ASA Use Doubles Death Rate From ICH
ASA Use Doubles Death Rate From ICH
Abstract & Commentary
By John J. Caronna, MD, Vice-Chairman, Department of Neurology, Cornell University Medical Center, Professor of Clinical Neurology, NewYork-Presbyterian Hospital. Dr. Caronna reports no consultant, stockholder, speaker's bureau, research, or other relationship related to this field of study.
Synopsis: Saloheimo and colleagues observed poor short-term outcomes and increased mortality, probably attributable to rapid enlargement of hematomas.
Source: Saloheimo P, et al. Regular Aspirin-Use Preceding the Onset of Primary Intracerebral Hemorrhage Is An Independent Predictor for Death. Stroke. 2006;37:129-133.
The recent hemorrhagic stroke of Ariel Sharon, who was on anticoagulation because of a previous embolic stroke related to a patent foramen ovale, underlines the dangers of anticoagulation in elderly patients. Many reports have demonstrated that patients on warfarin who have an intracerebral hemorrhage (ICH) show a high mortality rate due to rapid enlargement of their hematomas.1 The use of antiplatelet agents has not been shown to exert a similar effect. Therefore, in order to determine the role of Aspirin (ASA) premedication, Saloheimo and colleagues investigated outcome and hematoma enlargement in a population-based cohort of patients with ICH.
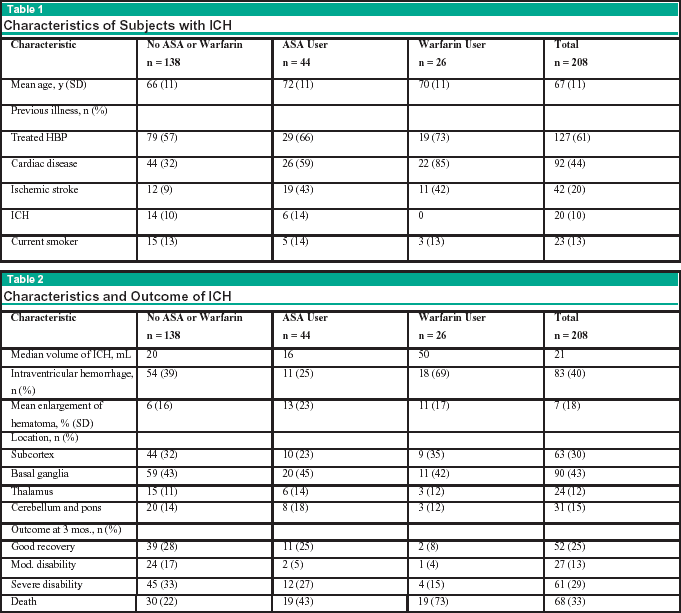
Saloheimo et al identified all subjects with intracerebral hemorrhage between January 1993 and September 1995 in a population of approximately 350,000 people in Finland. The study included all patients admitted with ICH to a university hospital during this period. Information about previous diseases, history of hypertension, medications, and smoking history were extracted from the hospital records. Complications during hospitalization such as infection, deep vein thrombosis, and pulmonary embolus were recorded, as were neurosurgical interventions. During the study period, 203 patients were admitted to the hospital with intracerebral hemorrhage, and the diagnosis was verified with a head CT scan. Five subjects succumbed elsewhere, and the diagnosis was verified at autopsy. The baseline characteristics of the study subjects are shown in Table 1. The study population included 101 men and 107 women. The ASA users were significantly older than the non-users of ASA or warfarin, and a history of either cardiac disease or ischemic stroke was more common in the group of ASA/ warfarin users than in non-users.
|
|
The clinical characteristics and outcomes of the 208 subjects are shown in Table 2. On admission, ASA users did not have larger hematomas than non-users, but warfarin users had significantly larger hematomas than ASA users and non-users. The groups differed significantly by outcome at 3 months and bleeding into the ventricles; the latter finding being most frequent in warfarin users. Primary ICH was the cause of death in 89% of the 19 fatalities in the group of ASA users. Among warfarin users, the cause of death was the primary ICH in 95% of the 19 fatalities. Thirty non-users of ASA and warfarin died within 3 months; the cause of death being the primary bleed in 24 patients (80%), and pneumonia, MI, or GI bleeding in the others. The overall mortality of all groups at 3 months was 33%.
