Is the LP necessary in SAH with new generation scanners?
Is the LP necessary in SAH with new generation scanners?
Authors: John W. Morehouse, MD, Attending Physician, Kaiser Permanente Medical Center, Oakland, CA; N. Ewen Wang, MD, Assistant Professor Surgery, Assistant Professor Pediatrics, Associate Director, Pediatric Emergency Medicine, Stanford University, Palo Alto, CA
Peer Reviewer: Robert E. Falcone, MD; Clinical Professor of Surgery, The Ohio State University, Columbus, OH
Introduction
Subarachnoid hemorrhage (SAH) affects approximately 30,000 individuals annually in North America and is present in nearly 1% of emergency department (ED) patients who present with headache.1,2 Case fatality rates range from 32% to 67%; of those patients who survive, 20% are severely disabled.3 SAH also tends to affect younger patients more than other stroke subtypes, with a mean age at presentation of 55 years.1 Symptoms of SAH can vary widely, making appropriate diagnosis difficult. Patients with milder symptoms are more likely to be misdiagnosed and correspondingly may have a higher risk of death and disability.4
Detection of sentinel bleeding or a 'warning leak' is thought to be essential in reducing morbidity and mortality from SAH because re-bleeding from a ruptured aneurysm can occur in 26% to 73% of untreated patients.5 The relative rarity of SAH in patients presenting with headache, coupled with the opportunity for early intervention and the morbidity of a missed diagnosis, creates uncertainty as to the extent of an evaluation for a patient presenting with a severe headache.
This article provides an overview of diagnostic approaches to SAH by reviewing selected recent articles. Papers were selected based on clinical relevance and applicability to the acute care environment from the literature published in the last three years identified by a search within the topics of "subarachnoid hemorrhage", "computed tomography", "acute headache", and "lumbar puncture".
The overall objective of this article is to familiarize the reader with the currently available diagnostic techniques, examine the controversies surrounding their use, and assist the reader to make informed diagnostic decisions when evaluating patients with severe, sudden-onset headache. The papers are organized in a way that should mirror clinical practice: a review of clinical characteristics and presentations, noninvasive diagnostic techniques, invasive diagnostic techniques, and appropriate review of current treatment practices.
Making the correct diagnosis
Source: Edlow JA. Diagnosis of subarachnoid hemorrhage. Neurocrit Care 2005;2(2):99-109.
This article provides a succinct review of the topic written by one of the field's most cited authors. SAH presentation follows a bell-curve distribution: the middle consists of patients with a classic, unique sudden-onset, severe headache; the tails are composed of patients with localizing neurologic findings and severe mental status changes on one end, and patients with transient headache and a normal neurologic exam on the other. Mildly affected patients stand to gain the most from early detection and treatment of aneurysmal SAH, but are often the most misdiagnosed.
A careful history and physical examination can help determine the difference between benign and serious causes of headache. Findings suggestive of SAH are abrupt onset of headache; worst-ever headache; headache distinct or unique for the individual patient; headache associated with nausea, syncope, or seizure; and physical exam abnormalities. Providers should perform a careful search for an alternate cause of headache.
Misdiagnosis of SAH results from three main errors: lack of recognition of the spectrum of presentation for SAH (from mild to severe), lack of understanding of the limitations of computerized tomography (CT) (sensitivity decreases with time and with anemia), and failure to perform or correctly interpret lumbar puncture (LP) results. The author posits an algorithm for evaluation once the decision is made to evaluate a patient for SAH. A noncontrast CT scan is the standard initial evaluation, and if negative or equivocal, LP with measurement of opening pressure is performed. Positive findings at any step, of course, necessitate prompt consultation and additional vascular imaging. When performing an LP, no specific threshold of red blood cells (RBCs) in the cerebrospinal fluid (CSF) below which SAH can be excluded but that cases where SAH was found with only "a couple hundred" RBCs are extremely rare. A threshold greater than 1000 RBCs with subsequent clearing between three tubes is suggested as the definition of a traumatic tap. An 'LP first' strategy cannot be broadly recommended without further study, but may be safe in carefully selected patients. LP should not be routinely delayed to improve the detection of xanthochromia.
