Drug Criteria & Outcomes: Paricalcitol (Zemplar) Formulary Evaluation
Drug Criteria & Outcomes
Paricalcitol (Zemplar) Formulary Evaluation
By Tim Barclift, PharmD Candidate, Harrison School of Pharmacy, Auburn (AL) University Written while on clinical rotation at Huntsville (AL) Hospital
Brand name: Zemplar (Abbott Laboratories)
Generic name: Paricalcitol
Similar drugs: Calcitriol (Rocaltrol), calcitriol injection (Calcijex)
Strengths/dosage forms available: Paricalcitol is available in 1 mL and 2 mL single-dose vials (5 mcg/mL) of aqueous solution for intravenous (IV) injection, as well as in 1 mcg, 2 mcg, and 4 mcg capsules. Calcitriol is available as 0.25 mcg and 0.5 mcg capsules and as an oral solution (1 mcg/mL). It also is available in an intravenous form (1 mL vials of 1 mcg/mL).
Mechanism of Action: Paricalcitol is a synthetic vitamin D analogue, which decreases parathyroid hormone (PTH) secretion in secondary hyperparathyroidism by binding to the vitamin D receptors on the parathyroid gland. It also binds to receptors in the kidney and bones. Calcitriol works by the same mechanism.
Indications: Paricalcitol is approved to treat secondary hyperparathyroidism in chronic renal failure patients. It was recently approved for use in patients ages 5-19. Calcitriol has the same indication, but is not approved for younger patients.
Storage: Paricalcitol and calcitriol should be stored at room temperature. Calcitriol also should be protected from light.
Pharmakokinetics
Injectable: Paricalcitol was studied in healthy adults and chronic renal failure patients receiving hemodialysis. It has a half-life of ≥ 7.3 hours in healthy individuals and 11-32 hours in chronic renal failure patients. It undergoes mainly hepatobiliary excretion, but is reliant on a small amount of renal clearance. The drug did not, however, accumulate in chronic renal failure patients (dosing is based on renal insufficiency). Paricalcitol does not appear to be cleared by hemodialysis, so it can be administered without regard to hemodialysis timing.
Capsules: Paricalcitol is well absorbed from the gastrointestinal (GI) tract. It has a time to maximum plasma concentration (Cmax) of 3 hours. Absorption is not significantly changed by consumption of food; however, a high-fat meal may delay Cmax by 2 hours. Paricalcitol’s distribution is affected by extensive binding to plasma proteins. Paricalcitol undergoes extensive hepatic and non-hepatic metabolism and is eliminated primarily by hepatobiliary excretion in feces. The half-life range is 4-6 hours in healthy individuals, but increases to 17 hours in chronic kidney disease (CKD) Stage 3 patients and to 20 hours in CKD Stage 4 patients.
Calcitriol(oral): Calcitriol also takes 3-6 hours to reach Cmax. It is extensively bound to plasma proteins (99.9%). It also is metabolized via hepatic and nonhepatic routes. It undergoes hepatic recycling and biliary excretion. The elimination half-life for calcitriol is 5-8 hours after single doses (in healthy patients).
Dosing
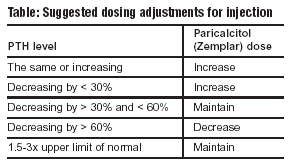
Paricalcitol injectable dosing: The starting dose of paricalcitol is 0.04-0.1 mcg/kg, given as a bolus injection. This dose is given no more frequently than every other day (or three times weekly). Doses of 0.24 mcg/kg have been administered safely. Doses may be increase 2-4 mcg every 2-4 weeks.
Paricalcitol capsule dosing: The dosing is based on baseline intact PTH levels. For a baseline ≤ 500 pg/mL, patients should take 1 mcg daily or 2 mcg three times weekly. For baseline levels > 500 pg/mL, patients should take 2 mcg daily or 4 mcg three times weekly.
Calcitriol (Rocaltrol) dosing: The initial dose for pre-dialysis and chronic dialysis patients is 0.25 mcg/day. If this is not a sufficient dose, it can be increased by 0.25 mcg every 4-8 weeks. The initial dose in children younger than 3 years old is 10-15 ng/kg/day.
Calcitriol injection dosing: The recommended initial dose of calcitriol injection, depending on the severity of the hypocalcemia and/or secondary hyperparathyroidism, is 1 mcg (0.02 mcg/kg) to 2 mcg administered three times weekly, approximately every other day. Doses as small as 0.5 mcg and as large as 4 mcg three times weekly have been used as an initial dose. If a satisfactory response is not observed, the dose may be increased by 0.5-1 mcg at two- to four-week intervals.
Special populations
Pediatrics: Injectable paricalcitol was recently approved for use in patients ages 5-19. Dosages should be started at 0.04 mcg/kg three times weekly. Paricalcitol capsules have not been evaluated in pediatrics. Oral calcitriol may be used in pediatric patients (even those younger than age 5); however, its safety and efficacy have not been well-established. It is supported by data from nonplacebo-controlled studies in pediatrics.
