The HIV-Positive Patient in the ED: HIV/AIDS Update for 2006. Part II
The HIV-Positive Patient in the ED: HIV/AIDS Update for 2006. Part II
Authors: Gary D. Hals MD, PhD, Attending Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC; Mark Muiznieks, MD, Resident Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC; and William Jennings, MD, Resident Physician, Department of Emergency Medicine, Palmetto Richland Memorial Hospital, Columbia, SC.
Peer Reviewers: Catherine A. Marco, MD, FACEP, Clinical Professor, Department of Surgery, Division of Emergency Medicine, Medical University of Ohio, Toledo; Attending Physician, Department of Emergency Medicine, St. Vincent Mercy Medical Center, Toledo, OH; and Steven M. Winograd, MD, FACEP, Attending Physician, Emergency Department, Adena Regional Medical Center, Chillicothe, OH.
Clinical Effects of HIV
HIV infection can be divided into three stages: the acute seroconversion reaction, latent phase, and active AIDS. Acute seroconversion produces a flu-like syndrome whose true cause easily can go unrecognized, as the symptoms overlap with many other acute viral syndromes. The latent phase is largely clinically silent as the immune system is able to suppress viral replication while maintaining CD4 levels that keep opportunistic infections at bay. Over time, an average of 8-10 years for adults (2 years for children < 5 years of age) without antiretroviral treatment, the immune system loses its battle with the virus, and CD4 counts drop below 200 cells/microL. Around this CD4 level, the body now is unable to fight off the many opportunistic infections that it normally can control. Be aware, though, that some HIV-associated diseases can arise independent of a normal CD4 count (i.e., pneumococcal pneumonia, tuberculosis [TB], Kaposi's sarcoma, and lymphoma). Median survival time does correlate with CD4 counts. Once CD4 counts drop to 200 cells/microL, it is 3.7 years, and only 1.3 years after diagnosis of an AIDS-defining illness.1 (See section on AIDS-defining Illness.) The virus also has direct effects on several organ systems besides those of the immune system (i.e., renal, central nervous system [CNS]). These direct effects of the virus also are related to CD4 levels, as lowered immune system function correlates with increased levels of viremia. The common opportunistic infections, major direct effects of the virus, and their treatment will be discussed by organ system.
Acute HIV Syndrome. The acute HIV syndrome is a flu-like or mononucleosis-like illness that usually occurs 2-6 weeks after exposure to the virus. Only 50-90% of individuals experience this reaction.2 The degree of symptoms experienced may be subclinical in some patients. The symptoms are caused by a burst of viremia that is second only to virus levels reached at the very end stage of AIDS. In the past it was thought that the initial degree of viremia correlated with the subsequent course of the disease, but this now is known not to be the case.
Acute HIV syndrome symptoms include fever, pharyngitis, myalgias, skin rash, fatigue, headache diarrhea, vomiting, and lymphadenopathy. (See Table 1.) Symptoms can last for 1-2 weeks (rarely up to 3 weeks). It is estimated that the actual diagnosis of the syndrome is "missed" in at least 75% of cases for a variety of reasons.3 The most common reason is that acute HIV syndrome resembles other viral infections, such as flu and mononucleosis. In other cases there may just be a low degree of suspicion. Since the majority of new HIV cases are in younger people, the diagnosis often is delayed in patients older than 50 years of age.4
| Table 1. Common Symptoms in Acute HIV Syndrome6 |
|
|
In most patients, the symptoms gradually subside as the immune response begins, antibodies to the virus are produced, and viremia is cleared. In some patients the viremia is strong enough to temporarily reduce CD4 counts below 200 cells/microL. Thus, a minority of patients also will have an opportunistic infection as part of their acute seroconversion. Further, 10% of all patients will not rebound from the initial burst of viremia, and will progress rapidly to clinical AIDS from which they may not recover. The other 90% of patients will have a strong immune response that adequately controls viral loads to allow CD4 counts to return to normal levels and begin the asymptomatic latent phase of HIV infection.
The Latent Stage. As mentioned above, once the immune system responds to the initial burst of viremia, begins antibody production, and largely clears the virus, the patient enters the latent stage of HIV infection. In this phase, the immune system is in constant battle with virus production. CD4 cells and viruses constantly are being produced and destroyed, with turnover numbers in the billions of CD4 cells daily until the immune system gradually is overwhelmed. The current goal of HAART is to prolong this phase of HIV infection by suppressing viremia. Interestingly, this latent phase is unique among human viruses. In nearly all other cases of viral infection, the virus is completely cleared or the host dies outright. The only exceptions are the hepatitis viruses B and C and herpes simplex virus. Yet in those cases, clinical latency arises from viral latency, which is not the case with HIV.
Without antiretroviral therapy, most patients will experience 8-10 years of clinically silent HIV infection before experiencing symptoms. The average patient's CD4 count will lower by approximately 50 cells/microL per year. The rate of decline of the CD4 count (or plasma HIV RNA levels) can be used to roughly predict the clinical course and estimate time remaining before AIDS symptom onset.5 As one would expect, those patients with higher viremia to start with lose CD4 cells at an accelerated rate and progress to AIDS more rapidly. It is worth noting briefly that even after more than 10 years, a very few patients do not advance from their latent phase to active HIV disease. They remain asymptomatic with normal CD4 counts, yet always have measurable plasma levels of HIV even though they have taken no antiretrovirals. These patients are called long-term nonprogressors. The basis of their apparent resistance to HIV infection likely is multifactorial, and based on infection with a defective virus and/or unrecognized variability in the patients' genetics.
Symptomatic HIV Disease. The symptoms of HIV vary widely and can appear in nearly every organ system. These illnesses usually arise when the patient's CD4 count has dropped below 200 cells/microL. In general, the lower the CD4 count goes, the more severe illnesses become. CD4 counts of fewer than 50 cells/microL are a marker of end-stage disease and this corresponds to the appearance of the more severe and life-threatening opportunistic infections (i.e., Mycobacterium avium complex or disseminated cytomegalovirus). Thus, it is important to be aware of the patient's recent CD4 count as it can help guide treatment, although many ED patients with HIV will not know their counts. CD4 counts are checked only every 4-6 months and counts 3-6 months old still can be used clinically. For example, a patient with a cough who has HIV but CD4 counts greater than 350 cells/microL three months ago usually can be treated as having a normal immune system. This approach must be balanced with the knowledge that an AIDS-defining illness still can arise at any point during the course of HIV infection. The next sections will list the recent CDC definitions on the current list of AIDS-defining illnesses and describe the common complications of HIV infection by organ system. As fever with vague symptoms (malaise, weight loss) is one of the most common presentations for HIV-positive patients, a suggested approach to these patients also will be discussed. Initial treatments of common AIDS complications are listed for convenience in Table 2.
AIDS-Defining Illnesses. The condition of AIDS is defined by the CDC when one of the indicator conditions (AIDS defining illnesses) arises in a HIV-positive patient. In 1993 the CDC expanded the definition of AIDS to include any patient with a CD4 count less than 200 cells/microL regardless of the presence or absence of AIDS-related disease. This CD4 level is taken as laboratory evidence of severe immunosuppression. In addition, they added pulmonary tuberculosis, recurrent pneumonia, or invasive cervical cancer to the list of AIDS-defining illnesses. (See Table 3.) The addition of invasive cervical cancer to the list of AIDS-defining illnesses may be a bit surprising, but it has been shown that HIV-positive women have higher rates of infection with human papilloma virus (HPV—the virus strongly associated with cervical dysplasia and cancer) and that immunosuppression associated with HIV infection increases the risk of cervical cancer.7
| Table 3. CDC 1993 Updated List of AIDS-Defining Illnesses |
|
|
Several diseases on the list could be confused with more common and benign conditions, especially when they present in young, apparently healthy adults. For example, the patient with persistent diarrhea who thinks he has giardia actually may have cryptosporidiosis or isosporiasis. The patient who has had "cold sores" in the past, but presents because this outbreak has lasted more than four weeks is another example. The young woman with unexplained weight loss may need more than screening for thyroid disease and anorexia. The young man with "bad reflux" actually may have esophageal candidiasis. While seeing upper lobe pneumonia certainly will make one suspect TB, the patient also will need to be screened for coexisting HIV infection. While not on the CDC list, patients with hepatitis B also are at higher risk for simultaneous HIV infection as up to 90% of people with HIV have evidence of past hepatitis B infection.8 Thus, physicians may need to consider HIV as well when they suspect acute hepatitis.
Remember that up to 23% of patients with HIV in the United States are not aware of their diagnosis. The patient presenting with the second or third lobar pneumonia in the past two years should prompt physicians to think of HIV infection, not just lung cancer, as a possible underlying cause. Thus, the ED physician needs to be aware of these AIDS-defining criteria so that undiagnosed patients (or those with known HIV infection) and one of these conditions can be identified, treated, and referred to proper follow-up.
The HIV Patient with Fever. Fever with vague constitutional symptoms (i.e., weight loss, malaise, and fatigue) is one of the most common reasons for HIV-positive patients to present to the ED.9 Unfortunately, they can be perceived as challenging as the "weak and dizzy" elderly woman. The differential varies widely from benign to deadly disorders, and the clinical evaluation may be of no help to identify the truly sick patients. Fortunately, HIV patients with fever can be approached systematically to screen for identifiable conditions in the ED.
First, the patient's recent CD4 counts and viral loads should be obtained, if available. Those with CD4 counts greater than 350 cells/microL and/or viral loads less than 50,000 typically are immunocompetent. In ED patients, these numbers frequently will be unavailable, but other clues may be useful. Total lymphocyte counts less than 1000 cells/microL have been shown to correlate strongly with CD4 count fewer than 200 cells/microL.1 Patients who are noncompliant with antiretrovirals or prophylaxis (e.g., Bactrim for PCP) are likely to be at risk for AIDS-related diseases. Patients who stop their antiretrovirals usually have their CD4 counts return to pre-therapy levels in only a few weeks.10 The patient may not admit to being noncompliant with antiretrovirals, as they never may have started it. Anyone who has a history of an AIDS-defining illness should be on a form of HAART and, if not, can be assumed to be immunocompromised.
