Prostate Cancer Screening Part I: The Screening Dilemma, Screening Tests, and the Role of Prostate Specific Antigen Testing
Prostate Cancer Screening Part I: The Screening Dilemma, Screening Tests, and the Role of Prostate Specific Antigen Testing
Author: Marc B. Garnick, MD, Clinical Professor of Medicine, Harvard Medical School, Physician, Beth Israel Deaconess Medical Center, Boston, MA.
Peer Reviewer: Durado Brooks, MD, MPH, Director of Prostate and Colorectal Cancers, American Cancer Society, Dallas, TX.
Prostate cancer is one of the leading causes of cancer in men, both in this country and in Europe. Due to the introduction of the prostate specific antigen blood test (PSA test), the ability to diagnose prostate cancer well before signs or symptoms of the disease develop has been realized. Following its introduction, the PSA was endorsed widely and recommended by many major public health organizations as a useful screening test. These included the American Cancer Society and American Urological Association, among others. However, despite the ability of the PSA test, either alone or in combination with the digital rectal examination (DRE), to detect earlier and earlier stages of prostate cancer, its routine use is no longer widely endorsed for screening populations of asymptomatic men. In great part, this is due to the lack of data from large randomized studies demonstrating a survival advantage for men who are screened vs. those who receive standard care without planned screening.
Because the natural history of prostate cancer is so variable, many patients with prostate cancer never develop any signs or symptoms of the disease during their lifetimes. Consequently, many men will die with prostate cancer rather than from prostate cancer. Because the disease is more prevalent in an elderly population who commonly carry concomitant medical diagnoses and co-morbidities, a screen-detected cancer, if definitively treated (e.g., with radiation or radical prostatectomy), may lead to substantial morbidity. Yet a subset of these cancers never would have caused clinical problems in the first place. From the primary care physician perspective, it is important to counsel patients about the complications arising from both the prostate biopsy procedure as well as definitive treatments for prostate cancer.
The primary care physician often is the first heath care professional who orders a PSA test. Thus, an understanding of the various characteristics of the testincluding sensitivity, specificity, novel isoforms of PSA, factors that can influence false-positive values, characteristics of the test that accompany benign conditions of the prostate glandmust be appreciated. Moreover, the changing policy recommendations of national organizations relating to prostate cancer screening that include internists and family physicians must be reviewed.
This two-part series first will focus on the issues surrounding the use of the PSA test as a screening test for prostate cancer. Part II will focus on understanding the definitive local therapies, including effectiveness, side effects, and quality of life, that are available for the treatment of a screen-detected prostate cancer as well as the natural history of untreated prostate cancer. Finally, the recent recommendations of relevant public health policy organizations will be summarized.
The Editor
Introduction
Prostate cancer is one of the most common cancers encountered in American men and will account for nearly 232,000 newly diagnosed patients in 2005. In that same year, nearly 30,000 men will die of prostate cancer. With the widespread introduction of the PSA test nearly 20 years ago, the complexity facing primary care physicians has intensified regarding whether to offer prostate cancer screening to asymptomatic patients. Because prostate cancer affects an elderly population of men, usually with a median age of diagnosis in the early 70s (as cited in Surveillance, Epidemiology and End Results [SEER] Program, released April 2004), competing causes of co-morbidity and mortality may occur well before a prostate cancer, even if histologically present, ever will become clinically active.
The Prostate Cancer Screening Dilemma
Because of the unique anatomic surroundings of the prostate gland, any effort to eradicate a cancer-containing prostate gland by either surgery or radiation (using various forms) has the potential to alter urinary function, erectile function, and bowel function. While multiple studies have shown that a substantial negative impact in the overall quality of life of the treated patient may result from treatment of screen-detected cancers, there is yet no unequivocal evidence that such screening, detection, and treatment improves overall survival.1,2
Prostate Gland Anatomy
The prostate gland anatomically lies anterior to the rectum, inferior to the bladder neck, and superior to the penile urethra. The tissue that surrounds the gland includes the bladder neck, anterior rectum, pubic arch, and base of the penis; hence any surgical extirpation of the gland and periprostatic tissues (e.g., seminal vesicles) requires skill to preserve continence and potency. Likewise, because of the close proximity of the prostate gland to the anterior rectum, radiation may cause some tissue damage to the distal rectum as a result of treatment. Thus, incontinence and impotence (aside from the operative risks of undergoing a radical prostatectomy, to be discussed fully in Part II of this series) are the two most important complications of treatment. These complicationsincontinence, impotence and in the case of radiation, proctitiscombined with the lack of data demonstrating a clear link between screening and decreasing cancer mortality have tempered enthusiasm for unlimited screening for prostate cancer.
What Are the Best Screening Tests for Prostate Cancer?
Screening studies traditionally have used a combination of the PSA blood test, the DRE, and transrectal ultrasound (TRUS), either alone or in varying combinations. Either DRE alone or TRUS alone generally is not useful. PSA commonly is used with DRE, with the TRUS providing supportive information at the time of anticipated prostate biopsy. The reader should appreciate that the sensitivity, specificity, and positive predictive value of detecting prostate cancer are improved when both the PSA and DRE are used as screening tests compared to either one alone.3-8 However, while both of the ongoing large international screening trials (the European Study of Screening for Prostate Cancer [ERSPC] and the American Prostate, Lung, Colorectal, and Ovarian [PLCO] Screening Trial) initially required both a PSA and a DRE as part of the screening procedures, the ERSPC subsequently has dropped the requirement for DRE, and lowered the PSA cutoff value from 4.0 ng/mL to 3.0 ng/mL.3 Thus, in the largest screening trial in Europe, PSA alone has become the only test used for initial screening and for determining the need for biopsy. The PLCO still requires both PSA and DRE as part of their entry criteria.
