Skin and Soft-Tissue Infections
Skin and Soft-Tissue Infections
Authors: Kenneth J. Robinson, MD, FACEP, Medical Director, Program Director, LIFESTAR Helicopter Program; Chief, Prehospital Services, Department of EMS/Trauma, Hartford Hospital; Associate Professor of Emergency Medicine, University of Connecticut School of Medicine, Farmington; and Sara Blomstrom, MD, Emergency Medicine Residency, University of Connecticut, Farmington.
Peer Reviewers: William J. Brady, MD, Professor of Emergency Medicine, Department of Emergency Medicine, University of Virginia, Charlottesville; and Catherine A. Marco, MD, FACEP, Clinical Professor, Department of Surgery, Medical University of Ohio; Attending Physician, Department of Emergency Medicine, St. Vincent Mercy Medical Center, Toledo, OH.
Several years ago I saw a patient with a "simple" paronychia of his thumb. Primarily because this was his third visit for this simple problem, I x-rayed the thumb. His osteomyelitis led to an amputation, and for me a new-found respect for simple hand infections.
We encounter soft-tissue infections commonly and, fortunately, most get better with antibiotics. A hundred years ago, however, soft-tissue infections were the cause of sepsis and death. We all recognize that antibiotic resistance is increasing. Therefore, we need to be certain of what disorder we are treating, what organisms are most likely, and what antibiotics are best.
—The Editor
Introduction
Skin and soft-tissue infections may be categorized in a number of different ways. This report begins with discussion of the most superficial lesions and progresses through the skin and soft tissue to the deeper and systemic disease processes.
Impetigo
Impetigo is a highly contagious, superficial skin infection that is most common among 2- to 5-year-old children. It classically is divided into two forms, nonbullous and bullous, with the nonbullous form accounting for about 70% of the cases.1,2 It most frequently occurs in warm and humid conditions.3 Other predisposing conditions for impetigo include poor hygiene, insect bites, pruritic conditions accompanied by scratching, crowded living conditions, and minor skin trauma.2,4,5
S. aureus now is considered the primary pathogen in both bullous and nonbullous impetigo.6,7 Although nonbullous impetigo may begin as streptococcal infection, staphylococci are isolated from the majority of lesions of both nonbullous and bullous impetigo. Skin surfaces that are subject to frequent trauma permit the pathogen to invade and are involved most commonly. Examples are the nares, perioral areas, and the extremities.2,3,6-8
Nonbullous impetigo begins as a small vesicle or pustule that ruptures and eventually forms a red, moist base under a characteristic honey-colored crust.1,2,6 In most cases the lesions are asymptomatic. Occasionally they may be pruritic and the subsequent scratching can lead to the formation of other lesions by autoinoculation and the spread to adjacent regions.5,6,7 The infection is self-limiting. Untreated cases usually last for 2-3 weeks and extend to involve a wider area; most heal without scarring.
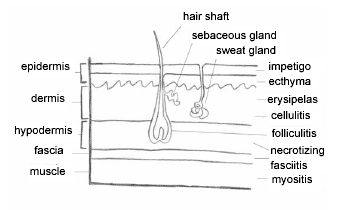
Bullous impetigo is characterized by large, superficial, fragile bullae that initially develop on the trunk, extremities, and intertriginous regions such as the axillae and neck folds.2,5,7 The vesicles rapidly enlarge and coalesce to form the bullae with clear to cloudy content.7 The flaccid bullae rupture easily and result in moist erythematous lesions that then form a thin, "varnish-like" light brown crust.3,5,7 When bullae occur, they result from specific staphylococcal exotoxins, usually made by the phage 2 group of S. aureus. These cause intraepidermal cleavage below or within the stratum granulosum.2,7 (See Figure 1 for a diagram of the skin and sections affected by skin conditions.)
