The Role of Diet in the Mitigation of Alzheimer's Dementia
The Role of Diet in the Mitigation of Alzheimer's Dementia
By Susan T. Marcolina, MD, FACP, Dr. Marcolina is a board-certified internist and geriatrician in Issaquah, WA; she reports no consultant, stockholder, speaker's bureau, research, or other financial relationships with companies having ties to this field of study.
Part 3 of a Series on Alzheimer's Disease
As the most common type of dementia in late life, Alzheimer's disease (AD) affects about 10% of persons older than age 65 and its prevalence increases with age. The pathologic sequelae of AD is neuronal cell death due to intracellular deposits of abnormally hyperphosphorylated tau protein (a microtubule assembly protein), which results in the formation of neurofibrillary tangles that are identifiable throughout the brain, but particularly in the temporoparietal regions, at autopsy. Extracellular deposits of amyloid B peptide (A beta) incorporate degenerating neurons to form the characteristic neuritic plaques.1
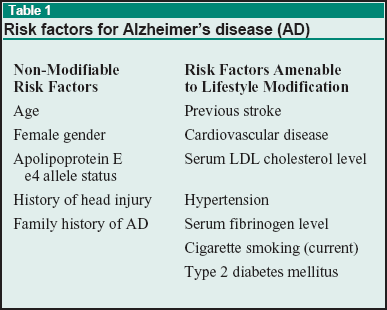
Risk factors, both genetic and environmental, have been identified for AD (see Table 1).2 Epidemiologic studies of dementia and cardiovascular disease show that both of these conditions are prevalent in elderly people and often co-exist.3,4 It is possible, then, that lifestyle modifications—such as specific dietary and exercise interventions that have been found to improve cardiovascular risk factors such as hypertension, hyperlipidemia, diabetes, and inflammation (also common risk factors for AD)—may also be useful in mitigating the clinical expression of AD, especially when initiated in mid-life or earlier.
 |
The drugs currently available for the secondary treatment of AD have only modest effects, are costly, and often are associated with problematic adverse reactions. Therefore, implementation of primary preventive strategies such as specific exercise (discussed in Part 2 of this series, see Alternative Medicine Alert August 2006) and dietary interventions to prevent comorbid diseases would be important steps to improve neurocognitive health in the context of overall improvement in cardiovascular and general medical health.
Possible Mechanisms of Disease in AD
Several studies have identified end products of lipid peroxidation5 and the presence of heme oxygenase-1 within the brains of AD patients, particularly within neurofibrillary tangles. Heme oxygenase-1 is a cellular enzyme upregulated in the brain and other tissues in response to oxidative challenges and other noxious stimuli.6
Immunohistochemical studies have shown widespread immunoreactivity for C-reactive protein (CRP) in the brains of patients with AD. Accumulated evidence reveals that CRP, rather than simply being an inflammatory marker, has a direct role in the atherogenic process. Torzewski et al localized both CRP and complement in human atherosclerotic lesions, suggesting a direct contribution to the development of atherosclerotic lesions.7
CRP is chemotactic for human monocytes and stimulates their recruitment during atherogenesis. CRP opsonizes LDL, thus mediating its uptake by macrophages, which results in the formation of foam cells. CRP also promotes vascular smooth muscle cell migration and proliferation.8 Endothelial function is further impaired by CRP by its direct attenuation of nitric oxide (NO, the key endothelium-derived relaxing factor) production and interference with endothelial NO synthase, thereby decreasing vasomotor reactivity.9
By virtue of these proinflammatory actions, elevated CRP levels in the brain are an additional cardiac risk factor responsible for cerebral macroangiopathy (large vessel disease or strokes) and microangiopathy (small vessel disease or lacunes). Both of these processes disrupt frontal-subcortical circuitry and result in dementia.10
Reactive oxygen species (ROS) are associated with neuronal damage in AD, and the possibility that ROS production is a primary event has led researchers to explore how antioxidants contained in foods and supplements may affect AD. Such supplements and dietary-based antioxidants include tocopherols (vitamin E), ascorbic acid (vitamin C), and carotenes. Vitamin E decreases amyloid beta peptide-induced lipid peroxidation and oxidative stress in vitro and down regulates inflammation-signaling cascades.9 Vitamin C blocks the formation of ROS and carotenes decrease lipid peroxidation.10 There also is evidence that intake of dietary antioxidants is associated with a decreased risk of stroke, and because stroke is a risk factor for AD, cerebrovascular disease may be an another pathway that links antioxidant vitamin intake with a decreased risk of AD.11,12
