Clostridium Difficile-Associated Disease
Clostridium Difficile-Associated Disease
Author: Cynthia A. Chan, MD, FAAP, Department of Emergency Medicine, Case Western Reserve University, MetroHealth Medical Center, Cleveland, OH.
Peer Reviewers: Larry B. Mellick, MD, MS, FAAP, FACEP, Professor, Department of Emergency Medicine, Residency Program Director, Department of Emergency Medicine, Medical College of Georgia, Augusta; and Ann M. Dietrich, MD, FAAP, FACEP, Department of Emergency Medicine, Children's Hospital, Columbus, OH.
We are all aware of the negative effects of unnecessary antibiotics. Antibiotic resistance is a current crisis in healthcare. MRSA and VRE are terms that were simply unheard of 20 years ago. Now they are part of our daily clinical life. Beyond antibiotic resistance, use of antibiotics may lead to Clostridium difficile. The diarrhea and associated dehydration caused by C. difficile can be more deadly than the original infection. Further, we have learned that C. difficile can be transmitted, often by healthcare workers.
—The Editor
Introduction
A 24-month-old boy comes into the emergency department (ED) with watery diarrhea and decreased oral intake for 3 days. He attends preschool, has been around other sick children and recently had an ear infection which was treated. Is this another case of viral infection, or should you be concerned about another cause?
Epidemiology
Clostridium difficile causes 10-30% of antibiotic-associated diarrhea and is the most common cause of nosocomial diarrhea.1 This spore-forming, gram-positive anaerobe also causes pseudomembranous colitis.
Classically associated with clindamycin, epidemic outbreaks recently have been reported in the United States, Canada, and Europe, causing more morbidity and mortality than in the past.2 There have been C. difficile epidemic strains resistant to clindamycin causing hospital outbreaks.3 Current strains now are resistant to fluoroquinolones, such as gatifloxacin and moxifloxacin, as reported in a six-state study, causing outbreaks from 2000-2003.4 The epidemic Quebec strain of C. difficile showed resistance to fluoroquinolones in 52% of the cases, alone or in combination with other antibiotics. Higher rates of severe outcomes in this epidemic also were reported with complications of toxic megacolon, shock, colectomy, and death.5
Extra health-care costs can amount to more than $3600 per case of Clostridium difficile-associated disease (CDAD), and in the United States, more than $1 billion per year.6
Studies from Quebec in the past four years have reported epidemics causing increased frequency and severity of CDAD. A retrospective review over a 13-year span reported increased incidence, 102 cases per 100,000 in 1991 to 866 cases per 100,000 in 2003, especially in patients older than 65 years and who had been exposed to fluoroquinolones. These severe cases also had higher mortality, 13.8%.7
Another report of an epidemic Quebec strain in 2002 documented that nosocomial cases were 5 times greater than in the past, with increased morbidity (2.3 times more likely to have severe outcomes such as intensive care unit (ICU) admission, colectomy, or death) and mortality up 400% since 1997, causing deaths in nearly 6% of CDAD cases.8 Healthcare-associated CDAD also has increased by 26% and in severity in 2000-2001.4
The usual risk factors no longer necessarily apply. Community-acquired CDAD, diarrhea with evidence of C. difficile by toxin, stool culture, or histopathology, with no history of a severe, chronic underlying illness or hospitalization in the past 3 months, also has increased in the past decade. Coincidentally, over that same time period, antibiotic prescriptions decreased and proton pump inhibitor use increased. In 2005, the CDC reported cases of severe CDAD in otherwise previously healthy patients (children and peripartum women), some of whom had little or no risk, e.g., no antibiotic exposure or just one prophylactic antibiotic dose in the prior 3 months. Some common features of these cases were young age, exposure to close contacts, lack of antibiotic exposure, bloody diarrhea, and a high recurrence rate of 39%.9 (See Table 1.)
