Management of the Sexually Active Adolescent in the ED: Vaginitis, STDs, PID, and Emergency Contraception
Management of the Sexually Active Adolescent in the ED: Vaginitis, STDs, PID, and Emergency Contraception
Authors: N. Ewen Wang, MD, Assistant Professor Surgery, Assistant Professor Pediatrics, Associate Director Pediatric Emergency Medicine, Stanford University, Palo Alto, CA; Alyssa K. Hamman MD, Resident in Anesthesia, Stanford University, Palo Alto, CA; Sangeeta K. Chona, MD; Attending Physician, Emergency Services, Clinical Assistant Professor, Department of Surgery, Division of Emergency Medicine, Stanford Hospital and Clinics, Palo Alto, CA.
Peer Reviewer: Angelo P. Giardino, MD, PhD, Clinical Associate Professor, Baylor College of Medicine, Houston; Medical Director, Texas Children's Health Plan, Houston, TX.
Acute care physicians play a very important role in preventing and managing sexually transmitted diseases (STDs). Frequently, the chief complaint is not straightforward, and optimal management may be challenging. The early identification and treatment of an STD may minimize complications unique to the adolescent population or prevent progression to a more serious disease. The authors provide a thorough review of STDs and related issues common to the adolescent population.
—The Editor
Introduction
Sexually active adolescents are commonly seen and evaluated in the emergency department (ED), although they are often not straightforward about the reason for their visit. It is vital for emergency physicians to inquire about sexual activity and specifically address signs and symptoms of STDs and pelvic inflammatory disease (PID), especially given the serious sequelae of these disease processes. This article is not meant to review all STDs, but instead focuses on issues commonly affecting adolescent patients.
We will discuss the diagnosis and treatment of several common sexually transmitted infections and vaginitis; the diagnosis of PID and management issues; medical management of possible human immunodeficiency virus (HIV) exposure and the basics of emergency contraception (EC). Finally, we briefly will discuss legal and reporting issues in caring for these patients in the ED setting.
Significance
STDs are common in adolescents in the United States, and female adolescents are especially affected. There were 18.9 million new cases of STDs in the year 2000; 48% of those infections occurred in persons aged 15-24 years, and a disproportionately high number of these were found in females.1 The Centers of Disease Control and Prevention (CDC) estimates that more than 1 in 10 sexually active female adolescents have chlamydial infections, and that females aged 15-24 years have the highest rates of gonorrhea.1 Female adolescents may face not only numerous obstacles to obtaining contraception and medical care, but they also are at risk for a number of serious sequelae from contracting STDs (e.g., infertility, ectopic pregnancy, chronic pelvic pain, and vertical transmission to newborns).1 STDs also are associated with increased HIV transmission,2,3 and many persons acquire HPV during their adolescent years.4
Unique Characteristics of the Adolescent
Adolescents are at higher risk for STDs secondary to the increased frequency of unprotected intercourse, limited duration of sexual partnership and specific biological issues that increase their vulnerability. The ectropion, or transformational zone at the border of the squamocolumnar junction regresses into the endocervical canal in adult women. In adolescents, the transformational zone is on the exocervix and exposed to the vaginal environment. It is hypothesized that this exposed transformational zone places adolescents at higher risk for primary infection by sexually transmitted pathogens and complications such as PID.2 Because adolescents have a high percentage of anovulatory cycles, especially at the beginning of menarche, they have a relative progesterone deficiency, which causes thinning of the cervical mucous. Because the cervical mucous acts as a defense against many STD pathogens, lack of this defense mechanism also increases the risk of PID.5,6 The emergency physician should always have a high index of suspicion for STDs and a low thres-hold for initiating empiric treatment.7
Considerations for the Pelvic Exam
A comprehensive evaluation of abdominal pain or possible STD in the ED may be the patient's first experience with a pelvic exam. Special considerations for the adolescent patient are important to make her experience as nonthreatening and comfortable as possible and to prevent the avoidance of reproductive care in the future.8 Start by explaining the rationale for the exam as well as the procedure and sequence of the pelvic exam. Be sure to choose the smallest speculum available and warm it with water before introducing it. A water-based lubricant also can be used, as long as a Pap smear is not being obtained. A Pederson or pediatric Graves speculum is usually tolerable for most adolescents. Cervical swabs can be collected for gonorrhea and chlamydia testing, which is often more sensitive than urine samples. Following the speculum exam, the bimanual exam should be performed with careful attention to cervical motion tenderness and adnexal masses. The technique of inserting only one finger is usually adequate and more comfortable for a teenager. Throughout the exam it is essential to explain to her what is being done, and to be gentle and deliberate. An adolescent who feels unduly traumatized by the ED pelvic exam may be deterred from obtaining health care in the future, putting her at risk for cervical cancer and untreated PID.
Like adults, adolescents who refuse to be examined for whatever reason cannot be forced to undergo a pelvic exam. The best approach is to explore the reason for refusal and address those concerns. Consultation with the gynecology service often can be helpful for a difficult examination. If an exam is painful, procedural sedation may be necessary.
Legal and Reporting Issues
The medical care of sexually active adolescents is complicated by their independent actions and volition in conjunction with their status as a minor. Emotions, beliefs about morality, the role of parental involvement, and ultimately, varying legal statutes all can affect management of the adolescent patient.
Patient confidentiality is a vital part of the adolescent ED visit. Adolescents often avoid seeking medical care because of fears of lack of confidentiality.9 Some adolescents may refuse testing for fear that their parents may find out.1 Adolescents may go alone to the ED in an attempt to obtain anonymous care. Assuring the patient of confidentiality is essential to encourage an honest history as well as appropriate follow-up care.
While adolescents younger than 18 years are considered to be minors and need parental consent for treatment, most states have minor consent laws that allow a physician to treat a minor without parental consent or notification for conditions involving treatment of STDs, contraception, pregnancy as well as for drug use and mental health.10 All states have statutes allowing minors to receive services related to STD testing and treatment without parental consent or involvement, but these statutes vary in detail among states.1,11 Some states explicitly allow an adolescent to be HIV tested without parental notification.12 An excellent resource is the Web site of the Allan Guttmacher Institute and the Center for Adolescent Health and the Law (http://www.guttmacher.org/index.html). The institute regularly monitors state laws governing access to confidential services and updates the information on its Web site.14 Twenty-one states explicitly allow teenagers to consent to contraceptive services, while many allow teenagers to consent to services under explicit circumstances. Again, a practitioner must know the specific laws of his/her state.
AIDS, gonorrhea, and syphilis are reportable diseases in every state, and chlamydia is reportable in most states.7 Trichomonas infection, genital herpes infection, and genital warts are not reportable diseases in adolescents or adults. Each state has slightly different reporting requirements, and the physician should be familiar with the reporting laws of his/her state. Adolescents should be informed about the need to report certain STDs to the public health department if a diagnostic test becomes positive.
Occasionally, a parent may request that his or her child be tested for an STD or pregnancy. If the adolescent is agreeable, then it is up to the ethical discretion of the physician. While familial involvement is ideal, if an adolescent does not consent, then he or she cannot be tested. The best procedure in this case, would be a confidential screening for symptoms and the development of a plan agreeable to the adolescent and family for follow-up care with a primary care physician or gynecologist.
As with all patients, adolescents should be screened for physical or sexual abuse. A thoughtful history with explicit screening for domestic violence and abuse—while always part of a thorough patient encounter—is especially vital for vulnerable populations such as adolescents. If a physician suspects abuse then he/she is legally required to report to the appropriate authorities. Often, the hospital social worker can be an invaluable resource in assessing the safety and appropriateness of an adolescent patient.