Apolipoprotein E (APOE), a gene associated with sporadic AD, influences lipid metabolism by modulation of cholesterol concentrations in response to dietary fat intake.13 It has been well established that replacement of most saturated and all dietary trans fat with monounsaturated and polyunsaturated fat, especially omega-3 fat (a polyunsaturated fat from marine or vegetable sources), lowers the risk of cardiovascular disease and could potentially lower the risk of AD via vascular and anti-inflammatory mechanisms. The marine-derived omega-3 fatty acids, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), are precursors to a group of eicosanoids (prostaglandins, leukotrienes, and thromboxanes) that have anti-inflammatory, antithrombotic, and vasodilatory effects (leukotriene B5, thromboxane A3, and prostaglandin E3, respectively).14
Dietary hydrogenated and saturated fat intake, on the other hand, promotes increased insulin resistance, which is associated with increased risk of AD by several mechanisms.15,16 Brain insulin-degrading enzyme, which regulates extracellular amyloid beta peptide levels, is inhibited by insulin. Elevated insulin levels, therefore, result in increased deposition of amyloid beta peptide in areas of the brain, such as the hippocampus, which are preferentially affected in AD. Insulin also has a role in the phosphorylation of the tau protein, the primary component of neurofibrillary tangles.17,18
Fish and Fish Oil Consumption
The human brain performs a vast array of cognitive and behavioral functions throughout a person's lifetime through a complex network of approximately 100 billion neurons and supporting glial cells (astrocytes and microglia). Despite the fact that it accounts for only 2% of adult body weight, the brain receives almost 15% of cardiac output and accounts for about 30% of the resting metabolic rate. As a result, dietary fat functions as an important source of energy that fuels cerebral metabolism. In addition, a primary component of membrane phospholipids in the brain is the omega-3 polyunsaturated fatty acid DHA (22 6n-3).19
Conquer et al in a case control study reported that the omega-3 fatty acid content of plasma phospholipids levels in patients with AD was only 60-70% of levels found in age-matched control subjects without AD. Morris et al in a prospective biracial cohort from the Chicago Health and Aging Project found that consumption of the omega-3 polyunsaturated fatty acids and at least one fish meal per week was associated with a 60% decrease in risk of AD compared to subjects who rarely or never consumed fish. Only DHA, however, was protective against the development of AD. Alpha linolenic acid (ALA), another omega-3 fatty acid derived from plant sources such as vegetable oil and flaxseed, also was protective against the development of AD, but only among persons with the APOE e4 genotype.20,21
Problems with Studies Relating Diet and AD
The biggest obstacle to relating dietary macronutrients (fats) and micronutrients (vitamins) with AD is that the length of the latency period in which dietary interventions can prevent or retard neurodegenerative disease progression is unknown, but probably spans several decades. Many studies of the association between diet and AD have been done in subjects 65 years or older, and individuals in this age group may be in an advanced stage of the latency period. In this case, the opportunity to change the course of the disease with diet may be limited. In addition, the dietary patterns reported in studies may result from preclinical cognitive problems and thereby reflect an inherent reverse causation bias. Although large, randomized, controlled prospective clinical trials overcome issues of bias and confounding and represent an ideal way to study diet and disease, the long preclinical latency of AD makes this type of study difficult to conduct.
Another important practical consideration is that it may not be just the nutrients but the whole foods, the interactions of nutrients within foods, or patterns of diet that are important in the modification of the vascular, inflammatory, and neurodegenerative processes that contribute to AD. One example of this type of synergism has been noted in the improved vascular outcomes for persons on a Mediterranean-type diet.18
Mediterranean Diet and Risk of Dementia
A Mediterranean-style diet (a diet that has olive oil as its primary fat source and is rich in fruits, vegetables, nuts, and beans) has been shown to have a significant effect on the progression of cardiovascular disease.22,23 Solfrizzi et al, in a population-based, cross-sectional study of 278 subjects aged 65-84 years, demonstrated that monounsaturated fatty acid (MUFA) intake was inversely related to cognitive decline.24 In a randomized, crossover, controlled trial of 200 healthy male volunteers 20-40 years of age, Covas et al showed that in addition to the monounsaturated fat content, the polyphenol content of virgin olive oil independently accounted for additive benefits with regard to elevation of HDL cholesterol levels and reduced oxidative damage as evidenced by reductions in oxidative biomarkers.25
Antioxidants in Supplements and in Fresh Fruit and Vegetables
There are several mechanisms by which dietary fruit and vegetable intake decrease blood pressure; in addition to this intervention being of benefit to overall cardiovascular health, benefits can be extended to neuro-cognitive health as well.