| Figure 1. Diagram of Skin and Sections Affected by Skin Infections |
|
The treatment is the same for both nonbullous and bullous impetigo. Uncomplicated cases that are local and superficial can be treated topically. (See Table 1.) In the case of multiple lesions or severe disease, systemic therapy is indicated.1 No topical antibiotic has been found to be superior to another, but mupirocin ointment or cream is the first topical antibiotic approved for the treatment of impetigo.7-9 Bacitracin also is widely used, but one study demonstrated an unacceptable rate of more than 50% treatment failures.1,2 When using a topical antibiotic, crusts should be removed with warm soaks before application because they block the penetration of the antibiotic. Mupirocin should be applied three times a day until all of the lesions have cleared.7 In most comparisons, there was no difference in cure rates for topical vs. oral antibiotics, and oral antibiotics resulted in more side effects, especially gastrointestinal effects, than topical treatment did.9 Treatment with beta-lactamase-resistant antibiotics such as dicloxicillin, cloxacillin, cephalexin, azithromycin, erythromycin, or amoxicillin/clavulanate traditionally has provided appropriate coverage for S. aureus.1,2,3,6,10 The oral antibiotic treatment of choice for children with impetigo appears to be cephalexin.2 In many locations, resistance of S. aureus to these antibiotics and methicillin (MRSA) exceeds 50% so that antibiotics such as trimethoprim/sulfamethoxazole (particularly in children younger than 8), doxycycline (contraindicated in children younger than 8 years), and minocycline (contraindicated in children younger than 8 years) should be used. Nasal carriers of S. aureus who suffer from recurrent impetigo can be treated with topical mupirocin ointment applied four times a day to the affected nares.1
Erysipelas
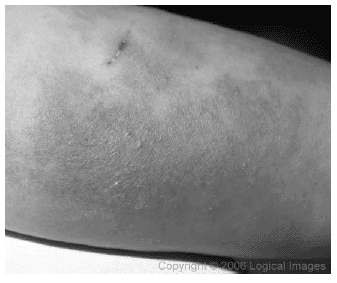
Erysipelas is a superficial cellulitis that includes prominent lymphatic involvement.3,7 (See Figure 2.) This condition is more common in infants, young children, and older adults. Historically, the face was the most commonly involved site. More recently, 70-80% of the lesions are on the lower extremities, with only 5-20% of the lesions on the face.3 The most common cause of erysipelas is group A streptococci, followed by group G streptococci.3,5,6,7 The bacteria gain entry into the skin via ulcers, local trauma, burns, psoriatic or eczematous lesions, or fungal infections. In the neonate, erysipelas may develop from an umbilical stump infection.1,3 Erysipelas tends to occur in association with venous stasis, ethanol abuse, diabetes mellitus, and in areas of preexisting lymphatic obstruction or edema.3,11
| Figure 2. Erysipelas |
|
| Image excerpted from VisualDx, the point of care visual diagnostic system for healthcare (www.logicalimages.com). |
A prodrome of malaise, chills, fever (101-104°F) and sometimes nausea and vomiting lasts 4-48 hours before the first lesions of erysipelas appear.6,7 Red, tender, firm spots appear at the site of infection. These lesions rapidly increase in size to form an elevated shiny patch with a well demarcated border.6,7,12 The resulting burning pain and redness, which sometimes can become severe, has led to the term St. Anthony's Fire being applied to this condition.6,7 Uncomplicated erysipelas is confined to the dermis and the lymphatics. Prominent adenopathy and a leukocytosis are common findings. Occasionally the infection may extend deeper to produce cellulitis, subcutaneous abscess, and necrotizing fasciitis.3 Without therapy, the rash may peak in 1 week and resolve in 1-2 weeks. In some patients, deep infection, which is potentially fatal, will result. Recurrent infections after antibiotic treatment occur in 18-30% of the cases and may lead to skin thickening and lymphatic obstruction.6,7
Early and mild cases of erysipelas can be treated with oral or intramuscular antibiotics. A penicillinase-resistant penicillin, first-generation cephalosporin, or erythromycin is an appropriate choice for treatment.3,6,10,12 More extensive infections may require parenteral antibiotics. Recurrent erysipelas may require long-term prophylactic treatment with low-dose penicillin or erythromycin.7
Erythrasma
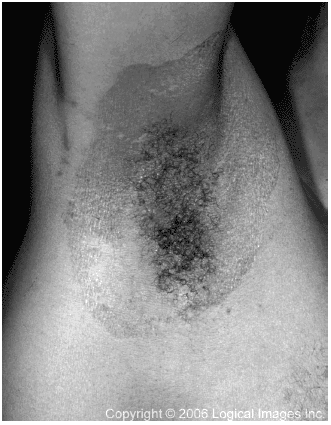
Erythrasma is a common superficial bacterial infection of the skin caused by Corynebacterium minutissimum. (See Figure 3.) It commonly occurs on moist, occluded intertriginous areas such as the intergluteal and crural folds, the axillae, the interspaces of the toes, and the inframammary areas.13 The lesions are more common in men and in obese individuals with diabetes mellitus.3 Other factors that may predispose a patient to erythrasma are a warm, humid climate, advanced age, compromised host status, hyperhidrosis, and poor hygiene.13
| Figure 3. Erythrasma |
|
| Image excerpted from VisualDx, the point of care visual diagnostic system for healthcare (www.logicalimages.com). |