There were significant differences between the survival curves of ASA users and non-users of ASA and warfarin, as well as between warfarin users and non-users of ASA and warfarin, and between ASA users and warfarin users. There was a 2-fold increase in 3-month mortality in ASA users compared with non-users of ASA and warfarin. On admission to the study, ASA-related ICH were of the same size as those seen in non-users of ASA and warfarin.
Saloheimo et al found that ASA use was significantly associated with hematoma growth, which is an independent factor increasing mortality after ICH.2 Saloheimo et al conclude that the effect of ASA use on 3-month outcome after ICH was attributable to early enlargement of the hematoma in ASA users. Alternatively, use of ASA may have emerged as a risk factor for death after ICH because of a proxy effect of the factors associated with its use (for example, patient age, history of cardiac disease, ischemic stroke and diabetes). In contrast, the high 3-month mortality in warfarin users reflects the fact that most of them showed large hematomas on admission and, in this series, no affective measure to reverse the anticoagulant effect was used.
Commentary
Previously recognized independent predictors of short-term mortality after spontaneous ICH include size and location of the hemorrhage, a midline shift on head CT, IVH, low Glasgow Coma Scale score on admission, and elevated blood glucose and blood pressure on admission.3 Previous studies, however, have not found recent use of anti-platelet drugs to be associated with an increased risk of either hematoma expansion or higher mortality.1,4-5 The present study offers a plausible explanation for the increased mortality of ASA users; namely, more frequent hematoma enlargement caused by impaired platelet mediated hemostasis during the first few days after the onset of ICH. In the present study, however, Saloheimo et al were unable to prove this hypothesis because a second CT scan was not available in all patients. Because hematoma volume has such a potent effect on mortality and functional outcome following ICH, it seems likely that any factors that promote increased volume, such as increased blood coagulability, thrombocytopenia, insufficient thrombin generation, elevated INR, and impaired platelet function all may promote hematoma enlargement and, thereby, affect mortality and morbidity after ICH. Saloheimo et al's findings are provocative, and they do not suggest that ASA use should be curtailed in patients with cardio and cerebrovascular disease, rather they indicate that prevention of hematoma growth may be needed by ICH patients who have previously used ASA. It is accepted that patients on warfarin need emergent reversal of anticoagulation. It, therefore, seems logical to reverse the antiplatelet effect of ASA in acute ICH patients by the use of platelet transfusions. Further studies are needed to determine whether the benefits off interventions such as platelet transfusion outweigh the risks in ASA associated ICH.
References
1. Rosand J, et al. The Effect of Warfarin and Intensity of Anticoagulation on Outcome of Intracerebral Hemorrhage. Arch Int Med. 2004;164:880-884.
2. Fujii Y, et al. Multivariate Analysis of Predictors of Hematoma Enlargement in Spontaneous Intracerebral Hemorrhage. Stroke. 1998;29:1160-1166.
3. Juvela S. Risk Factors for Impaired Outcome After Spontaneous Intracerebral Hemorrhage. Arch Neurol. 1995;52:1193-1200.
4. Flibotte JJ, et al. Warfarin, Hematoma Expansion, and Outcome of Intracerebral Hemorrhage. Neurology. 2004;63:1059-1064.
5. Nilsson OG, et al. Prediction of Death in Patients with Primary Intracerebral Hemorrhage: A Prospective Study of a Defined Population. J Neurosurg. 2002;97:531-536.
The recent hemorrhagic stroke of Ariel Sharon, who was on anticoagulation because of a previous embolic stroke related to a patent foramen ovale, underlines the dangers of anticoagulation in elderly patients.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.