In an epidemiologic study of 7,124 persons aged 18-79 from the German National Health Interview and Examination Survey, Beitz et al found that the systolic blood pressure (SBP) of women but not men showed a significant inverse association with fruit and vegetable intake but not with supplemental vitamin C intake after adjustment for age, BMI, and smoking. Since the vitamin C plasma concentrations correlated with the amount of consumed fruit and vegetables, other factors associated with fruit and vegetable intake could have been responsible for the greater decrease in SBP such as content of vitamin E, beta carotene, potassium, magnesium, calcium, dietary fiber, or phytophenols such as flavonoids.26,27
The results of Beitz et al are comparable with the DASH trial,28 which reported lower observed levels of 24-hour ambulatory SBP and diastolic blood pressure of persons consuming a diet rich in fruits and vegetables as compared with controls. The flavonoids in fruits, vegetables, nuts, legumes, and edible oils (olive) are responsible for their yellow, green, purple, red, and blue pigmentation and function as potent antioxidants. Epidemiologic studies reveal an inverse correlation between dietary flavonoid intake and coronary heart disease mortality.29
Commenges et al followed a cohort of 1,367 patients from the PAQUID (Personnes Agées Quid) study, a longitudinal study of community residents (older than age 65) in France that examined baseline and lifetime factors related to cognitive loss and AD. All subjects were evaluated for flavonoid intakes with a validated food frequency questionnaire. After adjustment for age, gender, education, weight, and vitamin C intake, the relative risk for dementia was found to be 0.49 for the highest two tertiles of flavonoid consumption vs. the lowest (P = 0.02).30
Data from prospective studies that relate intake of supplemental antioxidant vitamins and AD show conflicting results. In a study of more than 3,000 elderly Japanese-American men in Hawaii, Masaki et al found that intake of both vitamin C and E supplements was related to low risk of vascular dementia but not AD, and that intake of either supplement was related to improved cognitive function in people without dementia.31
Zandi et al studied 4,000 persons older than age 65 and found that a combination of vitamin C and E supplements (but not individually) was associated with a low risk of AD.32 Engelhart et al found in 5,395 persons aged 55 years and older that dietary intake of vitamins C and E but not supplemental intake was associated with a low risk of AD.33 Although there was no modification of this finding with education level or APOE allele status, this association was more pronounced in smokers. Likewise, Morris et al evaluated 815 community-dwelling individuals older than 65 years and found that dietary vitamin E, but not vitamin E supplements, was associated with a low risk of AD. However, this association was only present in individuals without the APOE e4 allele.34 On the other hand, Luchsinger studied 980 community-dwelling individuals older than age 65 and found no relationship between intake of dietary and supplemental vitamins C and E and carotenes.35
Association of Homocysteine, B vitamins, and Dementia
Data from studies relating vitamins B12, B6, and folate to cognitive decline and AD are inconsistent. Seshadri et al found that homocysteine concentrations higher than 14 micromol/L doubled the risk of AD in the Framingham study, even with adjustment for age and APOE allele status; however, concentrations of folate, vitamin B6, and vitamin B12 had no relationship to the risk of AD.36 Luchsinger et al, on the other hand, found no association between homocysteine concentrations higher than 14 micromol/L and AD risk after adjustment for age.37 Kado et al, in a cross-sectional and longitudinal (six years of follow-up) analysis of a cohort of 499 high-functioning, community-dwelling persons aged 70-79, found that after multivariate adjustment only low levels of folate (from 1.17-3.15 ng/mL) were significantly associated with cognitive decline (P = 0.01).38