| Table 1. Community-Acquired CDAD Features |
| • Young age • Exposure to close contacts • Lack of antibiotic exposure • Bloody diarrhea • High recurrence rate |
In a prospective study, a majority of community-acquired CDAD patients, 86.8%, were hospitalized in the previous 12 months.10
Not as many pediatric studies have been done as in the adult population. Almost 7% of inpatient children tested positive for C. difficile, but they either had mild antibiotic-associated diarrhea or were infants with high colonization.11
Etiology
C. difficile is commonly associated with antibiotic use. In 90-96% of the cases, patients had exposure to antibiotics, at times after one single prophylactic dose. Chances were higher if they were exposed to 3 or more antibiotics.12 The most common antibiotic classes implicated were cephalosporins (~75-84%), macrolides, clindamycin, and quinolones.10,13,14 Even metronidazole and vancomycin can contribute to antibiotic-associated colitis, but not as often.13 A study comparing oral to intravenous preoperative antibiotics prior to colorectal surgery showed that CDAD rates were higher in the patients who received oral antibiotics.15
In several studies, proton pump inhibitors (PPIs) as well as prolonged PPI exposure (> 6 months) were significant risk factors even if patients had not been hospitalized in the previous year.12,17 PPIs decrease the gastric acid, which is otherwise protective against gastrointestinal (GI) illnesses, such as Salmonella, and suppresses upper GI bacterial overgrowth.19 Lowering the gastric acidity helps C. difficile and its toxin survive.20 H2 blockers, though, were not found to be a significant risk for C. difficile, as they are not as potent a gastric acid suppressant as PPIs.17
Patients who have comorbidities or severe illness are at risk.16 Those having history of renal failure, malignancy, inflammatory bowel disease or MRSA culture positive have a greater chance of developing CDAD.11,17 Patients immunosuppressed with HIV or transplants cannot mount appropriate immune responses to C. difficile.13,21,22
With exposure to frequent antibiotics and the hospital environment, several factors put patients at risk for hospital-acquired C. difficile. These patients were more likely to be hospitalized in the prior 3 months.12 Sharing a hospital room with a patient who is culture positive puts the roommate at higher risk even if asymptomatic. In fact, sharing a room with an asymptomatic roommate poses a higher risk than sharing with a symptomatic roommate (61% vs 39%). Hospitalized patients acquire C. difficile 6 times faster with roommate exposure than without.18
In the hospital setting, other risks were found, including enteral tube feedings and ICU admission (in > 500-bed hospitals).23 ICU patients may have more severe underlying conditions, extended length of stay, medical devices, or admission during the winter months when there is a higher patient census and more antibiotics are used for respiratory infections. At smaller hospitals without ICUs, hospitalizations on the medicine services posed more risk with exposure to antibiotics and patients at risk for CDAD on those wards.24
Heathcare workers may actually help to spread the disease. A source of contamination is on the hands of hospital personnel. Fifty-nine percent of workers tested positive for C. difficile from hand cultures after coming into contact with C. difficile culture-positive patients, even if they were asymptomatic. Hand cultures, from personnel such as physical therapists, nurses, medical students, and physicians, tested positive from underneath fingernails, the palms, fingertips, and even under rings.18
C. difficile can transmit from patient to patient via contaminated instruments like stethoscopes and thermometers. It also persists on hospital environment surfaces after contamination when the room was occupied by symptomatic patients or asymptomatic carriers. Surfaces even tested positive in 8% of cultures taken where no C. difficile culture-positive patient had occupied the space for more than 48 hours.18