Vaginitis
Vaginal discharge, vulvar itching, and irritation are common complaints among women, especially those who are sexually active. The three most common types of infections associated with vaginal discharge in sexually active women are candidiasis, bacterial vaginosis (BV), and trichomoniasis. Along with these diagnoses, mechanical, chemical, allergic, or other noninfectious causes of vulvovaginal irritation should be kept in mind, especially if no vaginal pathogens can be isolated.14
Although each infection has general clinical characteristics, none are specific. Therefore, the cause of such infections must be determined by pH and microscopic examination of the discharge; pH paper can be used to detect an elevated pH (greater than 4.5), characteristic of BV or trichomoniasis, but not highly specific.4 Microscopic exam, "wet prep" or "wet mount," is done by collecting two small samples of discharge and placing each on a slide. The first is diluted with one to two drops of normal saline, and the second with one to two drops of 10% potassium hydroxide (KOH) solution. A cover slip is placed on the slides, and they are examined under the microscope. Findings specific to each disease are discussed in the following sections. See Table 1 for a summary of the most common causes of vaginitis and their characteristics.
| Table 1. Summary of Vulvovaginitis |
|
|
| Sources: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2006. MMWR 2006;55(No. RR-11):1-94. Zeger W, Holt K. Gynecologic infections. Emerg Med Clin North Am 2003;21(3):631-48. Sobel JD. What's new in bacterial vaginosis and trichomoniasis? Infect Dis Clin North Am 2005;19(2):387-406. Fredricks DN, Fiedler TL, Marrazzo JM. Molecular identification of bacteria associated with bacterial vaginosis. N Engl J Med 2005;353(18):1899-911. Schwebke JR. Trichomoniasis in adolescents: a marker for the lack of a public health response to the epidemic of sexually transmitted diseases in the United States. J Infect Dis 2005;192(12):2036-8. |
Vulvovaginal Candidiasis
Vulvovaginal candidal infections, or "yeast infections," are usually caused by Candida albicans, but occasionally are caused by other species of Candida or yeast. Patients typically present complaining of vulvovaginal pruritis, and white, "cottage cheese"-like discharge. Dyspareunia, external dysuria, and vaginal soreness also may be present.14,15 Vulvovaginal candidiasis is common, with an estimated 75% of women experiencing at least one episode in their lifetime, and 40%-45% experiencing two or more episodes.4 The risk of candidal vaginitis may be increased in women who are immunosuppressed, are using or have recently used corticosteroids or antibiotics, women with uncontrolled diabetes, or who are pregnant.14,15 Although vulvovaginal candidiasis is not transmitted sexually, it is more common in sexually active women. It is a common complaint in the ED, and often is diagnosed in adolescents being evaluated for STDs. Vulvovaginal candidiasis is currently divided into uncomplicated and complicated. Complicated is characterized by recurrence, severity, nonalbicans candidiasis or women with uncontrolled diabetes, debilitation, immunosuppression, or pregnancy.
An external genital exam should be performed first to evaluate the external genitalia, followed by a speculum exam. The vaginal mucosa and vulva are typically erythematous and edematous, and a thick, white discharge may be present. Occasionally extensive vulvar erythema, edema, excoriation, and fissure formation may be present. These findings suggest severe vulvovaginitis, and the patient should be treated with a longer course of therapy (7-14 days of topical azole or 150 mg of fluconazole in two sequential doses [second dose 72 hours after initial dose]) and referred to an obstetrician/gynecologist for follow-up care.4 Samples of the discharge should be collected during the speculum exam for microscopic evaluation. Findings consistent with candidiasis are a normal vaginal pH less than 4.5, and a wet prep with hyphae, pseudohyphae, or budding yeast. Treatment of the sample with a drop of 10% KOH solution improves the visualization of yeast and pseudohyphae by disrupting any cellular material obscuring the yeast.14 A Gram stain also can be done to evaluate for yeast.
Treatment consists of antifungal agents, usually administered topically, although an oral regimen does exist. The list of acceptable topical regimens is long and includes butoconazole, clotrimazole, miconazole, nystatin, tioconazole, and terconazole.4 Many are available as over-the-counter preparations. The topically applied azole drugs are more effective than nystatin.4 Short-course therapy, consisting of only 3 days, is usually sufficient.4 Oral fluconazole, given as a one-time dose of 150 mg, is also an accepted regimen. Monitoring liver functions is not necessary with a one-time dose of fluconazole. Only topical azoles, applied for 7 days, are recommended anytime during pregnancy. Follow-up care is not necessary unless the symptoms do not resolve after completing treatment or a recurrence of the same symptoms occurs within 2 months. As mentioned above, patients with severe vulvovaginal candidiasis should receive 7-14 days of topical azole or fluconazole 150 mg orally in two sequential doses (second dose 72 hours after first dose) as well as a referral to an obstetrician/gynecologist.4 Treatment of sex partners in not recommended because vulvovaginal candidiasis usually is not acquired through sexual activity, but may be considered in women who have recurrent infection.4 However, male sex partners may have balanitis, in which case treatment with topical antifungals is recommended.4
Bacterial Vaginosis
Bacterial vaginosis (BV) is the most prevalent cause of vaginal discharge or malodor worldwide and affects millions of women.14,16 It is present in 10%-20% of white women and 30%-50% of African American women; it has been found in 85% of female sex workers tested for vaginosis in Africa.16 Many affected women are asymptomatic.14,17 The cause of the microbial alteration of BV is poorly understood, but has been thought to be due to replacement of the normal vaginal flora by an overgrowth of anaerobic bacteria (e.g., Prevotella sp. and Mobiluncus sp.). Gardnerella vaginalis and Mycoplasma hominis are the most commonly present pathogens in the mixed infection.14,16 However, a recent study found that G. vaginalis frequently colonizes the vagina of women without BV.17 Furthermore, the researchers found that women with BV had greater bacterial diversity in vaginal samples with many newly recognized species, including three bacteria in the Clostridiales order that were highly specific for BV.17 Women without BV had a predominance of Lactobacillus species, thought to be "normal flora."17 These findings reiterate that there is no single causative agent for BV.16 BV is associated with having multiple sex partners, a new sex partner, douching, and lack of vaginal lactobacilli; whether BV results from acquisition of a sexually transmitted pathogen is unclear, and it rarely affects women who have never been sexually active.4 BV is associated with preterm labor, PID, and acquisition of HIV.14,16,18,19
BV can be diagnosed by the use of clinical criteria or Gram-stain criteria. For clinical diagnosis, three of the following four criteria must be met:14,15,17
- homogeneous, thin, white vaginal discharge that smoothly coats the vaginal walls;
- vaginal fluid pH > 4.5;
- positive "whiff test," an amine or fishy odor before or after addition of 10% KOH solution;
- presence of "clue cells" on microscopic exam (vaginal epithelial cells with small, adherent bacteria along the border).
Clinical diagnosis will most likely be employed in the ED, although the availability of a light microscope may be hospital-dependent. For diagnosis by Gram stain, differentiation of normal vaginal flora from bacterial vaginosis may be made by staining the vaginal fluid and then observing it under the microscope.17 There are also other commercially available tests, such as FemExamreg; test cards (Cooper Surgical, Shelton, CT) that work by detecting elevated pH and trimethylamine and report sensitivity and specificity greater than 90%, that may be of use in the ED.15,17 A rapid point-of-care diagnostic test is not currently available but would be extremely useful. Due to difficulty in diagnosis, especially in an ED setting, many women who complain of malodorous discharge are empirically diagnosed with BV without confirmation.16
All women with symptomatic disease require treatment. Because BV during pregnancy has been associated with adverse pregnancy outcomes, both asymptomatic and symptomatic pregnant women require treatment.4,18 Recommended regimens by the CDC include the following:4
- metronidazole 500 mg orally twice a day for 7 days;
- metronidazole gel 0.75% one full applicator (5g) intravaginally once a day for 5 days; or
- clindamycin cream 2% one full applicator (5g) intravaginally at bedtime for 7 days.
