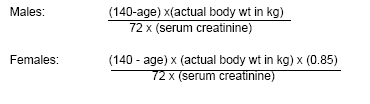Cardioprotection in the Emergency Department: Part I
Cardioprotection in the Emergency Department: Part I
Authors: Michael C. Plewa, MD, FAAEM, FACEP, Research Director, St. Vincent Mercy Medical Center, Toledo, OH; Kevin M. Casey, DO, FACEP, FAAEM, Assistant Director, Emergency Department, St. Vincent Mercy Medical Center, Toledo, OH; and Randall W. King, MD, FACEP, Director, Emergency Medicine Residency, St. Vincent Mercy Medical Center, Toledo, OH; Assistant Clinical Professor, University of Toledo College of Medicine.
Peer Reviewers: Brian Tiffany, MD, PhD, Chairman, Department of Emergency Medicine, Chandler Regional Hospital, Scottsdale, AZ; and Gregory A. Volturo, MD, FACEP, Professor of Emergency Medicine and Medicine, Vice-Chair, Department of Emergency Medicine, University of Massachusetts Medical School, Worcester.
Remember when the treatment for a heart attack was simply bed rest, morphine, and diazepam? Back then (not really that long ago), treatment of an acute infarction with nitrates, aspirin, heparin, or streptokinase was considered harmful. And cardiologists were fearful of performing coronary angiography in such an unstable patient. How much as changed! In fact, so many studies have been done on different patient populations, with diverse combinations of agents and procedures, and with various outcome measures, that it is difficult to know what is best for an individual patient. The authors of this two-part series have reviewed numerous clinical trials to develop a summary of the current medical management of acute coronary syndrome (ACS), concluding with their (bottom line) recommendations. I believe this information will be useful for emergency physicians to initiate dialogue and develop consensus with cardiologists and cardiothoracic surgeons regarding the treatment of ACS patients in their institution.
—J. Stephan Stapczynski, MD, Editor
Introduction
It's a daunting task to read from A to Z through the numerous clinical trials in cardiology in pursuit of a cure for acute coronary syndrome (ACS), to capture the essence of what is important, find some clarity on the topic, and to commit this information to memory and advance our daily practice caring for these patients.
Emergency physicians play an important role in the early diagnosis of myocardial infarction (MI) and ACS, as well as the early initiation of pharmacologic therapies that can salvage ischemic myocardium and save lives. These tasks are challenging since the definitive diagnosis may not be immediately available, and significant controversy exists over which of the many therapeutic agents are most effective in various patient settings. Should a patient receive one, two, or three antiplatelet agents? Is it safe to administer clopidogrel (Plavix) prior to results of coronary angiography? Which patients should receive glycoprotein IIb/IIIa inhibitors? Should patients scheduled for percutaneous coronary intervention (PCI) be treated with unfractionated heparin (UFH) or low-molecular weight heparin (LMWH)? Are oxygen, nitroglycerin, and beta-blockers essential? Should statins or angiotensin-converting enzyme (ACE) inhibitors be given in the ED? Does it make a difference to transfuse anemic patients or treat hyperglycemia? This review will attempt to clarify the reasonable use of antiplatelet, antithrombotic, anti-ischemic, and miscellaneous therapies for ACS.
Specific reperfusion strategies, such as thrombolysis, PCI or facilitated PCI, will not be specifically addressed. Throughout the article, results of various trials will be expressed both in terms of absolute risk reductions (ARR) or absolute risk difference (ARD) (which can be used to calculate "numbers needed to treat" [NNT] and "numbers needed to harm" [NNH]) and relative risk reductions (RRR). (See Table 1 for abbreviations.)
| Table 1. Abbreviations |
| ARR = Absolute risk reduction ARD = Absolute risk difference NNT = Numbers needed to treat NNH = Numbers needed to harm RRR = Relative risk reductions |
ACS encompasses unstable angina (UA), defined as a new symptom or a change from stable angina; non-ST segment elevation MI (NSTEMI), defined as positive cardiac markers with ST-depression or with nonspecific or normal electrocardiogram (ECG); and ST-segment elevation myocardial infarction (STEMI), defined as anginal symptoms with greater than or equal to 1 mm ST elevation in 2 contiguous leads on ECG. ACS is a spectrum of disease states with a broad range of etiologies including atherosclerotic plaque rupture, platelet aggregation, thrombosis, spasm and distal embolization of clot, with the final common outcome of myocardial ischemia. During ACS of any cause, some of these areas of at-risk ischemic myocardium may be salvaged by early therapy,1 and studies have demonstrated that long-term prognosis after acute myocardial infarction (AMI) is improved by limiting infarct size with the prevention of further ischemia or reperfusion injury.2,3
Numerous advances in therapeutic options have been discussed in the literature and between emergency physicians and cardiologists since the American College of Cardiology (ACC) and the American Heart Association (AHA) last updated their guidelines for the management of patients with NSTEMI ACS in 20024-6 and STEMI in 2005.7 According to these guidelines, antiplatelet (aspirin, clopidogrel, glycoprotein IIb-IIIa inhibitors), antithrombotic (unfractionated heparin [UFH], low-molecular weight heparin [LMWH]), and anti-ischemic (oxygen, nitrates, beta-blockers) therapy should be administered as soon as possible for all patients with high-risk ACS or STEMI. This early decision of which medications are to be used requires significant skill and is based on risk stratification, since definitive diagnosis may not available for hours (pending results of cardiac biomarkers and coronary artery imaging).
Risk Stratification
Various methods are available to risk stratify ACS patients, but prior to their use it is important to exclude the various non-cardiac causes of acute chest pain, some of which would have detrimental consequences if treated with antiplatelet or anti-thrombotic therapy (e.g., aortic dissection, pericarditis), as well as chronic stable angina.4 The ACC/AHA guidelines4 recommend risk stratification into low, intermediate, and high risk for death or nonfatal MI with historical features, pain character, and clinical, electrocardiogram (ECG), and cardiac biomarker findings. (See Table 2.) Important aspects of the high-risk criteria include the presence of left-sided heart failure, hemodynamic instability, rest angina with ECG changes (new bundle-branch block, ST-segment depression > 0.05 mV, ventricular tachycardia) or elevated troponin (TnT or TnI) > 0.1 ng/mL. Low-risk patients may have risk factors for coronary artery disease, but have fewer than 20 minutes of rest pain, normal or unchanged ECG, age younger than 70 years, and normal cardiac biomarkers. Therefore, by ACC/AHA guideline definition, all patients stratified into low-, intermediate-, or high-risk criteria must have at least an ECG and cardiac biomarkers measured. The authors believe that there exist patient sets that are "very low risk" and do not require cardiac marker measurements as part of the workup. We would suggest that a thorough history and physical, an ECG, and chest radiograph are often enough in the very low risk patients.
In the risk stratification of potential ACS patients it is important to consider a potential ischemic event, according to the ACC/AHA 2002 guidelines,4 as an episode of non-traumatic chest pain or severe epigastric pain described as central or substernal compression, crushing, pressure, tightness, heaviness, cramping, burning, or aching sensation, or unexplained indigestion, belching, or epigastric pain, especially if associated with radiating pain in the neck, jaw, shoulders, back or one or both arms, with dyspnea, nausea, vomiting, or diaphoresis.
Troponin elevation has become one of the most important measures of risk assessment since it is associated with a three- to five-fold increased risk of 30-day mortality or recurrent MI.8,9 Several clinical trials (such as CURE)10 have inclusion criteria for the high-risk group as elevated troponin or ST-depression on ECG. Others use multiple criteria, such as recurrent ischemia despite anti-ischemic therapy, sustained ventricular tachycardia, PCI within 6 months or prior coronary artery bypass graft (CABG) surgery, in addition to elevated troponin or ST-depression.
Another stratification method is the Thrombolysis in Myocardial Infarction (TIMI) Risk Score,11 a 7-point score based on various clinical findings (see Table 3), where the risk of adverse outcome, defined as death, reinfarction, or need for target vessel revascularization (TVR), increases from 5% (score 0, 1) to 41% (score 6, 7).11 For ACS, high risk is defined as a score of greater than 3 in studies on invasive strategy, or a score greater than 4 in studies on GP IIb/IIIa antagonists.
| Table 3. TIMI Risk Score |
| • Age > 65 • More than 3 coronary risk factors (male, dyslipidemia, smoking, hypertension, diabetes, obesity, family history) • Prior angiographic coronary obstruction (> 50%) • ST-segment deviation (persistent depression or transient elevation) • More than 2 angina events within 24 hours • Aspirin use within 7 days • Elevated cardiac biomarkers The total score is the sum of each of the previous components assigned a weight of one. High risk ACS is defined as a score > 3. The risk of adverse outcome (death, reinfarction, revascularization) increases from 5% with score of 0 or 1 to 41% with score of 6 or 7.11 |
Authors' Bottom Line Recommendations: High-risk patients with elevated troponin, ECG evidence of ischemia or left-sided heart failure, hemodynamic instability, or persistent rest pain are likely to benefit from combination antiplatelet, antithrombotic, and anti-ischemic agents.
Antiplatelet Therapy
Platelets are of primary importance in the pathogenesis of ACS. Erosion or rupture of atherosclerotic plaque, as often occurs in ACS or during PCI, leads to platelet activation and aggregation. Platelet activation results in alteration in platelet shape with an increase in receptor number, and platelet aggregation involves the cross-linking of platelets via the glycoprotein IIb/IIIa receptors and fibrinogen. Antiplatelet agents, therefore, play a central therapeutic role in ACS. The three classes of antiplatelet agents include aspirin, adenosine diphosphate receptor antagonists (thienopyridenes), and glycoprotein (GP) IIb/IIIa receptor antagonists.
