Pediatric Cancer Emergencies: Critical Diagnostic and Management Strategies
Pediatric Cancer Emergencies: Critical Diagnostic and Management Strategies
Authors: Emily Fontane, MD, FACEP, FAAP, Assistant Clinical Professor of Emergency Medicine and Pediatrics, Brody School of Medicine at East Carolina University; Attending Physician, Adult and Pediatric Emergency Medicine, Pitt County Memorial Hospital, Greenville, North Carolina; and Ronald M. Perkin, MD, MA, Professor and Chairman, Department of Pediatrics, Brody School of Medicine at East Carolina University; Attending Physician, Pediatric Intensive Care Unit, University Health Systems of Eastern Carolina Children's Hospital; Medical Director,University Health Systems of Eastern Carolina Children's Hospital, Greenville, North Carolina
Peer Reviewer: Jeffrey F. Linzer, Sr., MD, FAAP, FACEP, Assistant Professor of Pediatrics and Emergency Medicine, Emory University School of Medicine, Atlanta, Georgia
What percentage of previously healthy children presenting to the emergency department (ED) have an oncologic emergency? Hypothetically, 2 to 4 children younger than 15 years with an undiagnosed cancer may present to a medical facility with an annual pediatric census of 20,000. The probability that any of these children will present to the ED with an oncologic emergency is rare; however, a high index of suspicion and prior preparation are needed to manage an oncologic emergency in the pediatric patient successfully. An oncologic emergency can occur in a previously well child without known cancer, in a child with known cancer after cancer treatment, or in a child with a history of cancer who has tumor recurrence. The need for recognition and treatment of complications arising in children with cancer comes with the improved prognosis of children with cancer.1 The authors review the most critical pediatric oncologic emergencies and strategies to improve outcomes. — The Editor
Introduction
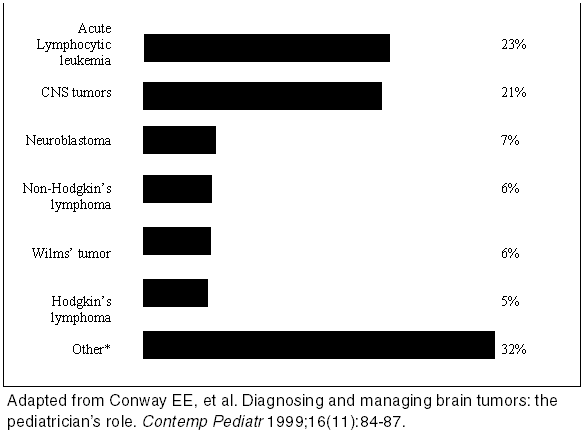
In the United States, an estimated 12,500 children and adolescents are diagnosed with cancer each year; 9,500 children are younger than 15 years.2,3 That is, for every 10,000 children, approximately 1 to 2 children younger than 15 years develop the disease.3 Leukemias (e.g., acute lymphoblastic leukemia and acute myeloid leukemia), central nervous system tumors (e.g., brain tumors and neuroblastoma), lymphomas (e.g., Hodgkin's lymphoma and non-Hodgkin's lymphoma), sarcomas (e.g., osteosarcoma, Ewing's sarcoma, rhabdomyosarcoma), and nephroblastoma are among the most common malignancies in the pediatric age group (Figure 1).4 Of the most common childhood malignancies, leukemias and brain tumors account for more than half of the new cases. Leukemia is the most common childhood malignancy, and the most common type of leukemia in children is acute lymphoblastic leukemia. The most common solid tumors in children are brain tumors, and brain tumors are the second most common childhood malignancy. Five-year survival rates for all childhood cancers is approaching 80%, reflecting the improvements in diagnosis, treatment, and long-term care.
| Figure 1. Distribution of Cancer in Children Younger than 15 Years |
|
|
Cancer is the leading cause of death by disease in children 1 to 14 years of age and remains responsible for more deaths from ages 1 year to adolescence than any other disease in the United States—more than asthma, diabetes, cystic fibrosis, and acquired immunodeficiency syndrome (AIDS) combined.3 Although the causes of childhood cancers are largely unknown, Down syndrome and other genetic syndromes, chemotherapy, radiation, and AIDS have been linked to an increased risk of specific childhood cancers. Notably, children with cancer and co-morbidities due to Down syndrome or other genetic syndromes, AIDS, prior chemotherapy or radiation may have more adverse events during therapy for their cancer.
The emergency physician must be able to recognize signs and symptoms associated with oncologic emergencies, and she/he must be prepared to manage these emergencies in children.1 Once a patient with an oncologic emergency has been identified and stabilized, immediate consultation with a pediatric hematologist-oncologist and transfer of the patient to a center with experience in treating children with cancer should be arranged. A multidisciplinary (including pediatric oncologists, pediatric intensivists, pediatric radiologists and pediatric surgeons) and specialized approach to the management of cancer and its complications in children is the standard of care. A brief discussion of the management of several oncologic emergencies in children that may present to the ED follows. All children who present to the ED with an oncologic emergency—be it neurological, respiratory, circulatory, metabolic, hematological, or infectious—are managed in the same manner as all patients with medical emergencies (i.e., with initial focus on resuscitation and stabilization of respiratory, circulatory, and neurological systems).
Superior Mediastinal Syndrome
Superior vena cava (SVC) obstruction by compression from mediastinal malignancies has been well described.5-23 The thin wall and low intraluminal pressure characteristic of the SVC factor greatly in the pathophysiology of the superior vena cava syndrome (SVCS) produced by a compressive mediastinal mass. SVC obstruction by malignancy in children is commonly accompanied by respiratory symptoms indicative of tracheal and/or bronchial compression as well. Because large mediastinal masses in children commonly compress, not only the SVC, but also the trachea and other superior mediastinal structures, the clinical condition in children has been referred to as superior mediastinal syndrome (SMS).
SMS in children constitutes a medical emergency.1,3 The mediastinum is the region of the thorax between the right and left pleurae. The superior mediastinum is the region bordered by the thoracic inlet and a plane, which passes through the sternal angle to the 4th – 5th vertebrae. Structures in the superior mediastinum include the thymus anteriorly; the great vessels, notably the superior vena cava, centrally; and the esophagus, trachea, and thoracic duct posteriorly.5 SMS appears at presentation in 12% of pediatric patients with malignant mediastinal tumors.3 It can present de novo or as a complication during therapy.6,7 Lymphoma and leukemia are the most common primary malignant causes of SMS in children.1 Nonmalignant causes of SVCS/SMS in children include teratoma, thymoma, and cystic hygroma. Iatrogenic or secondary causes include surgery for congenital heart disease, catheterization for venous access, and placement of ventricular shunts for hydrocephalus.