In children diagnosed with CDAD, common factors were hospitalization or antibiotics (70% of cases) in the previous 2 months, as antibiotics often are used for otitis media. Similar to adults, immunosuppressed children were more at risk: those receiving chemotherapy, solid organ transplant, frequent antibiotic use, or immunodeficiency, inhibiting response to infection.25
Pathophysiology
The C. difficile spores are found widely in the environment, including bathrooms, hospital wards, and nurseries. Exposure to antibiotics suppresses the gut's normal flora, mostly Bacteroides (> 90%), allowing pathogenic C. difficile to multiply and produce toxins. Not all strains of C. difficile produce toxins or are pathogenic.26 After ingestion, the spores germinate in the small bowel and colonize the lower GI tract.27 Toxin B damages the colonic mucosa, causing necrosis, shedding and cell injury. It also forms pseudomembranes, while toxin A causes the watery diarrhea.26,28 The epidemic C. difficile strain produces 16 times more toxin A and 23 times more toxin B and at a faster rate than controls.2,4
Infection, then, also depends upon a person's immune response and ability to develop antibodies to the toxins. People producing antibody to toxin A are better protected than those with low antibody titers, who are more susceptible to prolonged or relapsing CDAD infection.26,29 Asymptomatic carriers of C. difficile constantly shed toxigenic organisms, contaminating the environment. Three to five percent of the general adult population and up to two-thirds of hospitalized patients are colonized.18,26,28 The majority of infants become colonized and develop antibodies to the toxins in the first 6-12 months.26 In children, 3.5% are asymptomatic carriers.30
Clinical Features
While the clinical features of CDAD are straightforward, they also are somewhat non-specific. A low-grade fever (28%) can accompany lower abdominal pain or cramps (22%) and watery or soft diarrhea with possible mucus up to 20 times per day.27,31 Patients also may experience malaise, nausea, or vomiting.11 Leukocytosis occurs in 50% of the cases.27 Symptoms usually begin after 5-10 days of antibiotics, but can range from one day to 10 weeks after the antibiotic course.32 Severe cases of CDAD may present without diarrhea as with toxic megacolon, having ascites and low albumin.33 In 2005, the MMWR reported previously healthy pregnant women presenting with prolonged diarrhea that progressed to severe colitis, shock, and eventually death despite appropriate treatment.9 CDAD can manifest outside of the colon, usually in patients with comorbid illness, presenting with fever and local pain, without diarrhea. Such cases include bacteremia, intra-abdominal abscess (e.g., colon), or extra-abdominal abscess (e.g., foot infection, brain abscess).34 Risk factors and clues that might alert physicians to complicated CDAD are age greater than 65 years, history of recent hospitalization or surgery in the preceding 2 months, tube feedings in the past 2 months, immunosuppression, leukocytosis, and elevated creatinine.
Children also can have bloody stools.25 In the MMWR in 2005, more severe cases requiring hospitalization also were reported in children who had exposure to a sick contact with a self-resolving febrile diarrheal illness, but no exposure to antibiotics and up to 14 stools per day. Consider CDAD in a child with prolonged and intractable diarrhea. Pediatric CDAD has lower mortality than in adults, but can recur in 31% of cases.25
Nosocomial CDAD has been defined as: diarrhea, 5 or more bowel movements in 36 hours, as well as antibiotics in the previous 8 weeks; the detection of C. difficile toxin; and a response to therapy.13 Late-onset CDAD, is defined as occurring 2 weeks after exposure. Hospitalized children with CDAD have severe diarrhea for at least 2 days, most often accompanied by abdominal pain as well as nausea, vomiting, decreased appetite, abdominal distention, cramping, urgency, or tenesmus.11 Interestingly, the stool frequency is less for nosocomial CDAD or hospitalized patients than for community-acquired CDAD.
