Right Lower Quadrant Abdominal Pain in Women of Reproductive Age: An Algorithmic Approach
Right Lower Quadrant Abdominal Pain in Women of Reproductive Age: An Algorithmic Approach
Authors: Kenneth H. Butler, DO, Associate Residency Director, University of Maryland Emergency Medicine Residency Program, University of Maryland School of Medicine, Baltimore; Darlene K. Fidler, MD, Resident, University of Maryland Emergency Medicine, University of Maryland School of Medicine, Baltimore; Katherine A. Grundmann, MD, Faculty Member, University of Maryland Emergency Medicine, and Clinical Instructor, University of Maryland School of Medicine, Baltimore.
Peer Reviewers: Gary Hals, MD, PhD, Attending Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC; Allan B. Wolfson, MD, Professor of Emergency Medicine, University of Pittsburgh; Program Director, University of Pittsburgh Affiliated Residency in Emergency Medicine.
Women of childbearing age can present with a wide variety of abdominal disorders, some of which are of only minor concern and others that are potentially life-threatening. Patients who appear stable and do not have pain—or have only vague, nonspecific, non-localizing pain—can have true surgical emergencies as their underlying pathology.
The risk of litigation remains high for missed appendicitis and ectopic pregnancies. Often, women who present to the emergency department (ED) with abdominal pain are found to be pregnant. Most emergency medicine physicians, as well as some gynecologists, have difficulty appreciating adnexal or ovarian masses on pelvic examination. More importantly, a nonspecific finding such as adnexal tenderness can be a manifestation of any pelvic or abdominal abnormality.
This issue presents an efficient algorithmic approach to the female patient with right lower quadrant (RLQ) abdominal pain. This approach is designed to enable the physician to make expeditious distinctions between surgical and nonsurgical entities. Laboratory tests that may aid in decision making can be completed prior to the physical assessment. Unstable patients can be resuscitated by the ED staff working in tandem with surgical specialists. Stable, nonsurgical patients can be evaluated thoroughly, treated, and discharged.
Gynecologic, urologic, and gastrointestinal abnormalities are discussed in this article. Use of urinalysis and beta-human chorionic gonadotropin testing is reviewed. Recent trends in ultrasound and the choice between noncontrast and contrast computed tomography (CT) are discussed. Admission criteria, appropriate treatment regimens, and discharge are described.— The Editor
Introduction
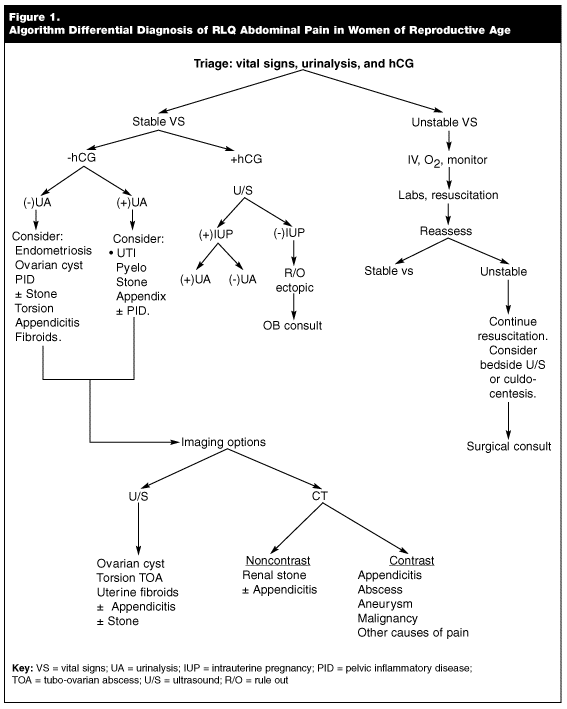
Abdominal pain accounts for 4-10% of all ED visits.1-3 It also is one of the most difficult presentations to assess; in up to 40% of patients with this complaint, no diagnosis is ever made.1 In women of childbearing age, the cause of abdominal pain in the RLQ can be especially difficult to discern. The causes range from the life-threatening to the benign. (See Table 1.) The algorithm introduced in this paper can be used to narrow the differential diagnosis. (See Figure 1.)
| Table 1. Differential Diagnosis of Right Lower Quadrant Abdominal Pain in Women of Reproductive Age | |
| • | Ectopic pregnancy |
| • | Adnexal torsion |
| • | Appendicitis |
| • | Pelvic inflammatory disease |
| • | Urinary tract infection (UTI) and pyelonephritis |
| • | Kidney stones |
| • | Ovarian cysts |
| • | Endometriosis |
| • | Uterine fibroids |
 |
History
The key to an efficient and accurate diagnosis is a detailed and thorough history. The following information should be obtained:
- Onset, location, radiation, and duration of pain;
- Aggravating or relieving factors;
- Severity of pain (constant or intermittent);
- Characteristics of the pain;
- History of the pain;
- Association with nausea, vomiting, anorexia, or diarrhea;
- Time of last bowel movement; and
- Recent use of analgesics, narcotics, or antibiotics.
In women of reproductive age, it especially is important to ask about urinary symptoms, the date of the last menstrual period (LMP), the presence of vaginal discharge, obstetric/gynecologic and sexual history (in detail), previous pelvic infections, birth control method, and use of fertility drugs. As with any patient, it is important to obtain a medical and surgical history, to ask about use of medications and about allergies, and to inquire specifically about previous abdominal surgery.
Physical Examination
A complete physical examination is warranted in every patient, but in women of childbearing age with the chief complaint of RLQ abdominal pain, certain areas should receive particularly close attention. A complete set of vital signs includes orthostatics to determine if the patient is volume depleted and assessment of fetal heart tones if the patient is known to be more than 10 weeks pregnant. After the cardiopulmonary status is evaluated and deemed stable, a thorough and complete abdominal examination is essential and should include inspection for masses, scars, or distention. Next, the abdomen should be auscultated for bowel sounds and bruits. Percussion and palpation should follow. The physician should begin by palpating away from the area that is painful and working toward the area that is most tender.
It also is important to palpate for any masses, organomegaly, rebound, or guarding. Equally important in this population is a detailed pelvic examination, which allows identification of external lesions, vaginal discharge, cervicitis, cervical motion tenderness, uterine size and tenderness, and adnexal masses or tenderness. Unfortunately, even under ideal conditions, with examinations performed under general anesthesia, the sensitivity of the bimanual examination is low for detection of adnexal masses, even by gynecologists.4 A rectal exam should be considered, and the presence of pain, masses, or blood should be documented.
Laboratory Tests
Since many women of childbearing age with abdominal pain are pregnant, it is imperative to diagnose or rule out pregnancy.1 Two mandatory tests—a urine pregnancy test and urinalysis—can narrow the differential diagnosis and help direct the remainder of the patient workup. A standard urine pregnancy test can be completed in fewer than five minutes, so the result often is available by the time the patient is examined by the emergency physician. Urine pregnancy tests are reliable and allow diagnosis of pregnancy within six days after conception.5 Qualitative urine pregnancy tests are positive when intact human chorionic gonadotropin (hCG) exceeds 25 IU/L.5 A urine sample should be sent for chemical screening (dipstick) as well as microscopic analysis.
A complete blood count (CBC) also commonly is done in women with lower abdominal pain. A CBC can be useful to check for significant blood loss, which is represented by low hemoglobin and hematocrit levels. It is important to remember, however, that if the blood loss is acute, the CBC may not yet be representative of the true hematocrit value. The white blood cell (WBC) count is neither sensitive nor specific for identifying the etiology of the abdominal pain, and the result rarely affects clinical decisions.1,6,7 Specific testing is discussed in detail under each topic listed.
Analgesia
Treatment should be individualized according to the presumed cause of the pain. In every case, however, early and adequate analgesia should be given.2 In the past, pain medications often were withheld in the mistaken belief that they would interfere with the physical examination and mask the diagnosis. Early administration of opiates is both safe and effective for patients with acute abdominal pain and actually facilitates the diagnosis.8 It allows the patient to relax, decreases voluntary guarding, and helps the examiner localize the area of maximum tenderness.8 Another advantage to using opiates for pain control is that they can be titrated to effect and easily reversed with naloxone hydrochloride (Narcan) if necessary.
Differential Diagnosis
Ectopic Pregnancy. The implantation of a fetus anywhere outside the uterine endometrium is considered an ectopic pregnancy.9 In 85-90% of cases, the implant is in the distal ampullary region of the fallopian tube.5,10 Sites of possible implantation also include other parts of the fallopian tubes, the uterine cervix, the ovary, and the peritoneal cavity.