All recommended regimens are equally efficacious. Alternative regimens include clindamycin 300 mg orally twice a day for 7 days, or clindamycin ovules 100 g intravaginally once a day at bedtime for 3 days. Metronidazole 2 g single-dose therapy has the lowest efficacy for BV and is no longer a recommended regimen. The FDA has cleared metronidazole 750 mg extended release tablets once daily for 7 days and a single dose of clindamycin intravaginal cream. No data have been published that compares the clinical or microbiologic equivalencies of these regimens with other regimens. Cure rates do not differ between intravaginal clindamycin cream and ovules. Pregnant patients should be treated with only oral regimens, either metronidazole 500 mg orally twice a day for 7 days, metronidazole 250 mg three times a day for 7 days, or clindamycin 300 mg twice a day for 7 days. It is important to warn adolescent patients to avoid consuming alcohol during treatment with metronidazole and for 24 hours after finishing treatment to prevent a disulfiram-like reaction. Adolescents should also be warned that clindamycin cream and ovules are oil-based and might weaken condoms and diaphragms. Follow-up care is unnecessary if symptoms resolve (except for pregnant patients), however recurrence is common and re-treatment may be necessary.4 Treatment of sex partners is not recommended.4
Trichomoniasis
Trichomoniasis is caused by the protozoan Trichomonas vaginalis. Trichomoniasis is the most common sexually transmitted infection in the United States, far exceeding either chlamydia or gonorrhea. The annual incidence in the United States has been estimated at 7.4 million cases, with an estimated rate of asymptomatic cases as high as 50%.16,20 Even more concerning are the recent findings of a study by Van Der Pol and colleagues evaluating 14- to 17-year-old adolescent females at high risk for STDs. During this longitudinal study, 6.0% were infected at enrollment, more than 23% had at least one episode of trichomoniasis, and more than 31% had multiple episodes.21 There are many potentially serious complications of infection with T. vaginalis. There is a significantly higher rate of PID among women with T. vaginalis infection compared with uninfected women.19 Despite this, trichomoniasis is not a reportable infection in the United States.20
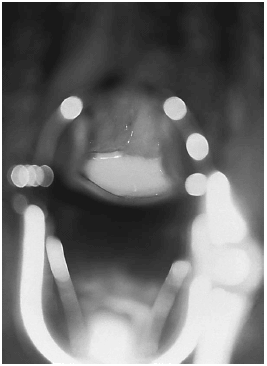
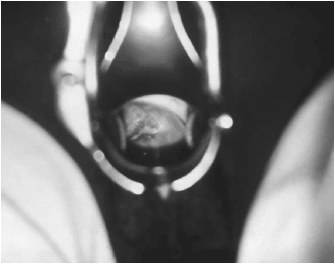
Trichomoniasis causes a diffuse, frothy, yellow-green, malodorous vaginal discharge, as well as irritation and inflammation.14,20 (Figure 1) Diagnosis is made by microscopy of vaginal secretions, looking for motile trichomonads on saline wet prep. However, even when performed by skilled microscopists, the sensitivity of this technique is only 60%.4,20 Other FDA-cleared tests for trichomoniasis in women include OSOM Trichomonas Rapid Test (Genzyme Diagnostics, Cambridge, MA), an immunochromatographic capillary flow dipstick technology, and the Affirm™ VP III (Becton, Dickinson & Co, San Jose, CA), a nucleic acid probe test that evaluates for T. vaginalis, G. vaginalis, and C. albicans. Both tests are point-of-care diagnostics and are performed on vaginal secretions and have a sensitivity greater than 83% and a specificity greater than 97%. The results of the OSOM Trichomonas Rapid Test are available in approximately 10 minutes, and results of the Affirm™ VP III are available within 45 minutes. Although these tests tend to be more sensitive than vaginal wet preparation, false-positive results might occur especially in low prevalence populations. Culture is the most sensitive and specific commercially available method of diagnosis. The recommended regimen for treatment of trichomoniasis includes metronidazole 2 g orally in a single dose or tinidazole 2g orally in a single dose.4 An alternative regimen is metronidazole 500 mg twice a day for 7 days.4 Again, patients should be warned to avoid consuming alcohol during and for 24 hours after treatment. Follow-up care is not necessary if symptoms resolve. Sex partners should be treated, although this is only possible if the original patient is willing to refer her partner(s).20 Patients should be instructed to avoid sex until they and their partners have been successfully treated.4
| Figure 1. Trichomonas vaginalis |
|
|
| Figure 1. This was a case of trichomonas vaginitis revealing a copious purulent discharge emanating from the cervical os. Trichomonas vaginalis, a flagellate, is the most common pathogenic protozoan of humans in industrialized countries. This protozoan resides in the female lower genital tract and the male urethra and prostate, where it replicates by binary fission. Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention. |
Genital Herpes
The differential diagnosis of genital ulcers in young, sexually active patients includes genital herpes, syphilis, and chancroid. Herpes virus is the most common cause of genital ulcers, with at least 50 million people infected in the United States.14,15 Acquisition of herpes virus is common in adolescent women, although symptomatic infection is relatively uncommon.22 Two serotypes of the Herpes simplex virus have been identified causing genital infections: HSV-1 and HSV-2. Most cases of genital herpes are caused by HSV-2, although now it is recognized that HSV-1 also can cause genital herpes and is responsible in 15%-30% of genital infections.15 While seroprevalence in the entire U.S. population has risen, the 12- to 19-year-old age group has the highest rates of increase with seroprevalence rates of 10%-11.7%.2, 3 The Herpes virus is spread through contact with infectious secretions from active lesions, and oral-to-oral, oral-to-genital, and genital-to-genital contacts are all associated with viral spread.15 However, many genital herpes infections are transmitted by asymptomatic carriers or those unaware they are even infected.23
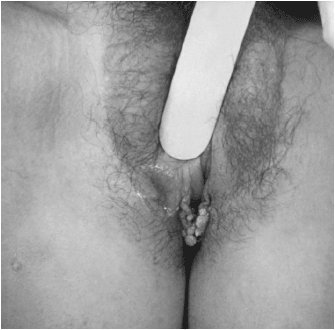
The typical outbreak of primary infection with genital herpes is described as multiple painful vesicular or ulcerative lesions, although clinical diagnosis is insensitive and nonspecific.4 Patients with a primary outbreak may be systemically ill with fever, myalgias, headache, and abdominal pain; the lesions may be longer lasting, more numerous, and more painful.15 Lesions may involve the external genitalia, perineum, buttocks, vagina, or cervix, (Figure 2) therefore, a speculum exam is indicated. Symptoms begin 1 to 4 weeks after exposure, first as vesicles and then as ulcers once the vesicles rupture. Ulcerated lesions persist for 1 to 2 more weeks and then crust and heal leaving no scars.15 After resolution of the primary episode, HSV remains latent in regional dorsal nerve root ganglia.7 Recurrent attacks are typically less severe and are the more common outbreaks seen in the ED.15 Often the patient will believe it is the primary outbreak. Immunodeficiency, trauma, fever, and persistent stressors can trigger an outbreak.7,15
| Figure 2. Cervicitis due to Herpes Simplex Virus |
|
|
|
Figure 2. This patient presented with a case of cervicitis due to what was differentially diagnosed as herpes simplex virus. Initially, this patient was believed to be suffering from a case of cervicitis due to Neisseria gonorrhoeae, which like herpes is a sexually transmitted disease. Note the erosive inflammation with accompanying paracervical purulency. Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention / Dr. Paul Wiesner. |
| Figure 3. Herpes Genitalis |
|
|
|
Figure 3. This was an outbreak of herpes genitalis manifested as blistering around the vaginal introitus due to the HSV-2 virus. The sexually transmitted herpes simplex virus type-2 (HSV-2) typically causes one or more blisters to form on, or around the genitals or rectum, which break, leaving tender ulcers that may take 2-4 wks. to heal after making their initial appearance. Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention./ Susan Lindsley. |
Clinical diagnosis of herpes infection should be confirmed by laboratory testing. The traditional Tzanck smear has been replaced by newer, more sensitive methods.15,23 Isolation of HSV in cell culture is now the preferred virologic test and gold standard, and results take from 1 to 3 days. The FDA-cleared glycoprotein G-based type-specific assays include the laboratory-based assays HerpeSelect™-1 enzyme-linked immunosorbent assay (ELISA) immunoglobulin G (IgG) or HerpeSelect™-2 ELISA IgG and HerpeSelect™ 1 and 2 Immunoblot IgG (Focus Technologies, Inc., Herndon, VA), and HSV-2 ELISA (Trinity Biotech USA, Berkeley Heights, NJ). Two other assays, Biokit HSV-2 and SureVue HSV-2 (Biokit USA, Lexington, MA, and Fisher Scientific, Pittsburgh, PA, respectively), are point-of-care tests that provide results for HSV-2 antibodies from capillary blood or serum during a clinic visit. The sensitivities of these glycoprotein G type-specific tests for the detection of HSV-2 antibody vary from 80% to 98%, and false-negative results might be more frequent at early stages of infection. The specificities of these assays are 96% or greater.4 False-positive results can occur, especially in patients with a low likelihood of HSV infection. Repeat or confirmatory testing might be indicated in some settings, especially if recent acquisition of genital herpes is suspected.