Diagnosis
Consider testing patients for C. difficile especially if they have any risk factors mentioned, exposure to antibiotics, hospitalization, etc. In the pediatric population, while other infectious or drug-induced causes are more common than C. difficile, if there is a high suspicion, test children older than 1 year who have had severe diarrhea for at least 2 days, especially if accompanied by any abdominal symptoms.11
The C. difficile cytotoxin assay tests for toxin B, having sensitivity of 67-100% and specificity of 85-100%, with results in 48-72 hours.35 False negatives have been found in 17-55% of cases, even in cases of pseudomembranous colitis.10,36 Positive stool cytotoxin assays are more likely in symptomatic patients rather than asymptomatic carriers.18 Therefore, it is recommended that asymptomatic patients not be tested, or to perform "tests of cure."27
Enzyme immunoassay is 70-95% specific and rapid, but has a sensitivity of 50-90% (it cannot detect low toxin amounts). If an initial enzyme immunoassay is negative, it is not likely that a repeat test would convert to a positive, so repeat testing is not recommended.13,37,38
Flexible sigmoidoscopy helps diagnose pseudomembranous colitis in the patient group having continued diarrhea despite a negative cytotoxin test. Some pseudomembranous changes can spare the rectum, so this procedure can check the sigmoid colon.10,39
Stool culture is not often used clinically.40 Leukocytosis is evident, especially in severe cases. Include CDAD in the differential in a patient having leukocytosis of unknown origin.41
Treatment
Healthy carriers without symptoms do not require treatment.42 Otherwise, conservative treatment starts with stopping antibiotics if possible and providing supportive therapy, fluids, etc. If the patient has other more serious infections requiring antibiotics, such as infectious endocarditis, he or she may need to continue that therapy. Antiperistaltic drugs are not advised as they may exacerbate or prolong the illness.
Patients with diarrhea, fever, and leukocytosis can start on oral metronidazole 500 mg 3 times daily (or 250 mg 4 times daily) for 10-14 days. Metronidazole and vancomycin are equally effective, with response rates of 90-97%. Because metronidazole is less expensive and vancomycin use potentially selects for vancomycin-resistant enterococci, metronidazole is recommended for first-line CDAD treatment.43,44
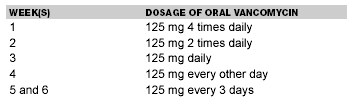
Vancomycin is indicated for patients with metronidazole allergy or intolerance, pregnancy, breastfeeding, and in children younger than 10 years. The oral vancomycin dose is 125 mg 4 times daily for 10 days.45 In pregnant women, metronidazole crosses the placenta and there is not enough evidence to support its safety for them, especially during the first trimester.44
If a patient has PO intolerance, intravenous metronidazole 500 mg 4 times per day effectively reaches appropriate concentrations in the intestine, whereas intravenous vancomycin does not.44,46,47
With an appropriate response to therapy, expect the patient to defervesce on day 1 and the diarrhea to resolve in 4-5 days.47 Treatment failure can be considered when the patient shows no improvement after 6 days of antibiotics.27 Resistance to metronidazole is rare and typically does not cause treatment failure.27
If CDAD recurs, the same antibiotic regimen should be used again. Use of either metronidazole or vancomycin has similar risk for recurrence of CDAD.48 If, however, a second or greater recurrence is being treated, several regimens have been recommended. One is metronidazole or vancomycin with Saccharomyces boulardii 500 mg (3 x 1010 colony-forming units) twice per day orally.45 Another regimen for multiple recurrences is vancomycin 125 mg 4 times daily with rifampin 300 mg twice daily, both oral, for 10 days.27 Tapering and pulsed antibiotic therapy are used to overcome persistent spores that produce toxins after an antibiotic course is stopped or completed. (See Table 2.) The days off antibiotics let the spores vegetate, then the antibiotic days kill the spores.42 Patients on this oral vancomycin regimen did not relapse in the subsequent 6 months of follow-up.
