Drug Criteria & Outcomes: Ramelteon (Rozerem) Formulary Evaluation
Drug Criteria & Outcomes
Ramelteon (Rozerem) Formulary Evaluation
By Patrick J. Phillips, PharmD Candidate
McWhorter School of Pharmacy, Samford University Birmingham, AL
Description
Ramelteon (Rozerem) is an orally active hypnotic agent with an empirical formulation of C16H21NO2.1,2
Melatonin is an over-the-counter agent also known at MEL, MLT, or pineal hormone. Its scientific name is N-acetyl-5-methoxytryptamine.3,4
Indications
Ramelteon is indicated for the treatment of insomnia characterized by difficulty with sleep onset.1-2 The FDA approval allows physicians to prescribe ramelteon for long-term use in adults.
Melatonin is used orally for the treatment of jet lag, insomnia, shift-work disorder, circadian rhythm disorders in the blind, and benzodiazepine and nicotine withdrawal.3-5 Oral, intravenous injections, and intramuscular injections of melatonin have also been used for the treatment of cancer.3,4
Mechanism of action
Ramelteon expresses high affinity for the MT1 and MT2 receptors thought to be involved in the maintenance of circadian rhythm underlying the normal sleep cycle.
Melatonin is synthesized endogenously in the pineal gland and secreted into the blood stream and cerebrospinal fluid. Its primary role appears to be regulation of the body’s circadian rhythm, endocrine secretions, and sleep patterns from its agonist properties on melatonin receptors.
Absorption
Ramelteon is absorbed rapidly, with peak median concentrations occurring at approximately 0.75 hour (range, 0.5-1.5 hours) after fasted oral administration. Although the total absorption of ramelteon is at least 84%, the absolute oral bioavailability is only 1.8% due to extensive first-pass metabolism.1,2
Absorption of the oral immediate release form of melatonin is 3-76%. Exogenous melatonin undergoes significant (up to 60%) first-pass metabolism.4
Distribution
In vitro protein binding of ramelteon is approximately 82% in human serum, independent of concentration.1 Approximately 70% of the drug is bound to the human serum albumin. The mean volume of distribution after an intravenous administration of ramelteon was calculated to be 73.6 L. This suggests substantial tissue distribution.
Melatonin distributes in various body fluids including saliva, seminal fluid, cerebrospinal fluid, ovarian follicular fluid, and amniotic fluid. The mean distribution half-life has been shown to be 1.4-3 minutes after an intravenous bolus dose, and the volume of distribution has been calculated to be 35 L.4
Metabolism
Metabolism of ramelteon consists primarily of oxidation to hydroxyl and carbonyl derivatives, with secondary metabolism producing glucuronide conjugates. CYP1A2 is the major isozyme involved in hepatic metabolism of ramelteon. The major metabolite of ramelteon, M-II, is active and has one-tenth to one-fifth the binding affinity of the parent structure of the human MT1 and MT2 receptors. M-II has a half-life of two to five hours and is independent of dose.
Inactive metabolites of melatonin, 6-hydroxy-melatonin and N-acetylserotonin, are formed via hepatic metabolism.3
Elimination
Ramelteon has a short elimination half-life, on average 1-2.6 hours. Radiolabeled ramelteon showed 84% of the drug was eliminated in the urine and 4% in feces, resulting in a mean recovery of 88%.1-2 Elimination was essentially complete by 96 hours post-dose.1
Elimination half-life of melatonin’s parent compound is 30-50 minutes. Up to 85% of an exogenous dose of melatonin is excreted in the urine as 6-hydroxymelatonin sulfate. Total body clearance is 630-960 mL/min.4
Contraindications
Ramelteon and melatonin are contraindicated in persons with hypersensitivity to any component of the formulations.
Warnings/precautions
- Symptomatic treatment of insomnia should only be initiated after a careful evaluation of the patient for physical and/or psychiatric disorders.
- Patients with severe hepatic impairment should NOT use ramelteon.
- No dosage adjustment of ramelteon is required in patients with renal impairment, including patients with severe renal impairment (creatinine clearance < 30 mL/1.73 m2).1
- Patients should avoid engaging in hazardous activities that require concentration after taking ramelteon or melatonin.
- Avoid melatonin use with other central nervous system depressants.
Drug interactions
Drugs that increase the level of ramelteon via CYP inhibition include fluvoxamine (Luvox), amiodarone (Pacerone), ketoconazole (Nizoral), and fluconazole (Diflucan). Rifampin reduces the level of ramelteon via CYP induction.