The incidence of ectopic pregnancy has increased steadily over the past 30 years, from 4.5 per 1000 pregnancies in the 1970s to 19.7 per 1000 in the early 1990s, which is equivalent to more than 100,000 cases per year in the United States.11-14 After a woman has had one ectopic pregnancy, her risk of having another is 7-15%. Early diagnosis is essential, and prompt treatment can greatly decrease a woman’s morbidity and mortality risk as well as increase her chances of future successful pregnancies.15
The multiple risk factors for ectopic pregnancies are listed in Table 2.2,9,10,12,14-16
| Table 2. Risk Factors for Ectopic Pregnancy | |
| Strongly Associated | |
| • | Previous ectopic pregnancy |
| • | Diethylstilbestrol (DES) exposure |
| • | Intrauterine device |
| • | Prior tubal surgery |
| • | Documented peritubal adhesions |
| Moderately Associated | |
| • | Pelvic inflammatory disease |
| • | Multiple induced abortions |
| • | Smoking |
| Slightly Associated | |
| • | Pelvic or abdominal surgery |
| • | Vaginal douching |
| • | First sexual intercourse before age 18 |
| Adapted from: Ankum WM, et al. Risk factors for ectopic pregnancy: A meta-analysis. Fertil Steril 1996;65:1093-1099. (Web: www.jr2.ox.ac.uk/bandolier/booth/hliving/Ectopreg.html. Accessed 12/12/2001.) |
|
Clinical Presentation. Ectopic pregnancy can present in a variety of ways, from the asymptomatic to the surgical abdomen.17 The ectopic pregnancy can be an incidental finding, or the patient may experience vaginal bleeding, irregular menses, amenorrhea, and/or abdominal or pelvic pain.2,9,10,15,16 The pain may be constant or crampy.9 The woman’s only complaint may be syncope; therefore, ectopic pregnancy should be considered in the differential diagnosis when a woman of reproductive age presents with this complaint.2
When abnormal physical findings are present, common findings on the physical examination are orthostasis, lower abdominal and adnexal tenderness (90%), an adnexal mass (60%), and cervical motion tenderness.10,13 Unfortunately, up to 4% of women with hemoperitoneum due to a ruptured ectopic pregnancy have no pain, although many will have other symptoms that will bring them to the ED.13 If rupture has occurred, the patient may have abdominal distention, guarding, and rebound tenderness;10 hemorrhagic shock may ensue.
Diagnosis. Regardless of history and physical findings, ectopic pregnancy needs to be considered in every woman in the ED who presents in her first 20 weeks of pregnancy. Transvaginal ultrasonography and quantitative measurement of the serum hCG level have become the standard of care in the workup of ectopic pregnancy. A woman with a positive urine hCG test should not be diagnosed merely as having early pregnancy and be discharged without further testing. Abnormal pregnancies usually are associated with abnormally low hCG levels.18 Normally, the serum level of beta-hCG should double every 2.3 days, although this number is not absolute.16 Up to 10% of normal pregnancies will have abnormal doubling times, and up to 15% of ectopic pregnancies will have normal times.19 Generally, however, the absence of this doubling time on repeat beta-hCG measurements or a leveling off of the serum concentration is highly suggestive of either an ectopic pregnancy or an abnormal intrauterine pregnancy (IUP), which will abort spontaneously in 85-95% of cases.9,16,18
The diagnosis of ectopic pregnancy has been improved greatly by high-resolution transvaginal ultrasound, which can detect a gestational sac 5-6 weeks after the patient’s last menstrual period.16,20 The endovaginal probe allows for a more detailed evaluation of the endometrium, endometrial canal, and adnexa.21 Different types of sonographic techniques and equipment have different hCG threshold levels or discriminatory zones above which gestational sacs are large enough to be seen on ultrasound. The threshold for a trans-abdominal ultrasound is greater than 1800 mIU/mL, and 500-1000 mIU/mL for endovaginal ultrasound.21 Sonographic indications of ectopic pregnancy include an empty uterus, a solid or cystic mass with free fluid in the cul-de-sac, the appearance of a gestational sac in the tube, or a pseudogestational sac in the uterus.18,21 Findings that do not support ectopic pregnancy include a live IUP or the intradecidual sign and double decidual sign of an early IUP. These two signs can be used to identify an IUP before visualization of the yolk sac.21 The double decidual sign needs to be distinguished from the pseudogestational sac of an ectopic pregnancy. The pseudogestational sac is an intrauterine fluid collection surrounded by a single decidual layer.21 A normal gestational sac is covered by the decidual capsularis and embedded within the decidual parietalis, giving the "double decidual sign."14 Endovaginal ultrasound provides better differentiation of the pseudogestational sac vs. the double decidual sign of early pregnancy. Misinterpretation of these early ultrasound findings is one of the most common causes of the misdiagnosis of ectopic pregnancy.21 (See Table 3.)
| Table 3. Risk Factors Associated with Delayed or Missed Diagnoses of Ectopic Pregnancy | |
| • | Absence of abdominal pain; atypical pain |
| • | Absence of adnexal mass on physical examination |
| • | Negative aspiration on culdocentesis |
| • | Nondiagnostic ultrasound |
| • | Misinterpretation of sonographic image |
| • | Falling hCG level |
It previously has been assumed that the detection of a viable IUP by ultrasound essentially excludes an ectopic pregnancy. This was based on the reported incidence of a combined ectopic and uterine (heterotopic) pregnancy at 1:30,000.18,22 This value, however, was calculated in the 1940s. The incidence of both ectopic pregnancy and heterotopic pregnancies has increased greatly in past decades as a consequence of the increased use of ovulation-inducing agents and the increased incidence of pelvic inflammatory disease (PID).12,18 Recent estimates of heterotopic pregnancies range from 1:4000-7000 in the general population to 1:100, or 1%, in the assisted-reproduction population.12,18,21-23
Although not readily available in every ED, determination of the patient’s progesterone level also can aid in the diagnosis or exclusion of ectopic pregnancy. The serum progesterone levels reflect the production of progesterone by the corpus luteum, which is stimulated by a viable pregnancy.14 Low serum progesterone levels (< 5 ng/mL) have been associated with ectopic pregnancy or impending abortion and require immediate evaluation by a gynecologic consultant.14,15,18 A level of 25 ng/dL or higher defines normal pregnancy and obviates further testing.1,9,14,24 With levels between 5 ng/dL and 25 ng/dL, further evaluation by ultrasound is required.
If the patient is unstable and an ectopic pregnancy is suspected, and if bedside ultrasonography is not available, culdocentesis can be done to check for intraperitoneal hemorrhage. In this procedure, the rectovaginal cul-de-sac is aspirated with a needle inserted through the posterior fornix of the vagina.13 If the aspiration is dry, it has no diagnostic value. If clear, non-bloody fluid is aspirated, it is considered a negative test.13,16 If non-clotting blood is aspirated, the tap is positive. A culdocentesis can be falsely negative if the bleeding is rapid and the intraperitoneal blood clots.17 False positives can occur due to technical errors, such as hitting a vascular structure with the needle, or the bleeding could be to the result of the rupture of a leuteum cyst.17
Treatment. If ectopic pregnancy is diagnosed, an intravenous line should be established; a blood sample should be sent for CBC, type, and screen; and an obstetric/gynecologic consult should be obtained immediately.2 Unstable patients need fluid resuscitation and may require emergent transfusion before surgical intervention by the consultant. If the patient is stable, the focus of treatment should be on preservation of fertility.16
Nonsurgical treatment is administration of methotrexate, a chemotherapeutic agent that is 95% successful in the treatment of ectopic pregnancy.18 It is a folic acid antagonist that inhibits the spontaneous synthesis of purines and pyrimidines, thus interfering with DNA synthesis and cell multiplication.24 The decision to use methotrexate must be made in consultation with a gynecologist. The drug can be given to women who are hemodynamically stable, if the ectopic pregnancy has not ruptured and the fetus is smaller than 4 cm when measured by ultrasound, no fetal cardiac activity is detected, and the serum hCG level is lower than 3500.9,18 The 5% failure rate has been associated with advanced gestation and hCG concentration of greater than 5000.11 Methotrexate is given in four doses of 1 mg/kg IM or IV on four alternating days (days 1, 3, 5, and 7), with 0.1 mg/kg of leucovorin given on intervening days.18 The most common side effect of methotrexate therapy is increased abdominal pain,25 presumably caused by either tubal abortion or stretching of the tube by hematomas. The pain usually is self-limited and should be treated by administration of nonsteroidal anti-inflammatory drugs (NSAIDs) every six hours.25 If sufficient doses of NSAIDs do not relieve the pain, the patient should be reevaluated. High-dose methotrexate also can cause bone marrow suppression, hepatotoxicity, pulmonary fibrosis, alopecia, and photosensitivity.25 These side effects are extremely rare in the doses required for treatment of ectopic pregnancy, and the risk is further lessened by the use of leucovorin.25 Patients who receive methotrexate should be monitored with weekly beta-hCG measurements until the level is undetectable.26 If any of the criteria for methotrexate therapy listed above are not met, or if the patient is unstable, surgery is required.