Recurrences are much less frequent with HSV-1 infection than HSV-2 infection, with 95% of patients infected with HSV-2 having a recurrence within a year compared with only 50% of patients infected with HSV-1.15 Because no results will be completed while the patient is in the ED, follow-up care is a necessity. Any patient with genital ulcers should also be screened for syphilis, and Haemophilus ducreyi in settings where chancroid is prevalent.14
There is no cure for genital herpes, and the mainstay of treatment is with systemic antiviral drugs. Although it is impossible to accurately differentiate between primary and recurrent outbreaks in the ED, treatment does differ slightly and the physician's best judgment should be employed. Patients with a primary outbreak of genital herpes should be treated with one of the following regimens:4
- acyclovir 400 mg orally three times a day for 7-10 days;
- acyclovir 200 mg orally five times a day for 7-10 days;
- famciclovir 250 mg orally three times a day for 7-10 days;
- valacyclovir 1 g orally twice a day for 7-10 days.
For recurrent episodes of disease, antiviral therapy can be administered either episodically to treat outbreaks or as suppressive therapy to reduce the frequency of recurrences. Suppressive therapy reduces the frequency of recurrences by 70% to 80% among patients with frequent recurrences, and reduces subclinical viral shedding.4 These options can be discussed with all patients in the ED with a suspected herpes outbreak, however, the patient would most likely be seeking treatment for a current outbreak and so suppressive therapy can be decided upon at an outpatient follow-up visit. Effective episodic treatment should be initiated within 1 day of lesion onset, or during the prodrome, with one of the following regimens:
- acyclovir 400 mg orally three times a day for 5 days;
- acyclovir 800 mg orally twice a day for 5 days;
- acyclovir 800 mg orally three times a day for 2 days;
- famciclovir 125 mg orally twice a day for 5 days;
- famciclovir 1000 mg orally twice a day for 1 day;
- valacyclovir 500 mg orally twice a day for 3 days;
- valacyclcovir 1 g orally once a day for 5 days.
There is no role for topical treatment for either primary or recurrent outbreaks. Patients with severe disease or complications (e.g., disseminated infection, pneumonitis, hepatitis, meningitis, or encephalitis) should be hospitalized and treated with intravenous acyclovir.4
Follow-up care is recommended for all patients for results of culture and type-specific tests and to discuss suppressive therapy. Counseling regarding the natural history of genital herpes, sexual and perinatal transmission, and methods to reduce transmission should be initiated at the ED visit and continued at the follow-up visit. Patients should be encouraged to inform their current sex partner(s), and to inform future partners before initiating a sexual relationship.4 Genital herpes is not a reportable disease.
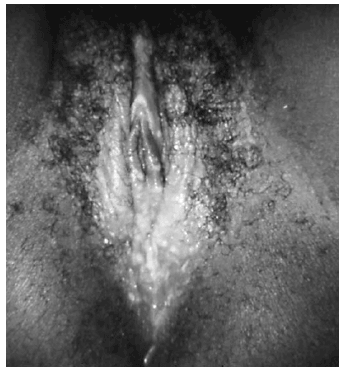
Genital Warts
HPV types 6 or 11 are commonly found before, or at the time of, detection of genital warts; however, the use of HPV testing for genital wart diagnosis is not recommended. It is documented that more than 5 million people develop HPV each year with two-thirds of these infections in young adults 15-24 years of age.24 Clinical presentation ranges from subclinical infection to clinically apparent lesions, (Figure 4) thus making precise determination of infection rates difficult. More than 40 types of HPV causing genital infection have been identified.25 Visible warts themselves, while perhaps prompting the ED visit or noticed during physical exam, may be of aesthetic concern to the patient but are not the main concern of the physician. HPV infections have been associated with cervical cancer with high-risk HPV types causing subclinical infection rather than clinical warts.6,14 Persistent HPV infections are necessary for the development of the precursor lesion of cervical cancer, and HPV type 16 is responsible for approximately half of all cervical cancers worldwide.25 Other HPV types associated with cervical cancer include 18, 31, 33, and 35.14 There is no evidence that treatment of visible warts affects the development of cervical cancer, and most patients will require a course of therapy rather than a single treatment.7 Thus, there is no reason to initiate treatment of genital warts in the ED; patients should be referred for follow-up with primary care for treatment as well as for a Pap smear for detection of cervical dysplasia and prevention of cervical cancer.
| Figure 4. Condyloma Acuminatum Genital Warts |
|
|
| Figure 4. A patient with a primary syphilitis vulvar chancre, and condyloma acuminatum genital warts. The primary stage of syphilis is usually marked by the appearance of a single sore called a chancre. The chancre is usually firm, round, small, and painless and is caused by the bacterium. Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention./ Susan Lindsley. |
Many HPV infections are contracted in adolescence, and cervical cancer is the third most common cause of cancer in women worldwide.26 Of interest, a recent article by Mao and colleagues demonstrating that a vaccine against HPV type 16 prevented the precursor lesion for cervical cancer for a follow-up period of 3.5 years is promising for the prevention of cervical cancer.25 It is believed that an even more effective vaccine including protection against other HPV types also will be developed in the near future.26
Chlamydial and Gonococcal Infections
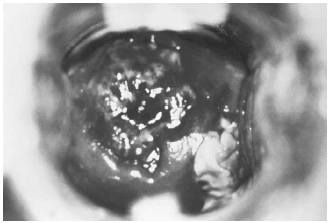
The CDC estimates that more than 1 in 10 sexually active female adolescents have chlamydial infections, and that females aged 15-24 years have the highest rates of gonorrhea.1 Chlamydia is the most common reportable STD in the United States. Age-specific rates are highest in adolescents aged 15-19 years. Prevalence rates in 15- to 24-year-old women range from 2.3% to 29.8%.3 Chlamydial infection can be asymptomatic in both males and females. Women who have an apparently uncomplicated cervical infection already may have a subclinical upper-genital tract infection. Chlamydial infection can cause dysuria, urinary frequency and urgency, pelvic pain, vaginal bleeding, and abnormal vaginal discharge.15 Mucopurulent cervicitis can be present, although it is usually asymptomatic and is not sensitive or specific for chlamydial infection.15 Neisseria gonorrhoeae infections also may be asymptomatic in women. Gonococcal genital infections, if symptomatic, have the same symptoms as chlamydial infections (Figure 5).