Interpreting PSA Tests
Prostate specific antigen is a glycoprotein of molecular weight of 33 kiloDaltons (kD) secreted by prostatic epithelium. It is prostate-gland specific, but not prostate-cancer specific. Until recently, PSA tests that showed a value of greater than 4 ng/mL were considered abnormal.4 Patients with values of 4-10 ng/mL often were considered for prostate biopsy regardless of the physical findings of the DRE; and values greater than 10 ng/mL often were associated with prostate cancer.5-8 Other common pathologic findings of the prostate gland were benign prostatic hyperplasia (BPH), chronic prostatitis, or the pre-neoplastic lesion, prostatic intraepithelial neoplasia (PIN); these often were detected if cancer was not found on prostate biopsy of men with values of PSA greater than 4 ng/mL.
PSA, after being secreted into the bloodstream by the prostatic epithelium, may circulate bound to serum proteins (e.g., alpha one anti-chymotrypsin, or beta two microglobulin) as well as in a "free" formunbound to circulating proteins. If the ratio of free PSA as a fraction of total PSA is less than 15% (when the total PSA is between 4 and 10 ng/mL), there is a greater likelihood that prostate cancer may be present. In contrast, ratios of free PSA as a fraction of the total PSA of greater than 25% generally are associated with BPH. Thus, the free vs. bound PSA ratios or simply the percentage free PSA test may be ordered to help distinguish PSA elevation due to cancer from elevations related to benign prostatic diseases.9 Other isoforms of PSA are emerging in research settings that may further distinguish PSA emanating from cancer vs. that generated by benign tissue.
Sensitivity and Specificity of Screening PSA Tests for Detecting Prostate Cancer
Published estimates of the sensitivity and specificity of PSA for cancer detection vary widely. These values are markedly impacted by the cutoff level selected to trigger prostate biopsy. In one study, for men younger than 60 years and using a cutoff of PSA of greater than 4 ng/mL, the sensitivity was only 0.18, with a specificity of 0.98. However, when a cutoff point of 2.6 was selected (that is, if biopsies were performed on all men with a PSA of greater than 2.6 as opposed to 4.0 ng/mL), sensitivity increased nearly two-fold to 0.36, with a specificity of 0.94, using statistical methodologies. The increase in sensitivity by lowering the cutoff point for consideration of biopsy men is known as "verification bias."10 Verification bias is the term that refers to the following dilemma: The specific number for determining sensitivity and specificity of PSA for values less than the 4 ng/mL cutoff point are subject to bias, since few if any of those individuals underwent a prostate biopsy. Using complex mathematical modeling, the potential sensitivity and specificity can be calculated for those who are not biopsied at varying cutoff points below the previously established value of 4 ng/mL.
Until recently, PSA levels less than 4.0 ng/mL were considered normal. A growing body of research is leading many experts to question this interpretation. Very recent data obtained from the Prostate Cancer Prevention Trial (PCPT), which contains information on men who were biopsied with "normal" PSA values and a normal DRE, indicate that in men older than 55 years, there really is no consistent cutoff PSA value that distinguishes normal from abnormal, even in the presence of a normal DRE. Indeed, these recent data indicate that nearly 26% of men with a PSA level between 3 and 4 ng/mL will harbor prostate cancer, even in the settings where the DRE is completely negative.11 In one large study of more than 10,000 screened men with PSA, the positive predictive value of PSA in detecting prostate cancer was 24% for PSA levels of 4.0 to 9.9 ng/mL. The likelihood of detecting prostate cancer continuously increases with PSA levels above 10 ng/mL.12
Recent findings notwithstanding, in many clinical settings PSA levels less than 4.0 ng/mL continue to be reported as "normal," and additional evaluation is reserved for patients with PSA values of 4.0 ng/mL or higher. These individuals often are considered for prostate biopsy, regardless of the physical findings of the DRE.
Other Considerations Regarding PSA Interpretation
PSA Velocity. In addition to its utility as a potential marker to enhance earlier diagnosis of prostate cancer in an otherwise asymptomatic man, changes in PSA values over time may be extremely helpful in determining whether prostate cancer exists and, if so, what the biology of the cancer is likely to be. The rate of change of PSA over time is called PSA velocity.13,14 In general, a man's prostate gland gradually increases in size due to benign causes after the age of 50, with a corresponding increase in PSA values. The normal yearly increase after the age of 50 years generally has been estimated to be less than 0.75 ng/mL per year. Thus, even if the PSA value is less than "normal" (i.e., less than 4 ng/mL) a change of greater than 0.75 ng/mL from one year to the next in a patient undergoing routine screening may raise concern and could prompt a biopsy.