Commentary
This is a well-written, comprehensive review of the available literature that is widely applicable to the acute care practitioner. It provides an approach to the problem of acute severe headache that is stepwise, logical, and to the best extent possible, evidence-based. Patients without an alternative hypothesis that explains their acute, atypical, and severe headache should undergo CT and LP with measurement of opening pressure. Those patients who have truly negative studies should be reassured, and further testing is rarely indicated.
How good are EPs at judging who has an SAH?
Source: Perry JJ, et al. Attitudes and judgment of emergency physicians in the management of patients with acute headache. Acad Emerg Med 2005; 12(1):33-37.
This study is the first to attempt to determine the accuracy of emergency physicians (EPs) for predicting SAH, their comfort level with not ordering testing, such as CT and LP, and physician comfort with not ordering CT imaging prior to LP. This study was a prospective, multicenter cohort study at three Canadian teaching hospitals, enrolling 747 of 1070 consecutive patients with nontraumatic acute headache or syncope associated with headache and normal neurologic examination.
Attending EPs and residents were asked to determine the pretest probability that the patient had SAH, and assess their comfort in either performing no diagnostic testing or performing LP prior to CT. Diagnostic testing was performed at the discretion of the EP. Patients who did not undergo testing were contacted at one- and six-month intervals and had their charts reviewed for repeat visits and testing.
Physicians were "uncomfortable" or "very uncomfortable" with not ordering any tests for 75% of patients, but were "very comfortable" with not ordering diagnostic testing in 10% of patients. Correspondingly, most patients underwent diagnostic testing; 83% had either CT imaging or an LP performed, and 3% had only an LP performed. Fifty patients were diagnosed with SAH. Using a pretest probability of 2% for SAH as a threshold for performing further testing, the sensitivity of clinical suspicion was 93% with a specificity of 49%. Three patients with a clinical pretest probability of less than 2% were diagnosed with SAH. There were no cases of missed SAH in the cohort.
The authors concluded that physician judgment performs relatively well, but still missed some patients with SAH. This study demonstrated the need for the development of a clinical decision rule to better discriminate between patients with SAH and those with more benign causes of headache.
Commentary
This study reinforced the notion that patients with a moderately low pretest probability for SAH (in this case 2% or greater) should undergo diagnostic testing, and demonstrated that Canadian physicians are uncomfortable not ordering diagnostic testing in this patient population; fewer still were comfortable performing an LP without prior CT imaging. This study was performed well, particularly regarding patient inclusion criteria, representing the patient with a normal neurologic examination, as well as the patient who would benefit the most from early detection. The statistical methods used are sound and easily applicable to clinical practice. Several patients had serious diagnoses other than SAH. We hope that the authors' next step will be to construct a decision rule to apply to this patient population.
Missed SAH: What happens?
Source: Kowalski RG, et al. Initial misdiagnosis and outcome after subarachnoid hemorrhage. JAMA 2004;291(7): 866-869.
The authors of this study examined the frequency, risk factors, and the clinical outcome among patients who were misdiagnosed on initial presentation and subsequently hospitalized with SAH. Of 482 patients enrolled in the study period, 56 (12%) were misdiagnosed initially. The patients who were initially misdiagnosed had smaller hemorrhages and were less likely to have lost consciousness. Other characteristics of misdiagnosed patients included: language spoken other than English, single marital status, and lower levels of education. The most common diagnostic error was failure to perform CT imaging (73%); 4 (7%) patients had CT imaging but no LP performed.
Outcomes of all patients initially misdiagnosed were not significantly different from those with an initially correct diagnosis of SAH. However, a subgroup multivariate analysis of those initially presenting with a normal neurological examination demonstrated that these patients were 4 times more likely to die within 12 months, and those surviving had poorer functional status and quality of life.