| Figure 5. Condyloma Acuminatum Genital Warts |
|
|
| Figure 5. This patient presented with a case of gonorrhea with symptoms including cervicitis and vaginal discharge. Gonorrhea is the most frequently reported communicable disease in the United States Disseminated gonococcal infection is most often the cause of acute septic arthritis in sexually active adults, and the reason for most hospitalizations due to infective arthritis. Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention. |
Diagnosis for either infection must be made by laboratory confirmation. Although culture remains the gold standard for definitive diagnosis, nonculture techniques such as polymerage chain reaction (PCR) and ligaste chain reaction (LCR) are becoming more common, and have a high sensitivity and specificity.7 Nonculture techniques have become the standard of care. Nucleic acid amplification techniques such as PCR and LCR provide the highest sensitivity (93%-99%) and specificity (99%-100%).7,15 These tests for gonococcal or chlamydial infection can be performed on cervical, urine, or urethral specimens, and even patient-collected vaginal specimens.15 In general, sensitivities are lower with urine tests.27 When collecting an endocervical sample, be sure to remove any excess mucus from the cervix first. Although each kit has unique instructions, the technique is usually the same: The collection swab is inserted into the endocervical canal, rotated 360°, and left in place for 10-30 seconds to ensure adequate sample collection. With urine tests, ideally the patient should not have urinated in the last hour prior to sample collection. Women should not clean the labial area, and the sample collected should be first-catch urine. Any single test has limitations, and the emergency physician must utilize the preferred test for a given hospital. The threshold should remain low because test results are often not available during the ED visit.
While noninvasive diagnostic tests for STDs are convenient, they do not take the place of a physical exam, including the pelvic exam. Each patient must be evaluated based on history and symptomology as well as physical exam. The complaint of vaginal discharge and discomfort necessitates a physical exam often with pelvic exam, to narrow the differential diagnosis.
Treatment of Gonorrhea and Chlamydia
Patients infected with N. gonorrhoeae are often also co-infected with Chlamydia trachomatis, and therefore dual therapy for both infections is now recommended in patients suspected to have either infection. This is especially important in the adolescent population, where follow-up care may be poor and infection rates are high. Treatment of infected patients can prevent the development of PID, relieve symptoms, and prevent transmission to sex partners. See Table 2 and Table 3 for regimens recommended by the Centers for Disease Control (CDC) in Sexually Transmitted Diseases Treatment Guidelines 2006. The ideal regimen for the adolescent patient is a single-dose treatment, consisting of ceftriaxone injection and azithromycin pill, both administered while still in the ED. If the patient is wary of injections, then an oral antigonococcal agent could be selected instead. Regardless of selected regimen, the first dose should be dispensed on site. Patients should be instructed to abstain from sexual intercourse for 7 days after completion of therapy.4,15 Patients do not need to be retested unless symptoms persist and re-infection is suspected.4
|
|
It is important to note that quinolone-resistant N. gonorrhoeae (QRNG) is continuing to spread worldwide, especially in parts of Asia and the Pacific rim.4 Fluoroquinolones should not be used to treat N. gonorrhoeae in Hawaii, California, and areas with high resistance rates.4 The CDC website (http://www.cdc.gov/std/gisp) or state health departments can provide the most current information. Although fluoroquinolones have not been recommended for persons younger than 18 years due to the possibility of joint damage seen in some young animals, the CDC recommendations state that children who weigh more than 90 lbs can be treated with any quinolone regimen recommended for adults.4
Treatment of Sex Partners
Patients with either C. trachomatis or N. gonorrhoeae infection should be instructed to refer their sex partners for evaluation and treatment. Re-infection is common in adolescent patients, and patients should be instructed to avoid sexual intercourse until 7 days of therapy is completed and until their partners also have been evaluated and treated. Adolescent patients also should be counseled about STD prevention and safe-sex practices.
Partner notification and health department reporting regulations vary from state to state. Gonorrhea and chlamydia infections are often reportable, therefore familiarity with the health department statute of the practitioner's state is essential. Empiric therapy may be warranted for partners, however individual evaluation of partners is preferable, given patient-specific factors.7 State and local health departments may be of use in partner notification and follow-up.
Screening for Other STDs
When an adolescent presents with an STD or PID, the decision must be made to screen for other STDs, including HIV. As stated previously, STDs are associated with increased transmission of HIV. The CDC estimates that up to 50% of new HIV infections occur in individuals younger than 25 years.28 In 2003, adolescent females accounted for 50% of HIV infections in persons aged 13 to 19 years.28 Because of these statistics, testing for HIV is recommended and should be offered to all patients seeking evaluation and treatment for STDs.1,4 Pretest and post-test counseling should be provided, or patients should be referred to such services. Additionally, HIV testing always requires informed consent from the patient, and written consent is required in some states. Emergency physicians should be familiar with local requirements, as well as services specializing in counseling adolescents, including those with positive test results. Occasionally an adolescent may request post-exposure prophylaxis (PEP) after having unprotected intercourse. There are very little data regarding PEP outside the context of occupational exposure or sexual assault. This decision should be left to the discretion of the provider after appropriate consultation with infectious disease or immunology expert.
Syphilis screening also is recommended for all patients evaluated for STDs, especially those with genital ulcerative lesions.1 Other adolescents who should be screened for HIV and syphilis include those who have had multiple sexual partners or a high-risk sexual partner, who have engaged in exchange of sex for drugs or money or intravenous drug use, who have been admitted to jail or other detention facility, or who live in areas with high prevalence of HIV or syphilis.1 Lastly, all pregnant adolescents should be offered testing if they haven't already been tested, and referred immediately for obstetric follow-up. Given the risk for cervical neoplasia, adolescents who have had sexual intercourse should have their initial Pap test approximately 3 years after becoming sexually active, but no later than age 21 years.1 Since this is not an emergent issue, the patient can be referred for outpatient follow-up if necessary.
