Tackling The MRSA Epidemic: How Should You Change Your Practice?
Tackling The MRSA Epidemic: How Should You Change Your Practice?
Author: Laura Pimentel, MD, Assistant Professor, University of Maryland School of Medicine, Chairman, Department of Emergency Medicine, Mercy Medical Center, Baltimore, Maryland
Peer Reviewer: Michael L. Coates, MD, MS, Professor and Chair, Family and Community Medicine, Wake Forest University School of Medicine, Winston Salem, North Carolina
Introduction
Community-acquired methicillin-resistant Staphylococcus aureus (caMRSA) is a microbe that has become increasingly prevalent and clinically significant to acute care practitioners worldwide. The genetic mechanism that confers methicillin resistance is associated with virulence factors that affect the clinical presentation and treatment of infections caused by the bacterium. In light of the rapid penetration of caMRSA into the population, physicians are faced with management dilemmas requiring difficult decisions at the point of care.
This literature survey provides an overview of recent research and case series' describing the exponential increase in caMRSA infections and the most successful management strategies. One review article provides a description of the microbiology and clinical impact of the organism. Data on management of skin and soft tissue infections (SSTI), including the importance of drainage and debridement, are reviewed. The role of adjunctive antibiotic treatment is addressed, including the most successful and cost effective regimens. The pediatric experience with caMRSA at one children's hospital is described. The survey concludes with a provocative report of four cases of necrotizing pneumonia contracted by generally healthy adults following viral respiratory infections.
Clinical and Genetic Characteristics
Source: Zetola N et al. Community-acquired methicillin-resistant Staphylococcus aureus: An emerging threat, Lancet Infect Dis 2005;5:275-86.
Zetola and colleagues from Johns Hopkins University systematically reviewed the literature on clinical and genetic characteristics of caMRSA. They emphasized those factors conferring antibiotic resistance and virulence in the context of clinical presentation and management.
The mechanism of methicillin resistance is production of altered penicillin-binding protein (PBP) known as PBP2a. PBP2a decreases the affinity of the staphylococcus bacillus for beta-lactam antibiotics. A mobile genetic element, the staphylococcal cassette chromosome (SCC), carries the mecA gene, which encodes PBP2a. The acquisition of this SCC by methicillin-sensitive S aureus (MSSA) is the mechanism to which most authorities attribute the rise of caMRSA.
Multiple virulence factors may be produced by S aureus and are associated with specific clinical syndromes. The SCC has introduced new virulence factors along with methicillin resistance. The most well known is the Panton-Valentine leukocidin (PVL) gene, which confers tissue necrosis properties to the bacterium. Another described virulence factor is production of bacteriocins, naturally synthesized antibiotics toxic to microbes closely related to caMRSA. These confer a competitive advantage to the producing strain. Others potentiate adherence to epithelial cells, adherence to collagen, and increase tolerance to salt.
Furunculosis, causing severe necrosis and abscess formation, is the most common clinical manifestation of caMRSA. Impetigo, bullous impetigo, and scalded skin syndrome have been attributed to a clonal group of caMRSA that produces exfoliative toxins in addition to the PVL genes. Fulminant necrotizing pneumonia is strongly associated with caMRSA. Pulmonary abscess formation occurs along with septic shock; respiratory failure is common and mortality is high. Infection associated with influenza is recognized, though not unique to MRSA. Toxin-mediated syndromes cause a toxic shock syndrome-like illness.
The antibiotic management of suspected staphylococcal infections should be guided by the prevalence of MRSA in the community. In areas with documented caMRSA, vancomycin should be empirically administered to patients with life-threatening infections. Non-life threatening caMRSA infections remain susceptible to some non-beta-lactam antibiotics; clindamycin, co-trimoxazole, linezolid, and minocycline may be empirically prescribed for skin infections in adults. Clindamycin has the potential for inducible resistance.
Non-antibiotic management is particularly important in caMRSA. Adequate drainage is definitive management of abscessed skin infections. Intravenous immuno-globulin therapy may be an appropriate adjunct to patients with life-threatening toxin-mediated illness. Preventive measures should be instituted in high-risk populations (e.g.,intravenous drug users, nursing home residents, dialysis patients). Eradication of caMRSA colonization is controversial but may be attempted with topical mupirocin or systemic trimethoprim-sulfametho-xazole (TMP-SMX).