Based upon autopsy findings of atherosclerotic plaque formation in young persons with homocystinuria, McCully initially hypothesized in 1969 that homocysteine was the causative factor of arteriosclerotic process. This theory was later altered to include a broader population, with the premise that mild elevations in homocysteine levels caused by dietary deficiencies of folic acid, vitamin B12, and vitamin B6, necessary cofactors required for the metabolism of homocysteine, were responsible for atherosclerosis and its sequelae. In developed countries, these B vitamins were partially removed from foods with processing and the typical diets contained large amounts of methionine, which is primarily derived from animal proteins.39
Homocysteine metabolism is complicated and levels are reduced as a result of its methylation to methionine, a process promoted by folic acid and vitamin B12, cofactors to the rate-limiting activity of the methionine synthase enzyme in nonhepatic cells throughout the body. In hepatic cells, the rate-limiting enzyme cystathionine B-synthase enzyme requires vitamin B6 as a cofactor in the conversion of homocysteine to cysteine.40
Hyperhomocysteinemia has been related to cerebral microangiopathy, endothelial dysfunction,41 impaired NO activity,42 and increased oxidative stress.43 All of these factors are associated with brain aging. Additionally, homocysteic acid, a metabolite of homocysteine, is an N-methyl-D-aspartate receptor agonist and may result in neuronal excitotoxic damage and apoptosis.44
There is compelling epidemiologic evidence of an association between elevations in plasma homocysteine levels and the risk of vascular disease in the coronary and carotid circulations,45,46 and it was believed that treatment with B vitamins to decrease homocysteine levels would prevent cardiovascular events in patients at risk. However, the results of three prospective interventional studies, the Vitamin Intervention for Stroke Prevention (VISP) trial,47 the Norwegian Vitamin (NORVIT) trial,48 and the Heart Outcomes Prevention 2 Investigators (HOPE-2)49 did not show any benefit in terms of prevention of composite cardiovascular disease endpoints (myocardial infarction [MI], stroke, or sudden cardiac death) in patients supplemented with folate, B6, and B12 to decrease serum homocysteine levels.
The VISP trial studied two groups of patients (n = 3,680) after a stroke. These patients were randomized to treatment with different daily doses of vitamin B12, folic acid, and vitamin B6. After two years, although a dose-dependent decrease in homocysteine levels was seen, there were no significant differences in the rates of vascular events between the two groups. The NORVIT study was a secondary prevention, double-blind trial involving 3,749 post-MI patients randomized to either placebo; folate and vitamin B12; vitamin B6; or the combination of folate and vitamins B12 and B6. After a median follow up of 40 months, the vitamin-treated groups had a 27% mean decrease in homocysteine levels from baseline. Despite this result, the vitamin groups did not benefit from a decreased incidence of the composite endpoints. Analysis of the combination vitamin group revealed that there was a concerning trend toward increased incidence of the composite endpoints of fatal or nonfatal MI, stroke, or sudden cardiac death.
HOPE-2 Investigators was a prevention trial of 5,522 patients with vascular disease or diabetes who were treated either with a daily folic acid, B6, and B12 combination, or placebo for an average of five years. Again, although vitamin treatment did result in a substantial decrease in plasma homocysteine levels, there was no significant reduction in the risk of the composite endpoints of MI, stroke, or sudden death from CV causes. This trial also showed a marginally significant trend for reduction of stroke in the patients receiving the vitamins vs. the placebo group.