Adolescent Males and STDs
Male adolescents are also at high risk for becoming infected with gonorrhea or chlamydia. Gonorrhea typically presents as urethritis with purulent urethral discharge and dysuria; 85%-90% of infected men develop symptoms within 10-14 days of exposure.7 Complications of untreated gonorrhea in men include epididymitis, prostatitis, and urethral stricture.7 Chlamydia is the most common cause of nongonococcal urethritis (NGU) in men, however, T. vaginalis infection is becoming increasingly recognized as a cause of NGU.20 Presentation is often delayed with Chlamydia, and symptoms may be less severe. However, it is impossible to differentiate among infections on clinical grounds alone, therefore, laboratory testing is diagnostic. As mentioned previously, nucleic-acid amplification tests are the most sensitive and specific for gonococcal or chlamydial infection, and either urethral or urine samples can be used. Urine sample collection has been described previously in the section Chlamydial and Gonococcal Infections. For urethral specimen collection, the patient should not have urinated in the hour prior to collection. The swab should be inserted 2-4 cm into the urethra and rotated for 2-3 seconds. The same treatment guidelines exist for male patients, with empiric treatment being recommended for patients, such as adolescents, who may be lost to follow-up care before test results are available.4
Pelvic Inflammatory Disease in Adolescents
PID is the most frequent gynecologic cause for ED visits (350,000/year) and incurs an annual total expense of more than $5 billion.29,30 PID is a polymicrobial infection of the upper genital tract (uterus, ovaries, and fallopian tubes) and often the peritoneal cavity. It is believed to be the result of an ascending infection from the cervix and vagina. Most cases are believed to be the sequelae of C. trachomatis and N. gonorrhoeae, however non-STD PID is a known entity.31 PID is not a reportable disease, therefore exact estimates of the incidence and prevalence are unknown. Most recent estimates of PID incidence from the CDC using discharge and outpatient database investigation stated that approximately 780,000 cases are diagnosed annually.31 It is estimated that nearly 70% of patients with PID are younger than 25 years, with 33% experiencing their first infection before the age of 20 years.5 Adolescents aged 15-19 years have the highest incidence of PID compared with all other age groups.5
There are behavioral, biologic, and systems-related risk factors that affect adolescent acquisition of PID.32 Adolescents have a high prevalence of STDs, thereby, increasing their risk for PID.31,32 Behaviors associated with this high prevalence of STDs include multiple sexual partners, unprotected sexual intercourse, and drug or alcohol use.1,32 Younger age incurs an increased risk of PID, both due to behavioral factors listed previously as well as biologic factors.31 A normal finding during adolescence is the presence of cervical ectopy, which provides large zones of columnar epithelium on the cervix for the targeted attachment of C. trachomatis and N. gonorrhoeae, leading to higher rates of PID.31,32 Adolescents also are believed to have a lower prevalence of protective antibodies and a greater penetrability of cervical mucus.31,32 Systems-related risk factors, or barriers to care, include decreased access to convenient, affordable, and confidential health care services, which can delay diagnosis and treatment of PID.31,32 This delay can be of serious consequence because women seeking care for PID-related abdominal pain of more than 3 days duration are three times more likely to experience infertility and ectopic pregnancy than women who seek care within 3 days.31,32
Clinical Characteristics of PID
PID has a varied presentation from asymptomatic to severe disease. Data indicate that a clinical diagnosis of symptomatic PID has a positive predictive value (PPV) for salpingitis of 65%-90% compared with laparoscopy.4 Also the PPV of a clinical diagnosis of acute PID depends on the epidemiologic characteristics of the population, with higher PPVs among sexually active young women (particularly adolescents), among patients attending STD clinics, or in other settings where the rates of gonorrhea or chlamydia infection are high. In all settings, however, no single historical, physical, or laboratory finding is both sensitive and specific for the diagnosis of acute PID. Many episodes of PID go unrecognized. The clinical diagnosis of acute PID is imprecise. No single historical, physical, or laboratory finding is both sensitive and specific for the diagnosis of acute PID. The symptoms can be mild, vague, or nonspecific, therefore, it is important for the emergency physician to maintain a low threshold for the diagnosis and empiric treatment of PID. Diagnosis and management of other common causes of lower abdominal pain (e.g., ectopic pregnancy or acute appendicitis) are unlikely to be impaired by initiating empiric antimicrobial therapy for PID.4 To aid with diagnosing PID, the CDC's Morbidity and Mortality Weekly Report (MMWR) has established guidelines.4 Empiric treatment of PID should be initiated in sexually active young women or women at risk for STDs if they are experiencing pelvic or lower abdominal pain, if no cause for the illness other than PID can be identified, and if one or more of the following minimum criteria are present on pelvic examination: cervical motion tenderness or uterine tenderness or adnexal tenderness.
The presence of signs of lower genital tract inflammation, in addition to one of the three minimum criteria, increases the specificity of diagnosis. In deciding upon the initiation of empiric treatment, clinicians also should consider the risk profile of the patient for STDs.
These guidelines have changed from past guidelines because they require that only one of the criteria be met. This change is to maximize sensitivity, reduce the chance of a missed or delayed diagnosis, and promote early treatment of PID.32 More elaborate diagnostic criteria may be needed to enhance specificity. These additional criteria that support the diagnosis of PID include:
- oral temperature > 101°F (>38.3°C);
- abnormal cervical or vaginal mucopurulent discharge;
- presence of abundant white blood cells (WBCs) on saline; microscopy of vaginal secretions;
- elevated erythrocyte sedimentation rate (ESR);
- elevated C-reactive protein (CRP);
- laboratory documentation of cervical infection with C. trachomatis and N. gonorrhoeae.
However, the most specific methods for diagnosing PID often are not performed in the ED, and often not on adolescents, and include the following:
- endometrial biopsy with histologic evidence of endometritis
- transvaginal sonography or magnetic resonance imaging techniques showing thickened, fluid-filled tubes with or without free pelvic fluid or tubo-ovarian complex, or doppler studies suggesting pelvic infection (e.g., tubal hyperemia);
- laparoscopic abnormalities consistent with PID.
Adolescent Patient Management
The treatment goals of PID in adolescents are to not only eliminate the infection, but also to prevent long-term sequelae. There remains a controversy about inpatient versus outpatient management, with recent trends moving toward mostly outpatient management.4,5,31 Currently, the CDC recommends that the decision of whether hospitalization is necessary should be based on the discretion of the health care provider.4 However, the following recommendations for hospitalization have been developed by the CDC, based on observational data and theoretical concerns to aid physicians in decision making: 4
- Surgical emergencies (e.g., appendicitis) cannot be excluded.
- The patient is pregnant.
- Tthe patient does not respond clinically to oral antimicrobial therapy.
- The patient is unable to follow or tolerate an outpatient oral regimen.
- The patient has severe illness, nausea and vomiting, or high fever.
- The patient has a tubo-ovarian abscess.
Of interest, there are no data available to suggest that adolescent women benefit from inpatient treatment of PID.4 Many clinicians continue to use the adolescent age group as an indication for hospitalization, often due to fears of poor follow-up and adherence to treatment regimens, especially given the severe morbidity associated with untreated or inadequately treated PID. All patients who are treated with an oral outpatient regimen should have follow-up care arranged within 72 hours, and should be given strict precautions for return.7 The ultimate decision of whether to treat an adolescent with PID as an inpatient or an outpatient must be individualized to each patient, taking into consideration not only clinical findings but also social situation and emotional status. When a presumptive diagnosis of PID has been made, empiric treatment should be initiated immediately. Whether inpatient or outpatient, antimicrobial coverage should be effective against C. trachomatis, N. gonorrhoeae, anaerobes, Gram-negative facultative bacteria, and streptococci.4,5 There have been no data directly comparing parenteral with oral regimens, however many trials have showed the efficacy of both regimens.4 See Table 4 and Table 5 for parental and oral regimens recommended by the CDC.
| Table 4. Parenteral Regimens for Treatment of Acute Pelvic Inflammatory Disease |
|
|
| ** Doxycycline should be administered orally whenever possible because of pain associated with infusion. Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2006. MMWR 2006;55(No. RR11):1-94 |
| Table 5. Oral Regimens for Treatment of Acute Pelvic Inflammatory Disease |
|
|
| Source: U.S. Dept. of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2006. MMWR 2006;55(No. RR11):1-94. |
Parenteral therapy can be discontinued 24 hours after a patient improves clinically. Clinical improvement is defined as afebrile, reduction in abdominal tenderness, and reduction in uterine, adnexal, and cervical motion tenderness.14 Continuing oral therapy with doxycycline 100 mg po BID or clindamycin 450 mg po QID, depending on original treatment regimen, should be administered for a total of 14 days of therapy. Limited data exist for the alternative regimens, however those regimens listed have been investigated in at least one clinical trial and have broad spectrum coverage.4
If treated as an outpatient, patients should show significant clinical improvement within 3 days of therapy. Patients who fail to improve will need admission and further evaluation at that time. Any patient treated as an outpatient must have follow-up care arranged within 3 days of initiating therapy.4 It is important for emergency physicians to be familiar enough with the regimens so that appropriate empiric treatment can be initiated in the ED for suspected cases of PID. Male sex partners of adolescents with PID also should be treated empirically for C. trachomatis and N. gonorrhoeae.