Erythrasma may be pruritic and uncomfortable, but more commonly the patient is asymptomatic and complains only of skin color changes in the affected areas. The lesions are finely scalded and finely wrinkled and may present as either well-defined or irregular patches.3,13-15 As the infection progresses, the lesions often become reddish-brown, are slightly raised from the surrounding skin, and may have central clearing.13-15 Included in the differential diagnosis are candidiasis, tinea versicolor, tinea cruris, psoriasis, intertrigo, and dermatophytosis. A coexisting dermatophyte or Candida albicans infection has been shown to be present in up to 30% of the individuals with interdigital erythrasma.13 The diagnostic procedure of choice is observation of fluorescence in a darkened room with a Wood's lamp. The C. minutissimum produce porphyrins that appear as coral red fluorescence under a Wood's lamp.3,13,14,16,17 Microscopic examination of skin scraping and cultures also may aid in the diagnosis.13
Treatment for cutaneous erythrasma can include topical and oral antibiotics and various adjunctive therapies. Interdigital infections have been shown to be more difficult to clear and usually require oral and topical antibiotics. The most commonly recommended first-line therapy is oral erythromycin 250 mg QID for 7-14 days. This therapy usually is efficacious.3,10,13 In a small set of patients, a single dose of clarithromycin (1 g) was shown to clear the infection (as evidenced by Wood's lamp) and none of the patients had a recurrence of their symptoms.16 Topical therapies such as erythromycin 2% gel, clindamycin 2% solution, and Whitfield's solution (benzoic acid 12% and salicylic acid 6%), and cleansing with an antibacterial soap and water also have been reported to have a role in treatment.3,10,13,14,17 The symptoms and lesions begin to resolve in approximately 48 hours.
Ecthyma
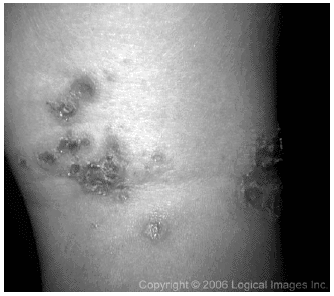
Ecthyma is a bacterial infection of the skin that can closely resemble impetigo. (See Figure 4.) It is more common in debilitated individuals (such as those with uncontrolled diabetes), and poor hygiene may predispose to this condition. Ecthyma is most common on the lower extremities, especially in children and the elderly, and often is the result of superficial lesions, insect bites, and trauma. Group A streptococcus is the etiology of most milder and initial infections, which often become contaminated with staphylococci.
| Figure 4. Ecthyma |
|
|
| Image excerpted from VisualDx, the point of care visual diagnostic system for healthcare (www.logicalimages.com). |
Ecthyma gangrenosum occurs in immunocompromised patients or those with hematologic malignancies and is caused by Pseudomonas aeruginosa. The lesions begin as painless red macules and enlarge into papular lesions and eventually hemorrhagic pustules and bullae. The initial vesicles and bullae are moderately painful, and enlarge and rupture to form an adherent crust that covers a punched-out ulcer.1,3,7 With progression, the bullae become necrotic, slough, and leave the characteristic erythema-surrounding gray-black eschar.1,6,18 Pathologically, the lesions begin superficially and eventually extend through the epidermis into the underlying dermis.1,3 Because of the underlying tissue involvement, the lesions may heal with scarring.7 If not treated, ecthyma can progress to lymphangitis and cellulitis.1
Treatment is different for the two forms of ecthyma. The common ecthyma, caused by streptococci and staphylococci, is treated similarly to impetigo, with a 10-day course of mupirocin, dicloxacillin, cephalexin, azithromycin, or clarithromycin (see Impetigo above).3,7,10 In areas where MRSA is prevalent, appropriate coverage would be trimethoprim/sulfamethoxazole (particularly in children younger than 8), doxycycline, and minocycline (both contraindicated in children younger than 8 years). Phenoxymethyl penicillin V could be added for better streptococcal coverage. Ecthyma gangrenosum is most appropriately treated with intravenous antibiotics, an aminoglycoside and an antipseudomonal penicillin, or a single antibiotic with pseudomonal coverage such as imipenem or ceftazidime.1,6
Staphylococcal Scalded Skin Syndrome
Staphylococcal scalded skin syndrome (SSSS) is a condition that usually is seen in infants and children, but rarely in adults. (See Figure 5.) Ninety-eight percent of those affected are younger than 6 years old, and 62% are younger than 2 years old.6 The disease is caused by exfoliative toxin producing S. aureus, most commonly from the phage 2 group.19 These exotoxins cause intraepidermal splitting through the granular layer.20 The condition spans a wide spectrum of severity from mild local reactions to severe life-threatening infections, with the mildest cases being similar to bullous impetigo.21 The mortality of children with SSSS is approximately 4%, whereas the mortality in adults can be as high as 60%.19 Antibodies to the exfoliative toxins protect against the effects of the toxins.21 Children are more likely to develop SSSS because they lack these antibodies. By the age of 10 years old, most people have antibodies to the exotoxins.6 Adults who develop SSSS have an excessive exotoxin burden, an increased S. aureus carriage rate, or an increased susceptibility to the toxin. A suppressed immune system from HIV, malignancy, severe sepsis, a genetic defect in the immune system, immunosuppressive medications, chronic ethanol abuse, or heroin abuse can predispose a patient to SSSS.19,21 Because the exotoxin is excreted via the kidney, renal insufficiency or renal failure are common in adults who develop SSSS.19,20 The toxin enters the circulation and affects the skin systemically.