The symptoms and clinical signs in children are similar to those in adults, however, the condition is considered more critical in children. Symptoms associated with SMS in children include swelling of the face, neck and upper torso; cough; stridor; dyspnea; wheezing; chest pain; headache; change in vision; ear fullness; anxiety; confusion; syncope. Clinical signs include prominence of neck and superficial chest veins, cyanosis, and plethora. SMS may be misdiagnosed as asthma. History, physical examination, and laboratory/imaging studies help determine the severity of obstruction. In leukemia and lymphoma, the complete blood count may reveal cell line cytopenia or leukocytosis, and serum uric acid, urea nitrogen, creatinine, and lactate dehydrogenase levels may be elevated. A chest radiograph will show a mediastinal mass or mediastinal widening with tracheal compression or deviation. Computed tomography (CT) scanning or magnetic resonance (MR) imaging of the chest will help assess the extent of SVC thrombosis and collateral circulation. An echocardiogram evaluates cardiac motility and the degree of venous return impairment.
Neuromuscular blockade and deep sedation in children with SMS has been associated with fatal deterioration of respiratory and cardiovascular systems.1,18 It has been suggested that the deterioration observed in children with SMS after paralysis and deep sedation is caused by the exacerbating negative effects of general anesthesia on a compressed airway. Lung volume is reduced by an increase in abdominal muscle tone and a decrease in inspiratory muscle tone; the tethering effect of expanded lungs on the airways is lost; large airway compressibility increases as a result of bronchial smooth muscle relaxation, which in turn leads to decreased expiratory flow rates; and paralysis of the diaphragm eliminates the caudal expansion of the lungs effected during spontaneous ventilation.19 All of this has the potential to exacerbate the negative respiratory effects of extrinsic compression caused by a mediastinal mass on the pediatric airway. Because neuromuscular blockade and deep sedation in children with SMS have been associated with fatal deterioration of respiratory and cardiovascular systems, the most recent literature recommends an approach utilizing flow volume loops to assess the airway and non-invasive airway support.6,21
The child suspected of having SMS due to a malignancy should have cardiorespiratory, pulse oximetry, and, if available, end-tidal carbon dioxide monitoring; maintaining spontaneous respiratory efforts is highly recommended to avoid the negative effects of muscle paralysis and deep sedation. Oxygen should be administered; non-invasive pressure support ventilation may be better tolerated than endotracheal intubation. Continuous positive airway pressure, bi-level positive airway pressure, and the laryngeal mask airway are potential airway adjuncts for the child requiring support. Endotracheal intubation for respiratory failure/arrest should be done without paralysis, and rigid bronchoscopy should be available. Dopamine, dobutamine, and epinephrine have been used in cases of circulatory collapse with generally good effects. All efforts in the ED should be directed toward patient stabilization. Anxiety treatment with midazolam is recommended.
After stabilization, the decision to perform a biopsy with local anesthesia or general anesthesia versus emergent radiation and/or chemotherapy to shrink tumor prior to definitive diagnosis depends upon the patient's condition and airway assessment; it is a decision made by the multidisciplinary cancer team.
Spinal Cord Compression
Spinal cord compression in childhood is the most common cause of lower limb paralysis in children.24 Cancer metastasizing to, or originating within, the spine may cause compression of the spinal cord. Lewis and co-workers studied 643 newly diagnosed pediatric cancer patients aged 3 months to 17 years during a 40-month period to better understand the incidence, presentation, and outcome of spinal cord disease in children with cancer.25 Twenty-one of 643 pediatric patients (approximately 3%) with newly diagnosed cancer developed spinal cord compression (consistent with report of incidence in other studies). Spinal cord compression occurred as the first sign of malignancy in 6 of these 21 children (4 had sarcomas and 2 had lymphomas). The remaining 15 children developed cord compression a median of 13 months after initial cancer diagnosis, a quarter of these at the time of first relapse. All 21 children had metastatic disease causing malignant spinal cord compression (MSCC).
Like SMS, the cause and mechanism of MSCC in children is different than the cause and mechanism of this entity in adults. The most likely cancers to cause acute spinal cord compression in childhood are neuroblastoma, Ewing's sarcoma, Wilms' tumor, lymphoma, soft-tissue sarcoma, and osteosarcoma. Interestingly, cord compression is more likely to be caused by paravertebral tumors that impinge on the spinal cord directly rather than by involvement of bony elements in the spine. Spinal cord compression in children by intramedullary cancers or cancers that originate within the spine (e.g., astrocytomas or ependymomas) are a less common cause of MSCC. Studies in animal models of cord compression show that white matter edema and axonal swelling led to white matter necrosis and gliosis.24
Studies in adults indicate that most adult patients are diagnosed only after they become unable to walk.24 Fortunately, children fare better than do adults. Pretreatment neurological status is the most important predictor of function and is, in turn, associated with duration of symptoms and time to diagnosis. Prompt diagnosis and emergent treatment aim, not only to preserve function, but also to prevent the development of neurological deficits.
Children may present with a complaint of local back pain (80% of patients in Lewis' study), neck pain, extremity weakness or change in gait, sphincter dysfunction, or sensory symptoms.24,26 In the Lewis study, symptoms were present 5 days to 4 weeks prior to diagnosis. Once neurological deficits develop, paraplegia or quadriplegia may evolve quickly.1 Although back pain is the most common symptom, and is typically worse with percussion, lying down, neck flexion, straight-leg raise, and valsalva, it may be poorly localized in very young children. Young children more commonly present with irritability and may present with loss of developmental milestones. Sphincter dysfunction is a late finding in children with MSCC, and in one series of pediatric patients urinary retention was not seen as a first symptom in any patient.27
Deep tendon reflex abnormalities have been noted in children with MSCC. A formal neurological evaluation is mandatory in any child with back pain and focal tenderness. A child's gait (if possible), reflexes, and entire spine (by palpation and percussion) should be examined carefully. MR imaging is the most useful diagnostic tool and is the imaging method of choice in the assessment of MSCC. It has an overall diagnostic accuracy of 95%.24 Imaging of the entire neuroaxis is recommended because lesions can be at more than one site.28 CT is a valuable tool where MR imaging is not available and for the assessment of the bony spine. Corticosteroids have been shown to improve outcome for patients with MSCC, however, there is no consensus regarding the optimum total dose and frequency of dosing.
For children with history and neurological examination findings consistent with MSCC, most resources recommend 1 mg/kg (up to a maximum of 100 mg) intravenous dexamethasone as a loading dose, followed by 1 to 2 mg/kg/day in four divided doses over 24 hours. Emergent MR imaging should be done following a dexamethasone loading dose. Surgery for decompression is done on an emergent basis in cases of unknown tumor. Treatment after patient stabilization may include surgery, radiation, or chemotherapy depending upon the tumor type, location, and extent of spread, as well as patient age and developmental stage.28
Children with history and complaint of back pain concerning for MSCC but no neurological findings on formal and complete neurological evaluation are given 0.25 to 0.5 mg/kg of dexamethasone orally every 6 hours and scheduled for MR imaging evaluation within the next 24 hours.1 Emergent consultation with the pediatric hematologist-oncologist to coordinate initial patient management and urgent neurosurgical, pediatric critical care, and pediatric radiological consultation is required. Other causes of spinal cord compression in children include abscess, hematoma, and blunt or penetrating trauma.