Complications of PID
Complications of PID can be acute or long-term, and can account for significant reproductive morbidity in the adolescent patient. Acute complications include development of a tubo-ovarian abscess and/or Fitz-Hugh-Curtis syndrome, while long-term complications include infertility, ectopic pregnancy, and chronic pelvic pain. In the United States, tubal damage following PID is believed to be responsible for 25%-35% of female infertility.5,32 The more episodes of PID a patient suffers, the higher the risk of infertility in the future. However, 30%-80% of women with infertility related to tubal damage have no history of recognized clinical disease, thus supporting the recommendations to maintain a high index of suspicion and treat empirically early. The rate of ectopic pregnancy ranges from 3 to 10 times higher in women with a history of PID.5,32 The rates of chronic pelvic pain from PID are difficult to assess, but have been estimated to be up to 3 times higher than in women with no history of PID.5,32
Tubo-ovarian Abscess
When PID goes untreated, the fallopian tubes can become damaged and blocked, and bacteria, leukocytes, and fluid can accumulate. When the inflammation and bacteria spill from the fimbriated end of the tube and into the ovary, a tubo-ovarian abscess can form. As the abscess is formed, an anaerobic environment is established. Tubo-ovarian abscesses are generally polymicrobial and anaerobic, and the predominant organisms isolated are Escherichia coli, Bacteroides fragilis, and Peptostreptococcus bacteria.5,15,32 It is a severe complication arising in up to one-third of patients with PID, and in 15%-20% of adolescents with PID.5,32 Tubo-ovarian abscess rupture can occur in up to 15% of patients and may be life-threatening. Mortality due to septic shock and the complications of peritonitis can approach 10%.15,33
Diagnosing tubo-ovarian abscess on clinical criteria is difficult and often inaccurate. Patients typically have symptoms similar to those seen in uncomplicated PID. A palpable adnexal mass is often absent on pelvic exam in the adolescent, and palpation may be difficult due to pain.34 A ruptured tubo-ovarian abscess may present with acute, progressive pelvic pain, eventually peritoneal signs on exam, and ultimately shock. Definitive diagnosis is made surgically or with imaging studies. Transvaginal ultrasound is the imaging modality of choice, and computed tomography or magnetic resonance imaging also may be of use.15,32,35 A patient with a tubo-ovarian abscess should be admitted, and antimicrobial regimens are identical to the parenteral regimens for PID.31 Medical versus surgical therapy for patients with tubo-ovarian abscess remains controversial, however all patients with a suspected ruptured tubo-ovarian abscess should receive prompt surgical therapy or drainage.33 It has been reported that up to 60%-80% of tubo-ovarian abscesses resolve with antibiotic treatment alone.36
Fitz-Hugh-Curtis Syndrome
Fitz-Hugh-Curtis syndrome, or perihepatic inflammation and adhesions, occurs in up to 10%-14% of patients with acute PID, however the incidence may be as high as 27% in adolescents with PID.33,37 Initially believed to be due exclusively to N. gonorrhoeae, most experts now believe that C. trachomatis is more often the culprit.37 Signs and symptoms include those of PID in addition to right upper quadrant pain, pleuritic pain, referred shoulder pain, and tenderness to palpation in the right upper quadrant.33,37 It is often mistaken for acute cholecystitis, and the differential diagnosis must be broad. The diagnosis is usually made clinically by eliminating other causes of right upper quadrant pain, along with the presence of pelvic symptoms.37 Radiographic studies, ideally ultrasonography, are most useful in eliminating other possible causes such as acute cholecystitis, and laboratory tests are generally nonspecific.37 Treatment consists of inpatient administration of parenteral antibiotic agents for treating PID, and the symptoms of perihepatitis usually resolve quickly. Laparoscopic evaluation should be performed in patients with persistent pain, and lysis of adhesions may be beneficial.37 The long-term consequences of this syndrome are unclear.31
Emergency Contraception in Adolescents
During 2000, 882,000 pregnancies occurred in adolescent women and 85% of those were unintended.38 It is thought that use of emergency contraception (EC) could prevent up to 70% of abortions.38,39 EC, also known as the morning-after pill and post-coital contraception, is intended for women who have had recent unprotected intercourse or failure of contraception. There are no contraindications for its use, even in patients with contraindications to oral contraceptive pills, given the short duration of use of EC.40 Most experts believe that any risks of EC are far outweighed by the benefits.40
There are currently a number of EC regimens available, and most must be initiated within 72 hours of intercourse, however EC has been shown to have efficacy up to 120 hours after intercourse.41,42 In general, the sooner the administration of EC the lower the rate of pregnancy. Plan B, perhaps the most common regimen, is a progestin-only regimen consisting of two tablets, each containing 0.75mg levonorgesterol.43 The first dose is taken within 72 hours of unprotected intercourse, and the second dose 12 hours later. This regimen has a pregnancy rate of 1.1%.43 The use of progestin-only pills results in markedly less nausea and vomiting than combined estrogen and progestin regimens.43 The major mechanism of action in inhibition or delay or ovulation, and other mechanisms include alteration of the endometrium, sperm penetration, and tubal motility; established pregnancies are not harmed.43
If a dedicated product for EC, such as Plan B, is not available, then oral contraceptive pills may be used instead. Most are progestin-estrogen combined pills and may cause more nausea and vomiting, but they are equally effective. The treatment schedule is the same as for Plan B, consisting of one dose within 72 hours after unprotected intercourse, and another dose 12 hours later. For a complete listing of treatment options and doses visit the EC Web site at www.not-2-late.com. This Web site is operated by the Office of Population Research at Princeton University and by the Association of Reproductive Health Professionals. It is dedicated to making accurate information from the medical literature available to the public and has no sponsorship from contraceptive manufacturers or sellers.
EC has been shown to be both tolerable and effective in adolescents, even those younger than 15 years.44 More importantly, there have been adequate data to support that even young adolescents can safely use EC, and the Society for Adolescent Medicine has placed no age limitations on access to EC.38,45 EC is currently available without prescription to women 18 years of age or older. Many emergency physicians are in the position to recommend, prescribe, and educate their adolescent patients about EC. Much of the debate about EC becoming available over-the-counter has centered on whether adolescents who use EC are more likely to be sexual risk takers.38 However, multiple studies have supported that adolescents, even young adolescents (younger than 16 years) with improved access to EC did not compromise their use of routine contraception nor increase their sexual risk behavior.38,46,47
The most common side effects of EC are nausea and vomiting.40,43 They are more common with the combined estrogen and progestin regimens, and less so with the progestin-only regimens such as Plan B. Only 4% of patients experienced vomiting with a progestin-only regimen.40 Antiemetic agents may be given prophylactically for the combined regimens and are needed only symptomatically for the progestin-only methods. Uncommon side effects may be treated symptomatically and include irregular bleeding, dizziness, fatigue, breast tenderness, and lower abdominal pain.40 There have never been any cases of death or serious complications associated with the use of EC.40
The adolescent patient should be advised that the risk of pregnancy still exists after the EC pills have been taken if they have unprotected intercourse again, and they should continue (or start) to use a method of birth control. Menstrual bleeding should occur within a week of the expected time. If bleeding does not occur, the patient should be evaluated for pregnancy. There is no increased risk for ectopic pregnancy after use of EC.40 Routine office follow-up care is not needed, but should be recommended, after prescribing EC, even for the adolescent patient.40
Conclusion
The sexually active adolescent provides a unique challenge to the emergency physician. The emergency physician should always maintain a low threshold for evaluation for STDs in the adolescent population and empiric treatment of STDs and PID given the significant reproductive morbidity caused by these diseases. All adolescents seeking evaluation for STDs or PID should also be offered testing for other sexually transmitted infections, including HIV. Adolescents can present with serious sequelae of PID, and prompt recognition and initiation of treatment can be life-saving.
References
1. ACOG Committee Opinion #301. Sexually transmitted diseases in adolescents. Obstet Gynecol 2004;104(4):891-8.
2. Braverman PK. Sexually transmitted diseases in adolescents. Med Clin North Am 2000;84(4):869-89, vi-vii.
3. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Sexually Transmitted Disease Surveillance, 2000. Atlanta, GA;2001.
4. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2006. MMWR 2006; 55(RR-11):1-94..