A recent study evaluated the outcomes of patients whose PSA increased by greater than 2 ng/mL in the year before prostate cancer was diagnosed. The study included approximately 1095 men who were part of a prospective screening program, who were diagnosed with T1c or T2a cancers and underwent radical prostatectomy. (See Table 1 for definitions of prostate cancer staging.) Of those with PSA increases of more than 2 ng/mL, a 2.2-fold increase in death from any cause and a 20-fold increase in death from prostate cancer was noted. Thus, even among a screened population that undergoes treatment, the kinetics of PSA change over time provides great insight into the biological behavior of the underlying prostate cancer.15 (In Part II of this series on prostate cancer, the doubling time of PSA in patients already treated for prostate cancer will be discussed, and will reiterate the importance of PSA changes over time as a surveillance measure.)
| Table 1. Glossary of Terms |
|
|
Age-Specific Reference Ranges for PSA. The concept of age-specific reference ranges for determining "normal" vs. "abnormal" PSA stems from the fact that the normal range may be higher for men as they age. Thus, the potential of using this measurement could be to avoid biopsy in men based solely on the absolute PSA value, but rather to evaluate the value based upon age considerations. By changing the criteria for biopsy in older men, one may perform fewer unnecessary prostate biopsies, but also may miss more potential cancers. Despite the initial enthusiasm for age-specific reference ranges, there is limited evidence to support the routine use of age-specific reference ranges for prostate cancer screening, especially given the more recent data indicating that even extremely low values of PSA with normal DRE can be associated with prostate cancer.11
What PSA Values Should Trigger a Prostate Biopsy?
The approach for recommending a prostate biopsy based upon an absolute level of PSA continually is undergoing re-analysis. As mentioned above, the largest randomized study of prostate cancer screening in Europe now is utilizing a PSA value of greater than 3 ng/mL to recommend a prostate biopsy. However, information is emerging from the Prostate Cancer Prevention Trial (PCPT), which included nearly 3000 men older than 55 years who were followed for seven years with a PSA of less than 4 and a normal DRE, and who underwent biopsy. The results showed that as the PSA values went from 0-0.5 ng/mL, 0.6-1, 1-2, 2-3, and 3-4, respectively, the likelihood of finding prostate cancer went from 6.6% to 10.1% to 17% to 23.9% and 26%, respectively. Even more significantly, the percentage of significant cancer, as defined by a Gleason score of 7 or more, increased from 12.5% to 25% in these four subgroups. (See Table 2 for an explanation of Gleason score.) These findings have placed into legitimate question what the trigger point is to consider a prostate biopsy.11,16 In general practice, however, a referral to a urologist from a primary care physician could be considered for any PSA above 3-4 ng/mL in a man older than 55 years, even if the DRE is non-suspicious. However, as now recommended by many national health organizations that set screening policy, the decision to have a PSA test in the first place should be a shared decision between health care provider and patient. Such an approach should include a full discussion of the pros and cons of screening before the test is undertaken.17
| Table 2. Gleason Score Definition |
|
|
Several organizations recently have provided guidance for prostate screening recommendations and are available on the respective web sites. Among them are the following:
American Urological Association. The American Urological Association states that early detection of prostate cancer should be individualized after discussion of the benefits and consequences of treatment.
This is a very major change in policy, issued since the publication of the USPSTF in December 2002. Previously, the AUA recommended screening with a PSA test and DRE for all men older than 50 years.
The American Medical Association. The American Medical Association states that screening programs for the early detection of prostate cancer are premature. However, high-risk groups that may benefit from early detection may include African American men older than 40 years of age with a first-degree relative with prostate cancer.
The American Cancer Society. The PSA test and the DRE should be offered annually beginning at age 50 to men who have a life expectancy of at least 10 years. Men at high risk should begin testing at age 45. Information should be provided to patients about benefits and limitations of testing. Specifically, prior to testing, men should have an opportunity to learn about the benefits and limitations of testing for early prostate cancer detection and treatment.
American Academy of Family Physicians. The American Academy of Family Physicians recommends that physicians counsel men between the ages of 50 and 65 years regarding the known risks and uncertain benefits of PSA screening. All men should be informed of the known harms and uncertain benefits of screening and should make an informed choice, rather than routinely undergoing PSA testing.
U.S. Preventive Service Task Force. The USPSTF found good evidence that PSA screening can detect early stage prostate cancer, but mixed and inconclusive evidence that early detection improves health outcomes. Screening is associated with important harms, including frequent false-positive results and unnecessary anxiety, biopsies, and potential complications of treatment of some cancers that may never have affected a patient's health. The USPSTF concludes that evidence is insufficient to determine whether the benefits outweigh the harms for a screened population.
The American College of Physicians. Recommendation 1: Rather than screening all men for prostate cancer as a matter of routine, physicians should describe the potential benefits and known harms of screening, diagnosis, and treatment; listen to the patient's concerns; and then individualize the decision to screen.
Recommendation 2: The College strongly recommends that physicians help enroll eligible men in ongoing clinical studies.
This policy about discussing the issues first, then individualizing the screening decision has been adopted by several public health policy advisory groups. A summation of the data suggests that screening may lead to earlier detection of potentially curable prostate cancer. However, not all cancers necessarily have to be treated, and treatment carries with it potential morbidities and mortality.
Additional help in determining the need for such a referral could be the PSA velocity, mentioned above, and consideration for performing one of the tests to determine PSA isoforms, thus assessing complexed or free PSA (see below).
What Are the Most Useful Isoforms of PSA?
PSA, when secreted into the systemic circulation, is found in both a bound form (i.e., circulates bound to various plasma proteins) and a free form unattached to circulating carrier proteins. In general, most studies have shown that a free PSA (fPSA) value that represents less than 15% of total PSA (t PSA) (when the tPSA is between 4 and 10 ng/mL) is associated with cancer in nearly two-thirds of cases if a prostate biopsy is obtained. When sending off a PSA level, the routine value reflects the total PSA, which in effect is the sum of the free PSA and the complexed (or bound) PSA. When the level of free PSA is greater than 25%, especially for total PSA values between 4 and 10 ng/mL, benign enlargement of the prostate gland is likely to be the explanation for the elevated PSA.9,18 In the next few years, additional isoforms of PSA such as pro-PSA may be introduced as potential markers of prostate cancer.