Brain Herniation/Intracranial Hypertension
Central nervous system (CNS) neoplasms are the second most common tumor of childhood, accounting for almost 21% of the total, and are a leading cause of death in children younger than 15 years. In the United States, incidence has risen from approximately 2.4 to 3.3 new cases per 100,000 children each year, including 1500 new pediatric brain tumors annually.4 Although incidence appears to be rising, CNS tumors simply may be diagnosed earlier than in previous years because of advances in neuroimaging techniques.3 During the past two decades, the mortality rate from pediatric brain cancer has declined. Nonetheless, brain and spinal cord tumors currently cause more deaths in children than either leukemia or lymphoma.4
Brain tumors can be classified by histology and anatomic location.4,28,29 About 50% to 65% of childhood brain neoplasms arise in the posterior fossa. Masses that arise above the tentorium are called supratentorial; those located below it are referred to as infratentorial.
Supratentorial masses are usually gliomas, principally astrocytomas, but ependymomas and primitive neuroectodermal tumors (PNETs) also can arise here. Supratentorial tumors include the so-called midline tumors. Among these deep-seated tumors are chiasmal biloma, craniopharyngioma, and pineal-region tumors (e.g., germ-cell tumors, PNETs, and pineocytomas), optic pathway/hypothalamic tumors (e.g., gliomas), craniopharyngiomas, and suprasellar region germ-cell tumors.
About 80% of brain tumors in children develop in the first decade of life.4 In children younger than 1 year, supratentorial tumors predominate. These are often gliomas, teratomas, medulloblastomas, or choroid plexus tumors. From ages 1 through 11 years, PNETs and other posterior fossa tumors are more common. Supratentorial and infratentorial tumors are equally common during late childhood and adolescence.
Many children with brain tumors present with signs of increased intracranial pressure.1 Central nervous system infection, hemorrhage, and infarction may present with similar symptoms. Four symptom complexes that may be seen in children presenting with brain tumors include cerebellar ataxia, acute deterioration of level of consciousness, acute onset cranial nerve palsies, and severe recurrent headaches with or without vomiting.4 Headaches on waking suggest intracranial hypertension (Table 1). Morning headaches associated with brain tumors in children are due to an increase in cerebrospinal fluid (CSF) volume and pressure that occurs in the recumbent position and an increase in intracranial blood volume as a result of the increase in blood flow that occurs during rapid eye movement sleep. Infants may present with vomiting, change in behavior, loss of developmental milestones, and increased head circumference. Older children most commonly present with headache. Other signs of acute intracranial hypertension that may be seen with brain tumors in children are diplopia, hemiparesis, speech disturbance, dizziness, and coma. Herniation of a cerebellar tonsil may present with head tilt and neck stiffness.
| Table 1. What Headaches Suggest Intracranial Pathology? |
|
|
Symptoms and signs of increased intracranial pressure due to a brain tumor may suggest the location of the tumor.1,30 For example, brain tumors near the third ventricle may present with visual loss. Parinaud's syndrome, describing obstruction of the aqueduct of Sylvius by a pineal tumor, causes impairment of upward gaze, convergence nystagmus, and alterations in pupillary response. Only about 14% of children with brain tumors have seizures, which are more likely in children with supratentorial tumors than in those with infratentorial tumors. The rate of seizures is three times higher in adolescents than in infants. Children with tumor-associated seizures also usually have personality abnormalities; their speech is impaired, and their school performance deteriorates.
The study of choice for the diagnosis of a brain tumor and increased intracranial pressure is a CT or MRI scan with contrast enhancement. Emergent treatment consists of elevation of the head, administration of 0.5 to 1 mg/kg intravenous dexamethasone followed by 1 to 2 mg/kg intravenous dexamethasone per day divided into four doses, consideration of prophylactic intravenous anti-seizure medication, 1 to 2 g/kg intravenous mannitol 20% solution, and 5 mg/kg per day acetazolamide.1 The child may be started on 75% maintenance intravenous fluids of normal saline; endotracheal intubation to control airway and partial pressure of carbon dioxide is recommended in severe cases. These patients require frequent neurologic status and vital sign monitoring. Immediate pediatric neurosurgical consultation for intra-cranial pressure monitoring may be part of the acute management.
Hyperleukocytosis
Leukemia in children is associated with hyperleukocytosis in 5% to 20% of cases.1 Hyperleukocytosis is commonly defined as a white blood cell count in excess of 100,000 per mm3.31 Mortality in leukemia with white blood cell counts equal to or greater than 100,000 cells per mm3 is increased by 20% to 40%. High counts of blasts in the circulation may lead to hemorrhage or infarction. Classically, hyperviscosity is thought to be due to sludging of blasts causing stasis; recent evidence suggests that interaction between leukemic blasts and the vascular endothelium mediated by cytokines results in leukostasis and vascular damage.
The incidence of hyperleukocytosis is greater in acute lymphoblastic leukemia, and tumor lysis syndrome due to hyperleukocytosis is seen most commonly with acute lymphocytic leukemia (ALL).1 The blasts in myelogenous leukemia are approximately twice the size of leukemic lymphoblasts. Stroke is seen more commonly with acute myeloid leukemia (AML). Diagnosis is made on clinical suspicion when signs of microvascular damage in the brain and lungs are noted on examination. Behavioral changes, headache, seizures, ataxia, visual changes, dyspnea, hypoxia, priapism, limb ischemia, dactylitis, and petechiae may be noted. Bleeding due to disseminated intravascular coagulation and thrombocytopenia is common. Promyelocytic leukemic blasts have been shown to contain heparin granules in their cytoplasm. Hyperleukocytosis associated with leukemia may be misdiagnosed as Rocky Mountain spotted fever, central nervous system infections, or sepsis.
Chest radiographs may be negative or may show diffuse interstitial infiltrates. Complete blood count with differential, blood smear, serum uric acid, and serum potassium, electrolytes, and creatinine measurements should be evaluated. Intravenous hydration at two times maintenance, urinary alkalinization with 30 to 40 mEq/L of bicarbonate added to intravenous fluids, and uric acid depuration with allopurinol at 200 mg/m2/day orally or intravenously is the recommended treatment. Cytoreduction in the form of leukopheresis and exchange transfusion is a way to rapidly reduce the white blood cell count and decrease the risk of tumor lysis syndrome, but it may exacerbate thrombocytopenia and requires large bore central venous access and anticoagulation. Platelets should be maintained above 20,000 per mm3 to prevent intracranial bleeding. Coagulopathy is treated with fresh frozen plasma and vitamin K. Packed red blood cell transfusions are avoided in patients with hyperleukocytosis because of the theoretical risk of increasing viscosity.