Drugs that may increase melatonin levels include fluvoxamine (Luvox) and amiodarone (Pacerone).
Drugs that deplete vitamin B6 may inhibit the body’s ability to synthesize melatonin. Beta-blockers and benzodiazepines may deplete melatonin by enzyme inhibition. Isoniazid alters B6 metabolism, limiting melatonin formation.
Melatonin significantly increased blood pressure and heart rate in 47 patients controlled by nifedipine.4 Four cases of reduced prothrombin time, two with bleeding complications, have been reported.4 Melatonin can stimulate immune function and may interfere with immunosuppressive therapy. Avoid use.
Adverse effects
The most frequent adverse events in ramelteon clinical trials were headache, somnolence, fatigue, dizziness, nausea, insomnia exacerbated, upper respiratory tract infection, and diarrhea.
Adverse effects of exogenous melatonin have been minimal. The following have been reported post melatonin dose: tachycardia, altered sleep patterns, confusion, disorientation, dysphoria, increased seizure activity, psychosis, sedation, fatigue, headache, gynecomastia, reduced body temperatures, decreased luteinizing hormone levels, autoimmune hepatitis, elevated liver enzymes, pruritis, vasodilation, and fixed drug eruption.3-4
Pregnancy/lactation
Ramelteon is Category C. There are no adequate and well-controlled studies in pregnant women. Use ramelteon during pregnancy only if the potential benefit justifies the potential risk to the fetus.1-2 Ramelteon has been shown to be a developmental teratogen in the rat when given in doses 197 times higher than the recommended human dose on a mg/m2 basis. Ramelteon is secreted into the milk of lactating rats. No clinical studies in nursing mothers have been performed. The use of ramelteon in nursing mothers is not recommended.1-2
Melatonin is possibly unsafe when used orally or parenterally. High dose might have a contraceptive effect. Until more is known, advise pregnant patients to avoid using melatonin at any dose.3 It is unknown whether melatonin is excreted in breast milk.4
Administration
The recommended administration and dosage of ramelteon is 8 mg taken within 30 minutes of going to bed.1 It is recommended that ramelteon not be taken with or immediately after a high-fat meal. After taking ramelteon, patients should confine their activities to those necessary to prepare for bed.
The usual oral dosage range for melatonin is 0.5-6 mg at bedtime.5 Geriatric patients may benefit from 1 mg or 2 mg oral sustained-release melatonin two hours before bedtime for up to two months.
Storage
Ramelteon should be stored at 25° C (77° F). Melatonin should be stored at 15-30° C (59-86° F).
Keep containers tightly closed and protect from moisture and humidity.
Clinical studies
Trial 1: Roth T, Stubbs C, Walsh JK. Ramelteon (TAK-375), a selective MT1/MT2 receptor agonist, reduces latency to persistent sleep in a model of transient insomnia related to a novel sleep environment. Sleep 2005;28:303-307.
Objective: To evaluate ramelteon efficacy for the treatment of transient insomnia in healthy adults.
Study design: Randomized, double-blind, placebo-controlled design using a model of transient insomnia related to sleeping in a sleep laboratory at 14 sleep centers in the United States. Eligible participants were stratified into two groups according to reported usual sleep duration (6.5 to < 7.5 hours or 7.5-8.5 hours) and then randomly assigned to one of three groups.
Intervention groups: Ramelteon 16 mg (n = 126) or 64 mg (n = 126), single dose 30 minutes prior to scheduled bedtime.
Control group: Placebo, single dose 30 minutes prior to scheduled bedtime (n = 123).
Participants: 375 healthy adults, ages 35-60 years.
Inclusion criteria
To be eligible, volunteers had to:
- Report usual total sleep duration of 6.5-8.5 hours.
- Have a usual sleep latency of < 30 minutes.
- Have a habitual bedtime between 8:30 p.m. and midnight.
- Be within 20% of their ideal body weight and be in good overall health as determined by medical history, physical examination, clinical laboratory values, and 12-lead electrocardiogram.
- Be able to give written informed consent.
Exclusion criteria
Volunteers were excluded if they had:
- Previously slept in a sleep laboratory.
- An Epworth Sleepiness scale score > 10.
- Changed sleep schedules within the preceding three months (shift work).
- Flown across three or more time zones within the preceding seven days.
- Symptoms of any primary sleep disorder.
- Any physical or psychiatric disorder (including substance abuse).