| Table 2. Tapering and Pulsed Antibiotic Therapy32 |
|
Probiotics are live nonpathogenic microorganisms that, when ingested, may produce some therapeutic or preventive health benefits by promoting and improving balance in intestinal microflora. S. boulardii, mentioned above, is one that decreases the risk of further recurrences of CDAD by 30%. Some of the side effects are dry mouth and constipation.45,49 Fungemia was reported with its use in patients with comorbid conditions, so caution should be used in these situations.50
In cases complicated by ileitis or toxic megacolon, the complication itself may inhibit antibiotics from achieving therapeutic levels. Higher doses of oral vancomycin, 500 mg every 6 hours, vancomycin by enema, nasogastric tube, or intravenous metronidazole may be used.13 With no response to these treatments, severe illness, or perforation, a colostomy may be necessary.27
In severe cases, other treatments may include immunoglobulin therapy and bacteriotherapy. Immunoglobulins aim to boost one's immune response to CDAD. For pseudomembranous colitis or multiple CDAD recurrences refractory to treatment, bacteriotherapy has had benefit. Bacteriotherapy infuses normal fecal flora, for example by nasogastric tube, to re-introduce the gut to normal flora, such as Bacteroides, which has an antagonistic effect on C. difficile.42,51 (This inexpensive method prevents further colonization and offers long-term remission.)43
Prevention
Infection control starts with prevention and careful use of antibiotics, including broad-spectrum ones.27 Care should be taken with contact precautions. C. difficile spores are resistant to alcohol-based hand hygiene solutions, so soap and water or chlorhexidine gluconate should be used to wash hands. The combination of both gloves and hand washing was found to be the most effective way to prevent transmission.18 If clothing soiling is likely, use gowns. Recommendations also include using dedicated instruments like stethoscopes,13 or in the ED with its high patient turnover, recommendations may necessitate cleaning one's own stethoscope with something other than an alcohol pad. Symptomatic patients also should have a private room, if available.9
Environmental cleaning needs special attention with approved hospital disinfectants because the hospital environment is frequently contaminated and the hardy spores persist on surfaces.52 Dilute bleach or phosphate-buffered hypochlorite help to eliminate C. difficile spores.18
Differential Diagnosis
Antibiotics themselves may cause a diarrhea without C. difficile infection. The antibiotics change the intestine's normal function of breaking down carbohydrates, causing an osmotic diarrhea. Typically, the diarrhea is not as severe as that with CDAD and resolves with stopping the antibiotics.12 (See Table 3.)
| Table 3. Clues to Differentiate Osmotic Diarrhea vs. CDAD53 |
|
|
In descending order of frequency, following antibiotic-induced diarrhea are: possible infectious gastroenteritis, unknown etiology, constipation with overflow diarrhea, Campylobacter, inflammatory bowel disease, Salmonella, ischemic colitis, and irritable bowel syndrome.10 If symptoms develop after a colectomy procedure, also consider mesenteric infarction and anastomotic leak.54
In the pediatric population, diarrhea often is secondary to dietary habits or changes, causing toddler's diarrhea or chronic nonspecific diarrhea of childhood. Osmotic diarrhea can be caused by consumption of a low-fat diet, high in carbohydrates and fluids, such as lots of fruit juice. A recent prospective study identified a pathogen in only 20% of community diarrheal cases. Of infectious causes of diarrhea in children, viruses are most common, e.g. enteric adenovirus and rotavirus. The most common bacteria isolated from diarrheal samples were atypical EPEC, EAEC, and C. perfringens. C. difficile was not found to be a significant cause of diarrheal illnesses in the community.30 Hospitalized pediatric patients can be exposed to therapies that could cause the side effect of diarrhea: laxatives, motility agents, tube feedings, and chemotherapy. Other possible causes are abdominal surgery and inflammatory bowel disease.11
Complications
Nosocomial CDAD itself prolongs illness, for which one may need to transfer to an "extended care facility" from the hospital.18 Risk factors for complicated CDAD are older age, leukocytosis, and if a second or more recurrence, history of renal failure with a second episode.48
A recurrence happens within 2 months of a prior episode caused by a relapse of the initial infection or reinfection, acquiring a different infection.48 Relapse occurs in 10-25% of patients treated with metronidazole or vancomycin, and of these, 65% have multiple relapses up to 10 or more episodes.42,51 Recurrent diarrhea, abdominal pain, cramps, or fever usually 1-2 weeks after stopping antibiotics or up to 1-2 months later suggests a relapse.42,47
Many of the risks for recurrence reflect those predisposing one to the initial episode, age older than 65 years, history of severe CDAD, severe comorbid conditions, diabetes mellitus, chronic renal failure, and immunosuppression. Other risks are history of prolonged antibiotics, other antibiotics after CDAD therapy, proton pump inhibitors or leukocytosis.12,16,48 Pediatric CDAD has fewer complications and less severity than adult cases. Few cases are complicated by prolonged hospitalization, ICU admission, exploratory laparotomy, or death from sepsis.25 Relapse occurs in 31% of children.