To generally screen HIV patients for AIDS-related conditions, five quick groups of questions can be used. The most common complications of HIV are pulmonary, neurologic, gastrointestinal, dermatologic, or ophthalmologic. Questions about shortness of breath, cough, and chest pain will screen for PCP, pneumonia, and TB. Asking about headaches, photophobia, confusion, seizures, and altered mental status will focus the exam on AIDS dementia, cryptococcal meningitis, toxoplasmosis, and CNS tumors. Complaints of persistent diarrhea, difficulty eating or swallowing, and abdominal pain likely will arise from candidiasis, bacterial or intestinal parasites, or may be a side effect of protease inhibitors (usually nelfinavir). Acute vision changes such as loss of acuity or visual field cuts, photophobia, eye redness, and eye pain should prompt one to think of CMV retinitis or ocular herpes. Lastly, new rashes can be from KS, herpes zoster, candidiasis, and scabies. If all of these symptoms are absent, the likelihood of common HIV complications is significantly lowered.
Tests may include electrolytes, complete blood count, urinalysis and culture, blood cultures (aerobic, anaerobic, and fungal), liver function tests, and chest films. Lumbar puncture for spinal fluid analysis and culture also may be indicated to evaluate for CNS infection. Specific serologic tests for cryptococcosis, toxoplasmosis, CMV, and coccidiomycosis may be indicated.
If no clear cause can be identified initially, consider the following conditions. Infection with the bacteria Mycobacterium avium complex (MAC) was a very common cause of disseminated disease in late-stage AIDS patients prior to the use of HAART. Patients who are not taking HAART medications and who have CD4 count fewer than 100 cells/microL should be considered high risk for MAC. Symptoms consist of fever, night sweats, weight loss, anorexia, diarrhea, and malaise. The diagnosis can be made with blood cultures or by acid-fast staining of body fluids. MAC should be treated with a combination of clarithromycin (Biaxin 500 mg PO BID), ethambutol (Myambutol 15-25 mg/kg PO Q day, max 1500 mg/day), and rifabutin (Mycobutin 300 mg PO Q day). The most common serious viral disease in AIDS is CMV, which also should be considered in these patients. Symptoms most commonly appear in the GI, CNS, and pulmonary organs. (See section on Ophthalmologic Disease.) HIV-positive patients with a history of injection drug use (IDU) and fever always should raise the suspicion for infective endocarditis. These patients should be admitted for echocardiography and blood culture results. Antibiotic coverage should be discussed with the admitting physician.
As with any ED patient, those HIV/AIDS patients with fever but no source who appear ill should be resuscitated, have appropriate laboratory studies drawn, receive antibiotic coverage initiated, and be admitted/transferred to an appropriate admitting physician. Ideally, patients should be admitted to their primary physician with infectious disease consultation or directly to the ID specialist once stabilized. The patient may require transfer to a facility to obtain specialty consultation, and this can be addressed on an individual basis.
Respiratory Disease. Over the course of their illness, at least 80% of AIDS patients develop some kind of pulmonary disease,12 with pneumonia being the most common. Unfortunately, the causes of pulmonary disease in the HIV patient are quite diverse. They include bacterial, fungal, protozoan, viral, and neoplastic causes. (See Table 4.) Only by correctly identifying the underlying cause will the physician be able to initiate the proper treatment. Signs and symptoms in these patients may be of little assistance. Most patients will present with cough, fever, shortness of breath, chest pain, and, in some cases, hemoptysis. Prolonged courses (2-weeks) are more likely to be from PCP, where acute presentations (3-5 days) are more likely to be bacterial in origin. Patients with productive coughs, though, are more likely to have bacterial sources, and hemoptysis will be seen more often in cases of TB or pneumococcal pneumonia.
Chest radiographs can help distinguish among the different causes, and these findings are summarized in Table 4. Patients with focal consolidation usually have bacterial pneumonia, but they also can have PCP or Mycoplasma. Diffuse interstitial infiltrates most often are seen with PCP, but also may occur with cytomegalovirus (CMV) in patients with advanced HIV disease. Though usually thought of as located on the skin, Kaposi's sarcoma can cause nodular lesions and adenopathy. Cavitary lesions not only occur with bacterial pneumonia but also with PCP or fungal infections. Adenopathy alone can be a marker of lymphoma. TB can cause any of the above findings in addition to just upper lobe disease. Chest CT may be helpful in select cases. The patient with suspected PCP and normal plain films will have findings on CT scan in 39% of cases.10 CT scan also may be of use in differentiating the cause of pulmonary nodules (neoplasm vs opportunistic infection) and adenopathy (TB or fungal disease).
Patients with CD4 counts of fewer than 200 cells/microL are at increased risk for PCP and Cryptococcal disease. Counts of fewer than 100 cells/microL increase risk of Pseudomonas aeruginosa and Staphylococcus aureus pneumonia, Toxoplasma gondii pneumonia (though rare), as well as KS. Counts fewer than 50 cells/microL often are associated with disseminated MAC, Aspergillus, CMV, histoplasmosis, and Coccidioidomycosis. Patients with disseminated disease often present with non-pulmonary symptoms as the primary complaints. It is important to remember that some diseases do not correlate well with the CD4 count and may present at any level. These include bacterial sources (i.e., Streptococcus pneumoniae, Haemophilus species, TB) as well as non-Hodgkin's lymphoma and nonspecific interstitial pneumonitis. The HIV-positive patient is 6-10 times more at risk for pneumococcal pneumonia, and 100 times more at risk for bacteremia than those without the virus.11
Blood cultures should be obtained to help identify bacteria, fungi, and mycobacteria. PCP is the most common diagnosis in patients with CD4 count fewer than 200 cells/microL. For patients with CD4 counts greater than 200 cells/microL and no clear diagnosis, treatment can be initiated with a third-generation cephalosporin (ceftriaxone [Rocephin]) and azithromycin (Zithromax). Aside from Staph and Strep, other bacterial causes include Haemophilus influenzae, Chlamydia pneumoniae, and Pseudomonas aeruginosa. Sputum cultures, blood cultures, and bronchial washings may be required for accurate diagnosis.
Pneumocystis pneumonia (PCP). The organism Pneumocystis carinii was changed to Pneumocystis jirovecii in 1999.13 Pronounced "yee row vet zee," the name change is in honor of the Czech parasitologist Otto Jirovec. Dr. Jirovec determined that Pneumocystis species actually are fungi (albeit strange ones). Fortunately, the acronym PCP still is accepted as it now stands for pneumocystis pneumonia.
PCP is one of the most common opportunistic infections seen in AIDS patients, though prophylaxis is helping to reduce the incidence. If not using HAART prophylaxis, up to 70% will acquire PCP during their HIV infection, and PCP is the original AIDS-defining infection in 60% of cases.14 Further, when a cause of death can be established, PCP is the most common cause of death in AIDS patients. Unfortunately, once the patient becomes ill enough to be admitted to the ICU, pre-existing use of HAART medications does not seem to provide any protection from PCP mortality. One study of 259 ICU admissions of AIDS patients over a three-year period found no difference between ICU or hospital mortality, need of mechanical ventilation, or ICU and hospital length of stay between patients previously treated with HAART or not.15
Classic presenting complaints include fever, non-productive cough (can be productive), burning retrosternal chest pain, and progressive shortness of breath. Symptoms generally progress slowly. Typically, chest films show diffuse interstitial infiltrates in 80% of patients, but the remaining 20% will have normal studies.16 Those on pentamidine nebulizer prophylaxis are more prone to cavitary upper lobe lesions.11 Other useful tests include the arterial blood gas, which will show hypoxemia and an increased alveolar-arterial (A-a) gradient. Lactate dehydrogenase (LDH) is elevated in PCP, but this has a low specificity and sensitivity for PCP vs. bacterial pneumonia/TB in the acute setting.17 Patients with HIV and unexplained hypoxia are presumed to have PCP in the ED. The final diagnosis may require bronchoalveolar lavage, sputum testing with monoclonal antibodies, sputum culture, or biopsy.
Treatment of PCP continues to be with trimethoprim (15-20 mg/kg/day) and sulfamethoxazole (75-100 mg/kg/day) (TMP-SMX) given either orally or intravenously in two daily divided doses for a total of 21 days. Treatment should be started in the ED (e.g., two Bactrim DS tablets every 8 hours) as a definitive diagnosis may not be made for several days. Unfortunately side effects of TMP-SMX are very common, and occur in 65% of AIDS patients.18 Multiple other drugs can be used (pentamidine isethionate, dapsone, clindamycin plus primaquine, atovaquone, or trimetrexate) and consultation with an ID specialist is recommended. Most will recommend intravenous pentamidine (4 mg/kg/d). Adjunctive treatment with a prednisone taper (21 days) also is used for patients with PaO2 less than 70 mmHg or A-a gradient greater than 35 mmHg.19 Begin with higher doses of 80 mg for 5 days, 40 mg for the next 5 days, and 20 mg for the remaining 11 days. Prophylaxis for PCP (Bactrim DS 1 PO qD) is advised for anyone with history of a previous infection or with CD4 count fewer than 200 cells/microL.