Commentary
This study reinforces the need for a high level of suspicion when evaluating patients with acute headache, especially those with normal neurological examination. Although well designed and performed, this study does have some limitations. Recall bias is expected with this type of study: patients (or surrogates) may erroneously connect an initial headache with their current diagnosis. Selection bias also occurred, as patients were included who were seen in a primary care physician's office and those who did not receive an evaluation by a physician. If only patients seen in an emergency department (ED) were included in the analysis, the percentage of misdiagnoses decreases to 5%. If only patients with normal mental status seen in an ED were included in the analysis, the rate of misdiagnosis decreases further to 3.7%.
In summary, patients with a normal neurologic examination and a concerning history should have CT imaging for SAH and an LP if necessary because patients with a more subtle presentation have the most to gain from early detection and intervention.
The true incidence of sentinel headaches
Source: Polmear A. Sentinel headaches in aneurysmal subarachnoid haemorrhage: what is the true incidence? A systematic review. Cephalalgia 2003; 23(10):933-934.
A systematic review was performed to determine the incidence of antecedent 'sentinel' headache in patients with SAH. Trials were included if they reported the number of patients diagnosed with SAH who had a history of unusually severe and sudden headache the 3 months prior to diagnosis. Nine studies met the inclusion criteria, yielding an incidence of sentinel headache from 10% to 43%.
The effect of recall bias was evaluated in five case-control studies by comparing the incidence of sentinel headaches in those patients with aneurysmal SAH to patients with nonneurological diagnoses or patients with nonaneurysmal bleeds or strokes. Since the frequency of apparent sentinel headaches (sudden and severe) in neurological controls was 4% and 5%, respectively and the incidence of sentinel headache was 0% in case-matched non-neurological controls, the investigator concluded that recall bias contributes approximately 5% of the rates of sentinel headaches in included studies. Higher rates of sentinel headache incidence (50%-60%) are reported in studies in which the definition and history of headache is less precise than required for inclusion in this review.
Commentary
Determining the true incidence of sentinel headache or 'warning bleed' in SAH is difficult, but important, to the acute care practitioner. If the rates of sentinel headache are low, the notion that SAH outcomes could be improved by early detection of aneurysmal bleeding should be questioned. However, if nearly half of patients have warning bleeds prior to the actual SAH, outcomes could perhaps be improved by more vigilant assessment of patients with symptoms of a sentinel headache. Unfortunately, despite the best efforts of this investigator, we do not yet have an accurate answer to the question at hand. The true incidence of sentinel headache probably lies somewhere between 10% and 43% based on the included studies, but these figures still are limited by significant biases, including recall bias, selection bias, assessor bias, and referral bias.
Are current scanners really better?
Source: Boesiger BM. Subarachnoid hemorrhage diagnosis by computed tomography and lumbar puncture: are fifth generation CT scanners better at identifying subarachnoid hemorrhage? J Emerg Med 2005;29(1):23-27.
This study sought to determine the sensitivity and specificity of modern (5th generation, multidetector) CT imaging for the diagnosis of SAH. The authors performed a retrospective analysis of 177 adult patients who were evaluated for SAH in their ED during a one-year period and who underwent CT imaging and LP if the CT scan was negative. All patients were scanned using a fifth-generation CT scanner and a standard protocol, and studies were interpreted by attending radiologists. All patients included with negative CT scans had LP performed with standard laboratory cell counts and visual inspection for xanthochromia. Patients were considered to have SAH if they had either a positive CT scan or xanthochromia. All patients without xanthochromia were followed up during a period of at least 3 months.
Seven patients were found to have SAH or interventricular hemorrhage by CT imaging, one of whom had an LP performed, which was believed to be negative based on cell counts. The remaining 170 patients with negative CT scans had LPs performed, none of whom had xanthochromia or elevated RBC counts. Patients with elevated first tube RBC counts and insufficient clearing (defined by the authors as less than a 10-fold decrease from first to fourth tubes) were later contacted by phone. None of the patients with a negative CT scan were found to have SAH by LP or patient follow-up. The authors concluded that the sensitivity of a fifth-generation CT scan for SAH is 100% (95% CI, 6I: 61.0 – 100%) and the specificity is 99.4%.