Neutropenia and Fever
Fever in children and adolescents with cancer and treatment-induced neutropenia is common and potentially life-threatening. Because of the high risk of invasive bacterial and fungal infections, these patients require immediate medical evaluation, prompt administration of empiric intravenous antibiotics, and usually, admission to the hospital for observation.32 Although all cancer patients with fever and treatment-induced neutropenia can develop life-threatening, infection-related illnesses, there is evidence that these patients constitute a heterogeneous group with variable risks.32-34 For instance, many pediatric cancer patients with fever and treatment-induced neutropenia do well with simple administration of antibiotics and 2 to 3 days of observation in the hospital, but others develop sepsis syndrome requiring administration of life-saving critical care therapies and admission to a pediatric intensive care unit (PICU).
Identifying which patients are at greatest risk for life-threatening illness at the time of presentation to an outpatient clinic or ED is difficult, in part, because neutropenia alters the inflammatory response to infections.32 Nevertheless, several groups of investigators have demonstrated that some clinical and laboratory characteristics are associated with patients at low risk for bacteremia or readmission to the hospital.32-39 This information has been helpful in identifying patients that might be candidates for early hospital discharge or treatment at home with antibiotics. However, little is known about patient clinical and laboratory factors associated with the development of life-threatening illness. Lack of this information is an important problem because such knowledge could allow for earlier identification of critically ill patients, more timely administration of life-saving therapies, and more cost-effective delivery of care and utilization of PICUs. Recently, West and colleagues concluded that children and adolescents with cancer, fever, and treatment-induced neutropenia who present to an ED or outpatient clinic with higher fevers (>39.5° C) or prolonged capillary filling times (>3 seconds) are at increased risk of developing life-threatening, infection-related illnesses requiring administration of critical care therapies.32
Although several studies have suggested that low-risk pediatric patients with fever and neutropenia can be clinically identified, there is no widespread support for initial therapy with oral antibiotics and outpatient treatment for pediatric patients. According to the Infectious Diseases Society of America (IDSA) guidelines for the use of antimicrobial agents in neutropenic patients with cancer, the data are insufficient to recommend initial empirical oral therapy for children with fever and neutropenia.39
Infection remains a leading cause of morbidity and mortality in children with cancer. The presence of an indwelling central venous catheter (CVC) and an abnormally low absolute neutrophil count (ANC) put the oncology patient at particularly high risk for a serious bacterial infection. The ANC is calculated by multiplying the percentage of neutrophils (segmented neutrophils plus bands) by the total white blood cell count. These data, along with the intensity of immune-suppressive therapy that the child is receiving, must always be considered when evaluating a child for potential infection, even if a common childhood illness (e.g., otitis media) is suspected.40 It is always advisable to confer with the child's oncologist should there be concerns about fever and/or infection.
Fever is defined as a single temperature of 38.5° C or higher within a 24-hour period taken axillary, orally, or by tympanic probe. Rectal temperatures should never be taken in neutropenic children. The initial evaluation of the febrile oncology patient should include a meticulous physician examination with attention to abnormalities in vital signs including blood pressure.40 Sites of occult infection (e.g., the mouth, skin, and the perianal area) should be inspected for even subtle evidence of inflammation (e.g., mild erythema, tenderness, or discharge). In children with CVCs, the catheter exit site and track should be examined for erythema, tenderness, skin breakdown, and discharge.
The laboratory evaluation of all febrile patients should include a complete blood count with differential to determine the ANC. Urinalysis and urine culture should be obtained from all patients, preferably with a clean catch technique. Bladder catheterization is avoided in all oncology patients due to cytopenias, risk of bleeding, and disruption of the mucosal integrity with subsequent potential introduction of infection. If respiratory signs or symptoms are present, a chest radiograph should be obtained.41,42 Other laboratory studies that are potentially relevant include culture and gram stain of the mouth, throat, or skin if signs or symptoms of infection are present. In addition, if patients have diarrhea, then stool samples for bacterial cultures should be obtained. The child with prolonged congestion or tenderness over the sinuses should have a diagnostic imaging study (computed tomography) to evaluate for sinusitis. Lumbar punctures are not performed routinely as part of an initial evaluation for sepsis. Meningitis in cancer patients is rare, however, it may present more subtly than in a well child.43 If lumbar puncture seems indicated, contact the child's oncologist for recommendations. Evaluation of increased intracranial pressure by neuro-imaging may be necessary prior to lumbar puncture.
The two most important factors that influence susceptibility to infection in a child with cancer are the number of circulating neutrophils and the duration of neutropenia.40 The threshold for defining neutropenia is an ANC <1000/mm3; moderate neutropenia is an ANC <500/mm3, and severe neutropenia is an ANC <200/mm3, Severe neutropenia for more than 2 to 3 weeks significantly increases the risk for overwhelming bacterial or fungal sepsis.44 Patients with long-standing neutropenia should be monitored closely and managed aggressively in consultation with a pediatric oncologist.
Because of the possibility of bacterial sepsis, children with cancer who present with fever and neutropenia are admitted to the hospital for observation and treated with broad-spectrum intravenous antibiotics.40 The neutropenic child may not exhibit the cardinal responses to infection and may have fever as the only sign of infection.44 For example, a neutropenic child with a skin infection may develop little induration or pus due to lack of circulating neutrophils, despite the presence of infection. Similarly, a neutropenic child with clinical pneumonia may have a negative chest radiograph until the ANC increases, at which time infiltrates may become apparent. Blunted immunologic response may be particularly true during treatment with high-dose steroids.
Several studies have attempted to identify low-risk febrile neutropenic children who may qualify for outpatient treatment or early hospital discharge.33-35,45,46 Currently, there is no consensus on either the appropriate criteria for outpatient management of febrile neutropenic patients or the criteria for hospital discharge prior to ANC reaching 500 cells/mm3. Thus, the standard of care remains hospitalization with administration of broad-spectrum antibiotics until the patient is afebrile and has evidence of bone marrow recovery.40
Empirical broad-spectrum antibiotics must be administered immediately to cancer patients with fever and neutropenia. The most common organisms responsible for sepsis in neutropenic children are gram-positive bacteria including Staphylococcus epidermidis, S. aureus and S. viridans, as well as gram-negative organisms including Escherichia coli, Enterobacter species, Klebsiella species, Serratia species, Acinetobacter species, and Pseudomonas species.40 Antibiotics should be chosen based upon microbial prevalence and antibiotic sensitivity patterns at each institution.39 Many combinations of antimicrobials are effective. Some examples are:
- Antipseudomonal ß-lactam drug plus an aminoglycoside (e.g., ceftazidime and tobramycin or gentamicin)
- Extended-spectrum penicillin plus antipseudomonal aminoglycoside (e.g., azlocillin, mezlocillin, piperacillin, or ticarcillin plus tobramycin or gentamicin)
- Monotherapy with ceftazidime, cefepime, imipenem, or meropenem
In a study of 136 episodes of fever and neutropenia in children, 34% of episodes had no focus of infection either clinically or microbiologically.37 Of those cases associated with a focus, the mucosal surface and the gastrointestinal system were most commonly involved. The researchers reported that neutropenic patients with fever had a greater than 60% chance of being infected and that use of central venous catheters, prophylactic quinolone use, and intensive chemotherapy in pediatric cancer patients have increased the incidence of gram-positive infection in these patients, especially with coagulase-negative Staphylococcus and Streptococcus viridans.37 The researchers noted that monotherapy and multi-drug therapy did not significantly differ in efficacy as empiric initial therapy.37
A severely neutropenic patient (i.e., ANC of 100 or less) may exhibit no or minimal symptoms and signs of inflammation. Expected induration, erythema, and pustulation in response to a bacterial infection may be lacking due to reduced numbers of circulating neutrophils. Hence, subtle clues (e.g., pain) should be addressed, especially if pain is elicited at sites at risk for infection. The mouth, pharynx, lung, perineum, anus, fundi, skin (especially catheter sites and biopsy sites), and periungual regions should be examined carefully. Although symptoms and signs may be few, neutropenic cancer patients with fever can have infections that progress rapidly and prove fatal. Blood samples are obtained for culture. The IDSA recommends that one set of blood samples be obtained from a previously placed central venous catheter (if the patient has one) as well as from a peripheral vein.39 Vascular devices may be left in place during antibiotic treatment even if there is an infection at the site or catheter related infection is suspected.39 However, if a patient has no obvious source and appears sick, obtaining pan-cultures of all possible sites of infection is reasonable. Approximately one-half of neutropenic patients who become febrile have an established or occult infection, and one-fifth of patients with neutrophil counts less than 100 cells per mm3 have bacteremia. Up to 70% of microbiologically documented infections are caused by gram-positive organisms, although gram-negative infections are increasing in some centers.