What Is the Optimal Interval for Screening?
Several important studies have looked at the optimal interval for re-screening men for prostate cancer detection. In great part, the initial PSA value obtained during the first screening test may help determine the frequency of subsequent testing. A recent study of more than 27,863 male subjects ages 55-74 years showed that if the initial screened PSA was less than 1 ng/mL, 98.6% of men would continue to have a PSA of less than 4 ng/mL during the next 4 years of screening; and 98.8% with an initial PSA of 1-2 ng/mL would continue to be negative during the next subsequent year of screening. Both categories were independent of the DRE findings. A recommendation emanating from this study is to screen patients with an initial PSA of less than 1 ng/mL every 5 years, and those with PSA of 1 to 2 ng/mL every 2 years; those with more than 2 ng/mL should be screened every year. This would reduce the PSA tests by 55% and only minimally result in a delay in detecting an elevated PSA test.19 Most studies that have evaluated screening with PSA testing, with or without DRE, have done so in men older than 50 years, and have repeated evaluations annually. Many ongoing studies are attempting to ascertain whether an increased screening interval may be recommended without leading to a significant worsening of outcomes.20
What Extraneous Factors Can Change the PSA Value?
Although elevations of PSA may occur after a variety of circumstances, there are several worth noting, especially given that men now are being biopsied with lower and lower PSA values than in the past. Ejaculation, perineal pressure during physical exercise (e.g., bicycle riding), and the performance of a DRE transiently may increase the PSA level, leading to false-positive screening results. In one study of 20 volunteers (mean age 59 years) total and free serum PSA levels obtained 1, 6, and 24 hours after ejaculation were measured. The proportion of men with PSA levels greater than the expected biologic variability at each time point then was assessed. Total PSA levels remained significantly increased 6 and 24 hours after ejaculation. Forty percent at 24 hours after ejaculation had total PSA levels above the baseline level, suggesting that PSA testing within 24 hours after ejaculation may lead to erroneous elevations.21
Whether the DRE alters PSA values was evaluated in 93 men following the examination. Increases in PSA within 1 hour of DRE occurred and could remain elevated for approximately one week.22 Other studies have shown that PSA is increased insignificantly following a DRE if the PSA is 0.1 to 4 ng/mL or 4.1–10 ng/mL. However, as the initial PSA increases from 10 to 20 ng/mL, or greater than 20 ng/mL, the effect of DRE did lead to increases that approached statistical significance, and was statistically significant in the latter group.23 A recent study24 further has evaluated the differential response of free vs. complexed PSA following DRE and found that most of the immediate increase is due to the increase of free PSA. The bound portion of PSA was less sensitive to DRE, and its changes after DRE were clinically modest. These authors recommend that free PSA measurement be performed before DRE, and the percentage of free PSA should be used when the value is obtained before the DRE. Thus, the practical point is that a PSA is best obtained before the performance of a DRE. Again, such a practice may lead to fewer elevations in the normal range, especially if men are being considered for biopsy at lower and lower cutoff values.
What Are the Risks of Prostate Biopsy?
Since performing a prostate biopsy is likely to be one of the most invasive tests to result for a patient undergoing PSA screening, the spectrum of risks and complications should be appreciated by the primary care physician. While most primary care physicians may not actively manage complications of prostate biopsy, they should be active participants in helping patients understand potential complications, especially in those men with co-morbidities.
A prostate biopsy usually is performed under transrectal ultrasound guidance via the transrectal approach. Other methods include transperineal biopsy or transurethral biopsy, as performed during a transurethral resection of the prostate for BPH. In general, the potential harms associated with prostate biopsy include infection (both localized infections including prostatitis and systemic infections of the urinary tract and sepsis), bleeding, pain, urinary retention, and hematospermia. The primary care physician should be aware that the use of prophylactic antibiotics, peri-prostatic local anesthesia, and enemas sometimes may be prescribed. Anxiety also may occur in men who have a false-positive screening test. (See Table 3.)