Thirty percent of relapses advance to pseudomembranous colitis.10 Other severe complications can occur despite appropriate therapy. CDAD can lead to sepsis with fever, hypotension, tachycardia, and leukocytosis.27 Fulminant colitis is rare and presents as an acute surgical abdomen without diarrhea.13,27 If a patient has a history of antibiotics in the prior 2 months with symptoms of obstruction, leukocytosis more than 25,000, bowel thickening, and ascites on CT scan, consider toxic megacolon.27,55
Other possible complications are ICU admission, acute renal failure, perforation, colectomy, shock, or death.12,48 Some other rare complications occur more in patients who have had history of colectomy, such as ileitis or ileal perforation.54
Summary
Clostridium difficile-associated disease with its recent epidemic strains has come to the forefront. Being aware of regional trends of infection and remembering CDAD in the differential when relevant may help to prompt early treatment and limit its spread. Suspect a patient for CDAD who has a history of any of the following risk factors: recent hospitalization, older age, antibiotic exposure, underlying illness.13 Consider checking fecal leukocytes and C. difficile cytotoxin. Stop antibiotics if possible and inform the patient of contact precautions. Good hand washing with soap and water is essential to stopping the spread of disease.
References
1. Thielman NM, Wilson RH. Antibiotic-associated colitis. In: Mandell GL, Bennett JE, Dolin R. Principles and Practice of Infectious Diseases, 6th ed. Philadelphia, PA: Churchill Livingstone; 2005:1252-1253.
2. Warny M, Pepin J, Fang A, et al. Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet 2005;366:1079-1084.
3. Johnson S, Samore MH, Farrow KA, et al. Epidemics of diarrhea caused by a clindamycin-resistant strain of Clostridium difficile in four hospitals. N Engl J Med 1999;341:1645-1651.
4. McDonald LC, Killgore GE, Thompson A, et al. An epidemic, toxin gene-variant strain of Clostridium difficile. N Engl J Med 2005;353:2433-2441.
5. Loo VG, Poirier L, Miller MA, et al. A predominantly clonal multi-institutional outbreak of Clostridium difficile-associated diarrhea with high morbidity and mortality. N Engl J Med 2005;353:2442-2449.
6. Kyne L, Hamel MB, Polavaram R, et al. Health care costs and mortality associated with nosocomial diarrhea due to Clostridium difficile. Clin Infect Dis 2002;34:346-353.
7. Pepin J, Valiquette L, Alary ME, et al. Clostridium difficile-associated diarrhea in a region of Quebec from 1991 to 2003: A changing pattern of disease severity. CMAJ 2004;171:466-472.
8. Eggertson L. Quebec strain of C. difficile in 7 provinces. CMAJ 2006;174:607-608.
9. Chernak E, Johnson CC, et al. Severe Clostridium difficile-associated disease in populations previously at low risk—four state, 2005. MMWR Weekly 2005;54:1201-1205.
10. Johal SS, Hammond J, Solomon K, et al. Clostridium difficile associated diarrhoea in hospitalised patients: Onset in the community and hospital and role of flexible sigmoidoscopy. Gut 2004;53:673-677.
11. Spivack JG, Eppes SC, Klein JD. Clostridium difficile-associated diarrhea in a pediatric hospital. Clin Pediatr (Phila) 2003;42:347-352.
12. Dial S, Alrasadi K, Manoukian C, et al. Risk of Clostridium difficile diarrhea among hospital inpatients prescribed proton pump inhibitors: Cohort and case-control studies. CMAJ 2004;171:33-38.
13. Bouza E, Munoz P, Alonso R. Clinical manifestations, treatment and control of infections caused by Clostridium difficile. Clin Microbiol Infect 2005;11 Suppl 4:57-64.
14. Pepin J, Saheb N, Coulombe MA, et al. Emergence of fluoroquinolones as the predominant risk factor for Clostridium difficile-associated diarrhea: A cohort study during an epidemic in Quebec. Clin Infect Dis 2005;41:1254-1260.