Although folate fortification of the U.S. grain supply, which began in 1996, did affect the difference in homocysteine levels between the two vitamin treatment groups in the VISP study and could have impacted the ability of the study to be sufficiently powered to detect a difference between the two groups, the consistency of the data and the results obtained in these three similar trials strongly shows that there is no apparent clinical benefit to B vitamin supplementation in patients with vascular disease; indeed, there is some suggestion that they might cause harm.47
One mechanism by which folate supplementation may exacerbate vascular disease is through its promotion of cell proliferation in the atherosclerotic plaque. This mechanism has been proposed as an explanation for the results of a study by Lange et al. In this study, 636 angioplasty patients post-stent placement were supplemented with folate, B12, and B6 or placebo in a double-blind fashion for six months. It was found that patients who received supplements had a greater degree of restenosis within the stent compared to the patients in the placebo arm.50
Warnings Regarding Fish Consumption
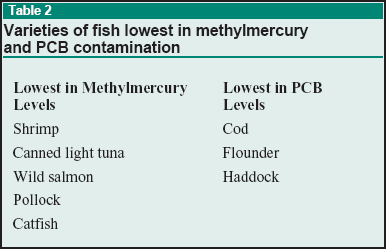
Fish are an important source of dietary protein and polyunsaturated fats. Industrial pollution, however, has resulted in contamination of waterways with organic methylmercury and organochlorine derivatives such as dioxin and polychlorinated biphenyls (PCBs), which can cause deleterious health effects, particularly for pregnant females and children. For this reason, the FDA has recommended limiting fish consumption to 12 ounces (two average meals) per week of a variety of fish with low levels of these contaminants (see Table 2). Fish affected by PCB and dioxin contamination vary by location in the United States; consumers are advised to follow local state advisories at www.epa.gov/waterscience/fish/states.htm.51 Of note, PCBs and dioxin are concentrated in the fat of fish. Therefore, the amount of contaminants ingested can be reduced by proper preparation and cooking procedures. The fish should be broiled, baked, or poached so that the fat drippings can be separated. These drippings should not be used to prepare sauces or gravies. Methylmercury, on the other hand, is distributed throughout a fish's muscle tissue. The only way to avoid contamination is to limit intake of known types of contaminated fish from specific regions/bodies of water.52,53
 |
Dietary and Multivitamin Dosage Recommendations
The American Heart Association recommends an intake of 1.5 g of omega-3 fatty acids daily for cardiovascular health.54 A general multivitamin supplement can be recommended but additional B vitamins should not be recommended for persons with established vascular disease given the trend noted in the NORVIT study for increased composite cardiovascular events (MI, stroke, sudden cardiac death) in patients given folate, B12, and B6 supplementation to decrease serum homocysteine levels. Everyone should try to follow the general dietary guidelines outlined in the Dietary Guidelines for Americans 2005 (www.healthierus.gov/nutrition.html). The optimal way for patients to obtain the daily requirements of essential vitamins is to eat a balanced diet that contains a variety of foods (including five servings of a variety of fruits and vegetables) from the Food Guide Pyramid, which can be individually customized at www.mypyramid.gov/mypyramid/index.aspx.
Until prospective interventional studies determine whether it is important to decrease homocysteine blood levels and by what means to safely decrease these levels, it is reasonable to recommend that patients with cardiovascular and neurodegenerative risk factors substitute a variety of plant-based proteins such as soy, legumes, seeds, nuts, and whole grains for certain animal-based proteins such as red meat as a means by which to decrease dietary saturated fat and methionine, a precursor for homocysteine.55
Conclusion
Diet and lipid metabolism are important modifiable factors in the complex etiology of AD and involve effects upon interdependent vascular, inflammatory, and antioxidant processes. Because there is evidence implicating vascular disease as an underlying etiology for AD, there are several potentially effective dietary interventions, such as:
- Replacement of dietary trans and saturated fat with polyunsaturated (primarily omega-3) and monounsaturated fats,
- Addition of a variety of fruits, nuts, and vegetables to the diet, which may have multiple effects upon blood pressure and systemic inflammation (especially if used in conjunction with monounsaturated fats, as in the Mediterranean diet), and
- Substitution of certain animal-based proteins such as red meat with a variety of plant-based proteins to limit intake of saturated fat and the homocysteine precursor methionine.
Genetic vulnerability relative to an individual's APOE e4 genotype may modify many of these risks and such interaction between environment and genetics may affect clinical outcomes.
Recommendation
Health care professionals should encourage all of their patients to adopt heart and brain healthy dietary practices including limitation of saturated and trans fatty acids and replacement with monounsaturated (especially virgin olive oil) and polyunsaturated (especially omega-3) fats; inclusion of five servings or more of a variety of fruits and vegetables; and replacement of certain animal-based proteins such as red meat with a variety of plant-based proteins to limit intake of saturated fat and methionine.
References
1. Dickson TC, Vickers JC. The morphological phenotype of beta-amyloid plaques and associated neuritic changes in Alzheimer's disease. Neuroscience 2001;105:99-107.
2. Luchsinger JA, et al. Aggregation of vascular risk factors and risk of incident Alzheimer's disease. Neurology 2005;65:545-551.
3. Elias PK, et al. NIDDM and blood pressure as risk factors for poor cognitive performance. The Framingham Study. Diabetes Care 1997;20:1388-1395.
4. Brayne C, et al. Vascular risks and incident dementia: Results from a cohort study of the very old. Dement Geriatr Cogn Disord 1998;9:175-180.