Mycobacterium tuberculosis (TB). TB is a significant problem for AIDS patients around the world; up to 33% of deaths are the result of TB.11 TB arises from both primary infection and reactivation disease. Approximately 14% of HIV-positive patients in the United States have co-infection with TB. Even more concerning, patients with HIV/AIDS progress more rapidly to active disease; the rate of reactivation for an HIV-positive patient is around 10% per year.11 Thus AIDS patients have relatively high rates of active TB, as confirmed by one study finding 5% of AIDS patients in the United States with active TB.20 A majority of these cases are in patients between the ages of 25 to 44 years, African American or Hispanics located in New York or Miami. In these groups, up to 70% of new cases of active TB are in HIV-positive patients.11 Further, incidence of multi-drug resistant TB is more common in AIDS populations, with 6% of TB in these patients being multi-drug resistant TB.20 Thus some of the TB outbreaks in AIDS patients in the early 1990s have led to mortality rates as high as 70%.21
In contrast to TB infections from atypical bacteria (MAC), pulmonary TB often strikes HIV patients early in the course of the disease. TB often is diagnosed in patients with CD4 count between 200-500 cells/microL.12 It is not unusual for pulmonary TB to be the initial AIDS-defining illness. These patients normally present with the classic pattern for pulmonary reactivation disease: fever, cough, weight loss, night sweats, and dyspnea on exertion. They also have the classic radiographic upper lobe cavitary lesion as well. Like with other disorders, disseminated disease is more common in patients with CD4 count fewer than 200 cells/microL. Patients with disseminated disease more often have lower lobe infiltrates and hilar/mediastinal lymphadenopathy. The classic upper lobe lesions occur less often and their chest films may not distinguish between TB and other opportunistic infections. TB commonly spreads to bone, brain, meninges, GI tract, viscera, and lymph nodes. Ultimately, 33% of HIV patients will have only pulmonary TB, 33% will have only extra-pulmonary TB, and the remaining 33% will have both.
In the ED, one must maintain a high degree of suspicion for TB in the HIV patient. Patients should be placed in respiratory isolation in a negative pressure room to prevent further exposure of staff and other patients. Negative PPD tests are common in AIDS patients with TB, and history of a negative test should not reassure one that TB has been ruled out. Like with any ED patient, admission decisions will be based on a multitude of factors, including the clinical condition, certainty of the diagnosis, availability of follow-up, and recommendations of the consultant. Most recommend a four-drug regimen (isoniazid, rifampin, pyrazinamide, and streptomycin) in AIDS patients with TB, but consultation is advised with treatment due to the drug interactions between rifampin and NNRTIs and PIs. These drug interactions complicate treatment and have led to new treatment guidelines from the CDC.22
Fungal. Fungal infections are common in the later stages of AIDS (lower CD4 counts), and can present with lung involvement. Patients will have symptoms similar to PCP, with fever, cough, dyspnea, weight loss, and hemoptysis. In at least 50% of these cases, the patient will be fungemic, and more than 90% also will have CNS infection.11 Sources can include Coccidioides immitis, Aspergillus, and histoplasmosis. Coccidioides is endemic to the southwest, and chest film findings can be useful as diffuse reticulonodular infiltrates are seen. Fluconazole 400 mg/d PO is the treatment for mild cases, and IV amphotericin B (0.7 mg/kg/day) is reserved for patients requiring hospitalization.
Neurologic Complications of HIV. Neurologic complications of HIV infection unfortunately also are very common. Neurologic disease is the initial AIDS-defining illness in up to 20% of cases,18 and up to 90% of AIDS patients will suffer neurologic problems during the course of their disease.23 These conditions can arise in both early and late HIV infection. Patients with acute HIV conversion can have focal or diffuse encephalopathy, ataxia, herpes zoster radiculitis, myelopathy, peripheral neuropathies, and aseptic meningitis.24 As with most complications of acute seroconversion, these symptoms usually abate with the immune response to HIV. Some patients will experience complications during the latent phase of HIV infection (CD4 count greater than 200 cells/microL) including segmental neuropathy, brachial plexopathy, and polyneuropathy with similarities to Guillian-Barré syndrome. AIDS patients most commonly suffer from HIV encephalopathy (AIDS dementia complex), cryptococcal meningitis, toxoplasmosis, and primary CNS lymphoma.18 These conditions will be discussed below. HIV/AIDS patients also can have infections that affect non-HIV patients, such as bacterial meningitis as well as more classic CNS disease of stroke and aneurysms. Further there are a number of disseminated HIV diseases that can affect the CNS, including histoplasmosis, CMV, herpes simplex, and TB.
AIDS Dementia Complex. Also termed HIV encephalopathy, AIDS dementia is a common neurologic complication of HIV infection. It is more common than toxoplasmosis or cryptococcal meningitis, and up to 33% of AIDS patients will be afflicted.25 Although the mechanism is not fully understood, AIDS dementia appears to be a direct effect of the virus on the CNS. Treatment with HAART has significantly lowered the frequency of severe dementia, and also can slow progression, indicating the risk for dementia is related to the viral load.26 As opposed to other neurologic complications (such as CMV encephalitis, cryptococcal meningitis), AIDS dementia slowly appears over several months, and differs from Alzheimer's dementia in that aphasia (inability to comprehend words), apraxia (difficulty speaking), and agnosia (inability to name or recognize objects) are relatively uncommon. Loss of memory, cognitive impairment, and gait problems are the primary complaints as opposed to focal neurologic signs or delirium, seizures, or headaches. It is important to realize that AIDS dementia is a diagnosis of exclusion, and that patients with a history of dementia still may develop opportunistic CNS infections. The appearance of delirium or focal neurologic signs should signal that a process other than dementia likely is behind these changes. MRI will show only diffuse symmetrical hypodensities in the white matter, and spinal fluid typically is normal. Without HAART, the deficits can progress with time to become profound with the patient eventually unable to move or speak.
Toxoplasmosis. Toxoplasma gondii is the second most common CNS infection in AIDS patients, although use of HAART is reducing the number of cases. T. gondii is a protozoan parasite that can infect a variety of mammals and birds. One of its three life cycle forms (tachyzoite) disseminates through the body of its host and deposits in various locations, including muscle, heart, and brain. The tachyzoite forms cysts in these tissues that can persist for the life of the individual. Cats are the definitive host of T. gondii and the source of most human infection. Oocysts passed from the cat's intestinal tract are accidentally ingested by humans through close contact with the cat's litter box, or by accidental ingestion of food (undercooked meat) or water contaminated with oocysts. The oocysts are infective for more than one year while in the environment, and thus may be present even after removal of the cat from the patient's house. In the absence of HIV infection, toxoplasmosis primarily is a concern for pregnant women who can become infected during pregnancy and pass the infection to their fetus.27 Exposure to T. gondii is actually quite common in the United States with seroprevalence as high as 67% of adults in some areas.27
As infection rates are high in the overall U.S. population, 30-50% of HIV-positive patients will progress to active toxoplasmosis if they are not receiving appropriate prophylaxis. Toxoplasmosis is thought to be a reactivation of previous disease, and is a late complication of HIV seen in patients with CD4 counts greater than 200 cells/microL. Patients present with headache, fever, and altered mental status. Seizures can be seen in 40% of patients.27 As the protozoan has predilection for the subcortical area of the cerebral hemispheres, basal ganglia, cerebellum, and brain stem, focal neurologic signs are common and are seen in 80% of cases.27 Although toxoplasmosis lesions will show ring-enhancement with contrast, non-contrast CT scan is used first because a normal initial scan is unlikely to show lesions when contrast is added.28 MRI is more sensitive and accurate in detecting lesions. The presence of multiple focal lesions, though not pathognomonic, strongly favors toxoplasmosis over other diagnoses such as CNS lymphoma or fungal infections. Spinal fluid antibodies alone are not diagnostic and may just represent previous exposure.
Patients with suspected toxoplasmosis should be admitted and started on pyrimethamine (100-200 mg PO loading dose, followed by 50-100 mg/day PO), plus sulfadiazine (4-8 g/day PO) with folinic acid (10 mg/day PO) to reduce the incidence of pancytopenia.18 High-dose steroids (Decadron 4 m IV q 6 hours) should be given in cases where mass effect is seen on CT or MRI scan, and seizure prophylaxis (phenytoin) should be discussed with the admitting physician. Maintenance therapy will be required after initial treatment to prevent relapse. Prophylaxis also is recommended for any patient with positive toxoplasmosis serology and CD4 count fewer than 100 cells/microL.
Cryptococcal meningitis. The fungus Cryptococcus neoformans is the organism responsible for the third most common CNS infection and most common type of meningitis seen in AIDS patients. C. neoformans is found in soil, especially soil contaminated by pigeon droppings. Transmission is thought to occur by inhalation of aerosolized spores. Up to 10% of AIDS patients will have cryptococcal disease,29 but numbers have decreased recently due to use of HAART. Most patients with cryptococcal meningitis have advanced disease with CD4 count fewer than 50 cells/microL.30 Presenting symptoms are those of subacute meningitis: fever, headache, nausea, and vomiting. Altered mental status may occur, but this is less common. Papilledema and cranial nerve palsies also can occur.
CT scan should be performed largely to rule out other diagnoses, as 50% of CT scans in cryptococcal meningitis are normal.29 When abnormalities are found, lesions consist of cystic clusters of fungi found mostly in the meninges, basal ganglia, and cortical gray matter. The most useful test is the cryptococcal antigen test using CSF as it is nearly 100% sensitive and specific.18 Serum cryptococcal antigen tests also are helpful, but slightly less sensitive. Identification of yeast with India ink staining of CSF is helpful when positive, but a negative test does not rule out cryptococcal disease. CSF pressures usually are greater than 25 cm H20. Admission is indicated for treatment with IV amphotericin B (0.7 mg/kg/day) and flucytosine (100 mg/kg/day PO). Bone marrow suppression is the most common complication of treatment. As with many other HIV infections, lifelong maintenance therapy (fluconazole 200 mg per day) is given to all patients treated for cryptococcal disease.