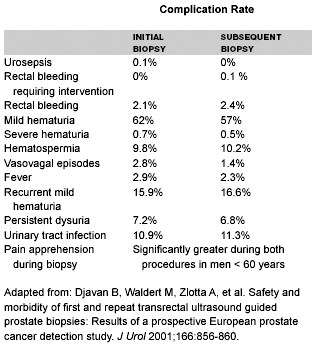
| Table 3. Comparative Rates of Biopsy-Related Complications in Subjects Undergoing an Initial and then Subsequent Prostate Biopsy |
|
|
General Complications. In one large European study of nearly 5802 men undergoing screening for the early detection of prostate cancer, the following complications after prostate biopsy were recorded (all received prophylactic antibiotics): hematuria lasting longer than 3 days, 22.6%; hematospermia, 50.4%; fever, 3.5%; urinary retention, 0.4%; hospitalization for prostatitis and/or urosepsis, 0.5%. A prior history of prostatitis was significantly associated with hospital admission and pain after biopsy. A history of prostatic hyperplasia and a higher International Prostate Symptom Score were predictors of urinary retention.25
Another study of 200 men (100 screened, 100 hospitalized) who underwent prostate biopsy demonstrated that there were similar complications regardless of whether the patients were evaluated as part of a screened population or whether they were referred for prostate biopsy with signs or symptoms suggestive of prostate cancer as hospitalized patients. The most frequent complications were pain at biopsy occurring in 52% and 63% of screened and hospital-based men, respectively; and persistent rectal bleeding and hematuria, occurring in 13% to 35%, respectively.26
Complications of Prostate Biopsy in Men with History of Prior Urinary Tract Infection. This clinical situation is of particular interest to the primary care physician. In one study of 2258 prostate biopsies performed between 1980 and 1997 in Olmsted County, MN, 377 (17%) were associated with at least one complication. A urogenital infection six weeks before biopsy was associated with an increased risk of a post-biopsy complication (odds ratio = 1.7, 95% confidence interval = 1.0 to 2.8) and an increased risk of a post-biopsy infection (odds ratio = 5.5, 95% confidence interval = 2.2 to 13.8). It should be appreciated that while the absolute post-biopsy complications in the community have increased tremendously because of the increased use of prostate biopsies, the complication rate per biopsy has stayed constant.27
Use of Antibiotics. The rate of urinary tract infections may be decreased if prophylactic antibiotics (e.g., with ciprofloxacin and metronidazole) are used. In one study of 289 patients undergoing core biopsy of the prostate gland, 5 (1.7%) developed a urinary tract infection, which was lower when compared to historical controls.28 Another study stratified and treated 400 men with a single dose of 500 mg of levofloxacin prior to prostate biopsy if they were considered to be at a low risk for infection post-biopsy; and an additional two days of single dose levofloxacin were administered if they were deemed at high risk. (High risk was defined as large prostate gland more than 75 cc, diabetes mellitus, recent steroid use, severe voiding dysfunction, or immune compromise.) No enemas were used. One of the 377 low-risk patients experienced a symptomatic urinary tract infection (0.27%). None of the 23 men at high risk who received additional doses of levofloxacin experienced a complication, for an overall infection rate of 1 out of 400 cases (0.25%).29
Use of Anesthesia. A randomized prospective study of periprostatic local anesthesia prior to TRUS in 100 men with a sterile urine culture demonstrated that rectal bleeding was significantly less in the patients who received anesthesia. Fever (greater than 37.8°C) was more frequent in the nerve block group, with two patients requiring rehospitalization. Bacteriuria in post-biopsy urine cultures was significantly more common in the anesthesia group. Urethral bleeding was similar in both groups.30
A second study of two groups of 100 patients each, one of which underwent a single injection of local anesthesia (10 mL of lidocaine) at the time of prostate biopsy and one that did not, showed that the level of pain in the group that did not receive anesthesia was greater with no additional complications.31
Use of Enemas. In one retrospective analysis of the utility of pre-prostate biopsy enemas, 225 patients who received enemas before biopsy were compared to 185 who did not. There was no difference in overall complication rate between the two groups. In this retrospective evaluation, all patients received identical antibiotic prophylaxis with ciprofloxacin.32
Complications in Immunosuppressed Patients. A study of 59 men receiving immunosuppressive agents after kidney transplantation with PSA values between 4 and 10 ng/mL underwent TRUS-guided sextant biopsy plus additional biopsies from the central or transitional portion of the gland. No differences were noted in pain apprehension or early and delayed morbidity (pain, rectal bleeding, hematuria, vasovagal episodes, fever, hematospermia, dysuria, urinary tract infections, and urospesis) compared to a control population studied from the European Prostate Cancer Detection Study, suggesting that TRUS-guided biopsies can be performed safely in these immunosuppressed patients.33
What Type of Cancers Are Detected with Screening Programs?
One important factor in considering a screening recommendation relates to the type of cancers that are detected as a result of screening. In general, the prostate cancers that are detected from screening programs are less likely to be metastatic; more likely to have a more favorable Gleason pathology score; and less likely to be treated with hormonal therapy compared to controls. If these screened cancers are treated with radical prostatectomy, they are more likely to be organ-confined and less likely to have nodal metastases compared to cancers detected clinically. In addition, if the cutoff point for performing a biopsy is lowered from a PSA of 4.0 ng/mL to values of 2-3 ng/mL, even more favorable pathological features result. The data supporting these pathological outcomes are derived from the European Randomized Study for Screening of Prostate Cancer. The ERSPC (Rottedam section) evaluated the clinical characteristics of screen-detected and clinically diagnosed prostate cancer in men aged 55 to 74. Out of 35,149 patients, 17,636 were randomized to be screened and 17,513 served in the control arm. Cancers detected by screening were less likely to be metastatic (0.6% vs. 6.7%); more likely to have a more favorable Gleason pathology score (84.1% with a Gleason of 5-7 vs. 69.8%); and less likely to be treated with hormonal therapy (2% vs. 17.4%).34
Another study of 845 screened patients who underwent radical prostatectomy correlated the rate of positive margins with preoperative serum PSA values and showed a significantly higher rate of organ-confined prostate cancers and a significantly lower rate of positive surgical margins in patients with preoperative total PSA values of less than 4 ng/mL compared with patients with higher preoperative total PSA levels.35
However, these data do not indicate that screening reduces mortality. It is hoped that with additional follow-up, these more favorable cancers that are detected by screening eventually will lead to survival benefits.