Aspirin. Aspirin inhibits platelet activation by irreversibly blocking (for the life of the platelet) cyclooxygenase 1, reducing thromboxane A2 production, with effects apparent within 30 minutes of oral ingestion. Between 5% and 19% of patients have aspirin resistance to cyclooxygenase 1 inhibition, perhaps because of genetic polymorphism or drug-drug interactions.12 Several non-steroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen, can diminish the protective antiplatelet effects of aspirin by competitively blocking cyclooxygenase 1.13,14 Aspirin resistance may explain the predictive ability of recent aspirin use (within prior 7 days) for adverse outcome in the TIMI risk score.11
Aspirin has been shown to be beneficial in the setting of ACS, including AMI, as well as in PCI, by decreasing the risk of re-occlusion.15 The pooled effects from four randomized, placebo-controlled, clinical trials in 2448 ACS patients demonstrated a 6% ARR (NNT=16), 49% RRR in early death or MI.4 In patients with suspected AMI, the ISIS-2 trial16 demonstrated a 2.4% ARR (NNT=42), 23% RRR in early death, and 1% ARR (NNT=100), 46% RRR in recurrent MI with 160 mg/day aspirin, without increased risk of bleeding. The Antithrombotic Trialists Collaboration17 meta-analysis of 15 trials with more than 19,000 acute MI patients demonstrated a 3.8% ARR (NNT=26), 27% RRR in vascular death, nonfatal MI, or stroke. In patients with stable angina undergoing PCI, the M-HEART II study found aspirin demonstrated a significant 4.5% absolute (ARR) (NNT=22), and 79% relative (RRR) risk reduction in post-PCI MI.18
There are a few contraindications for aspirin use.7 Absolute contraindications to aspirin include active bleeding (especially central nervous system, retinal, gastrointestinal, or serious genitourinary) and true aspirin allergy (urticaria or anaphylaxis), whereas relative contraindications include hemophilia, previous bleeding, anticoagulant use, and severe uncontrolled hypertension.
Aspirin can result in nausea, dyspepsia, gastrointestinal bleeding, and a small increased risk of major bleeding of 0.2%-4%. The CURE trial10 noted that major bleeding from aspirin alone increased from 2% with less than 100 mg aspirin, to 2.3% with 100-200 mg aspirin, and to 4% with more than 200 mg aspirin. Also, major bleeding with aspirin use increased with age from 2.1% with age younger than 65 years to 3.6% with age older than 75 years.
Aspirin is recommended for all patients (unless contraindicated) with ACS, regardless of risk, in an initial oral dose of 162-325 mg. Chewing the tablet, whether or not it is enteric-coated, provides the fastest onset and is especially recommended for patients with acute MI.7 When the patient is vomiting or unable to swallow, a 300-mg dose of rectal aspirin is a reasonable alternative. The best daily dose of aspirin, especially when combined with other antiplatelet agents, is uncertain. The Antiplatelet Trialists' Collaboration19 suggests that doses of 75-150 mg/day are as effective as 150-325 mg/day. In a study of 495 patients undergoing elective PCI, 80 mg was as effective as higher doses (up to 1500 mg/day).20 The CURE study10 demonstrated that when clopidogrel is used in combination with aspirin, the efficacy is independent of the aspirin dose, yet major bleeding is dose-dependent on aspirin, such that doses less than 100 mg result in 2.6% major bleeding event rate, 100-200 mg results in 3.5%, and more than 200 mg in 4.9%. This suggests that the optimal daily dose of aspirin when used in combination with clopidogrel may be less than 100 mg.
Because of an emphasis on the importance of aspirin therapy in ACS, many prehospital providers as well as emergency physicians will repeat a full dose of 325 mg orally, even if the patient has taken aspirin already that day, although the efficacy and safety of this practice has not been established and certainly may increase the bleeding risk.
Authors' Bottom Line Recommendations: Use an initial dose of 162-325 mg of chewed aspirin, 300 mg rectal dose for those unable to swallow, in all patients with ACS without active bleeding or true aspirin allergy.
Thienopyridines. Thienopyridines, such as ticlopidine (Ticlid) and clopidogrel (Plavix), irreversibly inhibit platelet activation by blocking the adenosine diphosphate (ADP) receptors (PsY1 and P2Y12), which would otherwise trigger platelet activation, expression of glycoprotein IIb/IIIa receptors, the release of granules, and formation of thrombin from prothrombin.
Ticlopidine has fallen out of favor for ACS primarily because of its slow onset of action where pretreatment for 3 days may be necessary,21 and lack of reduction in delayed restenosis,22 but also because of the risk of serious adverse events, such as severe neutropenia (1% of patients), bronchiolitis obliterans organizing pneumonia, and rare instances of thrombotic thrombocytopenia purpura-hemolytic uremic syndrome (TTP-HUS) (1 in 1600 to 4800 cases).23,24
Clopidogrel has been studied in several large trials of various settings, including elective PCI (CREDO25), in high-risk ACS (CURE10), high-risk ACS undergoing PCI (on average 6 days after presentation)(PCI-CURE26), and with fibrinolysis for STEMI (CLARITY,27 COMMIT28), as well as STEMI treated with thrombolysis followed by PCI on average 3 days after presentation (PCI-CLARITY29) (see Table 4). There are, as yet, no large trials supporting the early use of clopidogrel prior to emergent PCI for STEMI.
The CREDO25 trial found no difference in 28-day outcome unless the 300-mg loading dose was given more than 6 hours prior to PCI. Smaller reductions were noted in those who received a GP IIb/IIIa inhibitor, which did not increase risk of major bleeding. This suggests that clopidogrel loading is not necessary if the patient is going to PCI within 6 hours, especially if GP IIb/IIIa inhibitors are administered.
Clopidogrel may be beneficial in the whole spectrum of ACS patients, from low to high risk, since the CREDO25 study found benefit in stable patients with elective PCI (not ACS), and the CURE trial found clopidogrel improved outcome in TIMI low-risk groups (RR 0.71) as much as in the intermediate- (RR 0.95) and high-risk (RR 0.73) groups30 (although all patients had positive troponin or ECG changes on entry.) Only the COMMIT28 mega-trial was able to demonstrate significant mortality reduction in STEMI patients undergoing thrombolysis, whereas the others showed trends toward decreased death rate but were inadequately powered.10,25-27,29,30
Clopidogrel is absolutely contraindicated in hypersensitivity and active gastrointestinal or central nervous system bleeding. Caution should be used in those who have undergone recent trauma or surgery (exact length of time not defined), anticipated surgery fewer than 5 days, as well as previous reactions such as neutropenia or thrombotic thrombocytopenic purpura.
The most common complication of clopidogrel use is bleeding (discussed below). Less common adverse events include itching, purpura, diarrhea, dyspepsia, and rash. Rare events include severe neutropenia (0.05%) and thrombotic thrombocytopenic purpura-hemolytic uremic syndrome (4 cases in one million exposed).
Some clopidogrel resistance, measured with in vitro platelet aggregation studies, can occur in between 4% and 30% of individuals,31 more so in overweight or obese individuals.32 This clopidogrel resistance may result in more frequent coronary events in the months following PCI (40% in the lowest quartile of ADP-induced platelet aggregation vs none in the third and fourth quartiles).33
Except for the CURE trial, no differences in the rates of major, minor, or intracranial bleeding were noted between clopidogrel with aspirin vs. aspirin alone. Intracranial bleeding occurs on the order of 0.4%, and gastrointestinal bleeding occurs in less than 2% (CAPRIE).34 The CURE trial10 found increased rates of major and minor bleeding such that for every 100 patients treated with clopidogrel, there might be approximately 2 cases of cardiac death, MI, or stroke prevented, and one major bleed and 2-3 minor bleeds. (See Table 4.)
Bleeding is a significant concern in patients treated with clopidogrel within 5 days of CABG surgery (CURE). An analysis35 of 2858 ACS patients from the CRUSADE registry identified 852 (30%) receiving clopidogrel, and those with CABG surgery fewer than 5 days of clopidogrel experienced an 8.1% absolute (NNH=12), and 14% relative increase in blood transfusion. Several smaller, retrospective studies36-40 have also described the association of early CABG surgery within fewer than 5 days of clopidogrel therapy with increased morbidity,38,41 including increased chest tube drainage by 20% to 98% (averaging as high as 1485 mL in 24 hours),38 as much as an 11-fold increase in transfusion of blood products,39 and re-operation rates from 4.9% to 14.6%.38,39 Most of these studies have been underpowered to demonstrate increased mortality; however, a single study of 91 CABG patients found significantly increased mortality in those receiving clopidogrel within 72 hours of CABG surgery.40
As many as 15-17% of high-risk ACS patients10,41 and 6% of STEMI27 patients eventually require elective CABG. Fortunately, the incidence of urgent or emergent CABG following PCI has decreased significantly recently, and is now less than 2%.42-44 Although clopidogrel-induced bleeding during CABG may be minimized by platelet transfusion and intraoperative aprotinin,45 it is safest to avoid CABG early after clopidogrel administration, and the ACC/AHA guidelines4 suggest withholding clopidogrel for 5-7 days prior to elective CABG. Since patients requiring CABG are difficult to identify based on early presentations,41 the American College of Chest Physicians46 suggests withholding clopidogrel prior to PCI until the coronary anatomy is defined by cardiac catheterization (angiography) and the need for urgent CABG is excluded. The American College of Emergency Physicians6 clinical policy suggests a loading dose of clopidogrel be given in the ED prior to PCI in ACS patients considered "not at significant risk for urgent CABG," but ambiguously implies it could be given "cautiously" to those at greater risk for urgent CABG with unknown coronary anatomy.