Commentary
Zetola and colleagues have made an important contribution to the literature with this review of caMRSA. The content was systematically and comprehensively culled from the relevant English and Spanish literature from 1966 forward. It offers clinicians a readable summary of the clinically relevant features of this emerging public health problem. Their description of the genetic basis of methicillin resistance in community-acquired disease and why this less resistant strain has more successfully propagated in the community than the more highly resistant nosocomial strain is particularly interesting. Strength of the paper is the review of the molecular basis of virulence factors. The authors separated virulence from antibiotic resistance but further explain how they relate through carriage on the SCC.
Two tables are well constructed. One relates virulence factors to clinical syndromes and characteristics. The second provides first- and second-line antibiotic recommendations for patients based on epidemiologic characteristics and severity of clinical presentation. High quality images show a characteristic presentation of furunculosis and a series of computed tomography images of necrotizing pneumonia.
The pathophysiologic framework of caMRSA is well constructed. Descriptions of the clinical syndromes may be applied to primary care or hospital-based emergency medicine practice. High-risk patient groups are identified. The antibiotic and non-antibiotic management strategies are sound.
MRSA in an Outpatient Office
Source: Iyer S, Jones DH. Community-acquired methicillin-resistant Staphylococcus aureus skin infection: A retrospective analysis of clinical presentation and treatment of a local outbreak. J Am Acad Dermatol 2004;50:854-8.
Iyer and Jones described their clinical experience with caMRSA skin infections between December 2000 and March 2003. The case review is a retrospective analysis of all patients from their private Los Angeles office-based dermatology practice from whom cultures were positive for MRSA. Demographics, HIV status, infection type, antibiotic sensitivities, and response to treatment were described. The authors described changes in their antibiotic selections during the study period as the result of sensitivity data and clinical experience.
During the 28 months of the review, 39 patients (26.7%) were diagnosed with caMRSA infections, while 107 patients (73.3%) were diagnosed with MSSA infections. Of the 39 patients, 30 (97.4%) were male, 20 (51.3%) were HIV-positive, and 32 (69.6%) were diagnosed with cutaneous abscesses. Remaining diagnoses were folliculitis/furuncle, paronychia, impetigo, or cellulitis/wound infection. The caMRSA abscesses were characterized by significant inflammation and necrosis. Culture and sensitivity testing demonstrated susceptibility to gentamycin and vancomycin in all cases. Of the 39 caMRSA cultures, 38 (97.4%) were sensitive to TMP-SMX.
Iyer and Jones treated 30 of 32 abscesses with incision and drainage followed by antibiotics for 2 to 3 weeks. The other diagnoses were treated with antibiotics alone. Moxifloxacin was used empirically in 16 cases but successful in nine. The most successful antibiotic treatment regimens were linezolid (11 patients) and TMP-SMX combined with rifampin (6 patients). Success was 100%. Topical mupirocin alone successfully treated two cases of impetigo and one infected gluteal cleft.
The authors identified risk factors for caMRSA: use of contaminated instruments (e.g., hair clippers) and shared objects, systemic antibiotic use within 1 year, HIV disease, and recent hospitalization. Empiric antibiotic coverage shifted to linezolid after experiencing treatment failures with fluoroquinolones and TMP-SMX alone. Iyer and Jones concluded with these recommendations: nares treatment with topical mupirocin to eliminate carriage of caMRSA; systemic antibiotic coverage with TMP-SMX alone or in combination with rifampin, linezolid, or intravenous vancomycin.
Commentary
This retrospective chart review in one outpatient practice is of value to the practicing clinician because of its description and quantification of caMRSA infections. The culture and sensitivity data add to and generally reflect the experience of other researchers and clinicians encountering cutaneous caMRSA. The greatest strength of this paper was the authors' description of the real-world management challenges faced daily by clinicians when few evidence-based guidelines exist.
The paper has a number of weaknesses. The first is a low number of subjects (39) making extrapolations from the data and its subsets tentative. The authors noted success in 100% of patients treated with two antibiotic regimens. One was only initiated in 6 patients and the other in 11. Practice should not change on the basis of such numbers.