Adnexal Torsion. Adnexal torsion occurs when an ovary or a fallopian tube twists on its pedicle, cutting off blood supply. It is a true gynecologic emergency, requiring immediate surgical intervention. Conservation of the tube and ovary and preservation of fertility are the goals. With early diagnosis and treatment, it may be possible to untwist the pedicle and save the organ.27
Ovarian torsion can occur at any age, but is seen primarily in females of reproductive age. The average age is 26; 70-75% of cases occur in women younger than 30 years of age.28-30 Twenty percent of cases occur in pregnant women. It generally is unilateral, occurring more frequently on the right side than on the left.30-33
The onset of pain usually is sudden, corresponding to occlusion of the vascular supply to the torsed organ. Initially, the venous and lymphatic circulation is compromised, which leads to edema, hemorrhage, and enlargement of the adnexa. If this persists, the arterial circulation becomes occluded. Once the arterial supply is cut off, ischemic infarction and necrosis ensue.28,33,34 If left untreated, it can progress to infection, peritonitis, sepsis, and ultimately, death.29,33,35 The ovary will remain viable for only six hours from the moment torsion occurs. Time is fertility.29
Torsion can occur in normal adnexa, but more than 95% of cases are associated with adnexa enlarged by pathologic processes such as tumors and cysts.28,33,34 The most common growth associated with torsion is a benign cystic teratoma, present in 3.5-16% of cases.27,31,35 Risk factors include increased weight of the ovary, ovarian cysts larger than 4 cm, congenitally malformed tubes, excessive tortuosity of veins, and ovarian tumors.29
Clinical Presentation. The woman usually presents complaining of acute onset of lower abdominal pain that radiates to the back, pelvis, and thigh.29 Pain often is preceded by strenuous activity such as exercise, intercourse, or straining.29 The pain is constant and unilateral, localizes immediately, and often is associated with nausea and vomiting.28-30 About 10% of patients report a history of similar episodes in the past, suggesting previous torsion and detorsion.28-30,33
The patient may have tachycardia due to pain or, if presentation is delayed, fever and dehydration.29 The abdominal examination will show unilateral tenderness and decreased bowel sounds. Common findings on the pelvic examination are adnexal tenderness,2,28,29 a unilateral tender adnexal mass (in 50-80% of cases), and cervical motion tenderness (the same signs and symptoms of ectopic pregnancy). Unfortunately, these physical findings are nonspecific and difficult to differentiate from cases of ectopic pregnancy, tubo-ovarian abscess (TOA), or appendicitis, and necessitate further testing and imaging.
Diagnosis. The most sensitive imaging technique to date for diagnosis is color flow Doppler ultrasound.1,28 On ultrasound, an ovarian mass often can be visualized and may be cystic, solid, or mixed; the color Doppler determines whether there is circulatory flow to the adnexa. (See Figure 2.) Large ovarian cysts have poor vascularity at baseline, making color flow Doppler less than 100% sensitive. The hCG level always should be measured, not only to rule out an ectopic pregnancy as the cause of the pain, but also because 20% of cases of torsion occur in pregnant women.
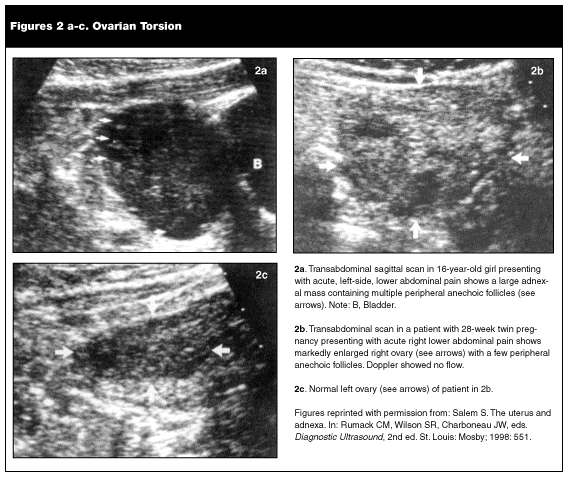 |
Treatment. ED treatment should include stabilization, pain control, antiemetics, and immediate gynecologic consult.2,29 Prompt laparoscopy is essential, as the tissue only remains viable for up to six hours. The procedure also allows interventions that help decrease the incidence and severity of complications.28,33 (See Table 4.) If a teratoma is present, the treatment usually is ovarian cystectomy, with preservation of ovarian tissue, if possible.36
|
Table 4. Complications of Adnexal Torsion |
|
| • | Infertility |
| • | Infection |
| • | Peritonitis |
| • | Sepsis |
| • | Adhesions |
| • | Chronic pain |
Appendicitis. Appendicitis is the most common non-traumatic abdominal surgical emergency.37 The lifetime risk of appendicitis is 6-7%. More than 200,000 cases of appendicitis occur each year in the United States.37-41 The incidence is 1.1 in 1000 per year and peaks in early adulthood.38-41 Mortality due to complications is 0.2-0.8%, with the highest numbers in patients with comorbid conditions.39 It remains an extremely difficult entity to diagnose (especially in women of childbearing age), and failure to do so remains a leading cause of litigation against emergency physicians.7 In the general population, a normal appendix is removed in 15-20% of appendectomies performed for suspected appendicitis, and this number increases to 35-45% in women of reproductive age, mostly because of the prevalence of PID and other gynecologic disorders in this population.7,35,37,38,42,43 On the other hand, patients who have appendicitis are misdiagnosed in about 33% of cases; these patients mistakenly are believed to have gastroenteritis, PID, or a urinary tract infection (UTI).7,37,39 Women with appendicitis often present more atypically than men, with diffuse lower abdominal pain, and they have a higher incidence of abnormal pelvic examination findings.37 Appendicitis is especially difficult to diagnose in pregnant women, because the location of the appendix changes as the size of the uterus increases. Because of these difficulties, female patients have an increased incidence of perforation and other complications.37
The appendix is a 10-cm, blind-ended pouch that arises from the posterior medial wall of the cecum. Its position in the abdominal cavity is variable and greatly influences clinical signs.7,38 The appendix contains a large number of lymphoid follicles, and it secretes about 5-6 mL of mucus per day, which empties into the cecum. Acute appendicitis begins with obstruction of the appendiceal lumen.7,38,39,41,44 The cause of lumenal obstruction is most commonly fecaliths, lymphoid follicle hyperplasia, foreign bodies, tumors, or calculi.38,39 Obstruction leads to an increase in intraluminal pressure and eventual obstruction of venous and lymphatic drainage. The epithelial mucosa begins to break down and bacterial invasion occurs. This progresses until arterial flow is compromised, and if left untreated, gangrene and perforation with peritonitis and abscess formation will occur.7,38,39,41 The initial appendiceal distention is received by visceral afferent pain fibers, which are vague and poorly localized in the periumbilical (T10) region. As the inflammation progresses, somatic pain fibers are activated and usually localize the pain to the RLQ, although the site can vary with the location of the appendix.41
Clinical Presentation. The presentation of appendicitis is notoriously inconsistent, adding to the difficulty of diagnosis. Only about 50% of patients present with the "classic" history of anorexia, periumbilical abdominal pain followed by nausea and vomiting, and then migration of pain to the RLQ.39 Vomiting, when present, almost always follows the onset of pain,45 and this is an important sequence to ascertain by asking the patient.7,38,39 Twenty-three percent of patients report a previous history of similar pain.7,39 If the appendix lies near the bladder or ureter, the patient may report urinary symptoms.39,41 Fever and chills are present in 10-20% of patients, and 5-10% have either diarrhea or constipation.40
Most patients with appendicitis have RLQ tenderness, most commonly at McBurney’s point, which lies 5 cm from the anterior superior iliac spine on an imaginary line drawn from the umbilicus.1,38,40 Other common findings include rebound tenderness, rigidity, guarding, rectal tenderness, and fever. Several other physical signs for which every intern is taught to check are rare. Rovsing’s sign—pain elicited in the RLQ by palpation of the left lower quadrant—is found in 5% of cases.38,40 The obturator sign—pain when passively flexing the right hip and knee and internally rotating the leg at the hip—is seen in 5-8% of cases.38,40 The psoas sign is an increase in abdominal pain when the patient, while lying supine, lifts his or her thigh as pressure is applied at the knee; it is found in 3-5% of cases.38,40 On pelvic examination, most women with appendicitis have nonspecific findings of cervical motion tenderness and right adnexal tenderness (as is evident in patients with other causes of RLQ pain).2
Diagnosis. In males, a diagnosis of appendicitis usually can be based on the history and physical examination alone. In females, further imaging studies are required to rule out a gynecologic/urologic cause prior to consulting a general surgeon. (See Figure 1.)
Appendicitis remains the most common extrauterine surgical emergency encountered in pregnancy.7 It may be difficult to diagnose, not only because the appendix may be displaced by the gravid uterus, but also because nausea and vomiting may be mistakenly attributed to pregnancy.41 The imaging studies of choice in the pregnant woman should start with ultrasound.