Results of Both Randomized and Non-randomized Studies for Prostate Cancer Screening and Results from Natural History Studies
At the current time, there is no randomized controlled trial evidence that screening asymptomatic patients for prostate cancer leads to a reduction in overall mortality, and those communities that have practiced more intensive screening for early detection of prostate cancer have not experienced an improvement in prostate cancer specific mortality compared to less intensively screened communities. The results from ongoing RCTs will not be known until 2006-2007 at the earliest. The ERSPC trial, which has now accrued 163,126 men between the ages of 55-69, has sufficient power to detect a significant difference in prostate cancer mortality between the two arms (screened vs. non-screened) if the true reduction in mortality by screening is 25% or more.1
A comparison of rates of performing PSA screening, radical prostatectomy, and radiation therapy was undertaken in two areas (Seattle and Connecticut) to assess differences in prostate cancer specific mortality. More intensive screening rates and subsequent treatments, as practiced in Seattle, were not associated with lower rates of prostate cancer specific mortality over 11 years of follow-up.36
The American PLCO trial will randomize nearly 75,000 patients, aged 60 to 74, and compare annual screening with DRE and PSA yearly for four years with "usual" care. The ERSPC trial will randomize nearly 200,000 male patients between 50 and 75 years with DRE, PSA, and transrectal ultrasound to "usual" care. A smaller study (Prostate Cancer Intervention Versus Observation Trial: The PIVOT Study) conducted in the VA system will randomize 1000-2000 men to either radical prostatectomy vs. observation, with the primary endpoint being all-cause mortality.37
In a retrospective cohort study conducted utilizing data from the Connecticut Tumor Registry, 767 patients diagnosed with localized prostate cancer between 1971 and 1984 were either not treated or treated with immediate or delayed hormonal therapy. Patients whose Gleason scores were between 2 and 4 had a lower chance (4-7%) of dying from prostate cancer within 15 years of diagnosis than those patients with more aggressive Gleason scores.38
A retrospective study of 828 men treated conservatively (with observation and delayed hormone therapy but no radical surgery or irradiation) for clinically localized prostate cancer showed that 10 years after diagnosis, disease-specific survival was 87% for grade 1 or 2 tumors, and 34% for grade 3 tumors, and metastasis-free survival was 81%, 58%, and 26 % for grade 1, 2, and 3 disease, respectively.39
A Swedish prospective cohort study of 642 patients with prostate cancer of any stage consecutively diagnosed between 1977 and 1984 demonstrated that the corrected 15-year survival rate for untreated localized prostate cancer was 81%, and did not differ from those who had received specific treatment. Survival rates were substantially lower for those with locally advanced or metastatic disease (57% and 6%, respectively).40
Several studies that evaluated comparative mortality rates among patients who were offered PSA screening (compared to a population where PSA screening was not offered routinely) found that those offered screening had relatively lower death rates.41,42 Although the attribution of decreased mortality rates due to PSA-based screening could not be made with certainty, the trend of detecting earlier stages of cancer and the associated more favorable shorter-term survival may, in the future, be correlated with larger patient populations undergoing screening.
For men with a life expectancy of at least 10 years, the physician should discuss the potential benefits of being screened for prostate cancer as well as the potential harms associated with treatment for prostate cancer, if detected. A detailed review of the potential adverse events associated with the treatment of localized prostate cancer will be fully discussed in Part II of this series. The main side effects include incontinence and impotence for those undergoing radical prostatectomy, and radiation proctitis and urinary irritative symptoms in those undergoing radiation therapy. Bowel disturbances, including fecal incontinence, may occur with treatments for localized prostate cancer.
Special Considerations
Populations at Higher Risk of Developing Prostate Cancer. African American Men. African American men have an increased incidence and risk of developing of prostate cancer; it is uncertain whether cancers detected in African Americans are associated with more unfavorable outcomes compared to non-African Americans, even though some studies have shown that these cancers may be associated with more unfavorable histological characteristics. African American men have a 1.6-fold higher likelihood of getting prostate cancer compared to white men, and are more than twice as likely to die from the disease. African American men with prostate cancer between the ages of 50 and 59 have higher PSA levels, more advanced Gleason scores, higher stage of disease, and greater likelihood of recurrence. Even after adjusting for socio-economic status, the Prostate Cancer Outcomes Study showed that more advanced stage for African American men persisted.43 Most authorities who do recommend screening suggest that screening for African American men begin at an earlier age (perhaps 40 to 45 years of age) than for average-risk men.
Hereditary Prostate Cancer. Men with a family history of prostate cancer (at least one first-degree relative affected) are at greater risk to develop prostate cancer than men in the general population and may be considered for screening before the age of 50. There are no appreciable differences in the clinical and biological behavior of familial prostate cancer compared to sporadic prostate cancer. The risk is further increased if multiple first-degree relatives are affected.
African American men with a family history have an increased risk of developing prostate cancer. A prospective cohort study compared the results of screening in 1224 black men, 1227 men with a family history of prostate cancer, and 63 black men with a family history (the latter three represent the high-risk population), to 15,964 non-black men with no family history of prostate cancer. Among the high-risk men in their 40s, 8% had suspicious screening tests and approximately 55% who underwent a biopsy had cancer detected. Of tumors detected, 80% were organ-confined. In men 50 years old or older, prostate cancer detection rates were 6.4% for controls compared with 10.3% (black), 10.5% (positive family history), and 17.5% (both black and positive family history), respectively, for the high-risk groups.44,45
What Are the Take-Home Points in Dealing with the Issue of Screening for Prostate Cancer?