Primary CNS Lymphoma. CNS lymphoma, or primary B cell non-Hodgkin's lymphoma, is another disease that occurs in much higher frequency than in non-HIV-positive patients. The incidence of primary B cell lymphomas in HIV-infected patients is up to 6%, which is at least 1000 times higher than in the general population.31 Primary CNS lymphoma makes up about 20% of the lymphoma seen in AIDS patients,11 but is the AIDS-defining illness in only abut 3% of cases.24 CNS lymphoma occurs late in the course of AIDS, with CD4 counts usually fewer than 50 cells/microL. Patients usually present with headaches and focal neurologic deficits (cranial nerve palsies), and seizures. CT or MRI often shows only 1-3 ring-enhancing lesions that range in size from 3-5 cm. Treatment with radiation therapy is palliative in nature, as the prognosis is poor. Median survival times are less than 1 year.11
Renal. Renal complications seen in HIV-positive patients can be divided into pre- or postrenal azotemia, acute renal failure, and chronic renal insufficiency. Prerenal azotemia is the most common disorder seen, typically caused by dehydration from opportunistic GI infections. Postrenal azotemia also can occur with obstruction from lymphoma, stones, fungus balls, blood clots, or sloughed papilla.18 Treatment involves identifying the cause of and removing the obstruction. Acute renal failure can be a complication of antiretroviral medications, and chronic renal insufficiency most often is caused by direct effects of the virus on renal tissue (HIV-associated nephropathy).
Acute renal failure in HIV patients can result from ischemic tubular necrosis as a complication of sepsis or hypotension just as it can in non-HIV-infected patients. Some cases, though, are a result of antiretroviral and other drugs' side effects. Some of the commonly used medications in AIDS patients with known nephrotoxicity include acyclovir, amphotericin B, dapsone, foscarnet, ganciclovir, pentamidine, rifampin, and trimethoprim. The following HIV medications also have known nephrotoxicity: indinavir (Crixivan), ritonavir (Norvir), cidofovir (Vistide), and yenofovir (Viread).32 HIV-positive patients presenting in acute renal failure should be assessed for need of urgent dialysis (potassium levels, volume overload). Discuss stopping potentially causative drugs with the admitting physician.
Early on in the HIV epidemic, signs of direct renal damage from the virus were seen in AIDS patients. Termed HIV-associated nephropathy (HIVAN), it occurs in up to 10% of HIV-positive patients. Of the patients with HIVAN, 50% are IV drug users and 90% are black.33 As blacks represent 51% of the new cases of HIV diagnosed between 2001 and 2004,34 HIVAN is a significant cause of renal disease in blacks. Indeed, it is the third leading cause of end-stage renal disease (ESRD) in blacks following only diabetes and hypertension.35
The reasons for these associations remain unclear as does the exact mechanism of renal damage by the virus. Current research suggests a direct infection of renal epithelial cells by HIV. Unfortunately, these patients develop ESRD at an alarmingly rapid pace. The average time to ESRD from initial diagnosis of proteinuria is only 4-16 weeks.33 Even so, peripheral edema and accelerated hypertension does not develop as frequently as in other causes of progressive renal failure. Thus in most cases noting proteinuria, elevated blood urea nitrogen, or creatinine levels will be an incidental finding in the work-up of HIV-positive patients. Stable HIV-positive patients with significant proteinuria, or evidence of declining renal function should have appropriate follow-up arranged with a nephrologist. Several options for treatment can be effective, including corticosteroids, angiotensin-converting enzyme inhibitors, and ultimately dialysis. Therapy with HAART has been reported to dramatically improve renal function and lower proteinuria in many cases.36 Unfortunately, it is likely that even effective use of HAART will not stem the tide of increasing ESRD AIDS patients. One study estimated that even with a 95% reduction in progression to AIDS from use of HAART, the number of ESRD AIDS patients still will increase.37
Gastrointestinal (GI). Nearly 50% of AIDS patients will suffer from GI opportunistic infections during the course of their illness and many of them will end up in the ED for evaluation.11 Most patients will present with abdominal pain, diarrhea, or GI bleeding. The differential diagnosis list for acute abdominal pain in a non-HIV-positive patient is long enough, but it is even more extensive for the patient with AIDS.38 These patients can have non-HIV-related disease (i.e., appendicitis, cholecystitis, ectopic pregnancy, pelvic inflammatory disease, diverticulitis, peptic ulcers, etc). Further, some antiretrovirals are known to cause side effects that can produce abdominal pain (i.e., renal stones from indinavir, pancreatitis from didanosine). HAART complications often include vomiting, diarrhea, and liver abnormalities.
Oral Lesions. In AIDS patients, oral lesions can result from fungal, viral, and bacterial sources. Neoplasms such as KS and Hodgkin's lymphoma also can cause oral lesions. Fungal causes consist of oral candidiasis, or thrush. Thrush is seen in 80-90% of AIDS cases.18 Patients complain of burning pain with the lesions, dysphagia, and taste change. Dysphagia with thrush is limited to the mouth and throat, whereas dysphagia that extends into the chest is more indicative of esophagitis. (See next section.) Whitish plaques can be seen on the tongue, soft palate, and gums. These plaques can be scraped off easily, which distinguishes them from those of hairy leukoplakia. Treatment is with clotrimazole troches (10 mg PO five times a day for 14 days) or with nystatin pastilles (2 dissolved in mouth QID). Relapse is common, and systemic treatment with fluconazole (200 mg single dose, or 100 mg PO q d for 14 days) may be required.
Viral causes include oral hairy leukoplakia (caused by the Epstein-Barr virus) or herpes ulcers. While it appears similar to thrush, hairy leukoplakia usually is asymptomatic and appears as thickened lesions on the lateral edges of the tongue. If treatment is desired, antiretrovirals such as famcyclovir (500 mg BID for 7 days) can be used. Herpes mouth ulcers are painful oral ulcers that seem to take longer to resolve in AIDS patients. Aphthous ulcers commonly are confused with oral herpes lesions, but these do not appear more commonly in AIDS patients than in the general population. Aphthous ulcers respond well to topical steroids.
Esophagitis. Esophagitis can be thought of as an extension of the oral lesions in AIDS patients, although esophageal lesions may be present without corresponding oral lesions. Patients will complain of dysphagia and chest pain. The causes and treatments are largely the same as with oral lesions. The most common cause is candidiasis, and therapy with fluconazole (100-400 mg PO daily for 2-3 weeks) or ketoconazole (200-400 mg PO daily for 2-3 weeks) should be initiated in the ED. One need not wait for a definitive diagnosis to begin presumptive treatment. Patients with dehydration or the inability to take anything by mouth may need admission and treatment with amphotericin B. Esophageal ulcerations also may be caused by CMV or herpes simplex, and endoscopy is required to accurately diagnose the cause. CMV esophagitis should be treated with ganciclovir (5 mg/kg/IV q 12 hours).
Liver Disease. Liver complications are common in AIDS patients for several reasons. Hepatitis B and C are common co-infections, especially among IDUs. Up 90% of people with HIV have evidence of past hepatitis B infection.8 Infection with CMV, MAC, and TB all can produce a hepatitis-like condition. Lastly, non-nucleoside reverse transcriptase inhibitors (NNRTIs such as efavirenz) often have hepatic complications such as hepatic necrosis, cholestatic hepatitis, and fulminant liver failure.
Diarrhea. Diarrhea is reported universally in HIV/AIDS patients, and may be the most common overall complaint with the virus. Symptoms can vary from a few loose stools per day to fever, chills, bloody stools, and dehydration. Nearly 100% of AIDS patients in third world countries report diarrhea.39 Diarrhea is a complication for 50-90% of HIV-positive patients in the United States.18 In about 30% of cases, the cause of diarrhea never is identified.1 About half of these cases are attributed to the direct effect of HIV on GI tissues. These late-stage AIDS patients have large-volume, watery diarrhea and frequently require admission for rehydration and electrolyte replacement.
Laboratory tests may include stool samples for leukocytes, ova, and parasites; bacterial culture; C. diff toxin; and acid-fast stain. In some cases, these results may not always be available during the evaluation in the ED. However, historical features can help differentiate the possible causes of diarrhea. Chronic diarrhea is defined by symptoms lasting more than three weeks. Small-volume stools containing WBCs and/or blood are more likely to be colonic (inflammatory) in origin. Diarrhea originating in the small bowel (secretory) typically is watery, occurs in large volumes, and lacks WBCs/blood.
The causes of diarrhea are many, and the differential depends in part on the CD4 count. When the CD4 count is greater than 200 cells/microL, the only pathogen that seems to be more common in HIV-positive patients is Salmonella. Salmonellosis in the HIV-positive patient always is treated with antibiotics, as they are at higher risk for bacteremia and sepsis.18 CD4 count fewer than 200 cells/microL suggest opportunistic infections, but these are most common with CD4 count fewer than 50 cells/microL. The most common opportunistic infections causing acute diarrhea in AIDS patients are Salmonella and C. difficile. Chronic diarrhea is caused by multiple opportunistic including Cryptosporidium parvum, Microsporidia, CMV, and MAC.
Several of these opportunistic causes of diarrhea in AIDS patients deserve further mention. Cryptosporidium parvum is a common intestinal parasite that produces persistent watery diarrhea. Interestingly, the organism spreads to the biliary tract and is responsible for 20% of the case of acute cholecystitis in AIDS patients.38 Oocysts in the stool can be identified with acid-fast staining. Treatment is with paromomycin (500-750 mg PO four times daily for 2-4 weeks) or azithromycin (2400 mg/day on day 1, followed by 1200 mg/day for 4 weeks). Expect frequent relapses as, once established, C. parvum responds well only to restored immune function. End-stage AIDS patients more commonly have MAC or CMV. Disseminated M. avium infection produces fever, abdominal pain, night sweats, and weight loss in addition to the diarrhea. Abdominal lymph nodes typically are enlarged and, at times, MAC enteritis can mimic acute surgical problems.40 Treatment is with azithromycin (600 mg PO q day) plus ethambutol (15-20 mg/kg/day) and plus rifabutin (300 mg PO q day). CMV is the most common viral GI infection and produces frequent bloody stools. If suspected, most patients will require admission for biopsy to prove the diagnosis.