| Figure 5. Staphylococcal Scalded Skin Syndrome |
|
|
| Image excerpted from VisualDx, the point of care visual diagnostic system for healthcare (www.logicalimages.com). |
SSSS usually presents with a prodrome of sore throat, conjunctivitis, or an infected umbilicus. Fever, malaise, and irritability develop shortly thereafter.6,19 One to two days later tender, erythematous lesions develop on the head and neck. The lesions evolve into large, flaccid, clear bullae and often involve the flexures. The patient will have a positive Nikolsky sign. The bullae enlarge and rupture, and new bullae appear over the next 2-3 days. The exfoliations result in lesions that have a moist erythamous base and account for the scalded appearance. Desquamation involving large areas of the skin occurs within 36-72 hours.5 These patients must be treated effectively as burn patients. In milder cases, once antibiotics are initiated, the condition resolves within a few days and heals without scarring.19
The diagnosis is based on clinical and histological features such as tenderness, erythema, and desquamation or bullae formation; histopathological evidence of intraepidermal cleavage through the stratum granulosum; and isolation of an exfoliative exotoxin producing S. aureus.22 Antibiotics should be administered as soon as SSSS is clinically suspected and should not be delayed awaiting confirmation of the diagnosis. The oral or parenteral route for the antibiotics is appropriate. Because the source of the infection is distant from the site of the lesions or not known, topical antibiotics are not effective.
There are many issues that should be considered in the treatment of SSSS. Antibiotics must have penicillinase resistance and also cover methicillin-resistant organisms. Severe cases should be treated with vancomycin given intravenously for 5-7 days.10 Once sensitivities return, the vancomycin can be changed to nafcillin or oxacillin where appropriate. Milder cases can be treated with oral antibiotics such as dicloxacillin, erythromycin, or cephalexin.6,23 Ciprofloxacin, cefuroxime, gentamycin, trimethoprim/sulfamethoxazole, doxycycline (contraindicated in young children), minocycline (contraindicated in young children), vancomycin, and clindamycin are appropriate options.21,22 Because large areas of skin can be involved, fluid management, thermoregulation, and pain management are all important aspects of care for the patient with SSSS.21,24 Skin care involves leaving the blisters intact and covering the denuded areas with petrolatum-impregnated gauze.19 Patients with extensive skin involvement often are treated as burn patients and should be admitted to a burn or intensive care unit. Corticosteroids are contraindicated and may worsen the condition.3,19,21
Cellulitis
Cellulitis is an infection of the skin and subcutaneous tissues.25 The infection may be preceded by a traumatic disruption of the skin, but also can arise from a deeper or underlying infection such as osteomyelitis or bacteremia.26 Cellulitis can affect anyone, but certain populations are at increased risk for the development of cellulitis. Chronic leg edema due to venous stasis or insufficient lymphatic drainage predisposes patients to developing cellulitis.27 Patients who have had previous CABG with saphenous vein grafting are particularly at risk because of the combination of compromised lymphatic drainage and venous insufficiency.3 The portal of entry in these patients usually is a break in the skin from tinea pedis, which is likely to go undiscovered and untreated.3 In patients with previous saphenous vein grafting, the presentation usually is more systemic and toxicity is prominent.3
Staphylococcus aureus and Group A beta hemolytic streptococcus are the most common pathogens, but Pasturella and Clostridium also can cause cellulitis.3,25,28 The condition presents as painful, indurated subcutaneous tissues with warmth and erythema of the overlying skin.28 The borders of the erythema and induration are nondistinct in contrast to erysipelas.3,28 The extremities are the most commonly affected site of the body.25 With previous saphenous vein grafts, the infection spreads rapidly along the course of the venectomy.3 The spread of infection also can be seen as erythematous streaking along lymphatic drainage paths and regional lymphadenopathy is common.3,26 Cellulitis is a serious disease because of the propensity for the infection to spread to the bloodstream via the lymphatics.3 Systemic symptoms such as fever and malaise suggest worsening infection and possible bacteremia.26,29