Concept of "Normal" PSA. The definition of a cutoff point of PSA level that helps determine the need for further evaluation and prostate biopsy is undergoing re-evaluation. There is a continuum of risk for prostate cancer based upon PSA values, and that newer thinking suggests that there is no absolute PSA value that should trigger further evaluations. The use of PSA isoforms may provide greater accuracy in the future.
Outcomes of Screening Studies. There is limited evidence of a demonstrated survival advantage conferred by screening, and the results of large screening trials in the United States and Europe are pending. Detecting and treating prostate cancer through PSA-based evaluations carries with it known harms.
Public Health Policy Recommendations. Recognize that most large public policy organizations or specialty medical societies that provide guidance of screening policies recommend that the physician and patient enter into a frank discussion regarding the known harms of prostate cancer treatment and the uncertainty of benefits before giving consent to screening, diagnosis, and treatment. As stated earlier, a systematic review of the positions of many professional societies and health technology assessment agencies reveals that the majority of these organizations do not recommend formal screening programs because there is a lack of definitive evidence for benefit.
References
1. de Koning HJ, Liem MK, Baan CA, et al; ERSPC. Prostate cancer mortality reduction by screening: Power and time frame with complete enrollment in the European Randomised Screening for Prostate Cancer (ERSPC) trial. Int J Cancer 2002;98:268-273.
2. Prorok PC, Andriole GL, Bresalier RS, et al; Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial Project Team. Design of the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial. Control Clin Trials 2000;21(6 Suppl):273S-309S.
3. Schroder FH, Roobol-Bouts M, Vis AN, et al. Prostate-specific antigen-based early detection of prostate cancervalidation of screening without rectal examination. Urology 2001;57:83-90.
4. Catalona WJ, Smith DS, Ratliff TJ, et al. Measurement of prostate specific antigen in serum as a screening test for prostate cancer. N Engl J Med 1991:234;1156.
5. Schroder FH, van der Maas P, Beemsterboer P, et al. Evaluation of the digital rectal examination as a screening test for prostate cancer. Rotterdam section of the European Randomized Study of Screening for Prostate Cancer. J Natl Cancer Inst 1998;90:1817-1823.
6. Vis AN, Hoedemaeker RF, Roobol M, et al. Tumor characteristics in screening for prostate cancer with and without rectal examination as an initial screening test at low PSA (0.0-3.9 ng/ml). Prostate 2001;47:252-261.
7. Beemsterboer PM, Kranse R, de Koning HJ, et al. Changing role of 3 screening modalities in the European randomized study of screening for prostate cancer (Rotterdam). Int J Cancer 1999;84:437-441.
8. Schroder FH, Roobol-Bouts M, Vis AN, et al. Prostate-specific antigen-based early detection of prostate cancervalidation of screening without rectal examination. Urology 2001;57:83-90.
9. Catalona WJ, Partin AW, Slawin KM, et al. Use of the percentage of free prostate specific antigen to enhance differentiation of prostate cancer from benign prostatic disease: A prospective multicenter clinical trial. JAMA 1998;279;1542-1547.
10. Punglia RS, D'Amico AV, Catalona WJ, et al. Effect of verification bias on screening for prostate cancer by measurement of prostate-specific antigen. N Engl J Med 2003;349:335-342.
11. Thompson IM, Pauler DK, Goodman PJ, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level < or =4.0 ng per milliliter. N Engl J Med 2004;350:2239-2246.
12. Schroder FH, van der Cruijsen-Koeter I, de Koning HJ, et al. Prostate cancer detection at low prostate specific antigen. J Urol 2000;163:806-812.
13. Ciatto S, Bonardi R, Lombardi C, et al. Analysis of PSA velocity in 1666 healthy subjects undergoing total PSA determination at two consecutive screening rounds. Int J Biol Markers 2002;17:79-83.
14. Fang J, Metter EJ, Landis P, et al. PSA velocity for assessing prostate cancer risk in men with PSA levels between 2.0 and 4.0 ng/ml. Urology 2002;59:889-893.
15. D'Amico AV, Chen MH, Roehl KA, et al. Preoperative PSA velocity and the risk of death from prostate cancer after radical prostatectomy. N Engl J Med 2004;351:125-135.
16. Makinen T, Tammela TL, Hakama M, et al. Prostate cancer screening within a prostate specific antigen range of 3 to 3.9 ng./ml.: A comparison of digital rectal examination and free prostate specific antigen as supplemental screening tests. J Urol 2001;166:1339-1342.
17. Chan EC, Vernon SW, O'Donnell FT, et al. Informed consent for cancer screening with prostate-specific antigen: how well are men getting the message? Am J Public Health 2003;93:779-785.
18. Djavan B, Remzi M, Zlotta AR, et al. Complexed prostate-specific antigen, complexed prostate-specific antigen density of total and transition zone, complexed/total prostate-specific antigen ratio, free-to-total prostate-specific antigen ratio, density of total and transition zone prostate-specific antigen: Results of the prospective multicenter European trial. Urology 2002;60(4 Suppl 1):4-9.
19. Crawford D, Chia D, Andriole GL, et al. PSA testing interval, reduction in screening intervals: Data from the prostate, lung, colorectal and ovarian cancer (PLCO) screening trial. J Urol 2002:167;99 suppl (abstract 397).
20. Grubb RL, Roehl KA, Antenor JV, et al. Delays in cancer detection using 2 and 4 year screening intervals for prostate cancer screening in men with initial PSA less than 2 ng/ml. J Urol 2005:173;1116-1120.