This chart review is purely descriptive. There was no protocol or control group. One can draw no conclusions about the effect of antibiotic treatment on patients with cutaneous abscesses because there was no control group treated with incision and drainage alone. The authors listed factors that they interpreted as risks or predispositions to caMRSA. Because no data were provided about the prevalence of these factors in patients with MSSA infections or in their general patient population, the reader cannot assess the correlation with MRSA. Strikingly, 97.4% of their caMRSA patients were male. With no information about the gender composition of their patient population, this statistic is meaningless. Finally, the authors did not address cost in their data or discussion. They reported and recommended empiric use of linezolid without noting its strikingly high price relative to alternatives.
In conclusion, this paper is an interesting description of one practice's experience with caMRSA. It adds to information about prevalence relative to MSSA and antibiotic resistance. Low numbers and absence of a clinical protocol limit further conclusions from the data.
MRSA Skin Infection
Source: Moran GJ et al. Methicillin-resistant Staphylococcus aureus in community-acquired skin infections. Emerg Infect Dis 2005;11(6):928-30.
Moran and colleagues trended the proportion of patients with purulent community-acquired SSTI culture positive for caMRSA from August 2001 through March 2004. The study location was a county-owned inner city emergency department in Los Angeles. Data from several antimicrobial clinical trials for SSTI were combined for this investigation. Of the 96 subjects, 24 (25%) were outpatients. The remaining patients were admitted to the hospital. Patients ranged in age from 20 to 60 years, and 77% were male.
From August 2001 through December 2002, 14 (29%) of 49 infections isolated caMRSA. From January 2003 through March 2004, 30 (64%) of 47 infections isolated the organism. No patient had traditionally recognized predisposing factors. Twenty percent of caMRSA patients had recently injected illegal drugs, and 20% were homeless; however, these factors were not statistically significantly different from those patients from whom other organisms were isolated. No studied epidemiologic characteristics differentiated patients with caMRSA from those with other causative pathogens. Antibiotic sensitivity data demonstrated 100% sensitivity to TMP-SMX, 98% sensitivity to clindamycin, 98% sensitivity to rifampin, and 82% sensitivity to tetracycline.
The authors concluded that the prevalence of purulent SSTI caused by caMRSA is increasing; the organism is now the most common cause at their institution. They noted a similar trend in Europe and other geographic regions of the United States. Important treatment implications are inferred. The authors stressed the importance of adequate drainage and debridement. In geographic areas with high prevalence of caMRSA, appropriate empiric antibiotic treatment includes clindamycin or the combination of TMP-SMX and rifampin.
Commentary
The data reported by Moran and colleagues suggest a steep upslope in the prevalence of skin abscesses caused by caMRSA.
Several caveats must be noted before extrapolating their conclusions to other settings. The first is that the number of subjects was small (96 patients enrolled over 32 months). This was a convenience sample of patients enrolled in four different antibiotic studies with differing eligibility criteria. One of the studies enrolled only uncomplicated infections treated as outpatients. Three other studies enrolled only complicated infections requiring admission. The first group is entirely contained within the first half of this study interval during which the percentage of caMRSA was significantly lower.
Analyzing the subgroups of outpatients vs inpatients during the first half of the study actually shows a higher proportion of caMRSA isolates in the outpatients, 8 (33.3%) of 24, vs 6 (24%) of 25 inpatients. This finding lends credibility to the authors' conclusion that the prevalence of caMRSA increased over time. One must use caution in extrapolating results from this study to other geographic areas and patient populations.
The authors noted that their results cannot be generalized to the entire group of SSTIs, only those with purulent material available for culture. It is not clear whether MRSA is increasingly the etiologic pathogen in patients with cellulitis without necrosis. It may well be that it is not; the authors noted the high proportion of caMRSA strains that carry the Panton-Valentine leukocidin factor associated with necrosis.
The antibiotic susceptibility data reported in this trial are consistent with other reports of caMRSA. Treatment recommendations correspond to the data. The importance of adequate surgical drainage and debridement is emphasized.
Community-Acquired MRSA
Source: Fridkin SK et al. Methicillin-resistant Staphylococcus aureus disease in three communities. N Engl J Med 2005:352:1436-44.