Protease inhibitors commonly are associated with diarrhea as a side effect, especially nelfinavir (Viracept), ritonavir (Norvir), and lopinavir/ritonavir (Kaletra). It tends to be secretory in nature, with large-volume, watery stools. This type of diarrhea usually responds to loperamide (Imodium 4 mg first dose, then 2 mg after each unformed stool, max 16 mg a day). Remember that Clostridium difficile can complicate antibiotic use. While HIV-positive patients are not at higher risk for this bacteria as they are with Salmonella, C. difficile diarrhea occurs frequently as AIDS patients often use antibiotics. Treatment for C. difficile diarrhea is with metronidazole (Flagyl 500 mg PO TID for 10-14 days).
Neoplasms. Kaposi's Sarcoma (KS). A cluster of cases of Kaposi's sarcoma in previously healthy homosexual men in 1981 was one of the original diseases that drew attention to the emergence of HIV in the United States.41 Before then, KS was primarily a rare disease of organ transplant patients or people of Mediterranean, Jewish, or African heritage. In 1981, KS was seen in 79% of AIDS patients while currently the number is 25%.11 The primary reason for this decline is not effective treatment, but that KS commonly is seen in homosexual and bisexual men with HIV, and in 1981 this was the primary group infected with HIV. The reasons for the predilection of KS to homosexual/bisexual men remains unclear. KS is caused by the virus Kaposi sarcoma-associated herpes virus (KSHV), also known as human herpesvirus 8.42 Patients with lower CD4 counts have increasing risk for KS.
KS lesions can appear anywhere on the body, but usually affect the mucus membranes, skin, lymph nodes, lungs, and GI tract. The lesions are vascular nodules, plaques, papules, or tumors. Patients may have KS internally without skin lesions and vice versa. They can present with GI bleeding or bowel obstruction, but most often patients complain of lymphedema of the lower extremities caused by KS infiltration of the lymph nodes. Treatment is varied with local cyrotherapy or radiation, systemic chemotherapy, or, in some cases, lesions respond to HAART.43
Non-Hodgkin's Lymphoma. Non-Hodgkin's B-cell lymphoma is another late complication of HIV, with the majority of patients having CD4 counts fewer than 50 cells/microL. It is an AIDS-defining illness and occurs in 10% of AIDS patients. The risk of non-Hodgkin's lymphoma in HIV is estimated to be 120 times that of the general population.11 Unfortunately, the risk of lymphoma increases exponentially with duration of HIV infection and lowered immune function, an obvious problem for those living longer with HIV.
Besides the lymph nodes, non-Hodgkin's lymphoma affects the GI tract, liver, soft tissues, CNS, and bone marrow. Patients can present with painless lymph node swelling, fever, weight loss, night sweats, and fatigue. Before HAART, the survival rate for non-Hodgkin's lymphoma in HIV patients was only 4-6 months. With HAART the number has increased to 2 years or more, but unlike many other HIV complications, HAART does not seem to decrease the incidence of non-Hodgkin's lymphoma.
Other Malignancies. HIV patients have about a five-fold increased risk for Hodgkin's lymphoma. Unfortunately, it is more difficult to treat in these patients as they do not tolerate chemotherapy as well as non-HIV-infected patients and have a much higher incidence of tumor relapse.
In 1993, invasive cervical cancer was added to the list of AIDS-defining illnesses.7 There appears to be a high prevalence of human papillomavirus (HPV) infection, the virus associated with dysplasia, in HIV-positive patients. Abnormal PAP smears are seen in 60% of HIV-positive women compared to a rate of 5% in the general population.11 The risk factors for HIV infection and HPV overlap, including a high number of sexual partners and history of sexually transmitted diseases. Thus, women who become infected with HPV also are at high risk for HIV infection. Preliminary data suggest that immunosuppression caused by HIV infection also increase the incidence of cervical dysplasia and cancer in these patients.44 Currently treatment is the same for HIV-infected and non-infected women with cervical dysplasia and cancer. Not enough data have been collected to determine if HAART medications have an effect on cervical cancer in HIV-positive patients as they do with KS and non-Hodgkin's lymphoma.7
Cutaneous Complications. Up to 90% of patients have skin disorders during the course of their illness.11 While not often dangerous, they can be uncomfortable and bring the patient to the ED. HIV-related skin disorders fall under five general categories: infectious, inflammatory, neoplastic, drug-related, and the acute exanthema of primary HIV infection.
Acute Exanthem of HIV Seroconversion. The acute exanthem of primary HIV infection is important for the emergency physician to recognize as it represents an opportunity to catch the patient early in the course of their HIV infection. Of all the other HIV-associated skin conditions, this is the only one directly caused by the virus rather than through lack of immune system function. The rash appears in the context of the acute seroconversion syndrome. The syndrome is similar to mononucleosis, with fever, pharyngitis, myalgias, fatigue, headache, diarrhea, vomiting, and lymphadenopathy. The rash appears with the other symptoms about 2-6 weeks after exposure to the virus, and dissipates as the immune system increases antibody production and clears the majority of the virus from the bloodstream. In most patients, the acute HIV syndrome lasts only 1-3 weeks.
The rash itself consists of maculopapular lesions similar to roseola, and mucosal ulcerations. The rash appears on the face, neck, and trunk more so than on the limbs, but it can appear on the palms and soles. The ulcers are seen on the oral and genital mucosa and can be confused with herpetic lesions. Anyone who is discharged from the ED with suspected acute HIV syndrome should be referred for HIV testing with good documentation of the referral and discussion of its importance with the patient. Be aware that ELISA and Western blot HIV screening tests look for the presence of antibodies to HIV, and that these tests may not be positive early in the acute HIV syndrome.
Infectious Conditions. Infectious skin conditions in HIV can be viral, bacterial, fungal, or parasitic in nature. Herpes simplex viruses are the most common and painful of these conditions. The incidence of herpes zoster in AIDS patients is 25 times that of a non-HIV-infected patient and is likely reactivation of previous controlled infection.18 It takes many forms and can appear locally, systemically, or can manifest in an atypical fashion (i.e., multiple dermatome involvement). Oral mucosa, genital, and rectal areas are commonly affected sites for the severely painful grouped vesicles with an erythematous base. Outbreaks often are recurrent and can be resistant to treatment. Acyclovir (800 mg five times daily) or famciclovir (5 mg PO bid or tid for 7 days) are the drugs of choice for outpatient treatment. Consider admission for patients with systemic infection, ophthalmic zoster, or those with extensive dermatomal involvement.
Molluscum contagiosum is caused by a pox virus and is normally present in healthy children. Immunosuppression increases the incidence of these small umbilicated papules, and is found in up to 20% of HIV cases.18 Simple warts caused by human papilloma virus may not be more common in AIDS patients, but when they occur, they often are diffuse infections. Condyloma also are more frequent, and both respond best to raising CD4 counts rather to than local treatment.
Various bacterial skin infections also occur. Simple bacterial folliculitis (acne) is common, and usual pathogens include Staphylococcus aureus, S. epidermidis, and Pseudomonas aeruginosa. Impetigo and skin abscesses also can be seen, but unlike in the non-HIV-infected patient, systemic antibiotics often are required. Rashes from secondary syphilis may be seen, but biopsy may be required for diagnosis as VDRL tests can be falsely negative. Anyone with suspected syphilis seen in the ED should raise suspicion for undiagnosed HIV infection, and should be referred for HIV testing. TB infection can manifest as folliculitis, abscesses, or simple dermatitis. Culture of biopsy samples are required for accurate diagnosis.
Fungal skin infections, especially those of the oral mucosa (thrush), are one of the hallmarks of HIV infection. White exudates can be scraped away easily, which differentiates them from oral hairy leukoplakia. (See section on GI Complications.) Cutaneous dermatophytosis commonly is seen in HIV infection as well and can have widespread involvement. In any individual with extensive tinea corporis or subungal onychomycosis, the presence of HIV infection should be suspected. In patients with advanced HIV disease, histoplasmosis and cryptococcosis can infect the skin. Clinical identification is difficult as presentation is nondescript and can consist of pustules, nodules, ulcers, or papules. Tissue biopsy and specialized antifungal therapy are required.
Scabies is encountered more frequently in people with HIV infection than in the general population. Classic scabies can be treated with ivermectin or permethrin, but cases may be resistant and difficult to fully cure. Norwegian scabies is associated with HIV infection as well, and produces widespread skin involvement with psoriasis-like lesions. Norwegian scabies treatment is referred to the consultant.
Inflammatory Conditions. Multiple inflammatory skin problems affect the AIDS patient, and in some cases these may raise the suspicion of HIV infection in undiagnosed patients. Most will respond to topical antihistamines and/or steroids. Seborrheic dermatitis is the most common of these conditions, and is seen in up to 85% of cases but in only 3% of the general population.11 Clinically, the disease is characterized by slightly indurated, diffuse or confluent plaques with yellow greasy scales and crusting in malar, nasolabial folds, eyebrows, and scalp. In severe cases, it can extend to the chest, neck, and other parts of the body. Psoriasis does not appear more frequently in AIDS patients, but is more severe. Atopic dermatitis and seborrheic dermatitis, if already present, will worsen with falling CD4 counts. Eosinophilic pustular folliculitis can appear as hives that develop on the upper trunk, head and neck.