5. Marcus DL, et al. Increased peroxidation and reduced anti-oxidant enzyme activity in Alzheimer's disease. Exp Neurol 1998;150;40-44.
6. Smith MA, et al. Heme oxygenase-1 is associated with the neurofibrillary pathology of Alzheimer's disease. Am J Pathol 1994;145:42-47.
7. Torzewski J, et al. C-reactive protein frequently colocalizes with the terminal complement complex in the intima of early atherosclerotic lesions of human coronary arteries. Arterioscler Thromb Vasc Biol 1998;18:1386-1392.
8. Hattori Y, et al. Vascular smooth muscle cell activation by C-reactive protein. Cardiovasc Res 2003;58:186-195.
9. Venugopal SK, et al. Demonstration that C-reactive protein decreases eNOS expression and bioactivity in human aortic endothelial cells. Circulation 2002;106:1439-1441.
10. Kuo HK, et al. Relation of C-reactive protein to stroke, cognitive disorders, and depression in the general population: Systemic review and meta-analysis. Lancet Neurol 2005;4:371-380.
11. Butterfield DA, et al. Vitamin E and neurodegenerative disorders associated with oxidative stress. Nutr Neurosci 2002;5:229-239.
12. Pitchumoni SS, Doraiswamy PM. Current status of antioxidant therapy for Alzheimer's Disease. J Am Geriatr Soc 1998;46:1566-1572.
11. Vermeer SE, et al. Silent brain infarcts and the risk of dementia and cognitive decline. N Engl J Med 2003;348:1215-1222.
12. Ascherio A. Antioxidants and stroke. Am J Clin Nutr 2000;72:337-338.
13. Berglund L. The APOE gene and diets—food (and drink) for thought. Am J Clin Nutr 2001;73:669-670.
14. Lands WEB. Biochemistry and physiology of n-3 fatty acids. FASEB J 1992;6:2530-2536.
15. Bray GA, et al. The influence of different fats and fatty acids on obesity, insulin resistance and inflammation. J Nutr 2002;132:2488-2491.
16. Luchsinger JA, et al. Hyperinsulinemia and risk of Alzheimer disease. Neurology 2004;63:1187-1192.
17. Park CR. Cognitive effects of insulin in the central nervous system. Neurosci Biobehav Rev 2001;25:311-323.
18. Luchsinger JA, Mayeux R. Dietary factors and Alzheimer's disease. Lancet Neurol 2004;3:579-587.
19. Connor WE, et al. Dietary effects on brain fatty acid composition: The reversibility of n-3 fatty acid deficiency and turnover of docosahexaenoic acid in the brain, erythrocytes, and plasma of rhesus monkeys. J Lipid Res 1990;31:237-247.
20. Conquer JA, et al. Fatty acid analysis of blood plasma of patients with Alzheimer's disease, other types of dementia, and cognitive impairment. Lipids 2000;35:1305-1312.
21. Morris MC, et al. Consumption of fish and n-3 fatty acids and risk of incident Alzheimer disease. Arch Neurol 2003;60:940-946.
22. Robertson RM, Smaha L. Can a Mediterranean-style diet reduce heart disease? Circulation 2001;103:1821-1822.
23. Chrysohoou C, et al. Adherence to the Mediterranean diet attenuates inflammation and coagulation process in healthy adults: The ATTICA Study. J Am Coll Cardiol 2004;44:152-158.
24. Solfrizzi V, et al. High monounsaturated fatty acid intake protects against age-related cognitive decline. Neurology 1999;52:1563-1569.
25. Covas MI, et al. The effect of polyphenols in olive oil on heart disease risk factors: A randomized trial. Ann Intern Med 2006;145:333-341.
26. Beitz R, et al. Blood pressure and vitamin C and fruit and vegetable intake. Ann Nutr Metab 2003;47:214-220.
27. Hermansen K. Diet, blood pressure and hypertension. Br J Nutr 2000;83(suppl 1):S113-S119.
28. Sacks FM, et al. A dietary approach to prevent hypertension: A review of the Dietary Approaches to Stop Hypertension (DASH) Study. Clin Cardiol 1999;22(7 suppl):III6-III10.