21. Herschman JD, Smith DS, Catalona WJ. Effect of ejaculation on serum total and free prostate-specific antigen concentrations. Urology 1997;50:239-243.
22. Ornstein DK, Rao GS, Smith DS, et al. Effect of digital rectal examination and needle biopsy on serum total and percentage of free prostate specific antigen levels. J Urol 1997;157:195-198.
23. Crawford ED, Schutz MJ, Clejan S, et al.The effect of digital rectal examination on prostate-specific antigen levels. JAMA 1992;267:2227-2228.
24. Lechevallier E, Eghazarian C, Ortega JC, et al. Effect of digital rectal examination on serum complexed and free prostate-specific antigen and percentage of free prostate-specific antigen. Urology 1999;54:857-861.
25. Raaijmakers R, Kirkels WJ, Roobol MJ, et al. Complication rates and risk factors of 5802 transrectal ultrasound-guided sextant biopsies of the prostate within a population-based screening program. Urology 2002;60:826-830.
26. Mkinen T, Auvinen A, Hakama M, et al. Acceptability and complications of prostate biopsy in population-based PSA screening versus routine clinical practice: A prospective, controlled study. Urology 2002;60:846-850.
27. Roberts RO, Bergstralh EJ, Besse JA, et al. Trends and risk factors for prostate biopsy complications in the pre-PSA and PSA eras, 1980 to 1997. Urology 2002;59:79-84.
28. Larsson P, Norming U, Tornblom M, et al. Antibiotic prophylaxis for prostate biopsy: Benefits and costs. Prostate Cancer Prostatic Dis 1999;2:88-90.
29. Griffith BC, Morey AF, Ali-Khan MM, et al. Single dose levofloxacin prophylaxis for prostate biopsy in patients at low risk. J Urol 2002;168:1021-1023.
30. Obek C, Onal B, Ozkan B, et al. Is periprostatic local anesthesia for transrectal ultrasound guided prostate biopsy associated with increased infectious or hemorrhagic complications? A prospective randomized trial. J Urol 2002;168:558-561.
31. Taverna G, Maffezzini M, Benetti A, et al. A single injection of lidocaine as local anesthesia for ultrasound guided needle biopsy of the prostate. Urol 2002;167:222-223.
32. Carey JM, Korman HJ. Transrectal ultrasound guided biopsy of the prostate. Do enemas decrease clinically significant complications? J Urol 2001;166:82-85.
33. Wammack R, Djavan B, Remzi M, et al. Morbidity of transrectal ultrasound-guided prostate needle biopsy in patients receiving immunosuppression. Urology 2001;58:1004-1007.
34. Van der cruijsen-Koeter IW, Roobol MJ, Damhuis RA, et al. Comparison of screen detected and clinically diagnosed prostate cancer in the ‘European randomized study of screening for prostate cancer'. J Urol 2002:167;102 suppl (abstract 407).
35. Berger AP, Volgger H, Rogatsch H, et al. Screening with low PSA cutoff values results in low rates of positive surgical margins in radical prostatectomy specimens. Prostate 2002; 53:241-245.
36. Lu-Yao G, Albertsen PC, Stanford JL, et al. Natural experiment examining impact of aggressive screening and treatment on prostate cancer mortality in two fixed cohorts from Seattle area and Connecticut. BMJ 2002;325:740.
37. Wilt TJ, Brawer MK. The Prostate Cancer Intervention Versus Observation Trial: A randomized trial comparing radical prostatectomy versus expectant management for the treatment of clinically localized prostate cancer. Urol 1994;152(5 Pt 2):1910-1914.
38. Albertsen PC, Hanley JA, Fine J. 20-year outcomes following conservative management of clinically localized prostate cancer. JAMA 2005;293:2095-2101.
39. Chodak GW, Thisted RA, Gerber GS, et al. Results of conservative management of clinically localized prostate cancer. N Engl J Med 1994;330:242-248.
40. Johansson JE, Andren O, Andersson SO, et al. Natural history of early, localized prostate cancer. JAMA 2004;291:2713-2719.
41. Horninger W, Berger A, Pelzer A, et al. Screening for prostate cancer: Updated experience from the Tyrol study. Can J Urol 2005;12 Suppl 1:7-13.
42. Roberts RO, Bergstralh EJ, Katusic SK, et al. Decline in prostate cancer mortality from 1980 to 1997, and an update on incidence trends in Olmsted County, Minnesota. J Urol 1999;161:529-533.
43. Baquet CR, Horm JW, Gibbs T, et al. Socioeconomic factors and cancer incidence among blacks and whites. J Natl Cancer Inst 1991;83:526-527.
44. Catalona WJ, Antenor JA, Roehl KA, et al. Screening for prostate cancer in high risk populations. J Urol 2002;168:1980-1983.
45. Bova GS, Partin AW, Isaacs SD, et al. Biological aggressiveness of hereditary prostate cancer: Long-term evaluation following radical prostatectomy. J Urol 1998;160(3 Pt 1):660-663.
46. Djavan B, Waldert M, Zlotta A, et al. Safety and morbidity of first and repeat transrectal ultrasound guided prostate needle biopsies: Results of a prospective European prostate cancer detection study. J Urol 2001;166:856-860.
Prostate cancer is one of the leading causes of cancer in men, both in this country and in Europe. Due to the introduction of the prostate specific antigen blood test (PSA test), the ability to diagnose prostate cancer well before signs or symptoms of the disease develop has been realized. Following its introduction, the PSA was endorsed widely and recommended by many major public health organizations as a useful screening test.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.