Commentary
This study raised questions about the long-standing diagnostic algorithm of CT imaging followed by LP for the patient who presents with symptoms suggestive of subarachnoid hemorrhage. This topic has been queried with each successive advance in imaging technology. There are significant limitations to a small retrospective study such as this one. One important parameter that is not recorded is the time of symptom onset. It has been demonstrated that while CT imaging becomes increasingly less sensitive with time, LP becomes increasingly more sensitive with time as CSF xanthochromia may take as long as 12 hours to develop after SAH. The study does suggest a trend toward increasing sensitivity with improved imaging technology, especially given the existing accepted sensitivity of earlier CT scanners of 92%-98%. A significant observation of this paper is that LP in patients with negative CT scans may provide alternative diagnoses to SAH. Larger, multicenter, prospective studies will need to be performed before multidetector CT imaging without LP becomes the standard of care in the evaluation of SAH.
CT angiography comes of age
Source: Carstairs SD, et al. Computed tomographic angiography for the evaluation of aneurysmal subarachnoid hemorrhage. Acad Emerg Med 2006; 13(5): 486-492.
Dr. Carstairs and colleagues hypothesized that the addition of computed tomographic angiography (CTA) to the traditional work-up of noncontrast CT imaging and LP for patients presenting with 'thunderclap headache' or 'worst headache of life' would improve detection of aneurysmal SAH. This is a prospective, controlled pilot study to assess the ability of CTA in combination with CT and LP to diagnose SAH in patients who presented to the ED with a headache concerning for SAH. Patients were eligible to participate if they presented with a severe headache fitting predetermined criteria. Patients with relative contraindications to CTA were excluded. Patients enrolled in the study underwent noncontrast head CT followed by CTA in the same multidetector CT scanner. All patients with a negative noncontrast CT scan underwent LP. Patients with positive findings on CT, CTA, or LP underwent confirmatory cerebral digital subtraction angiography (DSA) at the discretion of the treating neurosurgeon.
Five of the 116 enrolled patients (4.3%) were found to have SAH or aneurysm. Six patients were found to have an aneurysm or SAH on CTA. One of these patients had a positive noncontrast CT scan, and two patients had positive LP results. Each of these patients was found to have an aneurysm on both CTA and DSA. Three patients with normal noncontrast CT scans and negative LP results had an aneurysm detected by CTA. One of these was found to have a false-positive CTA, with an aneurysm not seen on conventional DSA. CTA detected other causes of headache including venous sinus thrombosis, stroke, and arteriovenous malformation. The authors concluded that although this was a small study, CTA in conjunction with CT and LP may be useful in the evaluation of patients who are believed to be at high risk for aneurysmal bleeding based on their presenting symptoms.
Commentary
This study suggests that CTA can greatly assist in the diagnosis, and ultimately, in the management of patients who present with symptoms consistent with aneurysm or SAH. Cost analysis, in addition to larger trials, should be performed before routine addition of CTA to the algorithm of CT followed by LP. In this carefully selected study population, no patients had complications related to contrast injection at a one-year follow-up. CTA does present much less risk of stroke and vascular injury than DSA, and can be performed easily with equipment that is becoming much more widely available. This study is well designed and executed and will serve as a prototype for the design of much larger, multicenter studies to examine this issue in the near future.
The bloody tap: Is it traumatic or not?
Source: Shah KH. Distinguishing traumatic lumbar puncture from true subarachnoid hemorrhage. J Emerg Med 2002;23(1):67-74.
In this review, the author responded to the significant diagnostic ambiguity that results from a 'bloody tap'. The incidence of traumatic LP has been reported to be between 10% and 20% depending on the RBC cutoff used, but there is no clear consensus on what exactly constitutes traumatic LP. Given that two-thirds of patients with untreated or undetected SAH will die or have significant morbidity, balanced with the risks of unnecessary angiography or unnecessary surgical intervention for over interpretation, it is clear that errors in determining what is and what isn't a traumatic tap can have weighty consequences.
