactivates AChE by splitting the phosphorus moiety bound to AChE, leaving the regenerated enzyme. However, in order for 2-PAM to be effective, it must be given before aging of the enzyme occurs. The effect of 2-PAM is most evident at the skeletal muscle motor end plate.
The recommended dose of 2-PAM is 1-2 g IV given over 30 minutes to minimize the side effects of hypertension, headache, and vomiting. A second dose may be administered in 60-90 minutes if improvement occurred with the first dose. If IV access is not available, 2-PAM may be given intramuscularly (IM). 2-PAM is excreted rapidly in the urine, so hydration should be assured. Lower doses may be used in patients with renal failure.
Laboratory. No laboratory study directly can measure nerve agent levels from from a blood sample. The acute effect of nerve agents roughly can be estimated by measuring the reduction in red blood cell (RBC) cholinesterase activity (this is affected as opposed to plasma cholinesterase, which preferentially is affected by insecticides). However, the activity of RBC cholinesterase is variable between individuals, making it difficult to estimate change without a known baseline value. Additionally, enzyme activity correlates poorly with severity of local effects after exposure to vapor.6 Enzyme activity level that decreases to 20-30% of normal levels tends to correlate with systemic symptoms, and activity approaches zero in severely poisoned patients. Most importantly, antidotes never should be withheld to wait for lab results.
Disposition. Patients who present to the ED after nerve agent vapor exposure have suffered peak effects by the time they arrive, and no further worsening or new symptoms should be expected. Resolution of major effects occurs within 24 hours, though weakness, miosis, and subtle mental changes may linger.2 Patients with dermal exposures may experience a delayed onset of effects. Therefore, these patients should be admitted to a monitored setting and observed for 24 hours. After this time, they may be safely discharged. Once again, it is important that enzyme activity levels not be used alone to guide treatment.
References
1. Robinson J. The Problem of Chemical and Biological Warfare: Vol I. The Rise of CB Weapons. New York:Humanities Press;1971.
2. Sidell FR,et al. Chemical warfare agents: II. Nerve agents. Ann Emerg Med 1992;21:865-871.
3. Velez-Daubon LI, et al. CBRNE—Nerve Agents, Binary: GB2, BX2. (Online at www.emedicine.com; accessed 01/08/02.)
4. Dunn MA, et al. Progress in medical defense against nerve agents. JAMA 1989;262:649-652.
5. Arnold JL. CBRNE- Nerve Agents, G-series: Tabun, Sarin, Soman. (Online at www.emedicine.com; accessed 01/08/02.)
6. Holstege CP, et al. Chemical warfare: Nerve agent poisoning. Crit Care Clin 1997;13:923-942.
7. Bowers M, et al. Some behavioral changes in man following anitcholinesterase administration. J Neurol Ment Dis 1964;138:383-389.
8. Benitez FL, et al. CBRNE—Nerve Agents, V-series: VE, VG, VM, VX. (Online at www.emedicine.com; accessed 01/08/02.)
Dr. Ufberg, Assistant Professor of Emergency Medicine, Department of Emergency Medicine, Temple University School of Medicine, Philadelphia, PA, is on the Editorial Board of Emergency Medicine Alert.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