5. Lawson MA, Blythe MJ. Pelvic inflammatory disease in adolescents. Pediatr Clin North Am 1999;46(4):767-82.
6. Moscicki AB. Human papillomavirus infection in adolescents. Pediatr Clin North Am 1999;46(4):783-807.
7. McKinzie J. Sexually transmitted diseases. Emerg Med Clin North Am 2001;19(3):723-43.
8. Daley AM, Cromwell PF. How to perform a pelvic exam for the sexually active adolescent. Nurse Pract 2002;27(9):28, 31-2, 4, 7-9, 43; quiz 4-5.
9. Ford CA, Millstein SG, Halpern-Felsher BL, Irwin CE, Jr. Influence of physician confidentiality assurances on adolescents' willingness to disclose information and seek future health care. A randomized controlled trial. JAMA 1997;278(12):1029-34.
10. Reif C, Warford A. Office practice of adolescent medicine. Prim Care 2006;33(2):269-84.
11. Jones RK, Boonstra H. Confidential reproductive health care for adolescents. Curr Opin Obstet Gynecol 2005;17(5):456-60.
12. Minors' Access to STD Services. In: State Policies in Brief; 2006. Available: http://www.guttmacher.org/statecenter/spibs/index.html. Accessed: July 1, 2006.
13. Minor's Access to Contraceptive Services. In: State Policies in Brief; 2006. Available: http://www.guttmacher.org/statecenter/spibs/index.html. Acceessed July 1, 2006.
14. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2002. MMWR 2002;51(No. RR-6):1-77.
15. Zeger W, Holt K. Gynecologic infections. Emerg Med Clin North Am 2003;21(3):631-48.
16. Sobel JD. What's new in bacterial vaginosis and trichomoniasis? Infect Dis Clin North Am 2005;19(2):387-406.
17. Fredricks DN, Fiedler TL, Marrazzo JM. Molecular identification of bacteria associated with bacterial vaginosis. N Engl J Med 2005; 353(18):1899-911.
18. Leitich H, Bodner-Adler B, Brunbauer M, Kaider A, Egarter C, Husslein P. Bacterial vaginosis as a risk factor for preterm delivery: a meta-analysis. Am J Obstet Gynecol 2003;189(1):139-47.
19. Moodley P, Connolly C, Sturm AW. Interrelationships among human immunodeficiency virus type 1 infection, bacterial vaginosis, trichomoniasis, and the presence of yeasts. J Infect Dis 2002;185(1):69-73.
20. Schwebke JR. Trichomoniasis in adolescents: a marker for the lack of a public health response to the epidemic of sexually transmitted diseases in the United States. J Infect Dis 2005;192(12):2036-8.
21. Van Der Pol B, Williams JA, Orr DP, Batteiger BE, Fortenberry JD. Prevalence, incidence, natural history, and response to treatment of Trichomonas vaginalis infection among adolescent women. J Infect Dis 2005;192(12):2039-44.
22. Fife KH, Fortenberry JD, Ofner S, Katz BP, Morrow RA, Orr DP. Incidence and prevalence of herpes simplex virus infections in adolescent women. Sex Transm Dis 2006;In press.
23. Lonergan S, Herg HG. Refresher course on sexually transmitted disease. Emerg Med 2006:33-44. Available: www.emedmag.com.
24. Weinstock H, Berman S, Cates W, Jr. Sexually transmitted diseases among American youth: incidence and prevalence estimates, 2000. Perspect Sex Reprod Health 2004;36(1):6-10.
25. Mao C, Koutsky LA, Ault KA, Wheeler CM, Brown DR, Wiley DJ, et al. Efficacy of human papillomavirus-16 vaccine to prevent cervical intraepithelial neoplasia: a randomized controlled trial. Obstet Gynecol 2006;107(1):18-27.
26. Noller KL. HPV vaccination: more questions than answers. Obstet Gynecol 2006;107(1):4-5.
27. Shrier LA, Dean D, Klein E, Harter K, Rice PA. Limitations of screening tests for the detection of Chlamydia trachomatis in asymptomatic adolescent and young adult women. Am J Obstet Gynecol 2004;190(3):654-62.
28. D'Angelo LJ, Samples C, Rogers AS, Peralta L, Friedman L. HIV infection and AIDS in adolescents: an update of the position of the Society for Adolescent Medicine. J Adolesc Health 2006;38(1):88-91.
29. Washington AE, Katz P. Cost of and payment source for pelvic inflammatory disease. Trends and projections, 1983 through 2000. JAMA 1991;266(18):2565-9.
30. Curtis KM, Hillis SD, Kieke BA Jr., Brett KM, Marchbanks PA, Peterson HB. Visits to emergency departments for gynecologic disorders in the United States, 1992-1994. Obstet Gynecol 1998; 91(6):1007-12.
31. Beigi RH, Wiesenfeld HC. Pelvic inflammatory disease: new diagnostic criteria and treatment. Obstet Gynecol Clin North Am 2003;30(4):777-93.
32. Banikarim C, Chacko MR. Pelvic inflammatory disease in adolescents. Adolesc Med Clin 2004;15(2):273-85, viii.
33. Rock JA, Jones HW. Te Linde's Operative Gynecology. 9th ed. Lippincott Williams & Wilkins; 2003.
34. Slap GB, Forke CM, Cnaan A, Bellah RD, Kreider ME, Hanissian JA, et al. Recognition of tubo-ovarian abscess in adolescents with pelvic inflammatory disease. J Adolesc Health 1996;18(6):397-403.
35. Moir C, Robins RE. Role of ultrasonography, gallium scanning, and computed tomography in the diagnosis of intraabdominal abscess. Am J Surg 1982;143(5):582-5.
36. Lawrence LL. Unusual presentations in obstetrics and gynecology. Emerg Med Clin North Am 2003;21(3):649-65.
37. Peter NG, Clark LR, Jaeger JR. Fitz-Hugh-Curtis syndrome: a diagnosis to consider in women with right upper quadrant pain. Cleve Clin J Med 2004;71(3):233-9.
38. Irwin CE, Jr. Emergency contraception for adolescents: the time to act is now. J Adolesc Health 2004;35(4):257-8.
39. Trussell J, Ellertson C, von Hertzen H, Bigrigg A, Webb A, Evans M, et al. Estimating the effectiveness of emergency contraceptive pills. Contraception 2003;67(4): 259-65.
40. ACOG Practice Bulletin. Clinical Management Guidelines for Obstetrician-Gynecologists, Number 69, December 2005 (replaces Practice Bulletin Number 25, March 2001). Emergency contraception. Obstet Gynecol 2005;106(6):1443-52.
41. Ellertson C, Evans M, Ferden S, et al. Extending the time limit for starting the Yuzpe regimen of emergency contraception to 120 hours. Obstet Gynecol 2003;101(6):1168-71.
42. Rodrigues I, Grou F, Joly J. Effectiveness of emergency contraceptive pills between 72 and 120 hours after unprotected sexual intercourse. Am J Obstet Gynecol 2001;184(4):531-7.
43. Cunningham GF, Leveno KJ, Gilstrap LC, Hauth JC, Wenstrom KD. Williams Obstetrics. 21st ed: McGraw-Hill Companies, Inc.; 2001.
44. Harper CC, Rocca CH, Darney PD, von Hertzen H, Raine TR. Tolerability of levonorgestrel emergency contraception in adolescents. Am J Obstet Gynecol 2004;191(4):1158-63.
45. Gold MA, Sucato GS, Conard LA, Hillard PJ. Provision of emergency contraception to adolescents. J Adolesc Health 2004;35(1):67-70.
46. Harper CC, Cheong M, Rocca CH, Darney PD, Raine TR. The effect of increased access to emergency contraception among young adolescents. Obstet Gynecol 2005;106(3):483-91.
47. Raine T, Harper C, Leon K, Darney P. Emergency contraception: advance provision in a young, high-risk clinic population. Obstet Gynecol 2000;96(1):1-7.
Sexually active adolescents are commonly seen and evaluated in the emergency department (ED), although they are often not straightforward about the reason for their visit.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.