Neoplastic Conditions. Kaposi's sarcoma is probably the best known of HIV-related skin conditions, and is the second most common AIDS complication.18 (See section on KS.) The incidence of Kaposi's sarcoma has markedly decreased with the advent of HAART,45 but only in patients who are compliant with therapy. Several other HIV-associated malignancies produce skin findings as well. Malignancies of both B and T cells can develop in HIV-positive patients and most are at advanced stages at the time of diagnosis. Non-Hodgkin's lymphoma, cutaneous T-cell lymphoma, basal and squamous cell carcinomas, and melanoma are all seen in HIV infection. Melanoma and squamous cell carcinoma can have a more aggressive course in patients with HIV, while basal cell carcinoma can appear more frequently but does not appear to be more aggressive.45
Drug-Related. Erythema multiforme and Stevens Johnson syndrome have been reported with antiretrovirals as well as sulfa drugs. The NRTIs, particularly abacavir (Ziagen), seem to more high risk than some of the other HAART medications. It also has been suggested that the incidence of warts caused by human papillomavirus in patients of HAART may be increased.46 Each of the antiretrovirals can have unwanted associated effects ranging from paronychias and hypertrichosis to vasculitis and alopecia. The term lipodystrophy refers to a symmetric loss of fat from the extremities with redistribution to the abdomen, neck, and supraclavicular area, and has been associated as a side effect of HAART therapy. Lipodystrophy usually occurs within 1 year of HAART therapy and is estimated to affect around 50% of those on combination therapy.47 It is thought to result from inhibition of NRTIs on mitochondrial oxidative phosphorylation.48 No therapies for this syndrome have been established.
Ophthalmologic Complications. Ocular problems represent another organ system commonly damaged by HIV; 75-90% of AIDS patients will have ocular complications during the course of their disease.1 Cotton wool spots are the most common finding on ocular exam in AIDS patients, and they represent retinal microvascular disease. These usually are asymptomatic and virtually never affect the patient's vision. Thus no treatment is required. However, they can be confused with findings of CMV retinitis and can only be differentiated by serial exam, as the lesions of CMV are progressive.
CMV retinitis is the most common sight-threatening ocular complication of HIV, seen in up to 30% of AIDS patients.18 CMV is a late findings of HIV infection, and usually is seen in patients with CD4 count fewer than 50 cells/microL. Whitish lesions with associated retinal hemorrhage are seen along side major retinal arteries. These lesions do affect vision, and patients complain of blurred vision, floaters, or visual field defects. The symptoms often are progressive and can lead to blindness. On retinal exam, CMV can be confused with other AIDS complications, including toxoplasmosis, retinal hemorrhages, and cotton wool spots. Diagnosis can be made only by an ophthalmologist. Treatment is with gancyclovir (5 mg/kg every 12 hours for 2 weeks, followed by 6 mg/kg/day maintenance therapy) or foscarnet (90 mg/kg every 12 hours) and likely will be started only in conjunction with the consultant. Fortunately, HAART has considerably decreased the incidence of CMV retinitis in HIV patients.49
Several other less common conditions also affect the eyes of AIDS patients. Besides toxoplasmosis, necrotizing retinitis can result from herpes simplex or varicella-zoster viruses. Herpes keratitis also occurs in the HIV patient, and may require IV acyclovir for treatment. Syphilitic retinitis can occur in HIV-positive patients with CD4 counts greater than 200 cells/microL, and is treated as secondary syphilis with IV penicillin. TB can affect the eye, as can as disseminated candidiasis.
Cardiac. Cardiac effects from HIV are not as rare as first thought. Besides side effects of protease inhibitors (lipodystrophy) that can greatly accelerate coronary artery disease (see section on Protease Inhibitors in Part I), HIV can directly affect the heart as well. One autopsy series found cardiac involvement in 73% of cases,50 though these were not always clinically evident. The most common findings were myocarditis (90%) and pericarditis (20%).50 Most of the data support that this is a direct effect of HIV on the heart and that the virus produces a myocarditis51 leading to dilated cardiomyopathy and congestive heart failure (CHF). Several large studies have found that up to 12% of HIV-positive patients have echocardiographic evidence of dilated cardiomyopathy, and that the incidence of severe CHF can be as high as 18% per year.51 Patients are treated in the same fashion as those with other causes of CHF. It is not yet clear the effect of newer antiretroviral therapy on HIV-associated cardiomyopathy.
Though less common, a number of other cardiac problems are seen in AIDS patients, including opportunistic infections of the myocardium (Toxoplasma gondii, Trypanosoma cruzi) or pericardium, infective endocarditis in patients with injection drug use (IDU), pericardial effusions, neoplasms, and HIV-associated pulmonary hypertension. Endocarditis has been seen in up to 34% of HIV patients with IDU.52 Pericardial effusions can have a variety of sources, including bacteria (M. tuberculosis, S. pneumoniae, S. aureus) as well as viruses (CMV), fungi (Histoplasma, Cryptococcus), or protozoa. Effusions are not always symptomatic, as one study found effusions in 22% of asymptomatic AIDS patients.53 Cancers affecting the heart usually are KS or non-Hodgkin's lymphoma, which can be clinically silent or symptomatic with CHF, dysrhythmias, or rarely tamponade.
Miscellaneous Complications. Lymphadenopathy is a well-known feature of the acute HIV syndrome, but it also can persist in some patients. It is defined by enlarged nodes (> 1 cm) in at least two non-inguinal sites for more than three months without obvious cause. Lymphadenopathy is due to follicular hyperplasia in response to increased anti-HIV antibody production. As disease advances, enlarged nodes may be a result of KS, TB, lymphoma, or systemic infection with atypical TB, toxoplasmosis, or fungal infection.
Anemia is the most common hematologic abnormality seen in HIV infection, and while mild in most cases it can require chronic blood transfusions.11 It can be a complication of systemic fungal or TB infections or can be drug-related. Zidovudine (AZT) and dapsone both are known to produce anemia. Thrombocytopenia is seen in about 10% of patients with CD4 count fewer than 400 cells/microL.11 It rarely is serious and responds well to antiretroviral treatment.
Arthralgias are seen in about 33% of HIV-positive patients, with reactive arthritis being diagnosed in about 10%.11 Symptoms worsen as CD4 counts fall. Arthritis is polyarticular, with knees, ankles, and other large joints commonly affected. NSAIDS often provide little relief, and intra-articular steroids are required.
General wasting syndrome is defined as an involuntary weight loss of more than 10% in conjunction with fever, chronic diarrhea and fatigue lasting more than 30 days in absence of other causes. It is surprisingly common and is the presenting AIDS symptom in 10% of HIV cases in the United States.11 Patients experience severe muscle wasting and, although growth hormone, androgenic steroids, and parenteral nutrition all have been tried with limited success.
HIV Infection in the Pediatric Patient
Worldwide, there are approximately 2.5 million children living with HIV younger than 13 years of age. The vast majority of these are in Africa, and most cases are the result of vertical transmission from mother to child during gestation, birth, or breast-feeding. Unfortunately, it follows that as children are becoming infected from HIV-positive mothers, many also are orphaned by the disease. Current estimates are that 10 million children in the world have been orphaned by HIV/AIDS. The World Health Organization expects this number to double to 20 million by 2010.
In the United States, about 10,000 children now are living with HIV.34 It is estimated that 1750 children are infected each year in the United States, although many of these cases likely could be prevented with proper care. Combination therapy with Cesarean section and antiretroviral maternal treatment reduces the rate of transmission during birth in the United States to less than 1%.54 Still, at present AIDS is the leading cause of death for children 1-4 years of age. Nearly 100% of these infections arise from perinatal transmission. Currently there are clusters of pediatric cases with 85% of children diagnosed in larger cities of population more than 500,000 persons.55 Further, 44% of these are in the Northeast cities and another 36% in the South.55 As these numbers rise, more ED physicians will be treating these young patients with an "adult" disease. This section will give a brief overview of the differences in HIV disease and treatment in the pediatric patient. An HIV-positive child with a fever in the ED in general can be approached as one does a neutropenic patient with a fever.
Diagnosis of HIV. Even though the majority of pediatric HIV patients will be infected in the perinatal period, clinical signs of infection will not appear for several years in at least 70% of cases. Thus the clinical exam is not likely to be of use for HIV screening in the first years of life. HIV testing in infants and children is complicated by the fact that all infants born of HIV-positive women initially will test positive for the presence of HIV antibodies. Maternal IgG antibodies readily cross the placenta and remain detectable in the infant until 6-12 months of age.55 In some cases the IgG antibodies can be present for up to 18 months, thus any testing in children younger than 18 months has to be directed at detection of the virus itself. Remember that standard ELISA and Western blot tests only test for the presence of anti-HIV antibodies. These two tests can be used for diagnosis in children older than 18 months as they are in adults.
Tests that detect HIV DNA or RNA by polymerase chain reaction (PCR) or HIV culture or tests detecting the p24 HIV antigen should be used in those children younger than 18 months. Of these, HIV culture is the gold standard for testing in young children.56 Still, two positive tests are required to make the diagnosis, as up to 7% of children later shown to be uninfected will have a positive HIV culture in the first 6 months.57 HIV screening tests are recommended in children of HIV-positive mothers at 2 days, 1-2 months, and at 4-6 months to ensure accuracy of diagnosis.
Accelerated Progression to AIDS. Another significant difference in the pediatric patient is that the progression of HIV to AIDS tends to occur much more quickly than in adults. In general, the immature neonatal immune system is unable to mount an immune response as effective as an adult's, thus accelerating the progression to AIDS. Unlike adults who experience a rapid decline in viral loads just 2-4 months after seroconversion, young children have a much slower decline. Most infected newborns maintain higher viral levels, and viral loads take two years on average to decline.55 The majority of infected newborns (80%) have a median survival time of only six years.
In a subgroup of children, roughly 25-30% of cases,58 progression is much swifter. Before HAART treatment, these children only had a median survival time of 6-9 months. It is thought that the rapid decline is because the child acquires HIV in utero, perhaps as early as the first trimester. If infected at such an early stage, it is postulated that the virus is already in the CD4 cells when they migrate to the bone marrow, spleen, and thymus during development. Thus these children never have a normal immune system and are born with systemic HIV infection.