The IDSA recommends that empirical antibiotic therapy be administered promptly to all neutropenic patients at the onset of fever because progression of infection can be rapid and infection cannot be reliably ruled out at initial presentation. Studies have shown no significant difference between monotherapy and multidrug therapy for empirical treatment of "uncomplicated episodes of fever" in neutropenic patients.39 Several choices for monotherapy include cefepime, meropenem, imipenem, Zosyn® (not as extensively studies as other choices). The addition of an aminoglycoside can provide synergy against gram-negative bacteria and decrease the incidence of emergence of drug-resistant strains. The use of vancomycin should be reserved for use on an individual basis indicated by clinically suspected serious catheter-related infections, patients known to be colonized with penicillin or cephalosporin-resistant pneumococci, MRSA, or Streptococcus viridans, or patients with hypotension or cardiovascular impairment.
The use of antiviral agents, antifungal agents, and granulocyte colony stimulating agents are not generally recommended for initial empiric treatment for children with cancer who have fever and neutropenia.47
Typhlitis, also known as neutropenic enterocolitis, is a necrotizing colitis that was first described in 1970 as an almost uniformly fatal complication of treatment in childhood leukemia.47 It is now known to occur in both children and adults with immune suppression of various etiologies (e.g., ALL, acute myelogenous leukemia, lymphoma, aplastic anemia, myelodysplasia, cyclic neutropenia, multiple myeloma, renal and bone marrow transplants, breast cancer, and AIDS).47,48 Most patients have received chemotherapy, but if the underlying disease causes neutropenia, typhlitis can occur without chemotherapy.
Patients present with nonspecific signs and symptoms including abdominal pain and distention, fever, and neutropenia. Symptoms and physical findings are often suppressed by treatment with steroids and other drugs.
The cause of typhlitis is unknown, but the number of cases diagnosed is increasing because of better diagnostic imaging and the use of more aggressive chemotherapy. The pathophysiology involves many factors, including dilated cecum with a tenuous blood supply, disruption of mucosal integrity secondary to chemotherapy or leukemic infiltrate, and proliferation of bacteria because of decreased host resistance. Bowel wall necrosis, hemorrhage, perforation, sepsis, or shock may occur. Even with optimum treatment, mortality rates are high.48
Other conditions (e.g., appendicitis, intussusception, abscess, obstruction, mural hemorrhage, and pseudomembranous colitis) can have similar clinical and plain radiograph findings.48
Barium enema, angiography, CT, and ultrasound have all been used to confirm the diagnosis of typhlitis. Ultrasound is now widely accepted as the preferred diagnostic modality. It provides excellent visualization of the pathology, usually can distinguish typhlitis from other entities in the differential diagnosis, is faster and less risky, and may be performed at the bedside in these often unstable patients.
The characteristic ultrasound appearance is a "target" lesion in the right lower quadrant with thickened bowel wall. In the other conditions in the differential diagnosis, the bowel wall thickening is hypoechoic. Because typhlitis can involve only the cecum, cecum and ascending colon, cecum and distal ileum, or cecum and isolated areas of more distal colon, the ultrasound examination should attempt to determine the extent of disease by following thickened bowel wall proximally and distally to normal bowel.
CT findings are similar to ultrasound appearance, with symmetric cecal wall thickening and pericecal inflammation and fluid collection if perforation has occurred.49 Early diagnosis and determination of extent of disease are necessary so that appropriate medical treatment (i.e., bowel rest, normalization of neutrophil counts, antibiotics administration, and fluid management) or surgical intervention can be instituted. Indications for surgery include perforation, persistent bleeding, sepsis that does not respond to medical management, and the need for abscess drainage (percutaneous drainage may be indicated).48
Tumor Lysis Syndrome
Tumor lysis syndrome is an oncologic emergency seen most commonly in children with lymphoproliferative and highly chemosensitive tumors (e.g., lymphoma and leukemia), especially those associated with high granulocyte counts or hyperleukocytosis.1,50,51 Infants with bulky tumors (e.g., neuroblastoma) are also at risk. Tumor lysis can occur before or up to 5 days after chemotherapy.4 When tumor cells lyse, large amounts of intracellular contents are spilled into the circulation. Blast cells have more intracellular potassium than do normal cells. A rapid rise in serum potassium and phosphorus levels, a precipitous fall in serum calcium level, and elevations of the serum uric acid, serum urea nitrogen, and serum creatinine levels characterize tumor lysis syndrome. Nephropathy due to overwhelming levels of uric acid and phosphorus, which encourages the precipitation of calcium in the kidneys, ensues.