29. Ness AR, Powles JW. Fruit and vegetables and cardiovascular disease: A review. Int J Epidemiol 1997;26:1-13.
30. Commenges D, et al. Intake of flavonoids and risk of dementia. Eur J Epidemiol 2000;16:357-363.
31. Masaki KH, et al. Association of vitamin E and C supplement use with cognitive function and dementia in elderly men. Neurology 2000;54:1265-1272.
32. Zandi PP, et al. Reduced risk of Alzheimer disease in users of antioxidant vitamin supplements: The Cache County Study. Arch Neurol 2004;61:82-88.
33. Engelhart MJ, et al. Dietary intake of antioxidants and risk of Alzheimer disease. JAMA 2002;287:3223-3229.
34. Morris MC, et al. Vitamin E and cognitive decline in older persons. Arch Neurol 2002;59:1125-1132.
35. Luchsinger JA, et al. Antioxidant vitamin intake and risk of Alzheimer disease. Arch Neurol 2003;60:203-208.
36. Seshadri S, et al. Plasma homocysteine as a risk factor for dementia and Alzheimer's disease. N Engl J Med 2002;346:476-483.
37. Luchsinger JA, et al. Plasma homocysteine levels and risk of Alzheimer disease. Neurology 2004;62:1972-1976.
38. Kado DM, et al. Homocysteine versus the vitamins folate, B6, and B12 as predictors of cognitive function and decline in older high-functioning adults: MacArthur Studies of Successful Aging. Am J Med 2005;118:161-167.
39. McCully KS. Vascular pathology of homocysteinemia: Implications for the pathogenesis of arteriosclerosis. Am J Pathol 1969; 56:111-128.
40. Loscalzo J. Homocysteine trials—clear outcomes for complex reasons. N Engl J Med 2006;354:1629-1630. Epub 2006 Mar 12.
41. Chao CL, et al. Effects of methionine-induced hyperhomocysteinemia on endothelium-dependent vasodilation and oxidative status in healthy adults. Circulation 2000;101:485-490.
42. McCann SM. The nitric oxide hypothesis of brain aging. Exp Gerontol 1997;32:431-440.
43. Beal MF. Aging, energy, and oxidative stress in neurodegenerative diseases. Ann Neurol 1995;38:357-366.
44. Lipton SA, et al. Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor. Proc Natl Acad Sci U S A 1997;94:5923-5928.
45 Stampfer MJ, et al. A prospective study of plasma homocyst(e)ine and risk of myocardial infarction in US physicians. JAMA 1992;268:877-881.
46. Selhub J, et al. Association between plasma homocysteine concentrations and extracranial carotid artery stenosis. N Engl J Med 1995;332:286-291.
47. Toole JF, et al. Lowering homocysteine in patients with ischemic stroke to prevent recurrent stroke, myocardial infarction, and death: The Vitamin Intervention for Stroke Prevention (VISP) randomized controlled trial. JAMA 2004;291:565-575.
48. Bonaa KH, et al. Homocysteine lowering and cardiovascular events after acute myocardial infarction. N Engl J Med 2006;354:1578-1588. Epub 2006 Mar 12.
49. Lonn E, et al, The Heart Outcomes Prevention Evaluation (HOPE) 2 Investigators. Homocysteine lowering with folic acid and B vitamins in vascular disease. N Engl J Med 2006;354:1567-1577.
50. Lange H, et al. Folate therapy and in-stent restenosis after coronary stenting. N Engl J Med 2004;350:2673-2681.
51. U.S. Environmental Protection Agency. Fish Advisories. Available at: www.epa.gov/waterscience/fish/states.htm. Accessed Sept. 10, 2006.
52. New York State Department of Health: 2006-2007 Health Advisories: Chemicals in Sportfish and Game. Available at: www.health.state.ny.us/nysdoh/fish/fish.htm. Accessed Sept. 8, 2006.
53. Wilson JF. Balancing the risk and benefits of fish consumption. Ann Intern Med 2004;141:977-980.
54. Krauss RM, et al. AHA Dietary guidelines: Revision 2000: A statement for healthcare professionals from the Nutrition Committee of the American Heart Association. Circulation 2000;102:2284-2299.
55 U.S. Food and Drug Administration. More People Trying Vegetarian Diets. Available at: www.fda.gov/fdac/features/895_vegdiet.html#animal. Accessed Sept. 2, 2005.
Marcolina ST. The role of diet in the mitigation of Alzheimer's disease. Altern Med Alert 2006;9(10):109-115.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