The authors recommended several approaches to better inform diagnostic certainty:
- Measurement of the opening pressure should be routine. Finding both bloody CSF and an opening pressure greater than 20 cm H2O can be compelling evidence of SAH.
- Declining numbers of RBCs in the CSF from the first to the last tube of collected CSF can help differentiate traumatic tap. Collecting a small amount of CSF in the first tube and a larger amount in the next-to-last tube can accentuate the difference in RBC count. An RBC count that trends close to zero in the last tube or is nearly zero in the first tube is most strongly suggestive of traumatic LP without SAH.
- The detection of xanthochromia, based on subjective visual inspection, has been attributed to the presence of bilirubin, which is formed only in vivo and suggests that blood was present in the CSF prior to LP. While bilirubin is never present in the traumatic tap, the absence of xanthochromia cannot be reliably used to exclude SAH in the first 12 hours after the onset of headache.
If xanthochromia is detected by any method, the patient should undergo further evaluation. Other tests (e.g., the white blood cell count, blood clot formation, and D-dimer assay) are not reliable for distinguishing traumatic LP from SAH. Repeating the LP an interspace higher can be used to rule out SAH if the second LP drains clear CSF.
Commentary
This is a well-written review, but it provides little helpful information to the practitioner caring for the patient with symptoms suggestive of SAH and bloody CSF in reliably distinguishing traumatic LP from SAH, reflecting the paucity of reliable methods for making this distinction. In general, patients with symptoms of SAH, with a negative head CT scan but bloody CSF not clearing to nearly zero, without xanthochromia in the first 12 hours or with xanthochromia after that time, should undergo further diagnostic imaging and neurologic or neurosurgical consultation.
Treatment with calcium antagonists
Source: Rinkel GJ, et al. Calcium antagonists for aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev 2005(1):CD000277.
Calcium channel blockers and calcium antagonists are often used to prevent secondary ischemia after SAH thought to be due to cerebral vasospasm. Previous experimental studies have indicated that calcium antagonists do show some neuro-protective benefit and can prevent or treat cerebral vasospasm, but there is a lack of definitive evidence. This article from the Cochrane Collaboration is a meta-analysis of the recent available evidence.
Twelve trials were included from a pool of 19 identified that studied the use of calcium antagonists in patients with SAH between 1983 and 2000, including a total of 2844 randomized patients. The calcium antagonists analyzed were nimodipine (both oral [PO] and intravenous [IV]), nicardipine, AT877 (an intracellular calcium antagonist), and magnesium. Patients with SAH or aneurysm rupture were included in the analysis if they were treated with any calcium antagonist (or control) within 10 days of SAH, but before symptoms of secondary ischemia. The primary composite outcome measured was "poor outcome" represented by death, vegetative state, or severe disability. Secondary outcomes measured were death, secondary ischemia, re-bleeding, and adverse treatment effects.
Poor outcome was adequately reported by eight trials. The relative risk (RR) of poor outcome was 0.82 (95% CI, 0.72 to 0.93), the absolute risk reduction was 5.1%, and the number needed to treat (NNT) was 20 patients. For the three trials that used only oral nimodipine, the RR for poor outcome was 0.70 (95% CI, 0.58 to 0.84). For trials using drugs other than nimodipine, the results were also inconclusive. Overall, the RR of case fatality rate was 0.90, but not statistically significant.
Secondary ischemia as measured by clinical signs was seen in 27% of patients in the treatment groups compared with 40% of patients in the control groups (RR 0.67; 95% CI, 0.60 to 0.76). When measured by CT imaging, 37% of patients in the treatment group were found to have cerebral infarction compared with 47% of controls (RR 0.80; 95% CI, 0.71 to 0.89). For nimodipine trials (PO and/or IV), the RR of clinical ischemia was 0.65 (95% CI, 0.51 to 0.82); for infarcts seen on CT, the RR was 0.70 (95% CI, 0.58 to 0.85).