If the appendix lies in a retrocecal position near the bladder or ureter, both red and white blood cells may appear in the urine. Bacteriuria is seen in 15% of patients.7,39,46 Urinalysis is abnormal in 20-40% of patients with appendicitis, and this number rises to 50% when the appendix has ruptured.1,41,47
The surgeon will ask for the results of the CBC, but the WBC count is unreliable and will add little to diagnostic decision making.7,41 In 85-90% of cases, the patient has a WBC count greater than 10,000 cells/mm3; in other words, 10-15% of patients will have a normal WBC count.1,7,40 However, an increase in the percentage of neutrophils or bands ("left shift") with a normal WBC count is more supportive of the clinical diagnosis of appendicitis.48
A C-reactive protein (CRP) is an acute-phase reactant synthesized by the liver in response to infection.39 An absolute elevation in the CRP, or an increase of 1 mg/dL when the CRP is rechecked in a few hours, suggests appendicitis.1,7 When viewed together, an elevated CRP level, a WBC count greater than 10, and bandemia markedly increase the sensitivity of diagnosis.7,40
A plain frontal supine radiograph of the abdomen (kidney/ureter/bladder [KUB] film) is neither sensitive nor specific and is rarely helpful, as only a small percentage will show a calcified appendicolith.1,38-40 This film may be useful to rule out the presence of obstruction or free air.41
Abdominal ultrasound has a sensitivity in adults of 80-90% and a specificity of 90-100% for appendicitis.37,39-41 Diagnostic findings of appendicitis on ultrasound include visualization of a noncompressible appendix that is greater than 6-7 mm in diameter with a dilated lumen, periappendiceal fluid, and lack of peristalsis.40,41 Advantages of ultrasound are that it is noninvasive, uses no radiation, and can suggest an alternate diagnosis if negative for appendicitis (i.e., gallstones, ectopic pregnancy). It remains the diagnostic test of choice in pregnant women because of the lack of radiation. The primary disadvantage of ultrasound is that the accuracy of the test depends on visualization of the appendix. The appendix often is not seen secondary to bowel gas and the patient’s body habitus. Other disadvantages include lack of availability in some institutions, the quality of the examination is operator-dependent, and it can be a very painful examination for the patient.7,37,39
Helical CT scan has become the test of choice in many institutions. When triple contrast is used, sensitivities are 97-100%, with specificity of 95%.39-41,49 Comparable sensitivity and specificity have been achieved with unenhanced CT, a modality that decreases the time needed for diagnostic studies and allows surgical treatment to begin more quickly.50,51 The CT scan is read as positive if a thickened appendix (> 6 mm) is visualized, there is secondary peri-appendiceal inflammatory changes, or an abscess or fluid collection is seen.7,41,49 (See Figures 3 and 4.) CT scanning also can facilitate the identification of alternative surgical diagnoses (e.g., abdominal aneurysms).52
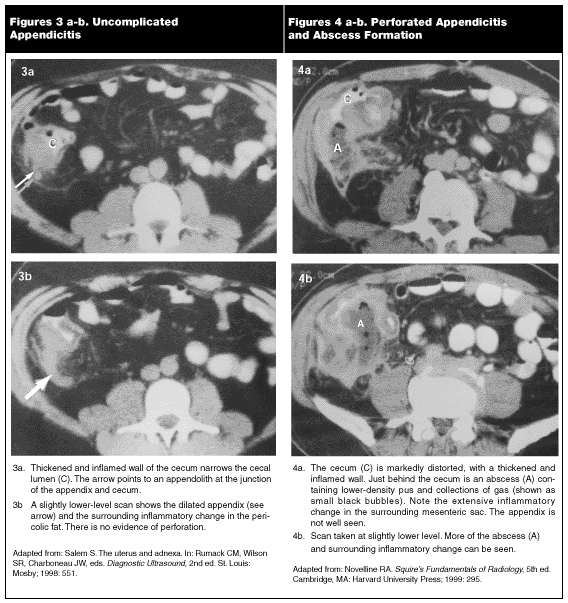 |
Treatment. Intravenous antibiotics that cover gram-negative aerobes and anaerobes are indicated in patients with acute appendicitis and should be given peri-operatively in consult with the surgeon.53,54 Early pain medication should be given. If suspicion of appendicitis is high based on the history and physical examination alone, then an immediate surgical consult should be obtained. If suspicion is low or moderate, the workup should include a CT scan or an ultrasound, serial abdominal examinations, and a surgery consult.40 Patients whose pain resolves in the ED, and who have a negative workup for other causes, may be discharged, to return for a recheck in 8-12 hours. Strict and explicit precautions need to be given to every patient discharged without a clear diagnosis.
Pelvic Inflammatory Disease. PID is an infection of the uterus, fallopian tubes, and adjacent pelvic structures.53,55,56 It is a diagnosis that should be considered in every female of reproductive age who presents with lower abdominal or pelvic pain.
PID is the single most frequent serious infection of women in the United States. More than 1 million cases occur each year, affecting 10-20 per 1000 women of reproductive age,53,55-57 and requiring more than 250,000 hospitalizations. The average annual cost of treatment exceeds $10 billion.28
PID results from an ascending spread of organisms from the cervix and vagina.53,57 Gonorrhea and/or chlamydia cause more than 50% of cases.5,53,56 Among the many pathogens, Chlamydia trachomatis is most invasive and can cause severe structural damage to the fallopian tube; unfortunately, many women with this type of infection may be asymptomatic. Other bacteria that can cause PID include Bacteroides, Peptostreptococcus, Gardnerella vaginalis, Escherichia coli, Haemophilus influenzae, Mycoplasma, and Ureaplasma. Mixed infections account for about 70% of cases.28,53,55,57,58 If the patient uses an intrauterine device (IUD), the infection might be caused by Actinomyces israelii.10,55
Risk factors for PID include multiple sex partners, a history of sexually transmitted diseases (STDs), frequent douching, smoking, and young age (> 75% of cases are in women < 25 years old).28,53,56,57 IUDs are associated with a two- to four-fold increased risk of PID. Menses, sex, or strenuous activity may exacerbate acute PID, possibly because of the breakdown of the antibacterial barrier of the cervical mucus, which allows bacteria from the cervix and vagina to ascend to the upper genital tract.10 Although pregnancy is protective against acquiring a new infection, PID can be present before pregnancy occurs and increases the risk of fetal loss.57
Clinical Presentation. A detailed sexual and gynecologic history should be obtained; most patients report a history of STDs.28,59 Patients usually complain of lower abdominal or pelvic pain, abnormal vaginal discharge, abnormal vaginal bleeding, dysuria, and dyspareunia. PID often is associated with nausea, vomiting, and fever. Symptoms usually begin during the first half of the menstrual cycle.2,10,34,55-57,59
A classic triad of PID is present in up to 90% of cases: cervical motion tenderness, lower abdominal tenderness, and adnexal tenderness.56 On speculum examination, a purulent discharge from the cervical os is seen in approximately 95% of women with PID.1,2 Vital signs may indicate the presence of fever, tachycardia, and orthostasis.
Diagnosis. For every patient assessed for PID, urinalysis, urine pregnancy test, wet prep, and cervical cultures should be done.10,55,57 The gold standard for diagnosis is laparoscopy, although this is not practical, and the diagnosis usually is made clinically.56 The emergency physician needs to maintain a high index of suspicion and a low threshold for treatment.53 The diagnosis is made if the patient meets all three major criteria and at least one minor criterion.28,53,55,57 (See Table 5.) If an adnexal mass is palpated and a TOA is suspected, pelvic ultrasonography should be performed.1,28,55 Laparoscopic findings diagnostic of PID include pronounced hyperemia of the fallopian tubes, edema of the tubal walls, and a sticky exudate on the tubal surface.60
| Table 5. Diagnostic Criteria for Pelvic Inflammatory Disease | |
| Major Criteria | |
| • | Cervical motion tenderness |
| • | Lower abdominal tenderness |
| • | Adnexal tenderness |
| Minor Criteria | |
| • | Temperature > 38°C |
| • | White blood cell count > 10.5 |
| • | Erythrocyte sedimentation rate > 15 |
| • | Gram stain of vaginal discharge positive for gram-negative diplococci |
| • | Cervical culture positive for gonorrhea or Chlamydia |
| • | Purulence on culdocentesis |
| • | Adnexal mass consistent with tubo-ovarian abscess |
Treatment. Antibiotic treatment should be aimed at coverage of the most common etiologies.56,61 (See Table 6.) Intravenous rehydration should be initiated if needed, and pain should be controlled; an IUD, if present, should be removed.55,57 The patient should be admitted for IV antibiotic treatment if any of the following criteria are met: 1) a surgical emergency causing the pain cannot be ruled out; 2) the patient is pregnant; 3) treatment failure with previously prescribed oral antibiotics; 4) the patient cannot tolerate the outpatient regimen; 5) the patient is severely ill with clinical signs of bacteremia; 6) a TOA is present; 7) the patient is immunodeficient; 8) the patient is nulliparous; or 9) there is poor likelihood of outpatient follow-up.28,38,53,55,61 If the patient is discharged, complete discharge instructions should be given, with the following recommendations: close follow-up in the next 48-72 hours, abstention from sexual intercourse for two weeks, and treatment of sexual partners.2,28,53,55,56 Patients should be encouraged to be tested for syphilis and the human immunodeficiency virus (HIV).
|
Table 6. Emergency Department Management of Cervicitis and Outpatient Treatment of Pelvic Inflammatory Disease (PID)33,60 |
||
| A. Cervicitis | ||
| Gonorrhea treatment: Ceftriaxone 125 mg IM or Cefixime 400 mg PO or Ciprofloxacin 500 mg PO or Ofloxacin 400 mg PO |
Chlamydia treatment: Azithromycin 1 gm PO single dose or Doxycycline 100 mg PO x 7 days |
|
| If pregnant or penicillin allergic: Azithromycin 2 gm po single dose or Erythromycin base 250 mg PO x 14 days or Erythromycin ethylsuccinate 800 mg PO x 7 days |
||
| B. Outpatient PID: | ||
| Choice for gonorrhea treatment from above list and Doxycycline 100 mg PO bid for 14 days or Ofloxacin 400 mg PO bid x 14 days plus clindamycin 450 mg PO bid x 14 days |
||
Complications. PID causes severe scarring of and damage to the fallopian tubes. This results in a six-fold increase in the risk of ectopic pregnancy and is the leading cause of infertility in women. The incidence of infertility after one episode of PID is 5%, 20% after two episodes, and 40% after three or more.53,56 Other complications include chronic pain, pelvic adhesions, and TOAs.10,28,53,57
Potentially fatal complications are the entrapment of purulent material in an occluded tube or pus leaking from a tube, which causes an abscess in the ovary or peritoneal cavity.28,53 Patients with a TOA are extremely ill, with severe pain, a high fever, nausea, vomiting, tachycardia, and possibly sepsis. They may be febrile, with high temperature, rigid abdomen, and palpable pelvic masses.10 Ultrasound is the most useful study for confirming the diagnosis of TOA.53 These patients need to be admitted with bed rest, IV fluids, IV antibiotics, and pain control.10,28 If the TOA has ruptured, the patient will have acute abdominal pain and require immediate surgery.62 If an unruptured TOA does not respond to conservative therapy with IV antibiotics and bed rest, then laparotomy is warranted for surgical drainage.