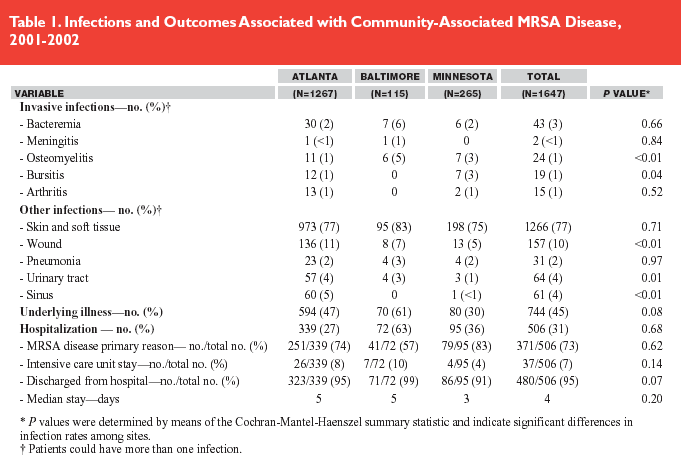
In this prospective population and sentinel surveillance study from three discreet geographic areas, Fridkin and colleagues documented the incidence of caMRSA, described epidemiological features, noted clinical characteristics, and reported antibiotic susceptibility data from 2001 and 2002. Population-based surveillance data were collected in Baltimore and Atlanta; hospital laboratory-based surveillance data were obtained from 12 hospitals in Minnesota. Medical records were reviewed, and subjects were interviewed to distinguish hospital-associated MRSA from caMRSA. The authors defined a community-associated infection as an MRSA isolate coming from a subject without established risk factors for MRSA; these included hospitalization, surgery, dialysis, or residence in a long-term care facility within one year of infection or the presence of a permanent indwelling catheter or percutaneous medical device.
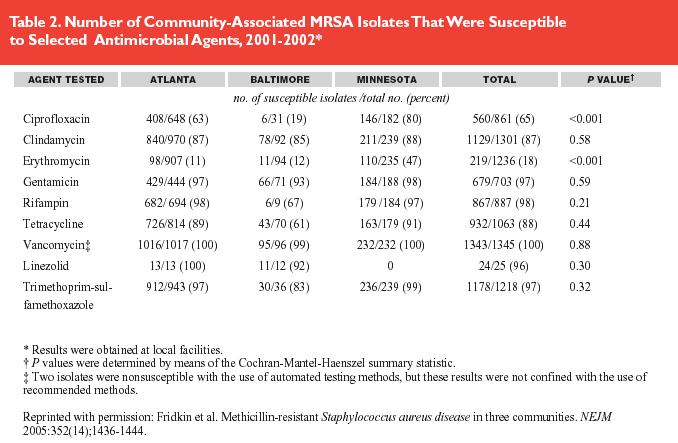
Of 12,533 MRSA infections isolated from the three locations, 17% were classified as probable or confirmed caMRSA. Prevalences ranged from 8% in Baltimore to 20% in Atlanta. The relative risk was 1.51 for patients younger than 2 years relative to older patients. Racial disparity indicating a relative risk of 2.74 for black patients relative to white was found in Atlanta but not in Baltimore. Seventy-seven percent of infections were manifested by skin and soft-tissue disease; 6% of subjects had invasive disease defined as a positive culture from a normally sterile site; infections included bacteremia, meningitis, osteomyelitis, bursitis, and arthritis. Pneumonia accounted for 2%. Antibiotic susceptibility data indicated greater than 95% sensitivity to gentamicin, rifampin, vancomycin, linezolid, and TMP-SMX. Seventy-three percent of patients received empiric antibiotic coverage to which the organism was not susceptible; however no adverse consequences were identified.
The authors concluded by exhorting clinicians to consider caMRSA as a potential pathogen, adequately drain and culture purulent material, and choose empirical antibiotics likely to cover caMRSA.
 |
 |
Commentary
This prospective surveillance study has a number of strengths that confer credibility to the results and conclusions: high numbers, rigorous methodology, and multiple data sources over three discrete geographic regions. The definitions of hospital associated and community-acquired infections are clear and specific. The article's Tables 1 and 2 collated the results such that they can be examined by region and in aggregate. The spectrum of severity of caMRSA infection is similar across the three regions with similar distributions of SSTI, invasive infections, and pneumonia.
Conversely, antibiotic susceptibility shows regional differences with Baltimore demonstrating generally lower percentages to the various antibiotics than Atlanta or Minnesota. The latter result suggests that clinicians should understand the local experience with caMRSA with respect to prevalence and antibiotic susceptibility to guide empiric treatment.