For as yet unexplained reasons, a small number (< 5%) of children do not experience a rapid progression of disease. They maintain relatively normal CD4 counts and low viral loads for longer than 8 years. These children may be similar to the very few adults, termed nonprogressors, who maintain good CD4 counts and low viral loads for more than 10 years even without treatment. The source of this lack of progression is likely multifactorial, and based on infection with a defective virus and/or unrecognized variability in the patient's genetics.
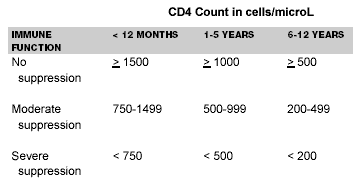
CD4 Counts. The CD4 count is naturally higher in children than in adults, requiring adjustment of which levels are considered "low." (See Table 5.) The CDC has classified CD4 counts as follows. In children younger than 12 months, a CD4 count fewer than 1500 cells/microL is abnormal and indicative of immunocompromise. A CD4 count of fewer than 750 cells/microL corresponds with an adult count of fewer than 200 cells/microL. The numbers change again for children between 1-5 years. Counts fewer than 1000 cells/microL are abnormal and levels fewer than 500 cells/microL correspond to adult levels fewer than 200 cells/microL. In children 6-12 years, any CD4 count fewer than 500 cells/microL is abnormal, and as with adults, counts fewer than 200 cells/microL correlate with severe immunosuppression. As in adults, low CD4 counts correlate with end-stage disease and are less common in infants.
|
Table 5. CDC Classification of CD4 Counts in Children < 13 years |
|
|
Clinical Effects. Besides undergoing a more rapid disease progression compared to adults, children with HIV have a different pattern of clinical effects as well. Infants born with active HIV infection initially may appear normal on physical exam but typically are of lower birth weight. In younger children (younger than 4 years) the most common findings are neurologic, with growth delays (failure to thrive), developmental delays, and microcephaly being the most frequent. These delays are seen in 50% of children by age 30 months and may be due to direct effects of the virus on the developing CNS akin to AIDS encephalopathy/dementia in adults.59
Like in adults, a low CD4 count (See Table 5) is associated with a highly increased risk of opportunistic infection. Opportunistic infections are common in children with HIV as well as adults, but recurrent serious bacterial illnesses occur much more frequently in children. Nearly 20% of AIDS-defining infections are bacterial in nature, and bacteremia, sepsis, and pneumonia make up more than 50% of these infections.55 Other dangerous infections like meningitis, urinary tract infections, deep abscesses, osteomyelitis, and septic arthritis are less common. Recurrent otitis media, sinusitis, cellulitis, and superficial abscesses occur frequently and may be atypical in presentation. Many of these infections are from encapsulated organisms like Streptococcus pneumoniae and Salmonella. Other common bacteria include H. influenzae type b, Staphylococcus, Enterococcus, and Pseudomonas aeruginosa. TB also is common in children with HIV, is more severe than in adults, and leads to death more often than in HIV-negative children.
PCP occurs in the child with HIV as it does in adults, but is more aggressive than in adults. Mortality from PCP in an infant is as high as 33%.56 PCP in a child represents a primary infection rather than reactivation disease as it does in adults. Thus the child lacks prior immunity and is prone to a more fulminant course. PCP occurs at younger ages with the peak age at onset of disease being only 3-6 months.60 Most children with PCP have lower CD4 counts as do adults, but still 30% of cases occur in children with CD4 counts greater than 1500 cells/microL.61 Symptoms are similar to adults with fever, tachypnea, and hypoxia. Lung exam may show wheezes or rhonchi. Chest films are normal or show interstitial or alveolar infiltrates. Like adults, bronchoalveolar lavage or lung biopsy is often required for final diagnosis, and treatment is with trimethoprim-sulfamethoxazole (15-20 mg/kg/24 hr of TMP and 75-100 mg/kg/24 hr of SMX q 6 hr IV) with adjunctive corticosteroids.
Lymphocytic interstitial pneumonitis (LIP) is a very common disorder in the pediatric AIDS patient, but is not seen frequently in the adult. It is theorized to be the result of primary infection with the Epstein-Barr virus. It is seen in nearly 25% of cases.55 It results from alveolar capillary blockage arising from chronic lymphoid hyperplasia in the bronchial epithelium. Patients present with tachypnea, cough, and moderate hypoxia but may not have fever. The chest film shows a characteristic reticulonodular pattern. Treatment is with corticosteroids, which usually results in clinical improvement.
Several conditions are much more common in adults than children with HIV. Crytopococcoisis and toxoplasma are relatively rare in children. HIV-associated cancers (KS, lymphomas) also occur in children with HIV, but again are seen infrequently compared to adults with HIV. On the other hand, oral candidiasis is common in pediatric patients, as are viral infections from the herpes group. Cardiomyopathies can be more common in children with advanced cases of AIDS. Nephropathy can cause renal complications in children as well, but do not seem to progress to renal failure as often as in the adult.
Immunizations. Although emergency physicians generally do not provide immunizations other that tetanus and rabies, they often see patients who are behind on their shots. It is valuable to know of any changes to the normal immunization schedule for the child with HIV. There is only one vaccine that is contraindicated in HIV-positive children. The oral polio vaccine is not recommended as live or attenuated vaccines carry a higher risk in children with HIV. However, MMR vaccine is given, as the mortality rate for measles in HIV-positive children is very high. Otherwise, the immunization schedule is similar to the HIV-negative child. Three vaccines that may be optional for HIV-negative children should be given in every child with HIV: influenza, pneumococcus, and varicella. These vaccines are given as these diseases are much riskier for the HIV-positive child.
Antiretroviral Treatment. While the ED physician will not be initiating these medications for children in the ED, it is useful to be aware that children are treated with antiretrovirals in a similar fashion to adults. Combination therapy (HAART) is the cornerstone of treatment, and treatment is started with AIDS-defining illness or low CD4 count for age. (See Table 5.) All the adult drug classes (NRTIs, NNRTIs, and PIs) can be used in children.
Vaccines and Future Treatments
Although more than 20 years have passed since the virus responsible for AIDS was isolated, there have been only two potential HIV vaccines to make it to phase III clinical trials, and in neither study was HIV transmission prevented.62
For a vaccine to be effective against HIV, several lofty requirements must be met.63,64 First, the vaccine must provide broad enough coverage to protect against all the HIV subtypes; currently there are 3 main groups of HIV-1 and 9 subtypes of just the main group. The rapid rate of mutation seen in HIV ensures that this number only will increase. Secondly, the vaccine will have to stimulate the immune system to evoke a response that is better than the native response induced by infection with the virus. The virus likely will have to be stopped at the mucosal level to prevent infection, thus strong cellular responses will be needed. Lastly, the vaccine will have to be affordable and easily mass-produced to be used in third world countries where the bulk of HIV infections occur.
Further study also is being conducted to find a way to eliminate the HIV reservoir(s) in the body. One of the main reasons antibodies to HIV or HAART medications cannot completely clear HIV from the body is that it is harbored in latent cells, for example memory T-cells.65 There are some examples of new combinations of medications that show some promise in small studies, such as adding enfuvirtide and valproic acid to HAART regimens,66 but the problem of total eradication of HIV reservoirs remains unsolved.
Finally, there are some recent efforts on prophylaxis among high-risk populations. In the past, efforts at prevention of HIV infection have been only through education and counseling especially of high-risk groups.67 The CDC currently is conducting a double-blind placebo-controlled study of the ability of daily oral tenofovir to prevent HIV infection in certain high-risk populations (IDUs, sex trade workers, homosexual men) in Thailand, Russia, Africa, and the United States.68 Studies of this nature have not been done before, and only animal data (SIV prevention in monkeys) show potential effectiveness.69 These studies are expected to last 2-4 years, and though 100% prevention is not predicted, perhaps significant reduction in transmission can be achieved.
References
1. Rothman RE, Marco CA, Kelen GD. HIV infection and AIDS. In: Tintinalli JE, ed. Emergency Medicine: A Comprehensive Study Guide, 6th ed. New York: McGraw-Hill; 2004: 925-936.
2. Schacker TW, Hughes JP, Shea T, et al. Biological and virologic characteristics of primary HIV infection. Ann Intern Med 1998;128:613-620.
3. Kahn JO, Walker BD. Acute human immunodeficiency virus type 1 infection. N Engl J Med 1998;339:33-39.
6. Perlmutter BL, Glaser JB, Oyugi SO. How to recognize and treat acute HIV syndrome. Am Fam Physician 1999;60:535-542.
4. Oncology (Williston Park). AIDS among persons aged > or = 50 years—United States, 1991-1996. MMWR Morb Mortal Wkly Rep 1998;47:21-27.
5. Lyles RH, Munoz A, Yamashita TE, et al. Natural history of human immunodeficiency virus type 1 viremia after seroconversion and proximal to AIDS in a large cohort of homosexual men. Multicenter AIDS Cohort Study. J Infect Dis 2000;181:872-880.
7. Steir E. Cervical neoplasia and the HIV-infected patient. Hematol Oncol Clin North Am 2003;17:873-887.
8. Shukla NB, Poles MA. Hepatitis B virus infection: Co-infection with hepatitis C virus, hepatitis D virus, and human immunodeficiency virus. Clin Liver Dis 2004;8:445-460.
9. Haukoos JS. Emergency department triage of patients infected with HIV. Acad Emerg Med 2002;9:880-888.
10. Huang L. Pulmonary manifestations of HIV/AIDS. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed. Philadelphia: W.B. Saunders; 2003: 2158-2169.
11. Longo DL, Fauci AS. Infections due to HIV and other human retroviruses. In: Kasper DL, et al. eds. Harrison's Principles of Internal Medicine, 16th ed. New York: McGraw-Hill; 2005: 1071-1139.
12. Rothman RE, Marco CA, Kelen GD. Human immunodeficiency virus infection and related disorders. In: Harwood-Nuss A, Wolfson AB eds. The Clinical Practice of Emergency Medicine, 3rd ed. Philadelphia: Lippincott Williams & Wilkins; 2001:926-934.