Urinary Tract Infection and Pyelonephritis. In patients with UTI, pathogenic microorganisms are detected in urine, but symptoms may not be present.63,64 The growth of more than 100,000 organisms/mL from a clean-catch urine specimen equals infection.64 Pyelonephritis is an infection of the renal parenchyma and pelvis, with inflammation resulting from local bacterial infection.
There are 6-7 million episodes of acute cystitis and 250,000 episodes of pyelonephritis each year in the United States. The cost of evaluation and treatment exceeds $1 billion annually.65 The vast majority of these cases are found in young, sexually active females. In fact, 25-35% of women ages 20-40 have had at least one UTI episode.65
Bacteria gain access to the bladder primarily via the urethra, causing acute cystitis, which can then ascend to the kidney, causing pyelonephritis.63 E. coli causes 70-95% of all UTIs. Other etiologic agents include Staphylococcus saprophyticus (5-20%), Proteus, Klebsiella, Enterococcus, and Pseudomonas.63-65 The etiology of pyelonephritis is similar to that of a UTI, with 90% of cases being caused by E. coli.
Females are very prone to UTIs because the urethra is short (~ 4 cm), close to the anus, and terminates beneath the labia. Sexual intercourse increases risk because urethral massage during intercourse introduces bacteria into the bladder. Voiding after intercourse lessens this risk. Any form of urinary obstruction, bladder dysfunction, or vesicoureteral reflux increases the risk of infection.63
The urinary tract is the most common site of infection during pregnancy, because of increased urinary stasis. Mild hydronephrosis is common after mid-pregnancy: right-sided in 75% of women and left-sided in 33%. Asymptomatic bacteriuria occurs in 2-10% of pregnant women. Acute cystitis occurs during 1% of pregnancies and acute pyelonephritis during 2%. The presentation of both cystitis and pyelonephritis is similar in pregnant and nonpregnant women.66
Clinical Presentation. Patients with UTI usually present complaining of dysuria, urgency, frequency, malodorous urine, and suprapubic abdominal pain.2,65 About 40% of patients will have hematuria, and many will report fevers. Patients with pyelonephritis often have flank pain, nausea, vomiting, temperature higher than 103°F, and back pain. They also may complain of rigors, headaches, general malaise, and myalgias. Many will have symptoms of cystitis as well, but more than 50% of cases of pyelonephritis occur in the absence of these symptoms.64,65,67
The physical examination of a patient with acute cystitis may be relatively unremarkable except for some suprapubic tenderness. A pelvic examination is indicated for any woman with abdominal pain unless the ED physician can be certain her symptoms are only related to a UTI. In addition, a pelvic examination provides the opportunity to look for herpetic ulcers as the cause of dysuria and pyruria.65 The presence of cervicitis suggests urethritis as the cause of dysuria.
Diagnosis. A urinalysis and pregnancy test should be obtained for every patient. The presence of leukocyte esterase in the urine is 75-95% sensitive and 94-98% specific for UTI.65 The urine also will show the presence of WBCs, bacteriuria, and nitrites. Hematuria is present in 40% of cases. Blood cultures will be positive in 25-50% of patients with pyelonephritis,67 but multiple studies have shown blood cultures in pyelonephritis to be unnecessary as they add cost without changing management.68-70 Imaging studies such as renal ultrasound or CT scan are indicated only for patients with complicated pyelonephritis.67
Treatment. The treatment regimen depends on whether the patient has a complicated or uncomplicated infection. A sexually active, nonpregnant woman with recent onset of symptoms, who has not had recent instrumentation or antibiotics and has no history of a genitourinary abnormality, has an uncomplicated infection. Left untreated, 50-70% of these cases will clear spontaneously. However, most doctors treat in response to the patient’s discomfort.65 Acute uncomplicated cystitis should be treated with a short course of antibiotics. This increases compliance, is cheaper, and decreases the incidence of adverse reactions compared with longer courses of antibiotics. Studies have shown that 85-95% of cases are eradicated by a single dose of trimethoprim/sulfamethoxazole (Bactrim) and 81-89% after a single dose of ciprofloxacin (Cipro). However, three days of treatment with trimethoprim/sulfamethoxazole has proven even more effective than a single dose, but there has been no proven benefit to treating longer than three days.65 Studies comparing three-day regimens of trimethoprim/sulfamethoxazole, nitrofurantoin (Macrobid), cefadroxil, and amoxicillin have demonstrated that trimethoprim/sulfamethoxazole has the highest cure rate and is the least expensive of this group of antibiotics.65 The patient should be encouraged to increase hydration to increase the removal of uropathogens. If the patient is having significant dysuria, she can be given a course of phenazopyridine (200 mg PO tid for one or two days) to alleviate her symptoms.
If the patient is pregnant and found to have a UTI or asymptomatic bacteriuria, she should be treated before it progresses to pyelonephritis.67 As mentioned above, up to 30% of pregnant women with asymptomatic bacteriuria will develop pyelonephritis, which is associated with increased risk of spontaneous miscarriage. Nitrofurantoin is the recommended antibiotic for pregnant women.
Patients with uncomplicated pyelonephritis can be treated as outpatients if they are not pregnant, if they have no complicating illnesses, if they are nontoxic, if they have good pain control, if they are tolerating oral intake, and if they have good follow-up.65,67 Fluoroquinolones are recommended for treatment and should be continued for 10-14 days.63,65 A patient with any underlying renal or neurologic condition predisposing her to infection is said to have complicated pyelonephritis. These patients should be admitted, as should any patient with the inability to tolerate oral intake, severe pain, high fevers, or questionable compliance.65,67 Any patient who is pregnant, diabetic, or immunocompromised should be admitted. These patients should be given IV antibiotics such as ceftriaxone, ciprofloxacin, or ofloxacin. Admission for IV administration of antibiotics decreases the incidence of complications such as sepsis, renal cortical scarring, and papillary necrosis.67
Kidney Stones. When a patient presents with acute, intense flank or side pain, ureteral stones are a primary consideration. Renal calculi arise due to papillary calcifications or precipitation of organized crystalline bodies of calcium salts, uric acid, cystine, or struvite (magnesium-ammonium-phosphate) in the urine.71
Kidney stones are very common in the United States, seen in 16 of 10,000 patients.72 Women have a 5-10% lifetime risk of developing stones.72-74 The age of presentation peaks in the third and fourth decades, and stones are three or four times more common in whites than blacks.71-75 Recurrence rates are very high: 10% after one year, 35% after five years, and 50% after 10 years.58,67,73,76
The precise cause of renal stones is unknown; however, theory suggests urinary supersaturation of solute followed by a crystal precipitate or a decrease in normal urinary proteins that inhibit crystal growth.77 Seventy-five percent of stones are made of calcium in conjunction with oxalate, phosphate, or both.77 Uric acid stones account for 5-10% of cases, struvite stones for 5-10%, and cystine for 1-5%.38,76 Etiologies for the formation of these different types of stones include hypercalciuria, hyperuricuria, hypocitraturia, hyperoxaluria, hypomagnesuria, cystinuria, chronic urinary tract infections, and low urine volume.58,78
Any systemic illness that leads to the above-mentioned conditions increases the risk of stone formation. For example, hypercalciuria is seen in patients with hyperparathyroidism and bowel disorders. Absorptive hypercalciuria is due to increased intestinal calcium absorption in the gut, but the precise cause is unknown. Oxalate excretion is enhanced in patients with bowel diseases such as Crohn’s disease and ulcerative colitis, as well as by small bowel bypass surgery.77 Chronic UTI by urea-splitting organisms leads to the formation of struvite stones.79 Nutritional and environmental factors also play a significant role. High dietary intake of salt, calcium, purine, and proteins increase risk, as does living and working in a hot, humid environment.58,76,77 Certain medications also increase risk by increasing urinary pH or by increasing excretion of calcium or phosphate. Examples include indinavir, diuretics, antacids, vitamin C, and allopurinol.77,79
Clinical Presentation. Patients with a stone usually present complaining of the acute onset of severe, intermittent, colicky pain, usually beginning in the flank and radiating to the lower abdomen, groin, and thigh.2,67,75,77,79,80 Twenty percent of patients also complain of nausea and vomiting.81 Many have gross hematuria. If the stone is lodged in the ureterovesical junction, urinary symptoms such as dysuria and frequency are present.58,71,75,79 The patient often reports that the pain makes her want to "roll around" to find a comfortable position.67,77 As renal colic is the most common misdiagnosis of an abdominal aortic aneurysm, this more severe condition must be considered when evaluating older patients with suspected renal colic.