Outcomes measured
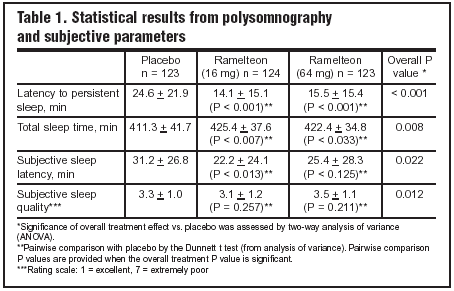
Primary endpoint was latency to persistent sleep (LPS). Secondary endpoints included total sleep time (TST), wake time after sleep onset (WASO), a subjective evaluation of sleep from a post-sleep questionnaire, number of awakenings, and percentage of time spent in each sleep stage.
Results
Overall statistical significance was noted in the above listed endpoints (see Table 1). When the 16 mg and 64 mg regimens were individually compared to placebo, some areas of interest were not statistically significant.
Strengths
- Trial design, multicenter, randomized, controlled trial.
- All participants were accounted for (ITT = intention-to-treat).
- Participants included both men and women (extrapolation of results to both sexes).
- Appropriate statistics were used.
Limitations
- Polysomnogram results for five patients were not included due to lost or unreadable recordings.
- Two active treatment arms were not directly compared (16 mg vs. 64 mg).
- Package insert: 8 mg 30 min. prior to bedtime.
Authors’ conclusion
Overall, ramelteon significantly improved both LPS and TST. The study demonstrates the potential efficacy of melatonin receptor agonists as novel treatment options for insomnia. Ramelteon may act as a "sleep on-off switch," and thus may be most useful in patients with difficulties in initiating sleep.
Trial 2: Zhdanova IV, Wurtman RJ, Regan MM, et al. Melatonin treatment for age-related insomnia. J Clin Endocrinol Metab 2001;86:4,727-4,730.
Objective: To evaluate the use of exogenous melatonin for the improvement of sleep efficiency in insomniac subjects older than age 50.
Study design: Randomized, double-blind, placebo-controlled trial.
Interventions/control: Melatonin (0.1, 0.3, and 3 mg) vs. placebo for improvement of sleep efficiency in insomniac subjects older than age 50. There was a washout period and data were gathered as subjects used all four forms of treatment.
Participants: 30 patients (15 normal sleepers, 15 insomniacs), older than age 50.
Outcomes measured
- Sleep efficiency (the ratio of total sleep time to sleep period, %) was the primary endpoint.
Results
- No significant increases in sleep efficiency were observed after subjects with normal sleep received any dose of melatonin.
- The sleep of insomniac subjects was significantly improved by all three melatonin doses, especially with the melatonin dose of 0.3 mg. It restored sleep efficiency (P < 0.0001) and elevated plasma melatonin levels to normal (P < 0.0008).
Strengths
- Randomized, double-blind, placebo-controlled.
- Appropriate statistics were used.
- Analysis was intention-to-treat, using mixed-models ANOVA.
Limitations/weaknesses
- No power given.
- Small sample size.
Authors’ conclusion
Physiologic doses (0.1 or 0.3 mg) raised plasma melatonin to levels within its normal nocturnal range (60-200 pg/mL) and can significantly improve sleep in people suffering from age-related insomnia. Supratherapeutic dose (3.0 mg) was less efficacious, exceeded normal plasma levels of melatonin, and produced hypothermia.
Recommendations
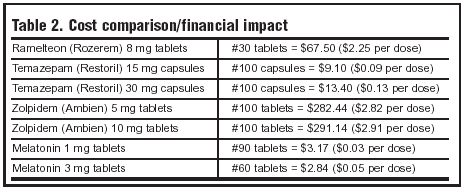
Ramelteon is only indicated for the treatment of insomnia characterized by difficulty with sleep onset. Some clinical data suggest its efficacy in reducing LPS and increasing TST. Due to cost parameters (see Table 2) and the available data, ramelteon should be continued in patients who have been admitted to the hospital already taking the medication. It should not be initiated within the hospital unless temazepam or zolpidem therapy fails.
References
- Takeda Pharmaceuticals. Rozerem [package insert]. Lincolnshire, IL; 2005.
- Efacts. Facts and Comparisons web site. Available at: www.efactsonline.com. Accessed Sept. 8, 2005.
- Jellin JM, ed. Natural Medicines Comprehensive Database. Stockton, CA: Therapeutic Research Faculty; 2002:876-880.
- DRUGDEX System. Thomson Micromedex web site. Available at: www.micromedex.com/index.html. Accessed Sept. 8, 2005.
- Lexi-Comp (Natural Products) [computer program]. Hudson, OH: Lexi-Comp; Sept. 14, 2002.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