The safety and effectiveness of calcitriol IV were examined in a 12-week randomized, double-blind, placebo-controlled study of 35 pediatric patients, ages 13-18 years, with end-stage renal disease on hemodialysis. Sixty-six percent of the patients were male, 57% were African-American, and nearly all had received some form of vitamin D therapy prior to the study. The initial dose of calcitriol was 0.5 mcg, 1 mcg, or 1.5 mcg, three times per week, based on baseline immunoreactive parathyroid hormone (iPTH) levels of less than 500 pg/mL, 500-1,000 pg/mL, or greater than 1,000 pg/mL, respectively. The dose was adjusted in 0.25 mcg increments based on the levels of serum iPTH, calcium, and Ca × P.
The mean baseline levels of iPTH were 769 pg/mL for the 16 calcitriol-treated patients and 897 pg/mL for the 19 placebo-treated subjects. The mean weekly dose ranged from 1 mcg to 1.4 mcg. In the primary efficacy analysis, seven of 16 (44%) subjects in the calcitriol group had two consecutive 30% decreases from baseline iPTH, compared with three of 19 (16%) patients in the placebo group (95% confidence interval for the difference between groups, -6%, 62%). One calcitriol-treated patient experienced transient hypercalcemia (> 11 mg/dL), while six of 16 (38%) calcitriol-treated patients vs. two of 19 (11%) placebo-treated patients experienced Ca × P > 75.
Geriatrics: Paricalcitol has not been well-evaluated in the elderly. Ten of the subjects in clinical trials were older than age 65 and experienced no discernable difference in results. However, there is not enough evidence to properly evaluate use in this age group. Calcitriol has not been specifically studied in a geriatric population.
Pregnancy/lactation: Both drugs are pregnancy Category C. Studies in animals have shown that paricalcitol can decrease fetal viability, but no studies have been performed in humans. Paricalcitol has not been studied in lactating mothers so the degree to which it may be excreted in breast milk is unknown. Calcitriol has been shown to cause problems in animal models, but sufficient information is not available in humans. Calcitriol is excreted in breast milk and should be avoided in nursing mothers.
Hepatic impairment: Paricalcitol’s pharmacokinetics are not significantly changed in patients with mild or moderate hepatic impairment. It has not been studied in patients with severe hepatic impairment. Calcitriol has not been evaluated in hepatic impairment.
Renal impairment: Paricalcitol dosing recommendations are based on patients with CKD Stages 3 and 4, so no adjustment of dosing is needed. Calcitriol’s elimination half-life is at least doubled in patients with renal insufficiency. Peaks were reached in 8-12 hours and half-lives were estimated at 16.2-21.9 hours. Calcitriol dosing recommendations are the same for dialysis, pre-dialysis, and hypoparathyroidism patients.
Adverse drug reactions
The most common adverse effects seen in both paricalcitol and calcitriol are hypercalcemia and hyperphosphatemia. Other adverse effects are wide-ranging and similar in all products (including headache, GI distress, pain, and increased sickness).
Drug-drug interactions
Paricalcitol injection: While specific drug interactions have not been studied, any condition that predisposes to hypercalcemia increases risk in patients taking digitalis.
Paricalcitol capsules: Paricalcitol does not appear to be affected by interactions involving the CYP 450 enzymes (in vitro studies only). In one study, administering omeprazole 2 hours before paricalcitol had no significant effect. Ketoconazole was found to have no effect on Cmax, but doubled the half-life in healthy patients.
Calcitriol: Cholestyramine may reduce intestinal absorption of fat-soluble vitamins and theoretically may reduce calcitriol’s absorption. Phenytoin and phenobarbital may accelerate metabolism of the active form of calcitriol. Thiazide diuretics may increase risk of hypercalcemia. Ketoconazole may reduce endogenous serum calcitriol concentrations. Corticosteroids may antagonize the calcium absorption effects of calcitriol.
Precautions/warnings: Paricalcitol and calcitriol may cause hypercalcemia and hyperphosphatemia. Patients, especially those with cardiac issues or taking digitalis compounds, should have calcium levels monitored closely (twice weekly). Staff should be educated on the signs and symptoms of hypercalcemia and vitamin D intoxication. In cases of acute overdose, emergency treatment for hypercalcemia may be required. Animal studies in mice and rats have shown increased rates of cancer and pheochromocytoma.
Contraindications: Neither paricalcitol nor calcitriol should be used in patients with a hypersensitivity to any part of the formulations or in patients with hypercalcemia or vitamin D toxicity.
Potential for medication error: Because of the small injection volume, patients could mistakenly be given subcutaneous or intramuscular paricalcitol injections. It also could mistakenly be given daily.
Look-alike/sound-alike: Zenapax (for Zemplar), Peri-Colace (for paricalcitol), calcitonin (for calcitriol).
Patient education: Patients should be counseled on the signs of hypercalcemia and vitamin D toxicity. They also should be counseled on special dietary considerations if necessary.
Special monitoring: Calcium and phosphate levels should be monitored closely in patients taking either drug.
Drug-food interactions: Intake of calcium, phosphate, vitamin D, and magnesium should be controlled. Doses of phosphate binders may need to be adjusted.