At institutions where cardiologists, cardiothoracic surgeons, and emergency physicians are in agreement to consider an early loading dose of clopidogrel to patients prior to coronary angiography and PCI, it is best to establish a form of risk-stratification for those likely to require urgent CABG. A score to predict the risk of CABG was derived47 from the TACTICS-TIMI-18 study of 2220 ACS patients (see Table 5) and found a risk of CABG of 6% for a score less than 3, 22% for a score of 3-5, and 55% for a score greater than 5. Others at high risk are those with ECG changes suggesting left main occlusion (see special situations below), those with prior angiogram results suggesting left main or triple vessel disease, and those with recent stress imaging suggesting diffuse, non-localized ischemia.
| Table 5. Predictors of Risk of CABG |
| Elevated troponin 3 Prior stable angina 1 ST-segment deviation > 0.5 mm 1 Male gender 1 Peripheral artery disease 1 History of previous CABG -2 The risk of CABG is 6% for score < 3, 22% for score of 3-5, and 55% for score > 5.47 |
Clopidogrel is usually given in an initial loading dose of 300 mg for all high-risk patients (up to 75 years of age) not undergoing immediate PCI, and when aspirin is contraindicated, and then followed by 75 mg daily. Alternatively, a 600-mg dose may be effective in 2-3 hours48-50 (in comparison to 6 hours for the 300-mg dose),26 is more effective than the 300-mg loading dose,51 with an 8% ARR (NNT=13), 67% RRR in comparison to 300 mg, and the 600-mg dose may diminish the likelihood of clopidogrel resistance.52
Authors' Bottom Line Recommendations:
- Initiate early dialogue with cardiologists and cardiothoracic surgeons regarding preferences for dose (300 mg vs 600 mg) and timing (before or after coronary angiography) to load clopidogrel in the PCI setting.
- Give a loading dose (300 mg or 600 mg) of clopidogrel several hours prior to planned PCI in high-risk patients with elevated troponin or ECG changes if these patients are not at high risk for emergent CABG surgery. Consider the 600-mg clopidogrel dose, or a GPIIb/IIIa inhibitor in combination with the 300-mg clopidogrel dose, when the time to PCI is expected to be less than 6 hours.
- Give clopidogrel by 300 mg oral loading dose in high-risk patients, with elevated troponin or ECG changes, undergoing conservative management (no PCI anticipated).
- Give 300 mg clopidogrel loading dose to STEMI patients treated with thrombolysis.
Glycoprotein IIb-IIIa Inhibitors. The glycoprotein IIb/IIIa receptor is the primary mechanism for platelet-platelet adhesion by binding fibrinogen. The GP IIb-IIIa inhibitors are more potent antiplatelet agents than aspirin or thienopyridenes and are effective within minutes of intravenous (IV) administration. Abciximab (ReoPro), eptifibatide (Integrilin), and tirofiban (Aggrastat) are the agents available for clinical use. Abciximab is a monoclonal antibody with a short serum half-life but prolonged clinical effect of approximately 24-48 hours.4 Eptifibatide, a peptide inhibitor, and tirofiban, a peptidomimetic inhibitor, are smaller molecules with very brief half-lives (15 minutes and 2 hours, respectively).
Numerous large trials have investigated the efficacy and safety of GP IIb/IIIa inhibitors in a variety of ACS settings (see Table 6). A meta-analysis53 of 21 trials of IV GP IIb/IIIa inhibitors show their effectiveness in reducing 30 day outcomes of death, MI, or TVR by 3.8% ARR (NNT = 26), 33% RRR in ACS with PCI; by 1.4% (NNT = 71), 11% RRR in the setting of NSTEMI/ACS; and by 3.9% ARR (NNT = 26), 49% RRR in STEMI with PCI.
Abciximab. Abciximab has been demonstrated to be useful in STEMI, NSTEMI, and ACS with PCI, but not in ACS without PCI.54 A meta-analysis55 of five major trials (RAPPORT56, ISAR-2,57 ADMIRAL,58 ACE,59 CADILLAC60) (see Table 6) of abciximab in AMI noted a 1% ARR (NNT=100), 29% RRR in 30 day mortality, with effects persisting to 6-12 months, 1.8% ARR (NNT=56), 29% RRR, without an increase in bleeding. Furthermore, the upstream use of abciximab for STEMI in the ED or ambulance improved 30 day death, MI, or TVR by 18.5% ARR (NNT=5), 88% RRR.58 The large trials of abciximab in ACS, including EPIC,61 EPILOG,62 EPISTENT,63 and CAPTURE64 (see Table 6) have also been favorable, with the EPIC trial61 noting a two-fold increase in major bleeding and transfusion. Another meta-analysis65 of eight trials and 9290 PCI patients found a significant reduction in 30-day (0.5% ARR), 6-month (0.7% ARR), one-year (0.9% ARR) and three-year (1.8% ARR) mortality.
Abciximab is indicated in ACS or STEMI undergoing PCI within 24 hours and is given as a 0.25 mg/kg IV bolus (10-60 min prior to PCI) followed by 0.125 micrograms/kg/min (maximum of 10 micrograms/min) for 12 hours. Abciximab can be given with aspirin and low-dose, weight-adjusted UFH.61-64 When patients with intermediate risk chronic stable angina (ISAR-REACT)50 are pretreated with 600 mg clopidogrel 2 hours prior to PCI, the addition of abciximab provides no further benefit.66 However, in patients with ACS undergoing PCI (ISAR-REACT 2),66 the addition of abciximab to 600 mg clopidogrel and aspirin decreased 30-day death, MI, or TVR by 3% ARR (NNT=33), 25% RRR with no increase in major or minor bleeding. This improvement is greater in patients with ACS and positive troponin, 5.2% ARR (NNT=19), 28% RRR and not significant in patients with negative troponin.
Eptifibatide. Eptifibatide has been studied in several large trials in the settings of high-risk ACS (positive biomarkers or ECG changes)(PURSUIT67), and elective, urgent, and emergent PCI (IMPACT II,68 ESPRIT69) with favorable effects on composite outcomes. (See Table 6.) In general, the risks of major and minor bleeding, intracranial hemorrhage, thrombocytopenia, and platelet transfusion are no different than placebo, yet an increase in bleeding adverse events (not meeting the definition of major or minor bleeding such as a decrease of Hb > 3 gm) of 1-10% have been noted. The ESPRIT trial69 found that early upstream use (as might be given in the ED prior to results of biomarkers) of a double bolus of eptifibatide, instead of administration in the catheterization lab, had a significant reduction in 6-month outcome that was sustained for 12 months.
Eptifibatide is indicated in the setting of high- or intermediate-risk ACS, whether managed medically or with PCI. For ACS without PCI the dose is 180 micrograms/kg followed by 2 micrograms/kg/min up to discharge, CABG, or 72 hours. For PCI, the recommended dose is 180 micrograms/kg immediately prior to PCI followed by 2 micrograms/kg/min, with second 180 micrograms/kg bolus 10 minutes after first bolus, and continued until discharge or 24 hours after procedure. The dose is decreased to 1 microgram/kg/min in patients with renal disease with creatinine clearance (CrCl) less than 50 mL/min or serum creatinine greater than 2 mg/dL.
Tirofiban. Tirofiban has been studied in several large trials, including ACS with PCI within 72 hours (RESTORE),70 ACS without PCI (PRISM,71 PRISM-PLUS72), and patients getting PCI, with or without ACS (ADVANCE73) (see Table 6). In the PRISM study,74 tirofiban did not improve 30 day composite outcome except in those with elevated troponin, whether treated medically or with PCI, with 4.6% ARR (NNT=22) 75% RRR in death and 4.1% ARR (NNT=24), 63% RRR in MI. Similarly, the ADVANCE trial73 confirmed that non-ACS patients did not benefit from tirofiban, whereas those with high-risk ACS (positive biomarkers or ECG changes) did. In the PRISM-PLUS study72 patients who benefited from tirofiban could be predicted by having more than 2 of the following: age older than 65, prior CABG, prior aspirin use, prior beta-blocker use, and ST depression.
For STEMI patients undergoing PCI, both the TIGER-PA75 and On-TIME76 trials found early ED dosing of tirofiban in 100 and 507 STEMI patients (average of 33 minutes or 79 minutes prior to PCI, respectively) improved angiographic evidence of flow without adequate power to detect differences in outcome or bleeding. In STEMI patients not candidates for PCI or thrombolysis (usually because of delayed presentation)(TETAMI),77 tirofiban failed to improve outcome over standard medical therapy. Tirofiban has not been studied with thrombolysis.
Tirofiban is indicated for high- or intermediate-risk ACS or NSTEMI, along with aspirin and UFH or enoxaparin, at 0.4 micrograms/kg/min for 30 minutes, then 0.1 micrograms/kg/min infusion for 12-24 hours after PCI (or up to 108 hours.) For renal impairment (CrCl < 30 ml/min) the infusion rate is 0.05 micrograms/kg/min. Tirofiban is equally effective with either enoxaparin or UFH,78 but has not been studied in combination with clopidogrel or thrombolytic agents. Tirofiban results in a 2.5% absolute (NNH=40), 24% relative increased risk of minor bleeding without increased risk of major bleeding events, and a risk of thrombocytopenia of less than 2%.
Early upstream use of abciximab is likely beneficial prior to PCI for STEMI.58 In a meta-analysis79 of 6 trials of PCI for STEMI in 931 patients, early upstream administration of abciximab or tirofiban in the ED resulted in improved TIMI flow, with a non-significant trend toward lower mortality.