15. Wren SM, Ahmed N, Jamal A, et al. Preoperative oral antibiotics in colorectal surgery increase the rate of Clostridium difficile colitis. Arch Surg 2005;140:752-756.
16. Kyne L, Sougioultzis S, McFarland LV, et al. Underlying disease severity as a major risk factor for nosocomial Clostridium difficile diarrhea. Infect Control Hosp Epidemiol 2002;23:653-659.
17. Dial S, Delaney JA, Barkun AN, et al. Use of gastric acid-suppressive agents and the risk of community-acquired Clostridium difficile-associated disease. JAMA 2005;294:2989-2995.
18. McFarland LV, Mulligan ME, Kwok RY, et al. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med 1989;320:204-210.
19. Williams C. Occurrence and significance of gastric colonization during acid-inhibitory therapy. Best Pract Res Clin Gastroenterol 2001;15:511-521.
20. Gurian L, Ward TT, Katon RM. Possible foodborne transmission in a case of pseudomembranous colitis due to Clostridium difficile: Influence of gastrointestinal secretions on Clostridium difficile infection. Gastroenterology 1982;83:465-469.
21. Altiparmak MR, Trablus S, Pamuk ON, et al. Diarrhoea following renal transplantation. Clin Transplant 2002;16:212-216.
22. Barbut F, Meynard JL, Guiguet M, et al. Clostridium difficile-associated diarrhea in HIV-infected patients: Epidemiology and risk factors. J Acquir Immune Defic Syndr Hum Retrovirol 1997;16:176-181.
23. Bliss DZ, Johnson S, Savik K, et al. Acquisition of Clostridium difficile and Clostridium difficile-associated diarrhea in hospitalized patients receiving tube feeding. Ann Intern Med 1998;129:1012-1019.
24. Archibald LK, Banerjee SN, Jarvis WR. Secular trends in hospital-acquired Clostridium difficile disease in the United States, 1987-2001. J Infect Dis 2004;189:1585-1589.
25. Morinville V, McDonald J. Clostridium difficile-associated diarrhea in 200 Canadian children. Can J Gastroenterol 2005;19:497-501.
26. LaMont T. Pathophysiology and epidemiology of Clostridium difficile infection. www.uptodate.com 2006 January 10; Available at: www.uptodate.com. Accessed Mar 16, 2006.
27. Gerding DN, Johnson S. Clostridium difficile-associated disease, including pseudomembranous colitis. In: Kasper DL, et al, editor. Harrison's Principles of Internal Medicine. 16th ed. New York: McGraw-Hill; 2005:760-761.
28. Levinson W, Jawetz E. Medical Microbiology & Immunology. 4th ed. 2006.
29. Kyne L, Warny M, Qamar A, et al. Association between antibody response to toxin A and protection against recurrent Clostridium difficile diarrhoea. Lancet 2001;357:189-193.
30. Vernacchio L, Vezina RM, Mitchell AA, et al. Diarrhea in American infants and young children in the community setting: Incidence, clinical presentation and microbiology. Pediatr Infect Dis J 2006;25:2-7.
31. Wanahita A, Goldsmith EA, Musher DM. Conditions associated with leukocytosis in a tertiary care hospital, with particular attention to the role of infection caused by Clostridium difficile. Clin Infect Dis 2002 June 15;34(12):1585-92.
32. Tedesco FJ, Gordon D, Fortson WC. Approach to patients with multiple relapses of antibiotic-associated pseudomembranous colitis. Am J Gastroenterol 1985;80:867-868.
33. Hurley BW, Nguyen CC. The spectrum of pseudomembranous enterocolitis and antibiotic-associated diarrhea. Arch Intern Med 2002;162:2177-2184.
34. Bedimo R, Weinstein J. Recurrent extraintestinal Clostridium difficile infection. Am J Med 2003;114:770-771.
35. Chang TW, Lauermann M, Bartlett JG. Cytotoxicity assay in antibiotic-associated colitis. J Infect Dis 1979;140:765-770.