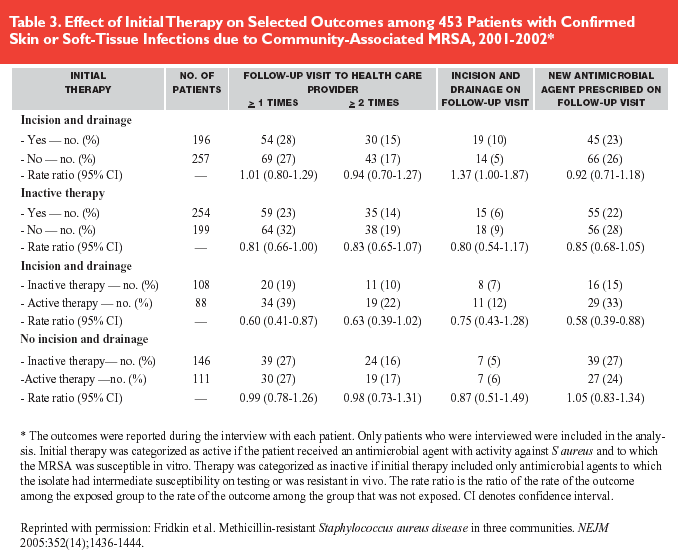
The clinical characteristics of caMRSA infection are well defined. The authors were unable to identify adverse outcomes resulting from empiric treatment with antibiotics resistant to the infecting organism. They underscored the importance of surgical drainage of skin abscesses when feasible.
The authors identified limitations to their results. Importantly, data collection was limited to cases where a culture was obtained. Their report may have underestimated incidence and prevalence because cases where patients were treated empirically with no culture would not have been included. Because their methodology required patient interviews, only 41% of eligible patients were included in the outcome analysis.
 |
 |
TMP-SMX for MRSA
Source: Grim SA et al. Trimethoprim-sulfamethoxazole as a viable treatment option for infections caused by methicillin-resistant Staphylococcus aureus. Pharmacotherapy 2005;25:253-264.
Grim and colleagues performed a literature review of studies reporting the efficacy of TMP-SMX for MRSA infections. The authors were particularly focused on the question of whether this antibiotic combination may be generally recommended as a cost effective, well-tolerated oral agent for empiric treatment. They reviewed articles identified by a MEDLINE search of pertinent keywords from January 1966 through December 2003 and abstracts from infectious disease meetings.
Mechanisms of MRSA resistance to TMP-SMX are not completely understood but are thought to be chromosomally or plasmid mediated. They are probably distinct from multidrug resistance mechanisms, but several reports suggest a relationship. Resistance rates of MRSA to TMP-SMX vary considerably in different geographic regions ranging from 0 to 74% in the United States. Institutions with large numbers of HIV patients treated with TMP-SMX for pneumocystis prophylaxis have particularly high rates. Clonal outbreaks of resistant MRSA have been described, with the Brazilian clone conferring 100% resistance to TMP-SMX.
Grim and colleagues reported several small studies and case reports of patients with documented MRSA infections treated or prophylaxed with TMP-SMX. The data suggest efficacy; the only randomized, double-blind trial found a 100% cure rate in hospitalized intravenous drug users with MRSA but significant resistance in patients with MSSA.1
The authors concluded that the available data supports the use of TMP-SMX for oral treatment of caMRSA but caution that vancomycin should be used for serious infections. Clinicians should be knowledgeable of local patterns of resistance and potential adverse drug effects and interactions before prescribing the combination drug. Finally, they highlighted the need for a randomized controlled trial comparing TMP-SMX to linezolid.
Commentary
The importance of this review is its focus on the value of TMP-SMX in the treatment of MRSA infections. The antibiotic combination is a potentially desirable agent for a number of reasons: low cost, oral and parenteral formulations, and an acceptable side-effect profile. The authors reviewed clinical, pharmacological, and microbiological data profiling the relationship between the antibiotic and MRSA.
The weakness of this review is the methodology. It is very poorly described, which leaves the reader unclear about inclusion and exclusion criteria for papers and abstracts reviewed. One cannot discern if the methodology was systematic or even the types of papers selected. The references suggest a range of source material including pharmaceutical package inserts, letters to the editor, and case reports.