A frequent question for emergency physicians, however, is whether it makes a difference in high-risk ACS patients without STEMI if a GP IIb/IIIa inhibitor is administered upstream in the ED or just prior to PCI by the interventional cardiologist in the catheterization lab. Although this question has been inadequately addressed by the literature, there is preliminary evidence that upstream use may be important. Early upstream use has been shown to improve coronary flow and minimize myocardial damage.80 Upstream use of eptifibatide in the ESPRIT trial69 demonstrated significant improvement in outcome in comparison to "bailout" use in the catheterization lab. A systematic review of the CAPTURE, PURSUIT, and PRISM-PLUS data suggests that GP IIb/IIIa inhibitors are effective at reducing 30-d composite outcomes, both upstream prior to PCI (if performed) (1.3% ARR, (NNT=77), 34% RRR), as well as when given after PCI (for PCI-related events) (3.1% ARR (NNT=32), 41% RRR).81
GP IIb/IIIa inhibitors also may be considered in combination with thrombolytic agents during acute STEMI, although results of clinical trials have not been impressive. Of 8 clinical trials in 25,275 patients using GP IIb/IIIa inhibitors (except tirofiban) and thrombolytic combinations (IMPACT-AMI,82 TIMI 14,83 SK-eptifibatide,84 SPEED,85 INTRO AMI,86 INTEGRITI,87 GUSTO V,88 ASSENT-389), almost all trials demonstrated improvements in TIMI flow, trends or significant increases in bleeding, yet none demonstrated improvements in survival.
Another common question for emergency physicians is how safe and effective is the use of triple antiplatelet therapy with aspirin, thienopyridene, and GP IIb/IIIa inhibitor in ACS? At least for abciximab, the ISAR-REACT50 and ISAR-REACT 266 demonstrated a significant benefit of abciximab in addition to aspirin and 600 mg clopidogrel prior to PCI only in ACS patients, especially those with elevated troponin. In contrast, the GRACE registry90 reported on 8081 ACS patients, and noted similar mortality rates, but a 1% absolute (NNH = 100) and 50% relative increase in major bleeding among the 37% (3011) of patients on triple antiplatelet therapy in comparison to aspirin and thienopyridine.
Although there are significant differences both in action and cost between the various GP IIb/IIIa inhibitors,91 the relative efficacy of these agents has been inadequately studied. One comparison trial has addressed clinical outcome between abciximab vs. tirofiban in 4809 non-emergent PCI patients (TARGET-ACS),92 with significant benefit with abciximab at 30 days (1.6% ARR, 21% RRR) but not at 6 months. With abciximab there was an increase in minor bleeding and thrombocytopenia, but not major bleeding. Another trial93 of 642 PCI patients demonstrated no difference in outcome between eptifibatide and abciximab, although thrombocytopenia was more frequent with abciximab. In a retrospective analysis of 267 PCI patients, no significant differences were noted between abciximab and non-abciximab-treated patients, except a trend toward increased length of stay and hemorrhagic complications was noted in the abciximab group.94 In a cost analysis (sponsored by Lilly, producers of abciximab)95 of 32,529 patients in 99 hospitals, abciximab had the greatest cost and the most significant benefit, but also the most favorable cost-effectiveness ratio, whereas eptifibatide had the fewest complications, and tirofiban had the lowest incremental cost.
Perhaps the most significant difference between the three agents is ease of reversibility for urgent or emergent CABG.96 The antiplatelet effects of both eptifibatide and tirofiban are short-lived, and discontinuation of their infusions at least 4 hours prior to surgery is recommended, whereas abciximab generally requires multiple units of platelet transfusions prior to surgery, although CABG can be performed safely in these patients.97
GP IIb/IIIa inhibitors are contraindicated in several conditions (see Table 7), as well as in aortic dissection, acute pericarditis, traumatic or protracted CPR, and pregnancy because of their increased risk of bleeding. Complications of GP IIb/IIIa inhibitors include bleeding and severe thrombocytopenia (< 50,000 /mL). Severe thrombocytopenia occurs more often (0.4-1%) with abciximab than eptifibatide (0.2%) or tirofiban (0.3%). Human anti-chimeric antibodies (HACA) are formed with abciximab, and these may increase risk of thrombocytopenia, and have the potential to interfere with efficacy or cause allergy or hypersensitivity reactions. Most clinical trials use the TIMI major and TIMI minor criteria. TIMI major bleeding involves a drop in hemoglobin more than 5 g/dL (with or without an identified site), intracranial hemorrhage, or cardiac tamponade. TIMI minor bleeding involves a drop of hemoglobin greater than 3 g/dL but less than 5 g/dL with bleeding from a known site or spontaneous gross hematuria, hemoptysis, or hematemesis. Most GP IIb/IIIa trials have not detected increases in TIMI major bleeding, although several have noted an increase in TIMI minor bleeding in the range of 2-8% with any agent.
| Table 7. Contraindications* to GP IIb/IIIa Inhibitor Use |
| • Hypersensitivity • Active internal bleeding • Recent clinically relevant (e.g., GI, genitourinary) bleeding within 30 days (tirofiban, eptifibatide) • Major surgery or trauma < 6 weeks (eptifibatide, tirofiban) • Major surgery or trauma < 2 months (abciximab) • Stroke < 2 years or with residual deficit (abciximab) • Stroke < 30 days (eptifibatide, tirofiban) • Intracranial disease (neoplasm, arteriovenous malformation, neurysm) • Hemorrhagic stroke (tirofiban, eptifibatide) • Bleeding diathesis (abciximab) or clotting disturbance such as INR > 1.5 (tirofiban) or > 2 (eptifibatide) • Thrombocytopenia (< 100,000 cells/microL) • Severe uncontrolled hypertension (> 200/110 mmHg) • Vasculitis (abciximab) • Hypertensive retinopathy (abciximab) • Renal insufficiency (eptifibatide) or requiring dialysis (abciximab) • Severe liver failure (abciximab, tirofiban) • Clinically significant liver failure (eptifibatide) • Concomitant or planned use of another IV GP IIb/IIIa inhibitor (eptifibatide) * All of these are absolute contraindications for abciximab, eptifibatide, and tirofiban, unless otherwise noted in parentheses indicating the medication to which they apply. |
Authors' Bottom Line Recommendations:
- Initiate early dialogue with cardiologists regarding the upstream use of a GP IIb/IIIa inhibitor prior to PCI; preference for individual agent used; use of GP IIb/IIIa inhibitor if patient has already received aspirin and clopidogrel (triple antiplatelet therapy); and use of GP IIb/IIIa inhibitor in STEMI patients treated with thrombolysis.
- Give abciximab, eptifibatide, or tirofiban, upstream in high-risk ACS patients with elevated troponin, even if pretreated with aspirin and clopidogrel, prior to PCI.
- Avoid abciximab in patients not undergoing PCI.
- Consider eptifibatide or tirofiban infusions in high-risk ACS patients (ongoing ischemia, elevated troponin, TIMI risk score > 4, or ECG changes) not undergoing early PCI.
Antithrombotic Therapy
Antithrombotic agents useful in ACS include unfractionated heparin (UFH); enoxaparin (Lovenox), a low molecular weight heparin (LMWH); and bivalirudin (Angiomax, previously named Hirulog), a direct antithrombin.
Unfractionated Heparin (UFH). UFH binds antithrombin III and potentiates inactivation of thrombin as well as several other circulating coagulation factors. Because UFH is a heterogeneous mixture of various sized molecules, with high and variable protein binding and low bioavailability, its antithrombotic activity is variable and difficult to predict. UFH has several disadvantages in that it is unable to inhibit clot-bound thrombin, is inactivated by circulating platelet factor 4 and heparinase, and can increase platelet activation by increasing platelet GP IIb/IIIa receptor protein and ADP-induced binding. A rebound increase in ischemia and thrombosis after UFH discontinuation98 can occur in fewer than 1% of patients, yet result in a two-fold increase in MI and eight-fold increase in mortality (PURSUIT,67 ESSENCE,99 TIMI 11B100). UFH results in bleeding in approximately 10% of cases, and serious bleeding in 2% of cases. UFH also can result in heparin-induced thrombocytopenia (HIT), with platelet counts typically less than 100,000/microL and paradoxically increased thrombosis, in less than 3% of patients.101,102 Although HIT typically occurs after approximately 5 days of heparin therapy, it can occur within 1 day in patients previously sensitized. HIT can be diagnosed by thrombocytopenia (less than 150,000/ microL or 50% decrease from baseline) in the presence of anti-platelet factor 4(PF4)/heparin antibodies. An advantage of UFH is that it can be reversed by protamine, and may be preferable when CABG or other surgery is anticipated.
UFH is now considered standard therapy for MI, especially STEMI, although the evidence is not strong. Studies in MI prior to the routine use of aspirin suggested 17% and 22% reductions in death and MI, respectively, with UFH.103 In the era of thrombolysis, both the International Study Group104 of 20,891 patients and the ISIS-3 study105 of 41,299 patients failed to show benefit in mortality, but did increase major bleeding episodes. A meta-analysis106 of these and other early trials suggested, however, that UFH with aspirin provided a 0.5% ARR (NNT=200), 6% RRR in mortality compared to aspirin alone. A systematic analysis107 of 6 trials with tissue plasminogen activator in STEMI found an insignificant trend toward (9% RRR) decreased mortality with UFH and aspirin, with a significant increase in bleeding complications.
UFH also is considered by many to be standard therapy for ACS undergoing PCI because of familiarity with its use and ability to monitor its effects on the activated clotting time (ACT; normal range 70-180 seconds). Early ACS studies, however, were inconsistent in demonstrating superiority of UFH and aspirin compared with aspirin alone. A trial of 479 ACS patients108 with UA found that in comparison to aspirin alone, the combination of aspirin and UFH provided a 1.7% ARR (NNT = 59), 52% RRR decrease in MI and 5.8% ARR (NNT = 17), 35% RRR in recurrent ischemia. In contrast, the RISC trial109 of 796 ACS patients found the combination of heparin and aspirin together trended toward improved outcome over aspirin alone, but not significantly. A meta-analysis110 of six randomized trials suggested a strong trend toward improved outcome in ACS with heparin and aspirin vs. aspirin alone, with decreased death or MI at 2-12 weeks post-randomization by 2.4% ARR (NNT = 42), 18% RRR.