Clinical trials
Trial 1: Teng M, Wolf M, Lowrie E, et al. Survival of patients undergoing hemodialysis with paricalcitol or calcitriol therapy. N Engl J Med 2003;349: 446-456.
Objective: To compare survival rates among hemodialysis patients receiving concurrent therapy with either paricalcitol or calcitriol.
Study design: Historical cohort study.
Intervention: Due to current information at the time of the study, there was no increased benefit with either drug. Therefore, the study did not randomize patients, but watched patients who were prescribed either drug and recorded the outcome.
Patient population
Inclusion criteria
- Patients receiving long-term hemodialysis at the U.S.-based dialysis facilities of Fresenius Medical Care North America.
- Patients were treated exclusively with paricalcitol or calcitriol tuntil they died, were transferred to another facility, received a renal transplant, or their data were censored (they received another vitamin D drug).
Exclusion criteria
- Patients receiving any form of injectable vitamin D prior to Jan. 1, 1999.
Outcomes measured: Survival rates and treatment-specific hazard ratios.
Results: For the 36-month period, the mortality rate was 0.180 per person year for the paricalcitol and 0.223 for the calcitriol group (P < 0.001). Discrepancy between therapies was evident at 12 months, then digressed further.
Strengths
- Large population observed (> 55,000).
- In-depth statistical analysis.
Weaknesses
- No randomization.
- Little control of other variables (meds, diets).
Authors’ conclusion: Long-term dialysis patients taking paricalcitol have a significant advantage in survival vs. patients taking calcitriol.
Trial 2: Sprague S, Lerma E, McCormmick D, et al. Suppression of parathyroid hormone secretion in hemodialysis patients: Comparison of paricalcitol with calcitriol. Am J Kidney Dis 2001;38:S51-S56.
Objective: To compare therapeutic efficacy of paricalcitol and calcitriol.
Study design: Double-blind, randomized, multi-institutional, comparative study.
Intervention: After a washout period, patients were randomized to paricalcitol or calcitriol. Doses were titrated to reach PTH goals (50% reduction). The maintenance period was 12 weeks.
Patient population
Inclusion Criteria
- Medically stable adults with end-stage renal disease undergoing chronic hemodialysis three times weekly for at least six months.
- No previous enrollment in paricalcitol trials.
Outcomes measured: PTH, calcium, and phosphate levels.
Results: Paricalcitol caused a greater and faster decrease in PTH levels from baseline compared to calcitriol. Occurrence of hypercalcemia was approximately the same, but because that effect is tied to decreases in PTH, paricalcitol would seem to be safer. There was a greater occurrence of hyperphosphatemia in patients receiving calcitriol therapy.
Strengths
- Double-blind, randomized, prospective.
- Included patients from three institutions.
- Specific goals for PTH, Ca, and Ca × P levels.
- Washout period was used to eliminate confounding variables.
Weaknesses
- Not controlled.
- Only included small subset of overall trial.
- No statistical analysis.
- Small adverse effect profile.
Authors’ conclusion: Paricalcitol provides several therapeutic advantages over calcitriol.
Cost
Paricalcitol (for injection) is available in 1 mL and 2 mL flip-top vials (5 mcg/mL). Twenty-five 1 mL vials cost $542.75. Twenty-five 2 mL vials cost $1085.50. Initial dosing of a patient (70 kg) would cost approximately $21.71 per dose. IV calcitriol (generic) is available in 1 mL vials (1 mcg/mL). Fifty vials cost $192.50. Initial dosing of a patient (70 kg) would cost approximately $3.85 per dose.
Paricalcitol capsules are available in 1 mcg, 2 mcg, and 4 mcg strengths. Thirty capsules cost $162.20, $324, and $648, respectively. If a patient is taking 1 mcg daily or 2 mcg every other day, this translates to a cost of $5.40/day.
Calcitriol is available in 0.25 mcg and 0.5 mcg capsules. It also is available as a liquid (1 mcg/mL). One hundred 0.25 mcg capsules cost $111.99, 100 0.5 mcg capsules cost $179.06, and 15 mL of the liquid costs $143.20. One 0.25 mcg capsule daily would translate to a cost of $1.12/day.
Recommendation: While paricalcitol appears more effective in the long-term suppression of PTH, calcitriol is much less expensive per dose and would be an acceptable alternative for short-term treatment. Calcitriol also is available in a liquid form.
Resources
- Abbott Laboratories. Zemplar [package insert]. North Chicago, IL; October 2004. Available at: www.rxabbott.com/pdf/zemplarivpi.pdf. Accessed Aug. 18, 2005.
- Abbott Laboratories. Zemplar Capsules [package insert]. North Chicago, IL; May 2005. Available at: www.zemplar.com. Accessed Aug.18, 2005.
- Roche Laboratories. Rocaltrol [package insert]. Nutley, NJ; July 2004. Available at: www.rocheusa.com/products/rocaltrol/pi.pdf. Accessed Aug. 18, 2005.
- Abbott Laboratories. Calcijex [package insert]. North Chicago, IL: November 2001. Available at: www.drugs.com/PDR/Calcijex_Injection.html. Accessed Aug. 31, 2005.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