The patient most likely will appear very uncomfortable and may be writhing around on the bed. A common finding is pain out of proportion to physical findings. The patient may exhibit costovertebral angle tenderness and suprapubic tenderness. If there is a concurrent infection, the patient may be febrile.78
Diagnosis. Each patient should have a urinalysis and urine pregnancy test. A CBC and measurement of creatinine and blood urea nitrogen (BUN) may be considered in patients with complicated presentations.72,75,79,80 The urinalysis commonly exhibits hematuria, but a normal urine does not rule out a stone (the urine will be normal in 10-15% of cases).72,77,82 A urine pH of less than 5.5 suggests a uric acid or cystine lithiasis, and a pH of greater than 7.5 suggests infection or a struvite lithiasis.67 It is imperative to rule out concurrent urinary infection.
Despite that 90% of all kidney stones are radiopaque, a plain KUB film has poor sensitivity and specificity for stones.1,16,80,83 This partly is because the stones are very small and can be obscured by overlying fecal content in the bowel or by the vertebral process or bony pelvis. Two better studies for diagnosis of stones are IVP and noncontrast helical CT. IVP consists of a scout film followed by a repeat KUB film 5, 10, and 20 minutes after injection of IV contrast. A stone is likely if there is delay in the appearance of the nephrogram or visualization of the entire ureter.77 If an abnormality is seen, films should be repeated after several hours. The sensitivity for kidney stones is 65-90%, and the specificity is 94-100%.77,80 Advantages of IVP include that it shows urinary system flow and function, is easy to perform, is inexpensive, and is universally available. Disadvantages include its invasiveness and the possibility of reactions to IV contrast; in addition, when the study is positive, it takes several hours to perform, and, when it is negative, it fails to provide an alternative diagnosis.83
Noncontrast helical CT is rapidly replacing IVP as the study of choice. CT is more effective than IVP in precisely identifying stones and is equally effective in determining the presence of obstruction.78 CT allows direct cross-sectional visualization of the entire genitourinary system. It is 97% sensitive and 96% specific in the diagnosis of stones.77,80 Advantages of CT include that it is complete in fewer than 5 minutes, no IV contrast is needed, it allows precise determination of size and location of the stone, and it can demonstrate nonurinary causes of pain if no stone is present.1,77,78,80,83 If one is unsure of the diagnosis but a kidney stone is high in the differential, a noncontrast CT should be obtained primarily, as contrast would obscure the visualization of a stone if present. If this study does not demonstrate a stone or other obvious cause of the pain, then a CT with contrast should be considered.
Treatment. The goals of ED management are adequate pain relief, IV hydration if the patient is dehydrated, ruling out infection, and determining if the stone will pass spontaneously or requires intervention.84,85 Pain control usually is well achieved with IV NSAIDs or narcotics, and antiemetics may be required. If an infection is present, antibiotics should be given. Management of the stone depends on its size, location, and composition. Stones smaller than 4 mm pass spontaneously 80% of the time, and stones 4-6 mm pass spontaneously 60% of the time.77,80 Stones located in the distal ureter pass much more frequently than those in the proximal ureter. Patients should be admitted for very large stones, persistent uncontrolled pain and emesis, infection, pregnancy, the presence of only one kidney, or persistent obstruction.16,79,80,85 If the patient can be discharged safely, she should be sent home with return precautions and adequate pain medicine and should be encouraged to drink 2-3 liters of fluid a day and be given a urine strainer.81,84 Every patient should be set up with urology follow-up within seven days.67
Ovarian Cysts. Physiologic or pathologic enlargement of an ovary is the most common cause of adnexal pain.28 Follicular cysts occur in the initial 2-3 weeks of the menstrual cycle. Follicular dysgenesis occurs when the hypothalamic-pituitary axis malfunctions or because of anatomic defects in the reproductive system.35 Risk factors include smoking, early menarche, infertility, and hypothyroidism.35 Follicular cysts can grow to 8-10 centimeters and generally regress spontaneously within 1-3 months. Stretching of the cyst wall can cause pain, or the cyst may rupture, leading to sharp, severe unilateral midcycle pain (mittelschmerz).28
Failure of degeneration of the corpus luteum leads to luteal cyst formation. These cysts are found in the final one or two weeks of the menstrual cycle, and they can become inflamed or hemorrhage spontaneously.35 Corpus luteum cysts can grow to 5-10 centimeters and can cause unilateral pelvic pain, amenorrhea, and if ruptured, peritoneal irritation and bleeding.28
Ovarian cysts occur in 30% of females with regular menses and in 50% of females with irregular menses.35 The majority occur during childbearing years, are benign, and have a predilection for the right side.
Clinical Presentation. Ovarian cysts are asymptomatic unless complicated by rupture, torsion, or hemorrhage. The patient will most commonly present with mid- or late-cycle lower abdominal pain that is sharp, intermittent, severe, and of sudden onset. It may have been preceded by strenuous activity or sexual intercourse. The patient also may complain of nausea, vomiting, or vaginal spotting. The physical examination most likely will elicit tachycardia, lower abdominal and adnexal tenderness, cervical motion tenderness, and possibly an adnexal mass.35 Although uncommon, hemorrhage may be significant, leading to signs of shock.
Diagnosis. If significant hemorrhage is suspected, the following laboratory tests should be obtained: CBC, type and crossmatch, and coagulation profile. All patients should have a urinalysis and urine pregnancy test. If the woman is unstable, culdocentesis can be done to diagnose or rule out peritoneal hemorrhage. If the patient is stable, transvaginal ultrasound is the study of choice to assess the size, structure, and complexity of the cyst and to rule out torsion.35
Treatment. If the woman is unstable, IV fluids and blood products should be given aggressively, and emergent gynecologic consult and surgery are required. If the patient has a simple, uncomplicated ovarian cyst, she can be given adequate pain control and gynecologic referral as an outpatient, because most cysts of this type resolve spontaneously.35
Endometriosis. Endometriosis is the presence of estrogen-dependent functioning endometrial glands and stroma outside the uterus and often is associated with pelvic pain and infertility.9,86,87 It can be a therapeutic challenge for emergency physicians, and it is a diagnosis of exclusion.
Endometriosis occurs in 10-15% of women, commonly presents in the third decade, and regresses after menopause.10,86,87 It is found in 60% of patients evaluated for infertility.86
The exact cause of endometriosis is unknown. It is postulated to occur secondary to the partial occlusion of the cervix, causing retrograde menstrual flow to pass into the fallopian tubes and into the abdominal cavity, where it implants and comes under the influence of cyclic female hormones.9,87 Other theories include transtubal dissemination or the iatrogenic deposition of endometrial tissue following invasive gynecologic procedures.87 The most common sites of deposition are the ovaries, the broad ligaments, and the peritoneal surfaces of the cul-de-sac. When endometriosis involves the peritoneal surfaces, it appears flat and brown and is described as "powder burns." As the disease progresses, numerous abdomen and pelvic adhesions form. The rectosigmoid colon can become tightly bound by adhesions to the posterior wall of the uterus, resulting in a fixed, retroverted uterus.10 Also possible is endometrioma of the ovary; that is, the ovary forms a cyst that becomes filled with a thick, chocolate-colored fluid consisting of aged, hemolyzed blood and desquamated endometrium.10
Risk factors for endometriosis among women of reproductive age are low parity, long intervals between pregnancies, short menstrual cycle (less than 27 days), long and heavy flow (> 7 days), and spotting before menses.26 Risk decreases with smoking cessation and regular exercise.26
Clinical Presentation. The most common complaint is severe dysmenorrhea and pelvic pain that generally begins two days before the onset of menses, lasts throughout menses, and extends several days after.26 As the disease progresses, pelvic pain occurs outside of menses and usually is described as dull, aching, deep, and radiating to the back, hips, rectum, or thighs.5 Many women also report dyschezia; premenstrual and postmenstrual spotting; and dyspareunia, especially with deep penetration. The intensity of the pain does not appear to correlate with the extent of disease.87
During pelvic examination of a patient with endometriosis, tender nodules can be felt along the thickened uterosacral ligaments, the posterior uterus, and in the cul-de-sac. These are felt most easily on the rectovaginal examination.10,86,87 The patient also may have diffuse, nonlocalized, abdominal tenderness and a retroverted uterus. If the patient has an endometrioma, a tender, fixed, adnexal mass may be detected.