In ACS without thrombolytic or GP IIb/IIIa inhibitor, UFH is given at 70 U/kg (up to 5000 U bolus) and then 15 U/kg/hr (up to 1200 U/hr), with a target aPTT of 60-100 sec. In STEMI treated with thrombolytic, or ACS treated with GP IIb/IIIa inhibitor, UFH is given at 60 U/kg (up to 4000 U bolus) and then 12 U/kg/hr (up to 1000 U/hr), with a target aPTT of 50-70 sec. Prior to PCI, UFH is adjusted (commonly by repeat bolus) in the catheterization lab based on results of the ACT, aiming for levels of 250-350 s (or 200-250 s when using a GP IIb/IIIa inhibitor).
Enoxaparin. LMWHs such as enoxaparin have several advantages over UFH, including more predictable antithrombotic effects, increased binding of factor Xa compared to factor IIa, reduced risk of thrombocytopenia, and lack of requirement for monitoring. Although monitoring of anti-Xa activity is possible, it is not practiced routinely. Approximately 8% of enoxaparin treated patients with ACS are under-anticoagulated (anti-Xa activity < 0.5 IU/mL), and these patients have a 13.9% ARD, 2.4-fold increase in 30-day death or MI.111 Enoxaparin also does not increase platelet GP IIb/IIIa receptor protein or ADP-induced binding.
The ESSENCE trial99 of 3171 ACS patients demonstrated 3.5% ARR (NNT=29), 15% RRR in 30 day death, MI, or recurrent angina with enoxaparin over UFH, without significant difference in major bleeding, but a 4.2% absolute (NNH=24), 30% relative increase in overall bleeding. PCI was not used routinely in ESSENCE patients. The TIMI 11B100 trial of 3910 ACS patients found a 2.4% ARR (NNT=41), 12% RRR in 43 day death, MI or TVR, without a significant increase of in major bleeding during hospitalization. Both the ESSENCE99 and TIMI 11B100 trials demonstrated an increase in minor bleeding. These two trials provided the evidence for the AHA/ACC guidelines4 to recommend enoxaparin over UFH in ACS.
The recent SYNERGY trial112 comparing enoxaparin and UFH in 10,027 ACS patients undergoing PCI after aspirin (95%), thienopyridine (66%), and GP IIb/IIIa inhibitor use (57%), and found an insignificant difference in 30 day MI or death (14% vs 14.5%).112 Similarly, the recent A to Z trial113 of 3987 ACS patients, when using tirofiban and aspirin, found no clinical benefit of enoxaparin over UFH. Both SYNERGY112 and A to Z trials113 suggest enoxaparin significantly increases major bleeding [1.5% absolute (NNH=67), 16% relative and 0.8% absolute (NNH=63), 36% relative, respectively.] In a systematic overview of 21,946 patients in 6 randomized controlled trials,114 there was no difference in death at 30 d, blood transfusion, or major bleeding at 7 days between groups, but enoxaparin had a 0.9% ARR (NNT=111), 8% RRR in 30 d death or MI over UFH.
Although the ACC/AHA guidelines recommend antithrombotic therapy for all patients with ACS regardless of risk, the TIMI 11B study noted improved outcome only in the group with elevated troponin values. In a meta-analysis115 of the ESSENCE and TIMI 11B subjects, the greatest benefit of enoxaparin over UFH is in patients with TIMI risk scores greater than 4.
Although UFH traditionally has been preferred in the setting of thrombolysis for STEMI, enoxaparin may be a reasonable alternative. The recent EXTRACT-TIMI 25 trial116 of 20,506 STEMI patients treated with thrombolysis, enoxaparin improved 30 day death or MI by 2.1% ARR (NNT=48), 17% RRR, in comparison to UFH, with an increase in major bleeding [0.7% absolute (NNH=143), 50% relative.] Despite a lower dose of 0.75 mg/kg for elderly patients, and once daily dosing for those with renal insufficiency, there was an increase in major bleeding in comparison to UFH.
It is important not to give UFH to those already receiving subcutaneous enoxaparin within 12 hours, and not to give enoxaparin to those getting IV UFH because of an increased risk of bleeding.114 In the SYNERGY open-label trial,112 almost 800 patients underwent post-randomization crossover with a significant 4.6% absolute (NNH=22), 33% relative increase in 30-day death or MI, and a 16.3% absolute (NNH=6), 107% relative increase in major bleeding requiring transfusion.
Enoxaparin is contraindicated in patients with active major bleeding, thrombocytopenia in the presence of anti-PF4/heparin antibody, and should be used with caution in those with history of HIT, bleeding diathesis, uncontrolled hypertension, hemorrhagic stroke, recent GI bleeding, retinopathy, renal insufficiency, indwelling epidural catheters, or recent spinal puncture or spinal surgery. Enoxaparin may result in HIT as infrequently as UFH102 (much less than 3%) in non-surgical patients, and cross-reacts with the anti-PF4/heparin antibody in about one-third of HIT patients.117 Enoxaparin is relatively contraindicated with previous HIT, but can be used cautiously if results of platelet aggregation studies are negative.
Enoxaparin is given in doses of 1 mg/kg (based on ideal body weight, rounded off to the nearest 5 mg, and without a maximum dosage) subcutaneous every 12 hours. For PCI patients, when enoxaparin has been given within 8 hours of procedure, further dosing is not necessary, yet when the dose is between 9 and 12 hours, another dose of enoxaparin 0.3-0.5 mg/kg can be given IV just prior to PCI in the catheterization lab, or a 0.75 mg/kg IV dose of enoxaparin if GP IIb/IIIa inhibitor is also being given. Patients weighing less than 45 kg are at increased risk of bleeding, and UFH may be preferred. In severe renal insufficiency with creatinine clearance less than 30 mL/min, UFH may be preferred, but enoxaparin can be given in daily dose of 1 mg/kg. Calculate creatinine clearance routinely in elderly (age older than 75 years) or low body weight (less than 80 kg) individuals, even when creatinine values are less than 2.0 mg/dL (see Figure 1). As an example, a 70-kg, 75-year-old female with creatinine level of 1.8 has a creatinine clearance of 29 mL/min.
| Figure 1. Cockcroft-Gault Equation for Creatinine Clearance |
|
Bivalirudin. Bivalirudin (Angiomax, previously named Hirulog), is an analogue of hirudin, a direct thrombin inhibitor with several theoretical advantages over UFH, including lack of dependence on antithrombin III for effect, ability to inactivate thrombin in soluble and fibrin-bound states, lack of platelet aggregating activity, predictable anticoagulant response without need for monitoring, and a wider therapeutic window.118
Bivalirudin was studied in the REPLACE-2 trial,119 along with provisional use (based on complicated findings at angiography) of GP IIb/IIIa inhibitor in comparison to heparin and GP IIb/IIIa inhibitor in 6002 patients undergoing elective PCI. Although there was an insignificant trend toward a higher rate of death, MI, or TVR (7.6% vs 7.1%) at 30 days, bivalirudin had a significantly lower rate of major hemorrhage, 1.7% ARR (NNT=59), 41% RRR.
In the BAT trial120 of 4312 ACS patients undergoing angioplasty, bivalirudin, in comparison to high-dose UFH, decreased the 7 day primary combined outcome of death, MI or TVR by 1.7% ARR (NNT=59), 22% RRR, which persisted at 3 months and 6 months, with a decrease in major bleeding of 5.8% ARR (NNT=17), 62% RRR.
Bivalirudin is indicated in ACS patients undergoing PCI as well as those with history or risk of heparin-induced thrombocytopenia (HIT) syndrome, and usually is given with aspirin and provisionally may be given with a GP IIb/IIIa inhibitor. Bivalirudin is contraindicated in patients with active major bleeding and hypersensitivity. The loading dose of 0.75 mg/kg bolus is followed by 1.75 mg/kg/hr through and usually up to 4 hours following PCI. The ACT obtained 5 minutes after the bolus is used to determine the need for second bolus of 0.2 mg/kg (with a target ACT of > 350 sec). In renal insufficiency with creatinine clearance < 30 mL/min, the infusion rate is decreased to 1 mg/kg/h, and in dialysis patients, to 0.25 mg/kg/h.
Authors' Bottom Line Recommendations:
- Initiate early dialogue with cardiologists regarding preferences for UFH, enoxaparin, or bivalirudin prior to PCI.
- Give weight-based UFH or enoxaparin for STEMI and ACS patients undergoing PCI (and adjust doses if giving a GP IIb/IIIa inhibitor or thrombolytic).
- Consider using enoxaparin for ACS patients undergoing medical management.
- Use bivalirudin in the setting of prior HIT or ACS undergoing PCI.
- Avoid crossover from enoxaparin to UFH, or UFH to enoxaparin, within 12 hours of prior dosing.
References
1. Maroko PR, Kjekshus JK, Sobel BE, et al. Factors influencing infarct size following experimental coronary artery occlusions. Circulation 1971;43:67-82.
2. Pfeffer MA, Braunwald E. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 1990;81:1161-1172.
3. Miller TD, Christian TF, Hopfenspirger MR, et al. Infarct size after acute myocardial infarction measured by quantitative tomographic 99mTc sestamibi imaging predicts subsequent mortality. Circulation 1995;92:334-341.
4. Braunwald E, Antman EM, Beasley JW, et al. ACC/AHA 2002 guideline update for the management of patients with unstable angina and non-ST-segment elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients With Unstable Angina). 2002. Available at: http://www.acc.org/qualityandscience/clinical/guidelines/unstable/update_index.htm.