Treatment with calcium antagonists showed a trend toward reduction of re-bleeding events, but did not reach statistical significance (RR 0.77; 95% CI, 0.58 to 1.02). Adverse treatment effects were reported in half of the 12 trials, but data were limited. Hypotension occurred in 2.1% of patients treated with nimodipine and 34% of those treated with nicardipine.
Overall analysis of the data suggests a reduction in poor outcomes and secondary ischemia, but case fatalities were not reduced significantly. This is particularly true with oral nimodipine. The authors concluded that treatment with oral nimodipine is widely regarded as standard; the evidence supports this position given the low risk and high potential for benefits of treatment.
Commentary
Calcium antagonists have long been a promising treatment for the reduction of vasospasm and ischemia after SAH. The methods of literature search, trial inclusion, and statistical analysis are quite elegant. The drawbacks of this paper are those that plague all meta-analyses — the results are dependent on the quality of the original works, and studies performed with similar questions in mind may not measure the same outcomes in the same way or over the same time period — both of which limit the general conclusions that one may draw from the results.
This meta-analysis' findings mirror what has been published previously on the topic. The use of calcium antagonists shows a trend toward benefit, but does not reduce overall mortality. Nimodipine 60 mg given orally every 4 hours remains the standard of care, as evaluated in five of the studies included in this review, but other therapeutic agents (e.g., magnesium and nicardipine) appear promising and worthy of further study.
A must-read: aneurysmal SAH
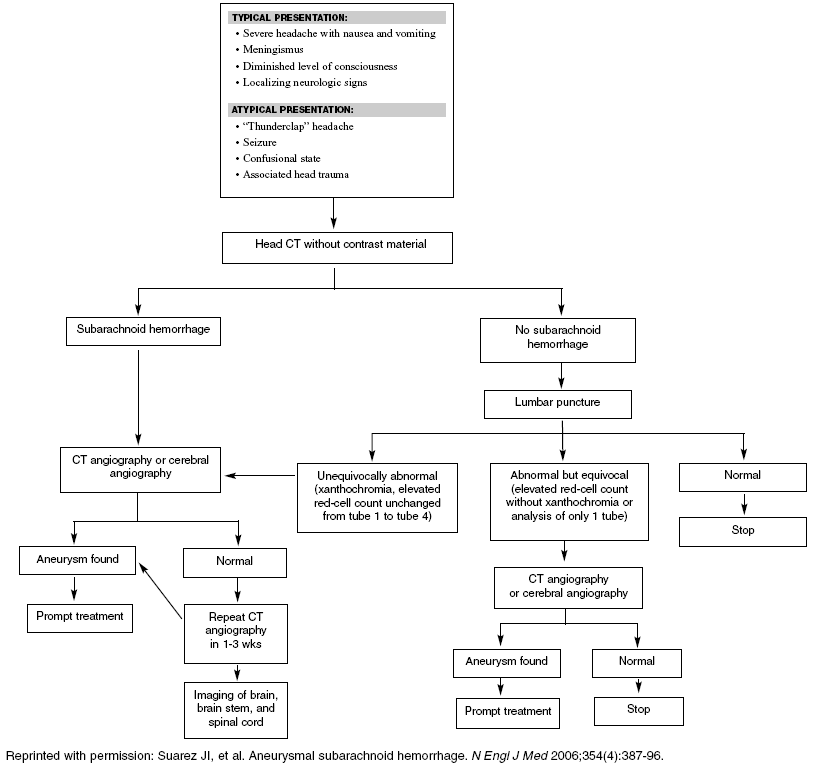
Source: Suarez JI. Aneurysmal subarachnoid hemorrhage. N Engl J Med 2006;354(4):387-396.