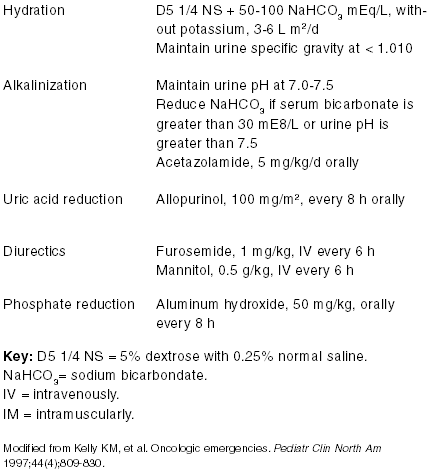
The treatment of tumor lysis syndrome starts with hydration (Table 2). Intravenous fluids of 5% dextrose with 0.25% normal saline and 50 to 100 mEq/L of sodium bicarbonate without potassium at two times maintenance are recommended. Urine specific gravity is maintained at less than 1.010, urine pH at 7.0 to 7.5, and serum bicarbonate below 30 mEq/L. Once euvolemia is achieved, diuresis with furosemide is the next step in the management of tumor lysis syndrome.
| Table 2. Management of Tumor Lysis Syndrome |
|
|
A number of other pharmacotherapies have been used to treat tumor lysis syndrome including allopurinol and rasburicase.52-56 Allopurinol and its active metabolite oxypurinol are potent inhibitors of xanthine oxidase, the enzyme responsible for conversion of hypoxanthine to uric acid. Allopurinol is available in both oral and intravenous forms; both forms are considered to be equally safe and efficacious.55 Adverse reactions noted with the use of allopurinol in children occur less than 1% of the time and include mild to moderate skin reactions and a more serious but rarer reaction associated with long-term treatment and compromised kidney function that is characterized by fever, hepatitis, skin rash, and eosinophilia.55 Rasburicase is a recombinant form of urate oxidase, a catalyst that turns uric acid to the urine soluble product allantoin.
Hyperkalemia is the most serious abnormality of tumor lysis syndrome. Hyperkalemia is treated with Kayexalate®, bicarbonate, dextrose and insulin, calcium, or dialysis.
Phosphate excretion may be aided with the addition of aluminum hydroxide, 50 mg/kg, administered orally every 8 hours. The level of serum phosphorus must be considered in the treatment of hypocalcemia because administration of calcium may increase calcium phosphate precipitation. Symptomatic hypocalcemia warrants replacement with calcium gluconate, 100-200 mg/kg/dose, by slow intravenous push while monitoring for bradycardia. Severe alkalosis may further reduce ionized calcium levels and, therefore, should be avoided.
When conservative measures are ineffective in correcting electrolyte disturbances and improving urinary flow, dialysis may be necessary.
Hypercalcemia
Hypercalcemia is regarded as a common, potentially life-threatening complication and a poor prognostic indicator of cancer in adult patients. These observations have not been made in children with hypercalcemia of malignancy. Children with hypercalcemia due to malignancies have not been shown to have a worse outcome than children who do not experience hypercalcemia due to their cancer.57-61 In 1993, McKay published results of a retrospective review of pediatric cancer patients, the goal of which was to determine the incidence of hypercalcemia in children with cancer. This study identified 25 children of all children diagnosed with cancer during a 29-year period. McKay postulated that the incidence of hypercalcemia in children with cancer at St. Jude Children's Hospital was 0.4% and stated that hypercalcemia of malignancy is extremely rare in children.60 Hypercalcemia developed in children with leukemia, sarcoma, lymphoma, hepatoblastoma, neuroblastoma, and brain tumor. The most common association was with leukemia. McKay also noted that children with ALL were likely to present with hypercalcemia at first diagnosis and that children with solid tumors presented later in the course of their disease.
In 1997, Hibi reported hypercalcemia in 4 of 83 pediatric ALL patients diagnosed during an 8-year period for an incidence of 4.8% in ALL patients.59 Hibi also noted that gastrointestinal (e.g., nausea, vomiting, abdominal pain) and musculoskeletal (e.g., leg pain, lower back pain, gait disturbance) complaints were common in the 4 patients with hypercalcemia and acute lymphoblastic leukemia.
Underlying mechanisms of hypercalcemia in pediatric cancer patients include increased osteoclastic activity, mediated by cytokines produced by the malignant cells as parathyroid hormone-related protein, tumor necrosis factor, and interleukins 1 and 6; decreased renal excretion of calcium due to decrease glomerular filtration; increased renal tubular absorption of calcium; and osteolysis due to bone metastases.1
The initial treatment of hypercalcemia thought due to malignancy in a child focuses first upon correcting dehydration and enhancing renal calcium excretion.57 Definitive treatment is treatment of the underlying malignancy.
Aggressive treatment generally is not indicated in patients with mild hypercalcemia (<12mg/dL), especially for children in whom hypercalcemia is generally better tolerated. The administration of a bisphosphonate or calcitonin in the ED should be done in consultation with a pediatric hematologist/oncologist. Ancillary interventions include control of nausea and vomiting and minimal use of sedating medications that can confound signs and symptoms.1
Normal saline solution should be started at a rate of 2 to 3 times maintenance. Once the patient has been adequately rehydrated and is considered euvolemic, furosemide is given at a dose of 1 mg/kg every 6 hours. Loop diuretics induce calcium renal excretion by inhibiting renal calcium resorption in the kidney. Electrolyte levels should be monitored frequently during treatment because loop diuretics promote the loss of potassium, sodium, and magnesium, as well as calcium. Pamidronate, a bisphosphonate, inhibits osteoclastic bone resorption and is one of the most effective agents for the treatment of hypercalcemia of malignancy.57 Pamidronate (1mg/kg over 2 hours IV) is given as a single dose over 2 to 24 hours. Pamidronate takes effect in 3 to 4 days with maximal effect in 7 days. A randomized study comparing these agents has shown pamidronate to be more effective and better tolerated that etidronate.61 Adverse effects noted with the use of bisphosphonates include transient temperature elevation, nausea, anorexia, dyspepsia, vomiting, especially with rapid administration and transient decreases in peripheral lymphocytes. Calcitonin (10 IU/kg/24 hours IV for 2 days) is a hormone that opposes the effects of PTH on bone and renal tubular calcium resorption. In patients responsive to calcitonin, onset of action is 2 to 4 hours but only lasts a few days. It is usually well tolerated, but potential adverse effects include nausea, transient cramping abdominal pain, cutaneous flushing and tachyphylaxis.57 Calcitonin is usually used in conjunction with pamidronate in patients with moderate to severe hypercalcemia with symptoms. The use of gallium nitrate and plicamycin (mithramycin) has been associated with nephrotoxicity. Steroids must be used cautiously due to potential to precipitate tumor lysis syndrome in undiagnosed sensitive cancers. Phosphate is contraindicated in renal insufficiency; intravenous use has been associated with precipitation of calcium into bone and soft tissue. The treatment of severe hypercalcemia in children has not been clearly defined. Specific therapy must be initiated as soon as possible. Serum calcium levels must be monitored for 7 days after administration of pamidronate due to risk of hypocalcemia.61 Recent reports of use of pamidronate alone or in combination with calcitonin in children with hypercalcemia of malignancy are encouraging.
Malignant Abdominal Mass
A palpable mass in the abdomen of a child is a serious finding. It requires urgent attention to determine if it is malignant and if there is compression of vital organs or internal hemorrhage.62
Abdominal masses are most often found in children younger than 5 years. In infants, most abdominal masses are retroperitoneal, of renal origin, and nonmalignant. The most common malignant abdominal tumor in infants is neuroblastoma, followed by Wilms' tumor. Other abdominal tumors in this young age range include soft-tissue sarcomas (e.g., rhabdomyosarcoma and non-rhabdomyosarcoma), germ cell tumors, and hepatoblastoma (Table 3). Beyond infancy, Wilms' tumor, sarcomas, and germ cell tumors predominate.
| Table 3. Malignant Abdominal Masses (Neonatal) |
|
|
Evaluation of an abdominal mass begins with a thorough history and physical examination. General signs and symptoms associated with malignant masses include increased abdominal girth, pain with movement or palpation, and constipation or decreased urination. Occasionally, there is associated weight loss, fever, or decreased appetite. Pallor or weakness may be caused by anemia from slow hemorrhage into the mass or from infiltration of the bone marrow.