5. Gibler WB, Cannon CP, Blomkalns AL, et al. Practical implementation of the guidelines for unstable angina/non-ST-segment elevation myocardial infarction in the emergency department. A scientific statement from the American Heart Association Council on Clinical Cardiology (Subcommittee on Acute Cardiac Care), Council on Cardiovascular Nursing, and Quality of Care and Outcomes Research Interdisciplinary Working Group, in collaboration with the Society of Chest Pain Centers. Circulation 2005;111:2699-2710.
6. Fesmire FM, Decker WW, Diercks DB, et al. Clinical policy: Critical issues in the evaluation and management of adult patients with non-ST-segment elevation acute coronary syndromes. Ann Emerg Med 2006;48:270-301.
7. Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation 2004;110;e82-e293.
8. Ottani F, Galvani M, Nicolini FA, et al. Elevated cardiac troponin levels predict the risk of adverse outcomes in patients with acute coronary syndromes. Amer Heart J 2000;140:917-927.
9. Fleming SM, Daly KM. Cardiac troponins in suspected acute coronary syndrome. A meta-analysis of published trials. Cardiology 2001;95:66-73.
10. Yusuf S, Zhao F, Mehta SR, et al; the Clopidogrel in Unstable Angina to Prevent Recurrent Events Trials Investigators. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med 2001;345:494-502.
11. Antman EM, Cohen M, Bernink PJ, et al. The TIMI risk score for unstable angina/non-ST elevation MI: A method for prognostication and therapeutic decision making. JAMA 2000;284:835-842.
12. Schulman SP. Antiplatelet therapy in non-ST-segment elevation acute coronary syndromes. JAMA 2004;292:1875-1882.
13. Kurth T, Glynn RJ, Walker AM, et al. Inhibition of clinical benefits of aspirin on first myocardial infarction by nonsteroidal anti-inflammatory drugs. Circulation 2003;108:1191-1195.
14. Kimmel SE, Berlin JA, Reilly M, et al. The effects of nonselective non-aspirin non-steroidal anti-inflammatory medications on the risk of nonfatal myocardial infarction and their interaction with aspirin. J Am Coll Cardiol 2004;43:985-990.
15. Meijer A, Verheugt FW, Werter CJ, et al. Aspirin versus coumadin in the prevention of reocclusion and recurrent ischemia after successful thrombolysis: A prospective placebo-controlled angiographic study: Results of the APRICOT Study. Circulation 1993;87:1524-1530.
16. ISIS-2 (Second International Study of Infarct Survival) Collaborative Group. Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2. Lancet 1988;2:349-360.
17. Antithrombotic Trialists' Collaboration. Collaborative meta-analysis of randomized trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ 2002;324:71-86.
18. Savage MP, Goldberg S, Bove AA, et al. Effect of thromboxane A2 blockade on clinical outcome and restenosis after successful coronary angioplasty. Multi-Hospital Eastern Atlantic Restenosis Trial (M-HEART II). Circulation 1995;92:3194-200.
19. Antiplatelet Trialists' Collaboration. Collaborative overview of randomized trials of antiplatelet therapy-I: Prevention of death, myocardial infarction, and stroke by prolonged antiplatelet therapy in various categories of patients. BMJ 1994;308:81-106.
20. Mufson L, Black A, Roubin G, et al. A randomized trial of aspirin in PTCA: Effect of high dose versus low dose aspirin on major complications and restenosis (abstract). J Am Coll Cardiol 1988;11:236A.
21. Steinhubl SR, Lauer MS, Mukherjee DP, et al. The duration of pretreatment with ticlopidine prior to stenting is associated with the risk of procedure-related non-Q-wave myocardial infarctions. J Am Coll Cardiol 1988;32:1366-1370.
22. Kastrati A, Schuhlen H, Hausleiter J, et al. Restenosis after coronary stent placement and randomization to a 4-week combined antiplatelet or anticoagulant therapy: Six-month angiographic follow-up of the Intracoronary Stenting and Antithrombotic Regimen (ISAR) Trial. Circulation 1997;96:462-467.
23. Bennett CL, Davidson CJ, Raisch DW, et al. Thrombotic thrombocytopenic purpura associated with ticlopidine in the setting of coronary artery stents and stroke prevention. Arch Intern Med 1999;159:2524-2528.
24. Steinhubl SR, Tan WA, Foody JM, et al. Incidence and clinical course of thrombotic thrombocytopenic purpura due to ticlopidine following coronary stenting. EPISTENT Investigators. Evaluation of platelet IIb/IIIa inhibitor for stenting. JAMA 1999;281:806-810.
25. Steinhubl SR, Berger PB, Mann JT III, et al, for the CREDO Investigators. Early and sustained dual oral antiplatelet therapy following percutaneous coronary intervention: A randomized controlled trial. JAMA 2002;288:2411-2420.
26. Mahta SR,Yusuf S, Peters RJG, et al, for the Clopidogrel in Unstable angina to prevent Recurrent Events trial (CURE) Investigators. Effects of pretreatment with clopidogrel and aspirin followed by long-term therapy in patients undergoing percutaneous coronary intervention: The PCI-CURE study. Lancet 2001;358:527-533.
27. Sabatine MS, Cannon CP, Gibson CM, et al, for the CLARITY-TIMI 28 Investigators. Addition of aspirin to fibrinolytic therapy for myocardial infarction with ST-segment elevation. N Engl J Med 2005;352:1179-1189.
28. Chen ZM, Jiang LX, Chen YP, et al; COMMIT (ClOpidogrel and Metoprolol in Myocardial Infarction Trial) collaborative group. Addition of clopidogrel to aspirin in 45,852 patients with acute myocardial infarction: Randomized placebo-controlled trial. Lancet 2005;366:1607-1621.
29. Sabatine MS, Cannon CP, Gibson CM, et al, for the Clopidogrel as Adjunctive Reperfusion Therapy (CLARITY)-Thrombolysis in Myocardial Infarction (TIMI) 28 Investigators. Effect of clopidogrel pretreatment before percutaneous coronary intervention in patients with ST-elevation myocardial infarction treated with fibrinolytics. The PCI-CLARITY Study. JAMA 2005;294:1224-1232.
30. Budaj A, Yusuf S, Mehta SR, et al. Benefit of clopidogrel in patients with acute coronary syndromes without ST-segment elevation in various risk groups. Circulation 2002;106:1622-1626.
31. Nguyen TA, Diodati JG, Pharand C. Resistance to clopidogrel: A review of the evidence. J Am Coll Cardiol 2005;45:1157-1164.
32. Angiolillo DJ, Fernandez-Ortiz A, Bernardo E, et al. Platelet aggregation according to body mass index in patients undergoing coronary stenting: Should clopidogrel loading-dose be weight adjusted? J Invasive Cardiol 2004;16:169-174.
33. Matetzky S, Shenkman B, Guetta V, et al. Clopidogrel resistance is associated with increased risk of recurrent atherothrombotic events in patients with acute myocardial infarction. Circulation 2004;109:3171-3175.
34. The CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 1996;348:1329-1339.
35. Mehta RH, Roe MT, Mulgund J, et al. Acute clopidogrel use and outcomes in patients with non-ST-segment elevation acute coronary syndromes undergoing coronary artery bypass surgery. J Am Coll Cardiol 2006;48:281-286.
36. Hongo RH, Ley J, Dick SE, Yee RR. The effect of clopidogrel in combination with aspirin when given before coronary artery bypass grafting. J Am Coll Cardiol 2002;40:231-237.
37. Gansera B, Schmidtler F, Spiliopoulos K, et al. Urgent or emergent coronary revascularization using bilateral internal thoracic artery after previous clopidogrel antiplatelet therapy. Thorac Cardiovasc Surg 2003;51:185-189.
38. Englberger L, Faeh B, Berdat PA, et al. Impact of clopidogrel in coronary artery bypass grafting. Eur J Cardiothorac Surg 2004;26:96-101.
39. Chu MW, Wilson SR, Novick RJ, et al. Does clopidogrel increase blood loss following coronary artery bypass surgery? Ann Thorac Surg 2004;78:1536-1541.
40. Ascione R, Ghosh A, Rogers CA, et al. In-hospital patients exposed to clopidogrel before coronary artery bypass graft surgery: A word of caution. Ann Thorac Surg 2005;79:1210-1216.
41. Mehta RH, Chen AY, Pollack CV Jr, et al. Challenges in predicting the need for coronary artery bypass grafting at presentation in patients with non-ST-segment elevation acute coronary syndromes. Am J Cardiol 2006;98:624-627.
42. Jamal SM, Shrive FM, Ghali WA, et al; Canadian Cardiovascular Outcomes Research Team (CCORT). In-hospital outcomes after percutaneous coronary intervention in Canada: 1992/93 to 2000/01. Can J Cardiol 2003;19:782-789.
43. Singh M, Nuttall GA, Ballman KV, et al. Effect of abciximab on the outcome of emergency coronary artery bypass grafting after failed percutaneous coronary intervention. Mayo Clin Proc 2001;76:784-788.
44. Shubrooks SJ Jr, Nesto RW, Leeman D, et al. Urgent coronary bypass surgery for failed percutaneous coronary intervention in the stent era: Is backup still necessary? Am Heart J 2001;142:190-196.
45. Akowuah E, Shrivastava V, Jamnadas B, et al. Comparison of two strategies for the management of antiplatelet therapy during urgent surgery. Ann Thorac Surg 2005;80:149-152.
46. Harrington RA, Becker RC, Ezekowitz M, et al. Antithrombotic therapy for coronary artery disease: The seventh ACCP conference on antithrombotic and thrombolytic therapy. Chest 2004;126:513S-48S.