The authors assembled the material in a readable fashion and were careful to focus on providing information of clinical relevance to practicing physicians. The most important portion of this review is the conclusion that the available data indicated that TMP-SMX is effective in the treatment of caMRSA infections. This is supported by many other recent and contemporaneous studies. Clinicians should understand that the drug is most useful for oral treatment of non-life-threatening infections managed in an outpatient setting. The end of the paper includes a brief but useful review of adverse reactions and drug interactions with appropriate cautions on patient selection. Finally, use of this common, cheap, and readily available drug may forestall development of resistance to linezolid and other newer antistaphylococcal antibiotics.
Pediatric Patients with MRSA
Source: Lee MC et al. Management and outcome of children with skin and soft tissue abscesses caused by community-acquired methicillin-resistant Staphylococcus aureus. Pediatr Infect Dis J 2004;23:123-7.
The goal of Lee and colleagues' prospective observational study was to identify the optimal management strategy for children with soft tissue abscesses caused by caMRSA. They specifically looked at the role of supplemental antibiotic therapy in patients primarily treated with incision and drainage. The patient population included children presenting to the emergency department or acute care clinic of a single institution, Children's Medical Center, Dallas, TX; eligible patients were treated for skin and soft-tissue abscesses that were culture positive for MRSA. Demographic and clinical data including management strategy were collected for the initial visit. Follow-up information was collected 1 to 6 days later and again about 1 week after the first follow-up contact.
Sixty-nine subjects were studied. All had antibiotics prescribed; 62 (90%) were treated with a drug to which the cultured pathogen was resistant, 5 (7%) were treated with an effective drug, and 2 were noncompliant. All of the isolates were susceptible to rifampin, TMP-SMX, gentamicin, and vancomycin; 88% were susceptible to clindamycin. Four (6%) patients initially treated with an ineffective antibiotic required hospital admission on the first follow-up visit. All hospitalizations were uncomplicated. The only identified predictor of hospitalization was the size of the initial lesion: an infected area greater than 5 cm in diameter was associated with a 33% chance of outpatient management failure. The susceptibility of the pathogen to the prescribed antibiotic was not associated with the success of outpatient treatment.
The authors concluded that children with skin abscesses less than 5 cm in diameter caused by caMRSA may be treated with incision and drainage without supplemental antibiotics.
Commentary
The importance of this study is its focus on identification of optimal management strategies for children with skin abscesses caused by caMRSA. There are advantages and disadvantages to the prospective observational methodology. An important advantage is identification of current common management practices; in this case, the authors identified 100% prescription of adjuvant antibiotic use in children with caMRSA abscesses. The main disadvantage is absence of the rigor of a clinical trial. In this study, management variation occurred with respect to use of incision and drainage, abscess packing, intravenous antibiotic administration on the first visit, and choice of antibiotic. Failure to control for these variables may affect interpretation of the data.
The authors were particularly interested in answering the question of the efficacy of antibiotics in abscesses caused by caMRSA. Their conclusion that they are not necessary in lesions less than 5 cm in diameter is consistent with previous studies of patients with skin abscesses prior to the common occurrence of caMRSA. Likewise, a 2001 study of children with caMRSA concluded that recovery was not dependent upon treatment with effective antibiotics.2
The reader may use this study's conclusion as a general guideline when treating children with skin abscesses suspected to be caused by caMRSA. As always, the clinical judgment of the treating physician must be employed when making individual treatment decisions.
Oral Linezolid vs Intravenous Vancomycin
Source: Sharpe JN et al. Clinical and economic outcomes of oral linezolid versus intravenous vancomycin in the treatment of MRSA-complicated, lower-extremity skin and soft-tissue infections caused by methicillin-resistant Staphylococcus aureus. Am J Surg 2005;189:425-8.
In this open-label trial, Sharpe and colleagues compared the efficacy of oral linezolid therapy to intravenous vancomycin in patients with documented MRSA skin infections of the lower extremity requiring surgical intervention; the cost of therapy with each agent was concomitantly tracked. The study enrolled 117 patients, but 57 were excluded. The 60 remaining subjects were randomized to one of the two treatment arms. All were initially hospitalized for acute infection control but then completed treatment as outpatients. The authors rigorously defined clinical cure, clinical improvement, and clinical failure.