Diagnosis. The evaluation of a woman suspected of having endometriosis should include urinalysis, urine pregnancy test, and cervical cultures. The only way to make a definitive diagnosis is by laparoscopy, which shows the hallmark "powder burn" blue-black lesions. An endometrioma can be visualized with transvaginal ultrasound, which will show a cystic mass with thickened walls and diffuse acoustic enhancement or scattered internal echoes.26 Magnetic resonance imaging may be helpful in making the diagnosis, as it can detect pigmented hemorrhagic lesions, loss of a clear margin of the uterus, and a tethered appearance of the rectum.26
Treatment. ED management includes excluding a life-threatening or other treatable cause of pain, appropriate analgesia, and outpatient follow-up with a gynecologist.87 Hormonal therapy relieves pain in up to 89% of patients, but should be initiated by a gynecologist.87
Complications. Common complications of endometriosis include infertility, pelvic adhesions, and chronic pelvic pain.87 An endometrioma can rupture, leading to a chemical peritonitis requiring emergent surgery.
Uterine Fibroids. Uterine fibroids, or leiomyomas, are the most common benign tumor of the uterus. Uterine fibroids are found in 20% of women older than age 35. They are twice as common in African-American women as in white women.88
The growth of fibroids, particularly multiple fibroids, may be an inherited tendency. Isolated leiomyomas more likely are due to gene mutations that cause overgrowth of the muscle cells of the uterus. Estrogen enhances the growth of fibroids; therefore, up to 20% of gravid women may experience growth during the latter half of pregnancy and in the postpartum and postabortion periods.88 Other factors associated with the growth of fibroids include 1) increased body mass index; 2) nulliparity; and 3) onset of menstruation before the age of 10.89
Clinical Presentation. Uterine fibroids may present clinically along a spectrum from an asymptomatic incidental finding, to alteration in uterine bleeding, to acute pelvic pain. Other symptoms may include pressure on the bladder, causing urinary frequency and urgency; pressure on the rectum, causing constipation; infertility; or increased abdominal girth or a change in abdominal contour.
Pelvic pain usually is caused by one of three complications, all of which are associated with ischemic necrosis. The first is ischemic degeneration, which is most common during pregnancy, when the fibroid can outgrow its blood supply. This can be difficult to differentiate from other causes of pelvic pain, particularly in the absence of a palpable mass. Signs include ileus, fever, and elevated WBC count. The second typical cause of pelvic pain is torsion of a pedunculated fibroid on its vascular stalk, which can cause colicky pain. The third is an aborting submucosal fibroid, which can cause profuse and prolonged vaginal bleeding and uterine cramping.
Diagnosis. ED evaluation of a patient with fibroids should include urinalysis and a pregnancy test. Palpation of a fibroid or finding on ultrasound can confirm the diagnosis. Uterine fibroids appear on ultrasound with typical sonographic patterns: they are hypoechoic and heterogeneous and have an echogenic rim.90 They can be submucosal, subserosal, or pedunculated. Degeneration is seen as small, cystic spaces within a fibroid. Pedulculated fibroids can appear like ovarian masses, so a thorough search for both ovaries must be done. Other tests, including endometrial biopsy, hysteroscopy, and hysterosalpingography, are beyond the scope of the ED workup.
Treatment. Treatment of uterine fibroids depends on the presenting symptoms and may include observation; NSAIDs for treatment of pelvic pain; oral contraceptives, which may decrease menstrual flow and pelvic pain; GnRH analogs or RU-486 to decrease estrogen levels; myomectomy; or hysterectomy. Uterine artery embolization, although beyond the scope of the emergency physician, is a promising new nonsurgical alternative. The technique is similar to that done for the treatment of emergent hemorrhage. It can be done as an outpatient procedure with a low same-day admission or delayed admission rate.91,92
Conclusion
Abdominal pain in women of childbearing age continues to be a major concern and difficult to assess in the ED. Patients who appear non-toxic actually may have an underlying surgical emergency, and some "sick" patients ultimately will be diagnosed with benign conditions. A patient’s history of pregnancy as well as the accuracy of a pelvic examination may be unreliable. STDs remain epidemic in the United States, increasing the risks of ectopic pregnancy. An algorithmic approach is presented, which distinguishes between pregnant and nonpregnant women; differentiates normal and abnormal urinalysis; and allows early surgical consultation and imaging, leading to rapid assessment, diagnosis, and appropriate treatment.
References
1. Colucciello SA, Lukens TW, Morgan DL. Assessing abdominal pain in adults: A rational, cost-effective, and evidence-based strategy. Emerg Med Practice 1999;1:1-20.
2. American College of Emergency Physicians. Clinical policy for the initial approach to patients presenting with a chief complaint of nontraumatic acute abdominal pain. Ann Emerg Med 1994;23:906-922.
3. Abbott J. Pelvic pain: Lessons from anatomy and physiology. J Emerg Med 1990;8:441-447.
4. Padilla LA, Radosevich DM, Milad MP. Accuracy of the pelvic examination in detecting adnexal masses. Obstet Gynecol 2000;96:593.
5. Jacobs DS, Demott WR, et al. Laboratory Test Handbook, 4th ed. Cleveland: Lexi-Comp Inc.; 1996:190.
6. Silver BE, Patterson JW, Kulick M, et al. Effect of CBC results on ED management of women with lower abdominal pain. Am J Emerg Med 1995;13: 304-306.
7. Salkin MS. Appendicitis: Avoiding failure to diagnose. ED Legal Letter 1996;7:113-124.
8. LoVecchio F, Oster N, Sturmann K, et al. The use of analgesics in patients with acute abdominal pain. J Emerg Med 1997;15:775-779.
9. Michalakes CJ, Pundt MR, Broderick KB. Obstetrics and disorders of pregnancy. In: Aghababian R, ed. Emergency Medicine, The Core Curriculum. Philadelphia: Lippincott-Raven; 1998:577-603.
10. Hacker NF, Moore JG. Essentials of Obstetrics and Gynecology, 2nd ed. Philadelphia: W.B. Saunders; 1992; 368-394, 425-435.
11. No authors listed. From the Centers of Disease Control. Ectopic pregnancy-United States, 1990-1992. JAMA 1995;272:533.
12. Pisarska MD, Carson SA. Incidence and risk factors for ectopic pregnancy. Clin Obstet Gynecol 1999;42:2-8.
13. Jehle D, Krause R, Braen GR. Ectopic pregnancy. Emerg Med Clin North Am 1994;12:55-71.
14. Pisarska MD, Carson SA. Ectopic pregnancy. In: Scott JR, et al, eds. Danforth’s Obstetrics and Gynecology, 8th ed. Philadelphia: Lippincott Williams & Wilkins; 1999:155-172.
15. Stovall TG, Kellerman AL, Ling FW, et al. Emergency department diagnosis of ectopic pregnancy. Ann Emerg Med 1990;19:1098-1103.
16. DeCherney AH, Jones EE. Ectopic pregnancy. Clin Obstet Gynecol 1985;28: 365-373.
17. Krause RS, Janicke DM. Ectopic pregnancy. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 686-693.
18. Ory SJ. New options for diagnosis and treatment of ectopic pregnancy. JAMA 1992;267:534-537.
19. Stovall TG, Ling FW. Some new approaches to ectopic pregnancy. Cont ObGyn 1992;5:35.
20. Barnhart KT, Simhan H, Kamelle SA. Diagnostic accuracy of ultrasound above and below the beta-HCG discriminatory zone. Obstet Gynecol 1999;94:583-587.
21. Rumack C, Wilson S, Charboneau JW. Diagnostic Ultrasound. St. Louis: Mosby; 1998,998-1004.
22. Reece EA, Petrie RH, Sirmans MF et al. Combined intrauterine and extrauterine gestations: a review. Am J Obstet Gynecol 1983; 146:323-330.
23. Rojansky N, Schender JG. Heterotopic pregnancy and assisted reproduction-an update. J Assist Reprod Genet 1996;13:594-601.
24. Carson SA, Buster JE. Ectopic pregnancy. N Engl J Med 1991;329:1174-1179.
25. Lipscomb GH, Stovall TG, Ling FW. Nonsurgical treatment of ectopic pregnancy. N Engl J Med 2000;343:1325-1329.
26. Duleba J. Diagnosis of endometriosis. Obstet Gynecol Clin North Am 1997;24:337-343.
27. Kim YH, Cho KS, Ha HK, et al. CT features of torsion of benign cystic teratoma of the ovary. J Comput Assist Tomogr 1999;23:923-928.
28. Brinsfield KH, DiBella M, Bennett MK, et al. Urogenital/gynecologic disorders. In: Aghababian R, ed. Emergency Medicine: The Core Curriculum. Philadelphia: Lippincott-Raven; 1998:1285-1294.
29. Blander M. Ovarian torsion. eMedicine.com 2000; 1-9. (Accessed 12/17/2001.)
30. Warner MA, Fleischer AC, Edell SL, et al. Uterine adnexal torsion: Sonographic findings. Radiology 1985;154:773-775.
31. Hibbard LT. Adnexal torsion. Am J Obstet Gynecol 1985;152:456-461.
32. Bider D, Mashiach S, Sulitzky M, et al. Clinical, surgical and pathological findings of adnexal torsion in pregnant and nonpregnant women. Surg Gynecol Obstet 1991;173:363-366.