47. Sadanandan S, Cannon CP, Gibson CM, et al. TIMI Study Group. A risk score to estimate the likelihood of coronary artery bypass surgery during the index hospitalization among patients with unstable angina and non-ST-segment elevation myocardial infarction. J Am Coll Cardiol 2004;44:799-803.
48. Hochholzer W, Trenk D, Frundi D, et al. Time dependence of platelet inhibition after a 600-mg loading dose of clopidogrel in a large, unselected cohort of candidates for percutaneous coronary intervention. Circulation 2005;111:2560-2564.
49. Kandzari DE, Berger PB, Kastrati A, et al. Influence of treatment duration with a 600-mg dose of clopidogrel before percutaneous coronary revascularization. J Am Coll Cardiol 2004;44:2133-2136.
50. Kastrati A, Mehilli J, Schuhlen H, et al. A clinical trial of abciximab in elective percutaneous coronary intervention after pretreatment with clopidogrel. N Engl J Med 2004;350:232-238.
51. Patti G, Colonna G, Pasceri V, et al. Randomized trial of high loading dose of clopidogrel for reduction of periprocedural myocardial infarction in patients undergoing coronary intervention: Results from the ARMYDA-2 (Antiplatelet therapy for Reduction of MYocardial Damage during Angioplasty) Study. Circulation 2005;111:2099-2106.
52. Gurbel PA, Bliden KP, Hayes KM, et al. The relation of dosing to clopidogrel responsiveness and the incidence of high post-treatment platelet aggregation in patients undergoing coronary stenting. J Am Coll Cardiol 2005;45:1392-1396.
53. Sabatine MS, Jang IK. The use of glycoprotein IIb/IIIa inhibitors in patients with coronary artery disease. Am J Med 2000;109:224.
54. Simoons ML. Effect of glycoprotein IIb/IIIa receptor blocker abciximab on outcome in patients with acute coronary syndromes without early coronary revascularisation: The GUSTO IV-ACS randomised trial. Lancet 2001;357:1915-1924.
55. De Luca G, Suryapranata H, Stone GW, et al. Abciximab as adjunctive therapy to reperfusion in acute ST-segment elevation myocardial infarction: A meta-analysis of randomized trials. JAMA 2005;293:1759-1765.
56. Brener SJ, Barr LA, Burchenal JEB, et al on behalf of the ReoPro and Primary PTCA Organization and Randomized Trial (RAPPORT) Investigators. Randomized, placebo-controlled trial of platelet glycoprotein IIb/IIIa blockade with primary angioplasty for acute myocardial infarction. Circulation 1998;98:734-741.
57. Neumann FJ, Kastrati A, Schmitt C, et al. Effect of glycoprotein IIb/IIIa receptor blockade with abciximab on clinical and angiographic restenosis rate after the placement of coronary stents following acute myocardial infarction. J Am Coll Cardiol 2000;35:915-921.
58. Montalescot G, Barragan P, Wittenberg O, et al, for the ADMIRAL Investigators. Platelet glycoprotein IIb/IIIa inhibition with coronary stenting for acute myocardial infarction. N Engl J Med 2001;344:1895-1903.
59. Antoniucci D, Migliorini A, Parodi G, et al. Abciximab-supported infarct artery stent implantation for acute myocardial infarction and long-term survival: A prospective, multicenter, randomized trial comparing infarct artery stenting plus abciximab with stenting alone. Circulation 2004; 109:1704-1706.
60. Stone GW, Grines CL, Cox DA, et al; Controlled Abciximab and Device to Lower Late Angioplasty Complications (CADILLAC) Investigators. Comparison of angioplasty with stenting, with or without abciximab, in acute myocardial infarction. N Engl J Med 2002;346:957-966.
61. Use of a monoclonal antibody directed against the platelet glycoprotein IIb/IIIa receptor in high-risk coronary angioplasty. The EPIC Investigation. N Engl J Med 1994;330:956-961.
62. Lincoff AM, Tcheng JE, Califf RM, et al, for the EPILOG Investigators. Sustained suppression of ischemic complications of coronary intervention by platelet GP IIb/IIIa blockade with abciximab: One-year outcome in the EPILOG trial. Circulation 1999;99:1951-1958.
63. Lincoff AM, Califf RM, Moliterno DJ, et al, for the EPISTENT Investigators. Complementary clinical benefits of coronary-artery stenting and blockade of platelet glycoprotein IIb/IIIa receptors. N Engl J Med 1999;341:319-327.
64. Randomised placebo-controlled trial of abciximab before and during coronary intervention in refractory unstable angina: The CAPTURE study. Lancet 1997;349:1429-1435.
65. Anderson KM, Califf RM, Stone GW, et al. Long-term mortality benefit with abciximab in patients undergoing percutaneous coronary intervention. J Am Coll Cardiol 2001;37:2059-2065.
66. Kastrati A, Mehilli J, Neumann FJ, et al, for the Intracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment 2 (ISAR-REACT 2) Trial Investigators. Abciximab in patients with acute coronary syndromes undergoing percutaneous coronary intervention after clopidogrel pretreatment. The ISAR-REACT 2 randomized trial. JAMA 2006;295:1531-1538.
67. Inhibition of platelet glycoprotein IIb/IIIa with eptifibatide in patients with acute coronary syndromes. The PURSUIT Trial Investigators. Platelet Glycoprotein IIb/IIIa in Unstable Angina: Receptor Suppression Using Integrilin Therapy. N Engl J Med 1998;339:436-443.
68. Randomised placebo-controlled trial of effect of eptifibatide on complications of percutaneous coronary intervention: IMPACT-II. Integrilin to Minimise Platelet Aggregation and Coronary Thrombosis-II. Lancet 1997;349:1422-1428.
69. O'Shea JC, Hafley GE, Greenberg S, et al, ESPRIT Investigators. Platelet glycoprotein IIb/IIIa integrin blockade with eptifibatide in coronary stent intervention: The ESPRIT trial: a randomized controlled trial. JAMA 2001;285:2468-2473.
70. Gibson CM, Goel M, Cohen DJ, et al, for the RESTORE Investigators. Six-month angiographic and clinical follow-up of patients prospectively randomized to receive either tirofiban or placebo during angioplasty in the RESTORE trial. Randomized Efficacy Study of Tirofiban for Outcomes and Restenosis. J Am Coll Cardiol 1998;32:28-34.
71. Platelet Receptor Inhibition in Ischemic Syndrome Management (PRISM) Study Investigators. A comparison of aspirin plus tirofiban with aspirin plus heparin for unstable angina. N Engl J Med 1998;338:1498-1505.
72. Platelet Receptor Inhibition in Ischemic Syndrome Management in Patients Limited by Unstable Signs and Symptoms (PRISM-PLUS) Study Investigators. Inhibition of the platelet glycoprotein IIb/IIIa receptor with tirofiban in unstable angina and non-Q-wave myocardial infarction. N Engl J Med 1998;338:1488-1497.
73. Valgimigli M, Percoco G, Barbieri D, et al. The additive value of tirofiban administered with the high-dose bolus in the prevention of ischemic complications during high-risk coronary angioplasty: The ADVANCE Trial. J Am Coll Cardiol 2004;44:14-19.
74. Heeschen C, Hamm CW, Goldmann B, et al. Troponin concentrations for stratification of patients with acute coronary syndromes in relation to therapeutic efficacy of tirofiban. PRISM Study Investigators. Platelet Receptor Inhibition in Ischemic Syndrome Management. Lancet 1999;354:1757-1762.
75. Lee DP, Herity NA, Hiatt BL, et al. Adjunctive platelet glycoprotein IIb/IIIa receptor inhibition with tirofiban before primary angioplasty improves angiographic outcomes: Results of the TIrofiban Given in the Emergency Room before Primary Angioplasty (TIGER-PA) pilot trial. Circulation 2003;107:1497-1501.
76. van't Hof AW, Ernst N, de Boer MJ, et al. Facilitation of primary coronary angioplasty by early start of a glycoprotein 2b/3a inhibitor: Results of the ongoing tirofiban in myocardial infarction evaluation (On-TIME) trial. Eur Heart J 2004;25:837-846.
77. Cohen M, Gensini GF, Maritz F, et al. The safety and efficacy of subcutaneous enoxaparin versus intravenous unfractionated heparin and tirofiban versus placebo in the treatment of acute ST-segment elevation myocardial infarction patients ineligible for reperfusion (TETAMI): A randomized trial. J Am Coll Cardiol 2003;42:1348-1356.
78. Blazing MA, de Lemos JA, White HD, et al. Safety and efficacy of enoxaparin vs unfractionated heparin in patients with non-ST-segment elevation acute coronary syndromes who receive tirofiban and aspirin: A randomized controlled trial. JAMA 2004;292:55-64.
79. Montalescot G, Borentain M, Payot L, et al. Early vs late administration of glycoprotein IIb/IIIa inhibitors in primary percutaneous coronary intervention of acute ST-segment elevation myocardial infarction: a meta-analysis. JAMA 2004;292:362-366.
80. Bolognese L, Falsini G, Liistro F, et al. Randomized comparison of upstream tirofiban versus downstream high bolus dose tirofiban or abciximab on tissue-level perfusion and troponin release in high-risk acute coronary syndromes treated with percutaneous coronary interventions: The EVEREST trial. J Am Coll Cardiol 2006;47:522-528.
81. Boersma E, Akkerhuis KM, Théroux P, et al. Platelet glycoprotein IIb/IIIa receptor inhibition in non-ST-elevation acute coronary syndromes. Early benefit during medical treatment only, with additional protection during percutaneous coronary intervention. Circulation 1999;100:2045-2048.