The treatment of cellulitis consists of antibiotic therapy against S. aureus and Streptococcus.26 First-line treatment is a first-generation cephalosporin such as cephalexin or a penicillinase-resistant penicillin such as oxacillin. Alternatives for treatment include clindamycin, macrolides, and fluoroquin-olones.26,28 If the infection is unresponsive to initial therapy, rifampin can be added to cephalexin.25 With minor infections, oral antibiotics are appropriate. If the infection is rapidly progressive, has systemic symptoms, or the patient has significant comorbidities, parenteral antibiotics and admission are indicated.25 Patients also should be admitted if significant portions of an extremity are involved or the infection involves the hands, feet, head, neck, or perineum because these areas are associated with a greater risk for complications.29 Routine blood cultures in uncomplicated cellulitis have been shown not to aid in the treatment or diagnosis of immunocompetent individuals.30 If the response to treatment is poor or an unusual organism is suspected, a fine-needle aspiration of the leading edge of infection for Gram stain and culture may aid in guiding the treatment, although this technique may not isolate an organism and is not widely recommended.25 Elevation and immobilization of the involved extremity will aid in healing.3 For patients with chronic dependent edema, proper hygiene and support hose will help prevent future occurrences.3,31
Cellulitis from Animal Bites. Cellulitis also may result from cat or dog bites, and in these cases most often is caused by Pasturella multocida.28 The cellulitis associated with Pasturella is a rapidly progressive cellulitis that presents within hours of contact as intense erythema, edema, tenderness, and sanguinous wound drainage.28 It is important to examine the deeper structures when animal bites are suspected as the infection can spread rapidly and cause deeper scarring. The antibiotic of choice for cellulitis associated with an animal bite is amoxicillin-clavulanate.28 Tetracycline, fluoroquinolones, TMP-SMX, and second- or third-generation cephalosporins are acceptable alternatives for animal bite related cellulitis. Patients with animal bite related cellulitis have to be monitored carefully because of the aggressive nature of the associated pathogens. If a patient has a rapidly progressive cellulitis, cellulitis on the hand, or may not be compliant to follow up in 24 hours, he or she should be admitted for parenteral antibiotics.28,31
Periorbital Cellulitis. Periorbital cellulitis is an infection of the tissues surrounding the eye but not involving the orbit.26 The infection is precipitated by trauma to the adjacent tissue or an infection including hordeola, chalazion, sinusitis, upper respiratory infection, or abrasion in greater than 50% of the cases.26,32,33 Hematogenous spread of an infection from a distant site can precipitate periorbital cellulitis.32,34 Historically, 80% of pediatric patients with periorbital cellulitis had associated Haemophilus influenzae type B bacteremia.35 As a result of the widespread administration of the H. influenzae B vaccine, the most common organisms are now S. aureus and Streptococcus.26 In children between 3 and 36 months old, periorbital cellulitis may be associated with S. pneumoniae bacteremia.34
Periorbital cellulitis presents as tender, erythematous, and indurated areas around the eye.26 There may be signs of trauma or previous infection (such as conjunctivitis, sinusitis, or trauma), and the cellulitis may extend to involve the upper cheek or brow area.26,36 The lids may be swollen closed, and the conjunctiva may be edematous and injected. Regardless of the appearance of the external tissue, the extraocular movements are intact and painless, and visual acuity is preserved.26,29
Periorbital cellulitis responds quickly to appropriate antibiotic therapy and rarely extends beyond the septum to cause orbital cellulitis.32 The antibiotics of choice are cephalexin or clindamycin.34 The patient with early periorbital cellulitis can be treated with oral antibiotics and daily follow-up.29 In children between 12-15 months who are ill-appearing and present with periorbital cellulitis, a septic work-up, including a lumbar puncture and intravenous antibiotics, are indicated due to the risk of S. pneumoniae bacteremia.34
Orbital Cellulitis. Orbital cellulitis is less common than periorbital cellulitis and involves an infection deep to the orbital septum, in contrast to periorbital cellulitis, which represents an infection superficial to the orbital septum.26,29,37 In 60-91% of orbital cellulitis, the infection arises from sinusitis of the ethmoid and maxillary sinuses.37 Children 6-15 years of age are most commonly affected, and there is a bimodal peak at 60-70 years of age.37 Males are more commonly affected, and there is a seasonal variance due to the increased incidence of upper respiratory tract infections between November and February.35 The most common organisms include S. aureus, S. pyogenes, H. influenzae, and S. pneumoniae.26
Orbital cellulitis presents as an edematous, erythematous eye with associated painful globe movement and decreased visual acuity or proptosis.26,34 These patients present differently from those with periorbital cellulitis in that patients with periorbital cellulitis have normal vision and painless, intact extraocular movements. Patients with orbital cellulitis are more ill-appearing than those with periorbital cellulitis, and may be febrile or appear lethargic.26 In the event that the lid is too edematous to determine visual acuity and extraocular movements, a contrast enhanced CT of the orbit may be indicated to differentiate orbital cellulitis from a subperiosteal abscess, ocular abscess, or concomitant sinusitis.34