13. Stringer JR, Beard CB, Miller RF, et al. A new name (Pneumocystis jiroveci) for Pneumocystis from humans. Emerg Infect Dis 2002;8:891-896.
14. Wolff AJ, O'Donnell AE. Pulmonary manifestations of HIV infection in the era of HAART. Chest 2001;120:1888-1893.
15. Khouli H, Afrasiabi A, Shibili M, et al. Outcome of critically ill human immunodeficiency virus-infected patients in the era of highly active antiretroviral therapy. J Intensive Care Med 2005;20:327-333.
16. Huang L, Stannell JD. AIDS and the lung. Med Clin North Am 1996;80:775-801.
17. Quist J, Hill AR. Serum lactate dehydrogenase (LDH) in Pneumocystis carinii pneumonia, tuberculosis, and bacterial pneumonia. Chest 1995;108:415-418.
18. Rothman RE, Marco CA, Kelen GD. AIDS and HIV. In: Marx, JA, ed. Rosen's Emergency Medicine: Concepts and Clinical Practice, 6th ed. St. Louis: Mosby; 2005:1843-1860.
19. Pareja JG, Garland R, Koziel H. Use of adjunctive corticosteroids in severe adult non-HIV Pneumocystis carinii pneumonia. Chest 1998;113:1215-1224.
20. Schneider E, Moore M, Castro KG. Epidemiology of tuberculosis in the United States. Clin Chest Med 2005;26:183-195.
21. Eldin BR, Tokars JI, Grieco MH, et al. An outbreak of multi-drug resistant TB among hospitalized patients with AIDS. N Engl J Med 1992;326:1514-1521.
22. Centers for Disease Control and Prevention. Updated guidelines for the use of rifabutin or rifampin for the treatment and prevention of TB among HIV-infected patients taking PIs or NNRTIs. MMWR 2000;49:185-189.
23. McArthur JC. Neurological complications of HIV infection. Lancet Neurol 2005;4:543-555.
24. Price RW. Neurologic complications of HIV-1 infection. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed. Philadelphia: W.B. Saunders; 2003:2153-2158
25. Lanska DJ. Epidemiology of human immunodeficiency virus infection and associated neurologic illness. Semin Neurol 1999;19:105-111.
26. McArthur JC. HIV dementia: An evolving disease. J Neuroimmunol 2004;157:3-10.
27. Montoya JG, Rosso F. Diagnosis and management of toxoplasmosis. Clin Perinatol 2005;32:705-726.
28. Barber CJ. Clinical utility of cranial CT in HIV positive and AIDS patients with neurological disease. Clinical Radiology 1990;42:164-165.
29. Perfect JR, Casadevall A. Cryptococcosis. Infect Dis Clin N Am 2002;16:837-874.
30. Pinner RW, Hajjeh RA, Powderly WG. Prospects for preventing cryptococcosis in persons with human immunodeficiency virus. Clin Infect Dis 1995;Suppl 1:S103-S107.
31. Parekh S, Ratech H, Sparano JA. Human immunodeficiency virus-associated lymphoma. Clin Adv Hematol Oncol 2003;1:295-301.
32. Izzedine H, Launay-Vacher V, Deray G. Antiviral drug-induced nephrotoxicity. Am J Kidney Dis 2005;45:804-817.
33. Saag M. Renal, cardiac, endocrine, and rheumatologic manifestations of HIV infection. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed. W.B. Saunders; 2003: 2177-2184.
34. Centers for Disease Control and Prevention. HIV/AIDS Surveillance Report, 2004. Vol 16. Atlanta: US Dept. of Health and Human Services, CDC; 2005. Available at: http://www.cdc.gov/hiv/stats/hasrlink.htm.
35. Eggers PW, Kimmel PL. Is there an epidemic of HIV Infection in the US ESRD program? J Am Soc Nephrol 2004;15:2477-2485.
36. Rao TK. Human immunodeficiency virus infection and renal failure. Infect Dis Clin North Am 2001;15:833-850.
37. Schwartz EJ, Szczech LA, Ross MJ, et al. Highly active antiretroviral therapy and the epidemic of HIV+ end-stage renal disease. J Am Soc Nephrol 2005;16:2412-2420.
38. Slaven EM, Lopez F, Weintraub SL, et al. The AIDS patient with abdominal pain: A new challenge for the emergency physician. Emerg Med Clin N Am 2003;21:987-1015.
39. Bartlett JG. Gastrointestinal manifestations of AIDS. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed. Philadelphia: W.B. Saunders; 2003: 2168-2170.
40. Davidson T, Allen-Mersh TG, Miles AJG. Emergency laparotomy in patients with AIDS. Br J Surg 1991;78:924-926.
41. Hymes KB, Cheung T, Greene JB, et al. Kaposi's sarcoma in homosexual men—a report of eight cases. Lancet 1981;19:598-600.
42. Bubman D. Pathogenesis of Kaposi's sarcoma. Hematol Oncl Clin North Am 2003;17:717-745.
43. Cattelan AM, Calabro ML, De Rossi A, et al. Long-term clinical outcome of AIDS-related Kaposi's sarcoma during highly active antiretroviral therapy. Int J Oncol 2005;27:779-785.
44. Duerr A, Kieke B, Warren D, et al. Human papillomavirus-associated cervical cytologic abnormalities among women with or at risk of infection with human immunodeficiency virus. Am J Obstet Gynecol 2001;184:584-590.
45. Wilkins K, Doley JC, Turner R, et al. Approach to the treatment of cutaneous malignancy in HIV-infected patients. Dermatol Ther 2005;18:77-86.
46. Berger T. Skin disease in patients with HIV infection. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed. W.B. Saunders; 2003: 2171-2172.
47. Vella S, Floridia M. Antiviral therapy. In: Cohen J, Powderly WG, eds. Infectious Diseases, 2nd ed. C.V. Mosby; 2003: 1387-1398.
48. Brinkman K, Smeitink JA, Romijn JA, et al. Mitochondrial toxicity induced by nucleoside-analogue reverse-transcriptase inhibitors is a key factor in the pathogenesis of antiretroviral-therapy-related lipodystrophy. Lancet 1999;354:1112-1115.
49. Roels P. Ocular manifestations of AIDS: New considerations for patients using highly active anti-retroviral therapy (HAART). Optometry 2004;75:624-628.
50. Yunis N, Stone VE. Cardiac manifestations of HIV/AIDS: A review of disease spectrum and clinical management. J Acquir Immune Defic Syndr 1998;18:145-154.
51. Barbaro G, Di Lorenzo G, Grisorio B, et al. Incidence of dilated cardiomyopathy and detection of HIV in myocardial cells of HIV-positive patients. N Engl J Med 1998;339:1093-1099.
52. Rerkpattanapipat P, Wongpraparut N, Jacobs LE, et al. Cardiac manifestations of acquired immunodeficiency syndrome. Arch Intern Med 2000;160:602-608.
53. Heidenreich PA, Eisenberg MJ, Kee LL, et al. Pericardial effusion in AIDS. Incidence and survival. Circulation 1995;92:3229-3234.
54. McGowan JP, Shah SS. Prevention of perinatal HIV transmission during pregnancy. J Antibicrob Chemother 2000;46:665-668.
55. Yogev R, Chadwick EG. Acquired immunodeficiency syndrome (HIV). In: Behrman RE, ed. Nelson Texbook of Pediatrics, 17th ed. W.B. Saunders; 2004: 1109-1121.
56. Havens PL. Pediatric HIV infection. In: Cohen J, Powderly WG, eds. Infectious Diseases, 2nd ed. C.V. Mosby; 2003: 1343-1356.
57. Newell ML, Dunn D, Maria AD, et al. Detection of virus in vertically exposed HIV-antibody-negative children. Lancet 1996;347:213-215.
58. Auger I Thomas P, DeGruittola V, et al. Incubation periods for pediatric AIDS patients. Nature 1988;336:575-577.
59. Cooper ER, Hanson C, Diaz C, et al. Encephalopathy and progression of human immunodeficiency virus disease in a cohort of children with perinatally acquired human immunodeficiency virus infection. J Pediatr 1998;132:808-812.
60. Simonds RJ, Oxtoby MJ, Caldwell MB, et al. Pneumocystis carinii pneumonia among US children with perinatally acquired HIV infection. JAMA 1993;270:470-473.
61. European Collaborative Study Group: Dunn D, Newell ML, et al. CD4 T cell count as predictor of Pneumocystis carinii pneumonia in children born to mothers infected with HIV. Br Med J 1994;308:437-440.
62. Slobod KS. HIV vaccines: Brief review and discussion of future directions. Expert Rev Vaccines 2005;4:305-313.
63. Joseph J, Etcheverry F, Alcami J, et al. A safe, effective and affordable HIV vaccine—an urgent global need. AIDS Rev 2005;7:131-138.
64. Garber DA, Silvestri G, Feinberg MB. Prospects for an AIDS vaccine: Three big questions, no easy answers. Lancet Infect Dis 2004;4:397-413.
65. Marcello A. Latency: The hidden HIV-1 challenge. Retrovirology 2006;3:7.
66. Lehrman G, Houge IB, Palmer S, et al. Depletion of latent HIV-1 infection in vivo: a proof-of-concept study. Lancet 2005;366:549-555.
67. Khalsa AM. Preventive counseling, screening, and therapy for the patient with newly diagnosed HIV infection. Am Fam Physician 2006;73:271-280.
68. Greenberg AE, Tappero J, Choopanya K, et al. CDC international HIV prevention research activities among injection drug users in Thailand and Russia. J Urban Health 2005;82(Suppl):24-33.
69. Bonn D. Tenofovir: A pill to prevent HIV? Lancet Infect Dis 2005 5:78.
HIV infection can be divided into three stages: the acute seroconversion reaction, latent phase, and active AIDS.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.