Defining the Susceptibility of Streptococcus pneumoniae to ß-lactam Antibiotics: Putting the Cat
Defining the Susceptibility of Streptococcus pneumoniae to ß-lactam Antibiotics: Putting the Cat Among the Pigeons
Abstract & Commentary
Synopsis: The MIC breakpoints for beta lactams against S pneumoniae have been revised, taking into account the different demands of therapy in meningeal and nonmeningeal infections.
Source: Musher DM, et al. Arch Intern Med. 2001;161: 2538-2544.
Until the 1970s, it had been assumed that all strains of Streptococcus pneumoniae were universally susceptible to penicillin and would remain so. The appearance of resistant strains in South Africa was an unwelcome surprise. The definition of Pneumococcal resistance was based primarily on the laboratory and clinical data arising from meningitis, but not the more common manifestations of pneumococcal disease such as sinusitis, otitis media or pneumonia. Now, almost a quarter of a century later, this has been rectified with a change in the definition of susceptibility (see Table 1).
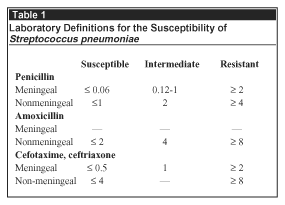 |
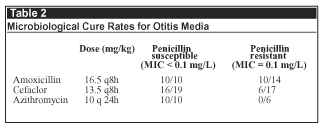
The choice of a low concentration of 0.06 mg/L was based on the fact that CSF levels of penicillin among children treated for meningitis 4 hours after the dose are around 1 mg/L and those of ceftriaxone are 3-4 fold higher. Hence, MICs that exceed this are unlikely to prove successful for treatment. However, it is plasma or serum levels that are more important in other forms of pneumococcal disease and these are higher than those obtained in CSF. For instance, in pneumonia an MIC of 2 mg/L of penicillin or 4 of ceftriaxone would be 2-4 fold lower than the peak serum concentrations and, hence, should prove amenable to treatment. Similar considerations apply to sinusitis and otitis media (see Table 2). While the S pneumoniae was eradicated after treatment with amoxicillin in 10 of the 14 cases with infection caused by a penicillin-resistant strain, only 6 of the 17 cases treated with cefaclor and none of the 6 cases treated with azithromycin responded. These results provide further motivation for setting different breakpoints for amoxicillin between meningeal and nonmeningeal disease or, rather, central nervous system disease and other diseases. The observation that the risk of dying within 4 days of starting treatment for bacteremia due to S pneumoniae was only increased for strains with an MIC of 4 mg/L or higher provides yet more evidence to support a change.
 |
Importantly, it is now explicitly acknowledged that, besides the MIC of the strain, the site of infection and tissue levels of the drug (which, in turn, depends upon the dosage of antibiotic and the route of administration) need to be considered when defining susceptibility or resistance of S pneumoniae.
Comment by J. Peter Donnelly, PhD
A change in the breakpoint MICs for S pneumoniae is a move in the right direction but reminds me of a discussion that surfaces from time to time—namely, how are these breakpoints actually arrived at and do they make any real sense. There are increasingly more exceptions to the rule which tacitly acknowledges the simple fact that the one-fits-all principle does not actually apply as often as was hoped and that perhaps the approach advanced by Ericsson and Sherris 30 years ago namely, that each genus should have its own breakpoint to any given antibiotic, was correct after all.1 Moreover, the susceptibility of bacteria to an antibiotic is bound to differ for infections in different body systems simply because bacterial growth in vivo is radically different from that found in a test tube. Microbiologists worry that complex rules for defining breakpoints might create confusion among clinicians, but my impression is that although a clear and unequivocal answer is usually demanded from the laboratory by clinicians, few actually expect a single breakpoint for the same bacterium under all circumstances. Indeed, some may argue that susceptibility testing is usually done after the fact because antibiotics are more often than not given empirically and the results only become of interest when treatment is considered to be failing. Perhaps it is time for clinicians to state directly whether they have a given drug in mind or whether they have already started treatment with the drug and only ask to be informed when the strain is found to be resistant with the microbiologist advising alternative drugs that might prove effective. The laboratory would still be able to use the result to refine the rates of susceptibility in order to judge whether the first-line treatment for a given indication needs to be reconsidered or not. For instance, if the incidence of S pneumoniae with MICs of penicillin exceeding 0.1 mg/L is low (as is still the case here in Nijmegen) knowing the susceptibility of a particular strain will not materially alter the odds of success or failure of treatment with a ß-lactam antibiotic. Conversely, high or rising rates of resistance would raise the alarm and necessitate changing the empirical regimen or at least adopting an approach whereby the initial regimen was changed accordingly should the offending strain prove less than fully susceptible given the site of infection. This seems to offer the best balance between meeting the needs of clinicians and allowing laboratories to exercise their expertise efficiently and economically.
Reference
1. Ericsson HM, Sherris JC. Acta Pathol Microbiol Scand [B] Microbiol Immunol. 1971;217(Suppl 27):1-20.
Dr. Donnelly, Clinical Microbiologist, University Hospital, Nijmegen, The Netherlands, is Associate Editor of Infectious Disease Alert.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