An abdominal mass in infants is first assessed by a plain abdominal radiograph to rule out gastrointestinal obstruction. Sonography can usually identify the organ of origin of the mass (e.g., the kidney or adrenal gland). Different types of tissue components (e.g., cysts, hemorrhage, calcification) can be identified. Vascular lesions (e.g., hemangiomas or hemangioendotheliomas) can be examined initially with special ultrasound techniques that allow real-time dynamic visualization to determine the amount and direction of blood flow in the lesion. After sonography, better resolution is usually necessary with CT scanning or MR imaging. If the mass is suspicious for a malignant tumor, CT scanning of the chest, abdomen, and pelvis are required to assess the size of the primary tumor and to determine the presence or absence of infiltration into vessels or vital organs. If cranial or peripheral neurologic signs or symptoms are present, MR imaging of the head and spine are indicated.62,63 Ideally, at the time of imaging, a pediatric surgeon and, if needed, a neurosurgeon should be involved.
Initial studies include a complete blood cell count (CBC) and chemistry panel (i.e., electrolytes, liver transaminases, total and direct bilirubin, albumin, calcium, phosphorus, uric acid, and lactate dehydrogenase levels). Subtle abnormalities in the CBC may give clues as to the underlying problem. An elevated white blood cell count with a left shift may be seen because of infection caused by obstructive effects of the underlying malignant process. Varying degrees of pancytopenia can be caused by extensive bone marrow infiltration by the malignancy or by marrow stressed by an infection. Anemia can be caused by chronic disease or may indicate hemorrhage into the tumor. Thrombocytosis is often seen with liver tumors. Coagulation studies are helpful to determine the presence of disseminated intravascular coagulopathy or liver dysfunction. Transient acquired abnormalities are associated with specific malignancies such as von Willebrand's disease with Wilms' tumor. A baseline urinalysis can detect the hematuria or proteinuria, which can be seen with a primary renal or bladder tumor.
Tumor markers are useful in determining the diagnosis and, sometimes, the prognosis of certain tumors.
Summary
The prognosis for the child with cancer has improved dramatically over the past decades. With this success comes the need for recognition and proper treatment of emergencies. Respiratory or circulatory failure may arise from compression of the SVC or airway. Epidural spinal cord compression by tumor may lead to irreversible paraplegia or urinary incontinence if intervention is not rapid. Raised intracranial pressure may be a life-threatening presentation of a brain tumor. Hyperleukocytosis carries a high risk of thrombotic events if not treated promptly. Life-threatening metabolic abnormalities are observed at presentation in children with leukemia and lymphoma. Hypercalcemia, although rare, may be a difficult situation to correct.
Immediate attention to these emergencies and appropriate treatment may save the life of a child with cancer.
References
1. Kelly KM, Lange B. Oncologic Emergencies. Pediatr Clin North Am 1997;44(4)809-30.
2. National Childhood Cancer Foundation and Children's Oncology Group. Available at: www.curesearch.org
3. American Cancer Society. Available at: www.cancer.gov
4. Conway EE, Asuncion A, DaRosso R. Diagnosing and managing brain tumors: the pediatrician's role. Contemp Pediatr 1999; 16(11):84-97.
5. Issa PY, Brihi ER, Janin Y, et al. Superior vena cava syndrome in childhood: report of ten cases and review of the literature. Pediatrics 1983;71(3):337-341.
6. Piastra M, Ruggiero A, Caresta E, et al. Life-threatening presentation of mediastinal neoplasms: reports on 7 consecutive pediatric patients. Am J Emerg Med 2005;23(1):403-8.
7. Ingram L, Rivera GK, Shapiro DN. Superior vena cava syndrome associated with childhood malignancy: analysis of 24 cases. Med Pediatr Oncol 1990;18(6):476-81.
8. Lokich JJ, Goodman R. Superior vena cava syndrome: clinical management. JAMA 1975;231(1):58-61.
9. Piro AJ, Weiss DR, Hellman S. Mediastinal Hodgkin's disease: a possible danger for intubation anesthesia. Int J Rad Oncol Biol Phys 1976;1:415-19.
10. Janin Y, Becker J, Wise L, et al. Superior vena cava syndrome in childhood and adolescence: a review of the literature and report of three cases. J Pediatr Surg 1982;17:290-5.
11. Neuman GG, Weingarten AE, Abramowitz RM. The anesthetic management of the patient with an anterior mediastinal mass. Anesthesiology 1984;60:144-7.
12. Northrip DR, Bohman BK, Tsueda K. Total airway occlusion and superior vena cava syndrome in a child with an anterior mediastinal tumor. Anesth Analg 1986;65:1079-82.
13. Ferrari LR, Bedford RF. General anesthesia prior to treatment of anterior mediastinal masses in pediatric cancer patients. Anesthesiology 1990;72:991-5.
14. Yellin A, Rosen A, Reichert N, et al. Superior vena cava syndrome. The myths – the facts. Am Rev Respir Dis 1990;141:1114-8.
15. Jeng MJ, Chang TK, Hwang B. Superior vena cava syndrome in children with malignancy: analysis of seven cases. Chin Med J 1992; 50:214-8.
16. Yellin A, Mandel M, Rechavi G, et al. Superior vena cava syndrome associated with lymphoma. AJDC 1992;146:1060-3.
17. Shamberger RC, Holzman RS, Griscom T. Prospective evaluation by computed tomography and pulmonary function tests of children with mediastinal masses. Surgery 1995; 118:468-71.
18. Vas L, Naregal F, Naik V. Anaesthetic management of an infant with anterior mediastinal mass. Paediatr Anaesth 1999;9:439-43.
19. Freud E, Ben-Ari J, Schonfield T, et al. Mediastinal tumors in children: a single institution experience. Clin Pediatr 2002;41:219-23.
20. American Cancer Society. Superior vena cava syndrome (PDQ) 2003; Available at: www.cancer.gov.
21. Ricketts R. Clinical management of anterior mediastinal tumors in children. Semin Pediatr Surg 2001;10(3):161-8.
22. Amaha K, Okutsu Y, Nakamura Y. Major airway obstruction by mediastinal tumour: a case report. Brit J Anaesth 1973;45:1082-4.
23. Laskin J, Cmelak AJ, Roberts J, et al. Superior vena cava syndrome. In: Abeloff MD, ed.: Clinical Oncology 3rd ed. Churchill Livingstone; 2004:1047-52.