82. Ohman EM, Kleiman NS, Gacioch G, et al. Combined accelerated tissue-plasminogen activator and platelet glycoprotein IIb/IIIa integrin receptor blockade with integrilin in acute myocardial infarction. Results of a randomized, placebo-controlled, dose-ranging trial. Circulation 1997;95:846.
83. Antman, EM, Giugliano, RP, Gibson, MC, et al, for the TIMI 14 Investigators. Abciximab facilitates the rate and extent of thrombolysis: Result of the Thrombolysis In Myocardial Infarction (TIMI) 14 trial. Circulation 1999;99:2720.
84. Ronner E, van Kesteren HA, Zijnen P, et al. Safety and efficacy of eptifibatide vs placebo in patients receiving thrombolytic therapy with streptokinase for acute myocardial infarction; A phase II dose escalation, randomized, double-blind study. Eur Heart J 2000;21:1530-1536.
85. [No authors listed.] Trial of abciximab with and without low-dose reteplase for acute myocardial infarction. Strategies for Patency Enhancement in the Emergency Department (SPEED) Group. Circulation 2000;101:2788.
86. Brener SJ, Zeymer U, Adgey AA, et al. Eptifibatide and low-dose tissue plasminogen activator in acute myocardial infarction. The integrilin and low-dose thrombolysis in acute myocardial infarction (INTRO AMI) trial. J Am Coll Cardiol 2002;39:377.
87. Giugliano RP, Roe MT, Harrington RA, et al. Combination reperfusion therapy with eptifibatide and reduced-dose tenecteplase for ST-elevation myocardial infarction. Results of the integrilin and tenecteplase in acute myocardial infarction (INTEGRITI) Phase II Angiographic trial. J Am Coll Cardiol 2003;41:1251.
88. Topol EJ. Reperfusion therapy for acute myocardial infarction with fibrinolytic therapy or combination reduced fibrinolytic therapy and platelet glycoprotein IIb/IIIa inhibition: The GUSTO V randomised trial. Lancet 2001;357:1905.
89. Efficacy and safety of tenecteplase in combination with enoxaparin, abciximab, or unfractionated heparin: The ASSENT-3 randomised trial in acute myocardial infarction. Lancet 2001;358:605.
90. Lim MJ, Eagle KA, Gore JM, et al. for the Global Registry of Acute Coronary Events Investigators. Treating patients with acute coronary syndromes with aggressive antiplatelet therapy (from the Global Registry of Acute Coronary Events). Am J Cardiol 2005;96:917-921.
91. Brown DL, Fann CS, Chang CJ. Meta-analysis of effectiveness and safety of abciximab versus eptifibatide or tirofiban in percutaneous coronary intervention. Am J Cardiol 2001;87:537-541.
92. Topol EJ, Moliterno DJ, Herrmann HC, et al; TARGET Investigators. Do Tirofiban and ReoPro Give Similar Efficacy Trial. Comparison of two platelet glycoprotein IIb/IIIa inhibitors, tirofiban and abciximab, for the prevention of ischemic events with percutaneous coronary revascularization. N Engl J Med 2001;344:1888-1894.
93. Suleiman M, Gruberg L, Hammerman H, et al. Comparison of two platelet glycoprotein IIb/IIIa inhibitors, Eptifibatide and abciximab: Outcomes, complications and thrombocytopenia during percutaneous coronary intervention. J Invasive Cardiol 2003;15:324-325.
94. Kimmelstiel C, Phang R, Rehman A, et al. Short-term comparative outcomes associated with the use of GP IIb/IIIa antagonists in patients undergoing coronary intervention. J Thromb Thrombolysis 2001;11:203-209.
95. McCollam PL, Foster DA, Riesmeyer JS. Cost and effectiveness of glycoprotein IIb/IIIa-receptor inhibitors in patients with acute myocardial infarction undergoing percutaneous coronary intervention. Am J Health Syst Pharm 2003;60:1251-1256.
96. Dyke CM. Safety of glycoprotein IIb-IIIa inhibitors: A heart surgeon's perspective. Am Heart J 1999;138:307-316.
97. Pang JT, Fort S, Della Siega A, et al. Emergency coronary artery bypass surgery in the era of glycoprotein IIb/IIIa receptor antagonist use. J Card Surg 2002;17:425-431.
98. Theroux P, Waters D, Lam J, et al. Reactivation of unstable angina after the discontinuation of heparin. N Engl J Med 1992;327:141-145.
99. Cohen M, Demers C, Gurfinkel EP, et al. A comparison of low-molecular-weight heparin with unfractionated heparin for unstable coronary artery disease. Efficacy and Safety of Subcutaneous Enoxaparin in Non-Q-Wave Coronary Events Study Group. N Engl J Med 1997;337:447-452.
100. Antman EM, McCabe CH, Gurfinkel EP, et al. Enoxaparin prevents death and cardiac ischemic events in unstable angina/non-Q-wave myocardial infarction. Results of the thrombolysis in myocardial infarction (TIMI) 11B trial. Circulation 1999;100;1593-1601.
101. Warkentin TE, Levine MN, Hirsh J, et al. Heparin-induced thrombocytopenia in patients treated with low-molecular-weight heparin or unfractionated heparin. N Engl J Med 1995;332:1330-1335.
102. Locke CFS, Dooley J, Gerber J. Rates of clinically apparent heparin-induced thrombocytopenia for unfractionated heparin vs. low molecular weight heparin in non-surgical patients are low and similar. Thomb J 2005;3:4-9.
103. MacMahon S, Collins R, Knight C, et al. Reduction in major morbidity and mortality by heparin in acute myocardial infarction. Circulation 1988;78:II-98 (Abstract).
104. The International Study Group. In-hospital mortality and clinical course of 20,891 patients with suspected acute myocardial infarction randomized between alteplase and streptokinase with or without heparin. Lancet 1990;336:71-75.
105. [No authors listed.] ISIS-3 Collaborative Group. ISIS-3: Randomised comparison of streptokinase vs tissue plasminogen activator vs anistreplase and of aspirin plus heparin vs aspirin alone among 41,299 cases of suspected acute myocardial infarction. ISIS-3 (Third International Study of Infarct Survival) Collaborative Group. Lancet 1992;339:753-770.
106. Collins R, Peto R, Baigent C, et al. Aspirin, heparin, and fibrinolytic therapy in suspected acute myocardial infarction. N Engl J Med 1997;336:847-860.
107. Mahaffey KW, Granger CB, Collins R, et al. Overview of randomized trials of intravenous heparin in patients with acute myocardial infarction treated with thrombolytic therapy. Am J Cardiol 1996;77:551-556.
108. Theroux P, Ouimet H, McCans J, et al. Aspirin, heparin, or both to treat acute unstable angina. N Engl J Med 1988;319:1105-1111.
109. The RISC Group. Risk of myocardial infarction and death during treatment with low dose aspirin and intravenous heparin in men with unstable coronary artery disease. Lancet 1990;336:827-830.
110. Oler A, Whooley MA, Oler J, et al. Adding heparin to aspirin reduces the incidence of myocardial infarction and death in patients with unstable angina. A meta-analysis. JAMA 1996;276:811-815.
111. Montalescot G, Collet JP, Payot L, et al. Anti-Xa activity relates to outcome in acute coronary syndromes treated with enoxaparin. Circulation 2004;108:IV-501.
112. Ferguson JJ, Califf Rm, Antman EM, et al, SYNERGY Trial Investigators. Enoxaparin vs unfractionated heparin in high-risk patients with non-ST-segment elevation acute coronary syndromes managed with an intended early invasive strategy: primary results of the SYNERGY randomized trial. JAMA 2004;292:45-54.
113. Blazing MA, de Lemos JA, White HD, et al, A to Z Investigators. Safety and efficacy of enoxaparin vs unfractionated heparin in patients with non-ST-segment elevation acute coronary syndromes who receive tirofiban and aspirin: A randomized controlled trial. JAMA 2004;292:55-64.
114. Peterson JL, Mahaffey KW, Hasselblad V, et al. Efficacy and bleeding complications among patients randomized to enoxaparin or unfractionated heparin for antithrombin therapy in non-ST-segment elevation acute coronary syndromes: A systematic overview. JAMA 2004;292:89-96.
115. Antman EM, Cohen M, Radley D, et al. Assessment of the treatment effect of enoxaparin for unstable angina/non-Q-wave myocardial infarction: TIMI 11B-ESSENCE meta-analysis. Circulation 1999;100:1602-1608.
116. Antman EM, Morrow DA, McCabe CH, et al, ExTRACT-TIMI 25 Investigators. Enoxaparin versus unfractionated heparin with fibrinolysis for ST-elevation myocardial infarction. N Engl J Med 2006;354:1477-1488.
117. Slocum MM, Adams JG Jr, Teel R, et al. Use of enoxaparin in patients with heparin-induced thrombocytopenia syndrome. J Vasc Surg 1996;23:839-843.
118. Bates ER. Bivalirudin for percutaneous coronary intervention and in acute coronary syndromes. Curr Cardiol Rep 2001;3:348-354.
119. Lincoff AM, Bittl JA, Harrington RA, et al, REPLACE-2 investigators. Bivalirudin and provisional glycoprotein IIb/IIIa blockade compared with heparin and planned glycoprotein IIb/IIIa blockade during percutaneous coronary intervention: REPLACE-2 randomized trial. JAMA 2003;289:853-863.
120. Bittl JA, Chaitman BR, Feit F, et al. Bivalirudin versus heparin during coronary angioplasty for unstable or postinfarction angina: Final report reanalysis of the Bivalirudin Angioplasty Study. Am Heart J 2001;142:952-959.
It's a daunting task to read from A to Z through the numerous clinical trials in cardiology in pursuit of a cure for acute coronary syndrome (ACS.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.