The treatment for orbital cellulitis is broad-spectrum IV antibiotics and possibly surgical decompression. Blood cultures should be drawn to guide antibiotic therapy. An ophthalmology consult should be obtained, and the patient should be admitted for parenteral antibiotics.26,34,35 The complications of untreated orbital cellulitis include an abscess, optic neuritis, retinal vein thrombosis, direct extension into the central nervous system (CNS), meningitis, epidural/subdural abscess, and cavernous sinus thrombosis.26 In developing countries, untreated cellulitis results in blindness in 52% of patients and death in 4% due to cavernous sinus thrombosis.37 Medical management is appropriate for the first 24 hours, but surgical decompression may be necessary if the patient's condition does not improve or if there are worsening features on CT scan.37 Findings that may indicate the need for immediate surgical drainage include an orbital abscess and a subperiosteal abscess. An orbital abscess occurs when the inflammation behind the orbit organizes into a necrotic fluid collection. It should be decompressed surgically to prevent further complications such as spread to the cavernous sinus.35 A subperiosteal abscess is an abscess between periorbita and the bony walls of the orbit and has risk for expansion.35 Ophthalmology consultation should be obtained urgently as most patients will require emergency surgery. One study, however, documented that in children with a subperiosteal abscess who were monitored closely for signs of worsening infection (i.e., vision and extraocular function), medical management was an acceptable option to immediate surgical drainage.38
Folliculitis, Furuncles, and Carbuncles
Folliculitis is the infection of hair follicles leading to very small abscesses with limited surrounding tissue inflammation.26 S. aureus is the most common pathogen but infections from Pseudo-monas may result from exposure to poorly maintained pools or hot tubs.3,26 Folliculitis presents as small (less than 5 mm) erythematous papules with a central pustule, usually is self-limited, and treatment is not indicated.3 Patients may require oral antibiotics if multiple sites are involved or if they are known to be chronic S. aureus carriers.39
A furuncle is a deeper infection of a single hair follicle, which presents with a more intense local tissue reaction than that of folliculitis.26 Furuncles often develop from preceding untreated folliculitis and tend to occur in areas where the hair follicles are subject to friction and perspiration, such as the face, buttocks, neck, scalp, thigh, perineum, breast, and maxilla.3,39 Predisposing factors include obesity, poor hygiene, and diabetes mellitus.3 A furuncle presents as a firm, red nodule that becomes painful and fluctuant, drains spontaneously, and resolves spontaneously.3 The treatment for furunculosis is moist heat compresses and sitz baths.3
Carbuncles are multiseptate, loculated abscesses that develop from infection in multiple hair follicles.3,26 The most common locations are the back and the neck.39 The infection spreads laterally into the subcutaneous fat rather than draining to the skin surface.3,39 Patients may have a systemic disease such as diabetes predisposing them to infection.39 S. aureus is the most common bacteria involved, but community-acquired MRSA is becoming more common.3 Patients with carbuncles appear more ill than those with furuncles and often present with fever, malaise, and bacteremia.3 Treatment includes incision and drainage of the abscess, including disrupting loculations if possible.3,39 Some carbuncles may be too extensive to be managed adequately in the emergency department.39 Dicloxacillin or cephalexin may be indicated for an extensive infection.3 Alternatively, TMP/SMX, clindamycin, or minocycline (contraindicated in small children) may be used in geographic areas with a high incidence of community-acquired MRSA.26,31
Abscess
An abscess is a localized collection of pus surrounded by erythema and indurated granulation tissue.29 Abscesses develop from high concentrations of pathologic organisms, occlusion of a follicle or skin opening preventing drainage, and trauma to skin that allows organisms to penetrate.25,39 The skin reaction usually begins as cellulitis, but as necrosis, liquefaction, and accumulation of debris occur, the cavity that forms becomes walled off.39 Abscesses represent 1-2 % of total ED visits, and the presence of a foreign body predisposes to abscess formation.39 Abscesses involving the head, neck, and perineum often are due to the obstruction of apocrine sweat glands, which increase in incidence after puberty.29 When abscesses present on the extremities, they usually arise from local trauma to skin.29 Microorganisms involved vary by site of the body involved. Perioral or perianal abscesses are associated more commonly with anaerobic organisms, whereas S. aureus is the most common organism in abscesses elsewhere on the body.25,29,39 Up to 5% of abscesses are sterile.29