24. Prasad D, Schiff D. Malignant spinal-cord compression. Lancet Oncol 2005;6(1):15-24.
25. Lewis DW, Packer RJ, Raney B, et al. Incidence, presentation, and outcome of spinal cord disease in children with systemic cancer. Pediatrics 1986;78(3):438-43.
26. Francel PC. Consultation with the specialist: extrinsic spinal cord mass lesion. Pediatr Rev 1998;19:389-94.
27. Gilbert RW, Kim JH, Posner JB: Epidural spinal cord compression from metastatic tumor: diagnosis and treatment. Ann Neurol 1978: 40(3).
28. American Cancer Society. Childhood brain tumors (PDQ): treatment. 2004; Available at: www.cancer.gov.
29. Ullrich NJ, Pomeroy SL. Pediatric brain tumors. Neuro Clin 2003; 21(4):897-913.
30. Gnanalingham KK, Lafuenta J, Thompson D, et al. The natural history of ventriculomegaly and tonsillar herniation in children with posterior fossa tumors – an MRI study. Pediatr Neurosurg 2003;39:246-53.
31. Porcu P, Cripe LD, Ng EW, et al. Hyperleukocytic leukemias and leukostasis: a review of pathophysiology, clinical presentation and management. Leuk Lymphoma 2000;39:1-18.
32. West DC, Marein JP, Mawis R, et al. Children with cancer, fever, and treatment induced neutropenia. Pediatr Emerg Care 2004;20(2):79-84.
33. Klaassen RJ, Goodman TR, Phram B, et al. "Low risk" prediction rule for pediatric oncology patients presenting with fever and neutropenia. J Clin Oncol 2000;18:1012-9.
34. Rackott WR, Gonin R, Robinson C, et al. Predicting the risk of bacteremia in children with fever and neutropenia. J Clin Oncol 1996; 14(3):919-924.
35. Lucas KG, Brown AE, Armstrong D, et al. The identification of febrile, neutropenic children with neoplastic disease at low risk for bacteremia and complications of sepsis. Cancer 1996;77(4):791-8.
36. Ammann RA, Hirt A, Luthy AR, et al. Predicting bacteremia in children with fever and chemotherapy-induced neutropenia. Pediatr Infect Dis J 2004;23(1):61-7.
37. Aquino VM, Tkaczewski I, Buchanan GR. Early discharge of low-risk febrile neutropenic children and adolescents with cancer. Clin Infect Dis 1997;25(1):74-78.
38. Mullen CA. Petropoulos D, Roberts WM, et al. Outpatient treatment of fever and neutropenia for low risk pediatric cancer patients. Cancer 1999; 86(1):126-134.
39. Hughes WT, Armstrong D, Bodey GP, et al. 2002 guidelines for the use of antimicrobial agents in neutropenic patients with cancer. Clin Infect Dis 2002;34(6):730-51.
40. Golden E, Beach B, Hastings C. The pediatrician and medical care of the child with cancer. Pediatri Clin North Am 2002; 49:1319-1338.
41. Korones DN, Hussong MR, Gullace MA. Routine chest radiography of children with cancer hospitalized for fever and neutropenia: is it really necessary? Cancer 1997; 80: 1160-4.
42. Feusner J. Routine chest radiography for pediatric oncology patients with febrile neutropenia: is it really necessary? Cancer 1997; 80:1009-10.
43. Sommers L, Hawkins D. Meningitis in pediatric cancer patients: A review of forty cases from a single institution. Pediatric Infect Dis J 1999;18:902-907.
44. Pizzo P. Management of fever in patients with cancer and treatment induced neutropenia. N Engl J Med 1993; 328:1323-1332.
45. Baorto EP, Aquino VM, Mullen CA, Buchanan GR, DeBaun MR. Clinical parameters associated with low bacteremia risk in 1100 pediatric oncology patients with fever and neutropenia. Cancer 2001;92:909-13.
46. Santolaya M, Alvarez A, Becker A, et al. Prospective, multicenter evaluation of risk factors associated with invasive bacterial infection in children with cancer, neutropenia and fever. J Clin Oncol 2001;19:3415-21.
47. Katz JA, Wagner ML, Gresik MV, et al. Typhlitis: An 18 year experience and postmortem review. Cancer 1990; 65:1041-1047.
48. Schlatter M, Snyder K, Freyer D. Successful nonoperative management of typhlitis in pediatric oncology patients. J Pediatr Surg 2002;37(8):1151-1155.
49. Merine D, Nussbaum AR, Fishman EK, et al. Sonographic observations in a patient with typhlitis. Clin Pediatr 1989;28(8):377-379.
50. Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Brit J Haematol 2004;127:3-11.
51. Davidson MB, Thakkar S, Hix JK, et al. Pathophysiology, clinical consequences, and treatment of tumor lysis syndrome. Am J Med 2004;116(8):546-54.
52. Bosly A, Sonet A, Pinkerton CR, et al. Rasburicase for the management of hyperuricemia in patients with cancer: report of an international compassionate use study. Cancer 2003; 98(5):1048-54.
53. Goldman SC. Rasburicase: potential role in managing tumor lysis in patients with hematological malignancies. Expert Rev Anticancer Ther 2003;3(4):429-33.
54. Goldman SC, Holcenberg JS, Finkelstein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis syndrome. Blood 2001;97:2998-3003.
55. Smalley RV, Guaspari A, Haase-Statz S, et al. Allopurinol: intravenous use for prevention and treatment of hyperuricemia. J Clin Oncol 2000;18(8):1758-63.
56. Macfarlane RJ. Rasburicase prevents tumor lysis syndrome despite extreme hyperleukocytosis. Pediatr Nephrol 2004;19(8):924-7.
57. Mathur M, Sykes JA, Saxena VR, et al. Treatment of acute lymphoblastic leukemia-induced extreme hypercalcemia with pamidronate and calcitonin. Pediatr Crti Care Med 2003;4(2):252-5.
58. Bruonuomo PS, Ruggiero A, Piastra M, et al. A case of acute lymphoblastic leukemia presenting as severe hypercalcemia. Pediatr Hematol Oncol 2004;21:475-9.
59. Hibi S, Funaki H, Ochiai-Kanai R, et al. Hypercalcemia in children presenting with acute lymphoblastic leukemia. Int J Hematol 1997;66:353-7.
60. McKay C, Furman WL. Hypercalcemia complicating childhood malignancies. Cancer 1993;72(1):256-60.
61. Gucalp R, Ritch P, Wiernick PH, et al. Comparative study of pamidronate disodium and etidronate disodium in the treatment of cancer-related hypercalcemia. J Clin Oncol 1992;10(1):134-42.
62. Golden CB, Feusner JH. Malignant abdominal masses in children: Quick guide to evaluation and diagnosis. Pediatr Clin North Am 2002;49:1369-92.
63. Merton DF, Kirks DR. Diagnostic imaging of pediatric abdominal masses. Pediatr Clin North Am 1985; 32:1397-1425.
In the United States, an estimated 12,500 children and adolescents are diagnosed with cancer each year; 9,500 children are younger than 15 years.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.