36. Wight N, Curtis H, Hyde J, et al. Clostridium difficile-associated diarrhoea. Postgrad Med J 1998;74:677-678.
37. Alfa MJ, Swan B, Van Dekerkhove B, et al. The diagnosis of Clostridium difficile-associated diarrhea: Comparison of triage C. difficile panel, EIA for toxin A/B and cytotoxin assays. Diagn Microbiol Infect Dis 2002;43:257-263.
38. Mohan SS, McDermott BP, Parchuri S, et al. Lack of value of repeat stool testing for Clostridium difficile toxin. Am J Med 2006;119:356.e7-8.
39. Tedesco FJ. Pseudomembranous colitis: Pathogenesis and therapy. Med Clin North Am 1982;66:655.
40. LaMont T. Clinical manifestations and diagnosis of Clostridium difficile infection. www.uptodate.com 2005 Dec:1-15. Accessed Mar 16, 2006.
41. Wanahita A, Goldsmith EA, Marino BJ, et al. Clostridium difficile infection in patients with unexplained leukocytosis. Am J Med 2003;115:543-546.
42. Kelly CP, LaMont JT. Clostridium difficile infection. Ann Rev Med 1998;49:375-390.
43. Wenisch C, Parschalk B, Hasenhundl M, et al. Comparison of vancomycin, teicoplanin, metronidazole, and fusidic acid for the treatment of Clostridium difficile-associated diarrhea. Clin Infect Dis 1996;22:813-818.
44. Zaleznik, LaMont T. Treatment of antibiotic-associated diarrhea caused by Clostridium difficile. www.uptodate.com 2005 September 14;1-6. Available at: www.uptodate.com.
45. Ohl ME, Stevermer JJ, Meadows S, et al. Clinical inquiries. What are effective therapies for Clostridium difficile-associated diarrhea? J Fam Pract 2005;54:176-178.
46. Bolton RP, Culshaw MA. Faecal metronidazole concentrations during oral and intravenous therapy for antibiotic associated colitis due to Clostridium difficile. Gut 1986;27:1169-1172.
47. Fekety R, Shah AB. Diagnosis and treatment of Clostridium difficile colitis. JAMA 1993;269:71-75.
48. Pepin J, Routhier S, Gagnon S, et al. Management and outcomes of a first recurrence of Clostridium difficile-associated disease in Quebec, Canada. Clin Infect Dis 2006;42:758-764.
49. McFarland LV, Surawicz CM, Greenberg RN, et al. A randomized placebo-controlled trial of Saccharomyces boulardii in combination with standard antibiotics for Clostridium difficile disease. JAMA 1994;271:1913-1918.
50. Lestin F, Pertschy A, Rimek D. [Fungemia after oral treatment with Saccharomyces boulardii in a patient with multiple comorbidities]. Dtsch Med Wochenschr 2003;128:2531-2533.
51. Borody TJ, Warren EF, Leis SM, et al. Bacteriotherapy using fecal flora: Toying with human motions. J Clin Gastroenterol 2004;38:475-483.
52. Wilcox MH. Cleaning up Clostridium difficile infection. Lancet 19961;348:767-768.
53. Rao SS, Edwards CA, Austen CJ, et al. Impaired colonic fermentation of carbohydrate after ampicillin. Gastroenterology 1988;94:928.
54. Hayetian FD, Read TE, Brozovich M, et al. Ileal perforation secondary to Clostridium difficile enteritis: Report of 2 cases. Arch Surg 2006;141:97-99.
55. Hurley BW, Nguyen CC. Drug therapy: Use of antimicrobial agents as first-line drug therapy or CDAD. Clinical Guidance from ACP 2006 June 9. Available at: pier.acponline.org/physicians/diseases/d320/drug.tx/d320-57.html. Accessed June 9, 2006.
We are all aware of the negative effects of unnecessary antibiotics. Antibiotic resistance is a current crisis in healthcare.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.