Written by a multidisciplinary team, this review provides a useful review of SAH diagnostic and treatment options. The authors emphasized that SAH should be suspected not only in patients with classically associated symptoms—but also in patients who present with less classic signs, particularly those patients with a 'first' or 'worst' headache. A noncontrast head CT scan should be the first study ordered, with thin cuts through the base of the brain. A sensitivity of 100% is quoted if performed within 12 hours, but the sensitivity drops significantly with time. LP should be performed in any patient with suspected SAH and a negative or equivocal CT scan. In patients with positive or equivocal LP results, CTA or DSA of the cerebral vessels should be performed. Patients with a negative imaging study should have a repeat study 7-14 days after initial presentation, followed by magnetic resonance imaging if no aneurysm is found.
SAH management includes stabilization and transfer to a center with dedicated neurologic critical care facilities and neurovascular expertise. Blood pressure should be normalized using labetalol or nicardipine, if necessary. Hyperglycemia and hyperthermia are associated with worse outcomes and should be controlled. Calcium channel blockers, particularly oral nimodipine (60 mg PO every 4 hours), should be administered to prevent ischemic complications. Antifibrinolytic therapy (aminocaproic acid) is optional for the first 24-48 hours. Definitive therapy for a ruptured aneurysm usually requires endovascular coiling or neurosurgical clipping. Complications (e.g., hydrocephalus and re-bleeding) are relatively common. Vasospasm should be managed with hypervolemia and induced hypertension, or angioplasty and local vasodilator injection for areas of focal narrowing. Hydrocephalus may require drainage or shunting. Seizures should be prevented with anticonvulsants for a week after initial bleed to reduce re-bleeding. Comatose patients should undergo electroencephalography because nonconvulsive seizures occur in 1 of 5 patients. Rehabilitation treatment should be initiated promptly to reduce long-term disability.
Commentary
This article provides a must-read summary of evaluation and treatment options based on recent available evidence. The authors suggested that early CT is 100% sensitive, yet recommend LP for all negative CT scans. That seems somewhat contradictory, but LP after negative CT scans remains the currently accepted practice.
Conclusions
The diagnosis of SAH, or more importantly, the exclusion of this diagnosis in the patient presenting with headache remains a difficult problem. A high level of suspicion for SAH should be maintained when evaluating patients with a new, their first, or their worst headache. Patients with missed diagnoses, particularly those with initially normal examinations suffer higher morbidity and mortality than those diagnosed initially.
Sentinel headaches probably do occur at a rate of 10% to 43%. Sensitivity for CT imaging to detect SAH may be increasing to nearly 100%, especially if performed within 12 hours of symptom onset, but further study is needed before CT imaging without LP becomes the standard of care. CT angiography likely will be used more commonly in the near future as a rapid noninvasive alternative to cerebral angiography in addition to CT imaging and LP. But again, it cannot yet be routinely recommended.
When performing LP, the incidence of traumatic tap is probably around 15%. Opening pressure should be measured. A threshold of 1000 RBCs per mm3 with subsequent RBC clearing can help define a traumatic tap, particularly if the count trends close to zero. Patients with xanthochromic CSF should undergo further testing. In addition to the usual treatment, evidence suggests that nimodipine 60 mg should be given orally every 4 hours. Patients who undergo CT imaging and LP and have truly negative studies for SAH should be reassured, but an alternative diagnosis for the cause of their headache should be carefully considered.
References
1. Mayberg MR, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage. A statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke 1994;25(11):2315-2328.
2. Edlow JA. Diagnosis of subarachnoid hemorrhage in the emergency department. Emerg Med Clin North Am 2003;21(1):73-87.
3. Hop JW, et al. Case-fatality rates and functional outcome after subarachnoid hemorrhage: a systematic review. Stroke 1997;28(3):660-664.
4. Edlow JA, et al. Avoiding pitfalls in the diagnosis of subarachnoid hemorrhage. N Engl J Med 2000; 342(1):29-36.
5. van Gijn J, et al. Subarachnoid haemorrhage: diagnosis, causes and management. Brain 2001; 124(Pt 2):249-278.
| Figure 1. Diagnostic Algorithm for Subarachnoid Hemorrhage |
 |
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