Patients usually appear well and are without systemic symptoms.29 Abscesses usually develop individually, unless associated with intravenous drug use or MRSA.29,39 An abscess presents as an erythematous, fluctuant mass with surrounding warmth and cellulitis.29 There may or may not be evidence of local trauma.29
The proper treatment of an abscess includes incision and drainage.25,29,39 Special situations including the hand, breast, pilonidal abscess, Bartholin's abscess, and abscesses in intravenous drug users will be discussed separately. Studies have shown that simple incision and drainage alone is adequate therapy for an abscess, but with the rising incidence of community-acquired MRSA, antibiotics may be indicated.25 The most common limiting factor for incision and drainage in the ED is inadequate anesthesia.39 The infected tissue has a low pH, and local anesthetics such as lidocaine are less effective in an acidic environment.39 Regional anesthesia is very useful when appropriate. After sufficient analgesia, the abscess should be incised and any loculations disrupted, the cavity then should be irrigated and loosely packed with gauze.29,39 Antibiotic therapy may be indicated for abscesses in certain sites or if an anaerobic infection is suspected.29 Patients should be seen by a surgeon for follow-up in 1-3 days for packing change and reevaluation.39
Community-Acquired Methicillin Resistant Staphylococcus Aureus (MRSA)
Methicillin-resistant Staphylococcus aureus (MRSA) was once only a nosocomial infection.40 In the 1980s-90s it became a disease of individuals in the community who were particularly at risk (i.e., recent hospital admission, HIV infection, intravenous drug users), and now it is a common infection in healthy individuals with no risk factors.40,41 Outbreaks have occurred in universities, daycare centers, and sports centers.40 Community-acquired MRSA is genetically different than hospital-acquired MRSA and is susceptible to a broader array of treatments.41,42 The most common presentation of community-acquired MRSA is an abscess (up to 70-100% of cases), followed by furunculosis/carbunculosis.40-42 In one study in Los Angeles, the incidence of community-acquired MRSA in all soft-tissue infections was 49.6%.41
The abscesses associated with community-acquired MRSA are significantly inflamed with cutaneous necrosis secondary to direct invasion of the organism and tissue destruction.40-42 This virulence is associated with the presence of the Panton-Valentine-Leukocidin (PVL) gene, which encodes an exotoxin that creates pores in the leukocyte cell membrane and contributes to cell death.41,42 Community-associated MRSA infections usually localize to soft tissue, but also have been found to cause septic shock and necrotizing pneumonia.42
The treatment of community-acquired MRSA consists of incision and drainage of the abscess, cultures of the discharge, and an appropriate antibiotic for a minimum of 7-10 days or until the patient is asymptomatic.25,40-42 It is important to completely drain the abscess because any residual infection can lead to further antibiotic resistance.42 Linezolid has been shown to bind the 50s subunit of the ribosome and block protein synthesis and has been associated with a cure rate of 100%.40 The dose of linezolid is 400-600 mg IV/PO q12 hours.43 Clindamycin also can be used in the treatment of community-acquired MRSA.44 There may be inducible resistance to clindamycin in strains that are resistant to erythromycin.42 Case reports have shown treatment failures or recurrences associated with clindamycin, but most patients have been shown to recover.44 One study demonstrated that clindamycin can be used successfully for invasive community-acquired MRSA in children.44 TMP-SMX has been shown to effectively treat MRSA.40,44 For MRSA associated with cellulitis, cephalexin may be added to TMP-SMX.44 Adding rifampin to TMP-SMX has been shown to help eradicate nasal carriage of MRSA and appears to reduce the incidence of recurrent furunculosis.44 Mupirocin applied topically to nares also has been advocated by some to eradicate the carrier state, but others recommend that it be used only for recurrent problematic infections.40,42 Other antibiotics that may have a role in the treatment of MRSA include quinupristin-dalfopristin, daptomycin, and fluoroquinolones.43 If a patient is very ill, has systemic signs of infection, and is being admitted to the hospital, it is important to empirically treat for MRSA with vancomycin.43