The results of the trial showed strikingly better clinical outcomes with oral linezolid: 97% of patients demonstrated clinical cure or improvement; the treatment failure rate was 3% with no amputations. The median length of inpatient therapy was 3 days followed by 7 days of outpatient treatment. Of subjects treated with intravenous vancomycin, 43% demonstrated clinical cure or improvement; 57% were treatment failures 7% of whom required amputation. The median length of inpatient therapy was 6 days followed by 4 days of outpatient intravenous treatment. The cost of inpatient linezolid therapy averaged $6438 less than vancomycin because of the lower length of stay. Outpatient costs were less with linezolid because it was administered orally and vancomycin required intravenous administration.
The authors concluded that oral linezolid offers definite economic advantage over intravenous vancomycin and may provide superior clinical results. They stated that vancomycin will remain first-line therapy for MRSA until a large, multicenter trial confirms their results.
Commentary
This study is an important contribution to the caMRSA literature for several reasons. The methodology was rigorous and definitions precise. The author's inclusion of economics in the outcome data is important; the decided cost advantage adds weight to the compelling clinical case for further study of oral linezolid for patients with complicated soft tissue caMRSA infections. These authors are the first to look at amputation as a discreet subset of treatment failure in patients with lower extremity infections treated with intravenous vancomycin. The 23% amputation rate for this treatment arm contrasted with 0% for the oral linezolid group is striking.
The study has several notable limitations. The first is the low number of subjects (60 patients). It is hazardous to extrapolate percentages obtained from such a limited data set to the whole population of complicated lower extremity caMRSA patients. It was a single site investigation and subject to the limitations of a narrow patient population. Finally, the authors only studied adult patients with lower extremity infections meeting their narrowly defined criteria.
The study's results are consistent with those of Weigelt and colleagues who conducted a large (1200 randomized patients) multicenter study of patients with complicated SSTIs randomized to treatment arms of vancomycin or linezolid.3 They found that treatment with linezolid was well tolerated, produced superior outcomes in patients with caMRSA and equivalent outcomes in all patients with complicated SSTIs, and required an average of 5 fewer days of intravenous therapy than vancomycin.
Linezolid should be strongly considered for treatment of complicated SSTIs, particularly in patients with lower extremity infections believed secondary to caMRSA.
MRSA Pneumonia
Source: Francis JS et al. Severe community-onset pneumonia in healthy adults caused by methicillin-resistant Staphylococcus aureus carrying the Panton-Valentine leukocidin genes. CID 2005;40:100-107.
Outbreaks of caMRSA, increasingly recognized worldwide, have primarily manifested as SSTIs. A more ominous expression is described by Francis and colleagues in their report of four cases of necrotizing caMRSA pneumonia presenting to one of two Baltimore hospitals in a two-month period during the 2003-2004 winter. Bacterial isolates were typed by strain; the presence of staphylococcal cassette chromosome mec (SCCmec) type IV was identified by polymerase chain reaction (PCR). PVL virulence factor, associated with tissue necrosis, was detected by PCR.
All of the cases occurred in previously healthy adults. Patients 1, 3, and 4 were female; Patient 2 was male. Patients 1 and 3 tested positive for acute influenza A; the other two had influenza-like prodromes but were not tested for influenza. Two patients were initially treated with oral antibiotics as outpatients; they subsequently deteriorated and were admitted to the hospital. Clinical features common to all four patients included fever, shock, and cavitary lesions on chest imaging; all required intensive care. Three of the four had hemoptysis. Two patients had pneumothoraces requiring chest tubes, and one had an empyema. One patient required bilateral below-the-knee amputations for lower extremity necrosis. Patient 2 died on hospital day 2. The other three were hospitalized from 41 to 108 days. Patients 3 and 4 were discharged to rehabilitation facilities. Patient 1 was the only one discharged to home; she spent 4 weeks in the intensive care unit.
The microbiological analysis of the caMRSA cultured from all four subjects revealed PVL virulence factors and the SCCmec type IV element. All isolates were sensitive to clindamycin, TMP-SMX, and gentamicin.
The authors noted that post-influenza staphylococcal pneumonia is likely the result of influenza-mediated damage to the respiratory epithelium. PVL is associated with necrotizing staphylococcal pneumonia. Currently, the incidence of staphylococcal pneumonia in healthy adults is low. If the prevalence of caMRSA carrying the PVL gene continues to increase, the percentage of severe pneumonia cases may increase. Clinicians should keep caMRSA in their differential diagnosis when treating otherwise healthy adults with fulminant pneumonia, particularly during influenza outbreaks.
