Clinical Trials: Voriconazole
Clinical Trials
Voriconazole
By Stan Deresinski, MD, FACP
Voriconazole was recently recommended for FDA approval for use in the treatment of invasive aspergillosis. This approval was largely based on the pooled analysis of 2 multicenter clinical trials, one performed in Europe and one in the United States, in which a total of 392 patients were randomized to treatment with either voriconazole or amphotericin B deoxycholate.1 In these studies, the global response rate at 12 weeks was 52.8% (95% CI for the difference, 9.6-33.6%) in the voriconazole recipients and only 31.6% in those randomized to amphotericin B. Furthermore, survival rates at 84 weeks were, respectively, 71.8% and 57.9% (P = 0.015). The few cases of A flavus infection did not respond to therapy. The results of an open, noncomparative experience in the treatment of invasive aspergillosis also appear generally favorable.
Separately, voriconazole was compared to liposomal amphotericin B in a randomized trial of empiric therapy of febrile neutropenic patients with hematological malignancy who remained febrile despite at least 96 hours of empiric broad spectrum antibacterial therapy.1-3 Voriconazole (barely) failed to meet the predetermined criterion for noninferiority and, as a consequence, has not received FDA approval for this indication.4 Nonetheless, there was no difference between the treatment arms with regard to breakthrough fungal infections or mortality. In fact, the apparent failure of voriconazole was solely due to less frequent resolution of fever during neutropenia.5 Thus, despite the lack of FDA approval, and taking into account the lesser infusion related and renal toxicity relative to liposomal amphotericin B, many clinicians will come to the conclusion that voriconazole is an effective option in this setting.
In addition to its activity against Aspergillus spp., this bistriazole congener of fluconazole has antifungal activity in vitro against Candida spp., including many resistant to fluconazole, with MICs against these organisms similar to those of itraconazole. Thus, voriconazole has also been compared, in randomized trials, to both fluconazole and amphotericin B deoxycholate in the treatment of esophageal candidiasis. In each case, outcomes between patients receiving voriconazole and those receiving the comparator were similar. Reports of case series of open noncomparative administration indicate that voriconazole may also be effective in some cases of infection due to less commonly encountered fungi, such as Scedosporium apiospermum, for which previously available therapies have frequently ineffective.
Fungicidal or Fungistatic?
Voriconazole, like other azoles, does not have fungicidal activity against Candida spp. It has been claimed that, in contrast to itraconazole, voriconazole is fungicidal against Aspergillus spp. One study found that the concentration of voriconazole required to achieve a 95% reduction in colony forming units of A fumigatus was 0.5 mcg/mL, compared to 1.0 mcg/mL for itraconazole. Another study found a 95-99% reduction at a concentration of 5 mcg/mL when read at 24 hours.6 It is likely, however, that delay of the evaluation until 48 hours would sharply reduce the apparent efficacy of this drug. Furthermore, while these studies were performed using a conidial inoculum, reported experiments indicate that hyphae are significantly more resistant to the fungicidal activity of this agent.6 Nonetheless, while the fungicidal activity of voriconazole against A fumigatus remains subject to debate, the clinical trial data discussed above leave little room for doubt about the efficacy of voriconazole in the treatment of patients with neutropenia and invasive aspergillosis.
Tolerability
Voriconazole has, in general, been well tolerated, although transient visual disturbance has been reported in every clinical trial with incidences that ranged from approximately 11-52%. In the empiric therapy in febrile neutropenia, the incidence was 22% in the voriconazole recipients and 1% in those given liposomal amphotericin. This adverse event appears to be plasma concentration-related, with an approximately 5% increase in risk for each 1.0 mcg/mL increase in plasma voriconazole concentration. At plasma concentrations > 3.0 mcg/mL, the incidence of visual complaints was > 25%. In addition, 4.3% of voriconazole recipients in the Aspergillus studies developed visual hallucinations, thought to be distinct from the more common visual disturbances.
The incidence of transaminase elevations was low and was similar in the recipients of voriconazole and of liposomal amphotericin. Data from compiled clinical trials suggest that serum ALT and AST elevations are associated with plasma concentrations of voriconazole in excess of 5 mcg/mL. However, a threshold concentration for hepatotoxicity could not be discerned on examination of data from early Phase I/II trials. Nonetheless, reversible hepatotoxicity was observed in 3 of 7 subjects given 400 mg b.i.d. in a Phase I study.