33. Nichols DH, Julian PJ. Torsion of the adnexa. Clin Obstet Gynecol 1985;28: 375-380.
34. Jain KA, Quam JP, Ablin DS, et al. Imaging findings in patients with RLQ pain: Alternative diagnoses to appendicitis. J Comput Assist Tomogr 1997; 21:693-698.
35. Kassi AA, Roberts R, et al. Ovarian cysts. eMedicine.com 2000; 1-16. (Accessed 12/17/2001.)
36. Kuo DY-S, Jones JG, Runowicz CD. Diseases of the ovary and fallopian tubes. In: Scott JR, et al. Danforth’s Obstetrics and Gynecology. Philadelphia: Lippincott Williams & Wilkins; 1999: 865-908.
37. Rothrock SG, Green SM, Dobson M, et al. Misdiagnosis of appendicitis in nonpregnant women of childbearing age. J Emerg Med 1995;13:1-8.
38. Wagner JM, McKinney WP, Carpenter JL. Does this patient have appendicitis? JAMA 1996;276:1589-1594.
39. Craig S. Appendicitis, acute. eMedicine.com 2000; 1-14. (Accessed 12/17/2001.)
40. Hals G. Acute appendicitis: Meeting the challenge of diagnosis in the ED. Emerg Med Reports 1999;20:71-82.
41. FitzGerald DJ, Pancioli AM. Appendicitis. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 535-539.
42. Whitworth CM, Whitworth PW, Sanfillipo J,et al. Value of diagnostic laparotomy in young women with possible appendicitis. Surg Gynecol Obstet 1988;167:187-190.
43. Wenstrom KD, Malee MP. Medical and surgical complications of pregnancy. In: Scott JR, et al. Danforth’s Obstetrics and Gynecology. Philadelphia: Lippincott Williams & Wilkins; 1999: 327-361.
44. Silen W. Acute appendicitis. In: Fauci AS, et al. Harrison’s Principles of Internal Medicine, 14th ed. New York: McGraw-Hill; 1998: 1658-1660.
45. Johnson GA, Farnsworth WJ, Heisig DG, et al. Abdominal and gastrointestinal disorders. In: Aghababian R, ed. Emergency Medicine: The Core Curriculum. Philadelphia: Lippincott-Raven; 1998: 1-3.
46. Arnbjornsson E. Bacteriuria in appendicitis. Am J Surg 1988;155:356-358.
47. Scott JH, Amin M, Harty JI. Abnormal urinalysis in appendicitis. J Urol 1982;129:1015.
48. Townsend CM, et al. Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice, 16th ed. Philadelphia: W.B. Saunders; 2001: 920-921.
49. Rao PM, Rhea JT, Novelline RA, et al. Helical CT technique for the diagnosis of appendicitis. Radiology 1997;202:139-144.
50. Lane MJ, Liu DM, Huynh MD, et al. Suspected acute appendicitis: Nonenhanced helical CT in 300 consecutive patients. Radiology 1999;213:341-346.
51. Malone AJ Jr, Wolf CR, Malmed AS, et al. Diagnosis of acute appendicitis: Value of unenhanced CT. Am J Radiol 1993;160:763-766.
52. Horton MD, Counter SF, Florence MG, et al. A prospective trial of computed tomography and ultrasonography for diagnosing appendicitis in the atypical patient. Am J Surg 2000;179:379.
53. Stewart C, Bosker G. Pelvic inflammatory disease: Diagnosis, disposition, and current antimicrobial guidelines. Emerg Med Reports 1999;20:163-172.
54. Andersen BR, Kallehave FL, Andersen HK. Antibiotics versus placebo for prevention of postoperative infection after appendectomy (Cochrane Review). Cochrane Database Systemic Rev 2001; 2:CD001439.
55. Abbuhl S, Reyes I. Pelvic inflammatory disease. Emedicine.com 2000; 1-15. (Accessed 12/17/2001.)
56. McCormack WM. Pelvic inflammatory disease. N Engl J Med 1994;330: 115-118.
57. Behrman AJ, Shepherd SM. Pelvic inflammatory disease. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 719-723.
58. Tierney L, et al. Current Medical Diagnosis and Treatment. Old Tappan, NJ: Appleton and Lange; 1995: 802-806.
59. Bongard F, Landers DV, Lewis F. Differential diagnosis of appendicitis and pelvic inflammatory disease. Am J Surg 1985;150:90-95.
60. Najem AZ, Barillo DJ, Spillert CR, et al. Appendicitis versus pelvic inflammatory disease: A diagnostic dilemma. Am Surg 1985;51:217-222.
61. Eschenbach DA. Pelvic infections and sexually transmitted diseases. In: Scott JR, et al. Danforth’s Obstetrics and Gynecology. Philadelphia: Lippincott; 1999: 579-600.
62. DeCherney AH, Pernoll ML. Current Obstetrics and Gynecologic Diagnosis and Treatment. Norwell: Appleton and Lange; 1994: 774-776.
63. Howes DS, Young WF. Urinary tract infections. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 625-631.
64. Stamm WE. Urinary tract infections and pyelonephritis. In: Fauci AS, et al. Harrison’s Principles of Internal Medicine, 14th ed. New York: McGraw-Hill; 1998: 817-824.
65. Hooton TM, Stamm WE. Diagnosis and treatment of uncomplicated urinary tract infection. Infect Dis Clin North Am 1997;11:551-570.
66. Bienstock JL, Fox HE. Comorbid diseases in pregnancy. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 702-707.
67. Hong RD, Gomella LG, Brunetto AD, et al. Renal disorders. In: Aghababian R, ed. Emergency Medicine: The Core Curriculum. Philadelphia: Lippincott-Raven; 1998: 847-871.
68. Pasternak EL 3rd, Topinka MA. Blood cultures in pyelonephritis: do results change therapy? Acad Emerg Med 2000;7:1170.
69. Thanasssi M. Utility of urine and blood cultures in pyelonephritis. Acad Emerg Med 1997;4:797-800.
70. Wing DA, Park AS, Debuque L, et al. Limited clinical utility of blood and urine cultures in the treatment of acute pyelonephritis during pregnancy. Am J Obstet Gynecol 2000;182:1437-1440.
71. Myers AR. Medicine, 2nd ed. Philadelphia: Harwal Publishing; 1994: 259-261.
72. Press SM, Smith AD. Incidence of negative hematuria in patients with acute urinary lithiasis presenting to the emergency room with flank pain. Urology 1995;45:753-757.
73. Balaji KC, Menon M. Mechanism of stone formation. Urol Clin North Am 1997;24:1.
74. Skolnick AA. Guidelines issued for treatment of ureteral calculi. JAMA 1997;278:1392.
75. Andreoli TE et al. Cecil Essentials of Medicine, 4th ed. Philadelphia: W.B. Saunders; 1997: 222-224.
76. Pak CY. Kidney stones. Lancet 1998;351:1797-1800.
77. Moll J, Peacock WF IV. Urologic stone disease. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 640-645.
78. Smith RC, Rosenfield AT, Choe KA, et al. Acute flank pain: Comparison of non-contrast-enhanced CT and intravenous urography. Radiology 1995;194:789-794.
79. Plantz S. NMS Emergency Medicine. Baltimore: Williams & Wilkins; 1997: 405-407.
80. Begun FP, Foley WD, Peterson A, et al. Patient management, laboratory and imaging studies. Urol Clin North Am 1997;24:97-116.
81. Morse RM, Resnick MI. Ureteral calculi: Natural history and treatment in an era of advanced technology. J Urol 1991;145:263-265.
82. Stewart DP, Kowalski R, Wong P, et al. Microscopic hematuria and calculus-related ureteral obstruction. J Emerg Med 1990;8:693-695.
83. Smith RC, Dalrymple NC, Neitlich J. Noncontrast helical CT in the evaluation of acute flank pain. Abdom Imaging 1998;23:10-16.
84. Barker LR, Burton J, Zieve P. Principles of Ambulatory Medicine, 5th ed. Baltimore: Williams & Wilkins; 1999: 565-575.
85. Singal RK, Denstedt JD. Contemporary management of ureteral stones. Urol Clin North Am 1997;24:59-61.
86. Morrison L, Spence J. Vaginal bleeding and pelvic pain in the nonpregnant patient. In: Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2000: 669-680.
87. Hsu K, Doak S. Endometriosis. eMedicine.com 2000; 1-7. (Accessed 12/17/01.)
88. Thack AM, Young GP. Pelvic pain. In: Rosen P, et al. Emergency Medicine. New York: Mosby; 1998: 2300.
89. Givens M. Fibroids. eMedicine.com 2001:1-13. (Accessed 12/17/01.)
90. Lee W, Comstock C. Pelvic ultrasonography. In: Tintinalli JE et al. Emergency Medicine: A Comprehensive Study Guide, 4th ed. New York: McGraw-Hill; 1996: 1408.
91. Siskin GP, Tublin ME, Stainken BF, et al. Uterine artery embolization for the treatment of adenomyosis: Clinical response and evaluation with mr imaging. Am J Roentgenol 2001;177:297-302.
92. Klein A, Schwartz ML. Uterine artery embolization for the treatment of uterine fibroids: An outpatient procedure. Am J Obstet Gynecol 2001;184: 1556-1563.
Want to earn free OB/GYN CME courses? With FreeCME you can earn accredited CME courses for free online. Get started today!
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