Commentary
Francis and colleagues have made an important contribution to the body of literature; their case series is a readable and comprehensive description of the clinical and molecular features of caMRSA necrotizing pneumonia. The article's Table 1 summarized salient clinical features of the four cases. The methods section depicts the methodology for patient selection, case definitions, identification and characterization of caMRSA, and molecular analysis of virulence factors and SCCmec typing. The descriptions are clear. The results are concise but complete.
The discussion makes several interesting points. The first is citation of literature documenting the increasing prevalence of caMRSA in the United States. The second is a review of case reports of necrotizing caMRSA pneumonia in previously healthy children and young adults. Those cases are consistent with the severity of this disease documented in this case series. The authors emphasized the association of severe necrotizing pneumonia with identification of the PVL virulence factor. They expressed concern that the rising incidence of caMRSA may herald an increase of necrotizing pneumonia in healthy patients. Finally, treatment recommendations for suspected caMRSA pneumonia were given based on common antibiotic susceptibility in vitro.
Conclusion/Recommendations
The prevalence of caMRSA infections has rapidly escalated in many geographic regions in the United States and throughout the globe. Fridkin and colleagues demonstrated the importance of understanding the local experience with respect to prevalence and antibiotic susceptibility. SSTIs are the most common clinical expression of caMRSA, representing at least 75% of infections; the remainder comprise invasive diseases.
The data presented in this review support the following management guidelines in the opinion of this author at this time:
1. The primary treatment of uncomplicated skin abscesses less than 5 cm in diameter is incision and drainage. In the absence of surrounding cellulitis, systemic symptoms, or immunocompromising conditions, initial supplemental antibiotics are not indicated. Purulent material should be cultured so that the organism and sensitivities are identified if the infection does not resolve as expected. Close follow-up and monitoring are recommended. These recommendations apply to children and adults.
2. If empiric antibiotic treatment is necessary for outpatient treatment of skin infections, the clinician should understand local patterns of resistance to caMRSA. In most cases, TMP-SMX with or without rifampin is an excellent initial choice. Clindamycin or minocycline may be used in patients with contraindications to TMP-SMX. Oral linezolid is efficacious and well tolerated; however, the cost is prohibitive for routine use.
3. For complicated infections requiring surgery or inpatient treatment, linezolid is emerging as the antibiotic of choice. In their small series, Sharpe and colleagues suggested that oral linezolid offers economically and clinically superior outcomes to intravenous vancomycin. Weigelt and colleagues validated their findings in a multicenter trial. Currently, the therapies are equivalent for empiric coverage of all complicated SSTIs. Linezolid is superior in those caused by caMRSA.
4. Intravenous vancomycin is the antibiotic of choice for patients presenting to acute care settings with life-threatening caMRSA infections. Linezolid may be considered for patients with known resistance or contraindications to vancomycin.
Necrotizing pneumonia is a life-threatening manifestation of caMRSA. The presence of the PVL gene, conferring tissue necrosis properties, is probably responsible for the fulminant destruction of lung tissue.
Community-acquired MRSA should be considered in otherwise healthy patients presenting with life-threatening pneumonia, particularly in the post-influenza period. Intravenous vancomycin should be added to empiric antibiotic therapy for community-acquired pneumonia; standard antibiotic coverage will not provide adequate coverage for this pathogen.
Community-acquired MRSA is an emerging infection. The epidemiology and microbiological characteristics are dynamic. Acute care physicians and other practitioners are urged to follow the literature and local experience closely. Those communities with high prevalence of this microbe in SSTIs are at risk for increasing incidence of necrotizing pneumonia.
 |
References
1. Markowitz N, Quinn EL, Saravolatz LD. Trimethoprim-sulfamethoxazole compared with vancomycin for the treatment of Staphylococcus aureus infection. Ann Intern Med 1992;117:390-8.
2. Fergie JE, Purcell K. Community-acquired methicillin-resistant Staphylococcus aureus infections in south Texas children. Pediatr Infect Dis J 2001;20:860-3.
3. Weigelt J, Itani K, Stevens D, et al. Linezolid versus vancomycin in treatment of complicated skin and soft-tissue infections. Antimicrob Agents Chemother 2005;49: 2260-6.
Community-acquired methicillin-resistant Staphylococcus aureus (caMRSA) is a microbe that has become increasingly prevalent and clinically significant to acute care practitioners worldwide.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