Cardiac toxicity has not been reported in the clinical trials. However, in preclinical studies, QT interval prolongation was observed in dogs with peak serum concentrations of 24 mcg/mL. The reported association of itraconazole and the development of congestive heart failure must also be kept in mind.
Voriconazole, like other azoles, is teratogenic.
Pharmacokinetics
In the absence of a loading dose, steady state trough concentrations are achieved after 5 days of dosing. The plasma kinetics of voriconazole are nonlinear as the consequence of saturable metabolism. Thus, a 2-fold increase in dose from 200 mg to 400 mg results in a 2.8-fold increase in Cmax and a 3.9-fold increase in AUC, together with an increase in T1/2 from 6 hours to 12 hours. The saturable metabolism also leads to plasma accumulation with significant increases in Cmax and AUC with repetitive dosing. Females have significantly higher drug exposure and greater drug accumulation than males. Only 1.5% of the drug is excreted unchanged in the urine. Its major metabolite, N-oxide voriconazole, has minimal antifungal activity.
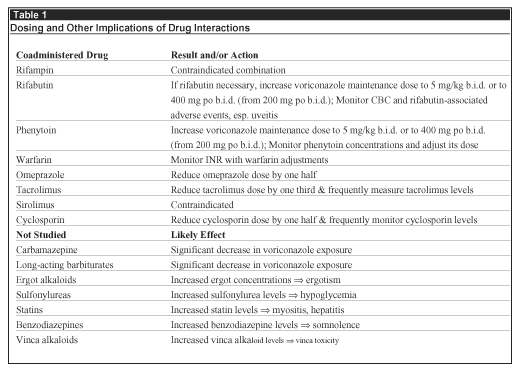 |
Voriconazole is a substrate, as well as an inhibitor of the CYP450 enzymes, CYP2C19, 2C9, and 3A4 and at least 30% of the variability in its pharmacokinetics is accounted for by widespread human polymorphisms in CYP2C19. Approximately 5% of Caucasians and 10-20% of Asians are deficient in this enzyme and, thus, are poor metabolizers of voriconazole. Analysis of the genotypes of the patients participating in the Phase I trials of this drug, 28% of whom were Japanese, found that 9% were PM and 29% were heterozygotes. Despite this reported variability, patients in the Aspergillus trials maintained mean plasma voriconazole levels between 2 and 4 mcg/mL throughout the 12-hour dosing interval during which approximately 75% of individually measured levels were between 1 and 7 mcg/mL. These results are not inconsistent with data compiled from Phase I studies in which, with repetitive dosing, 22% of patients had a voriconazole Cmax = 1.0 mcg/mL, while in 12% the Cmax was = 6.0 mcg/mL.
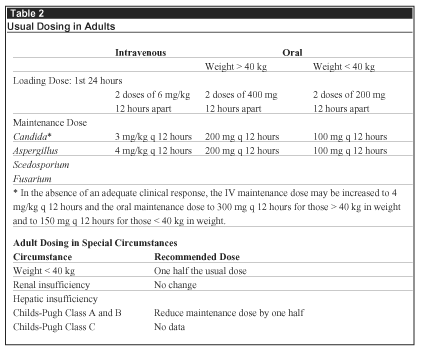 |
The voriconazole AUC is increased more than three-fold in the presence of mild-to-moderate hepatic impairment (Child-Pugh Class A and B). As a consequence, after administration of the usual initial dose, subsequent doses of voriconazole should be reduced by one-half. There are no data in patients with Child-Pugh Class C liver disease. Voriconazole kinetics are not altered in the presence of renal insufficiency. However, voriconazole for IV administration is formulated with sulpho-butyl-ether-cyclodextrin and this solubilizing excipient is renally excreted and, in addition, is associated with nephrotoxicity in experimental animals. As a consequence, the IV formulation of voriconazole should not be administered to patients whose creatinine clearance is < 50 mL/min.
References
1. www.fda.gov/ohrms/dockets/ac/01/transcripts/3792t2.htm
2. Denning DW, et al. Clin Infect Dis. 2002;34:563-571.
3. Walsh TJ, et al. N Engl J Med. 2002;346:225-234.
4. Powers JH, et al. N Engl J Med. 2002;346:289-290.
5. Marr K. N Engl J Med. 2002;346:278-280.
6. Lass-Florl C, et al. Antimicrob Agents Chemother. 2001;45:124-128.
Dr. Deresinski, Clinical Professor of Medicine, Stanford; Director, AIDS Community Research Consortium; Associate Chief of Infectious Diseases, Santa Clara Valley Medical Center, is Editor of Infectious Disease Alert.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