Abscess in the Intravenous Drug User. Skin and soft-tissue infections are the most common reason for hospital admission and ED visits in IV drug users.45 This population is predisposed to abscess formation, and abscesses are the most frequent soft-tissue infection.45,46 The mechanisms of infection include injection technique, poor hygiene, contaminated drugs, needle sharing, and direct toxic effects of some injectable drugs.45 Skin popping, or injecting the drug into the subcutaneous tissue, has been shown to be the strongest risk factor for abscess for-mation.45,46 "Black tar" heroin is especially toxic and increases the risk of tissue necrosis from a direct toxic effect.45 Black tar quality of heroin comes from its production and "cutting" with black or brown substances in Mexico where it is made.45 When the final product is made, it looks like tar or coal.45 Other mechanisms may include tissue ischemia from direct vasospastic effects of drugs such as cocaine, or repetitive trauma to areas used frequently for injection.46,47 The microbiology of abscesses in IV drug users differs from that of the general population.45,46 S. aureus is the most common organism, but there is an increased prevalence of oral flora in skin abscesses of IV drug users.45 The oral flora is thought to originate from using saliva to prepare the skin or syringe. The presence of oral flora may be as high as 50-70%.39,45,46 The anaerobes that are present from oral flora include fusoform bacteria, Eikenella corrodens, and Prevotella.46,47 Gram-negative organisms also may be involved, and Pseudomonas is the most prevalent.45
Abscesses in IV drug users are more commonly on the extremities, but they can occur anywhere on the body.45 One study found that up to 31% of IV drug user patients with an abscess presented with a skin area that appeared to be simple cellulitis, with the abscess occurring below the subcutaneous tissue in muscle or fascia.46 Another study documented that fluctuance was absent in 25% of patients with abscess.47 Therefore, it is important to have a high index of suspicion for an abscess even in the absence of fluctuance.46,47
The treatment of an abscess in an IV drug user includes incision and drainage and broad-spectrum antibiotics that cover anaerobes and S. aureus.46 Appropriate antibiotics include clindamycin, ampicillin-sulbactam, or vancomycin and flagyl.46 A prolonged antibiotic course may be necessary.47 There may be situations in which an abscess is too complicated to incise and drain in the ED, such as complicated infections; abscess in the hand, neck, or groin; presence of sanguinous fluid on needle aspiration; and a history of vascular puncture. In these situations, a surgical consult should be obtained for possible incision and drainage in the operating room under controlled circumstances.45 Early incision and drainage is essential to the treatment of abscess in the IV drug user, and multiple drainage procedures may be required.47
Abscess/Infection of the Hand
Infections of the hand can be particularly debilitating because of the high incidence of loss of function with untreated hand infections.48 The infections that are encountered and treated most commonly in the ED include paronychia, felon, and herpetic whitlow.48 Flexor tenosynovitis will be discussed briefly as it is important to recognize.
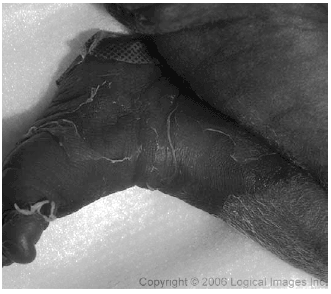
Paronychia. A paronychia is an infection of the paronychium (also known as eponychium), which is the epidermis bordering the nail.48 This infection is caused by localized trauma around the fingernail and/or cuticle and may occur secondary to dishwashing, manicure, an ingrown nail, or a hangnail.39,48 The infection presents as painful, erythematous swelling at the base of the fingernail.48 There may be local fluctuance, draining, or evidence of surrounding cellulitis.39 A mobile nail bed indicates extension of the infection forming a subungual abscess.39 The usual organisms include S. aureus, S. pyogenes, anaerobes, gram-negative bacilli, and rarely Pseudomonas.48
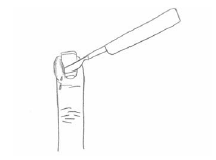
The treatment of a paronychia includes incision and drainage of the fluctuant area or warm water soaks in the absence of fluctuance or purulent discharge.39,48 After administration of a digital nerve block using a lidocaine/bupivocaine mixture, the finger should be soaked initially to soften the skin. Then an 18-gauge needle or scalpel blade is advanced parallel to the nail at the site of maximal swelling, and the tissue is gently lifted off of the nail.39 (See Figure 6.) In the case of a deep cavity being opened, gauze may be placed under the eponychium for 24 hours to facilitate drainage.39 A subungal abscess may necessitate the removal of a portion of the nail to facilitate drainage.39 The finger should be splinted and elevated, and warm soaks should be applied daily to prevent a recurrent abscess.39 An oral antibiotic usually is indicated and an appropriate agent is a first-generation cephalosporin such as cephalexin or a penicillin such as amoxicillin-clavulanate. In complicated cases, a hand consult should be made to facilitate follow-up.48
| Figure 6. Paronychia |
|
|
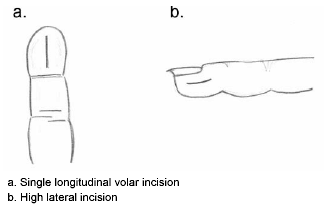
Felon. A felon is an abscess of the distal pulp or pad of fingertip.48 The pulp is divided into 15-20 compartments by fibrous septa, which run from the periosteum to the skin.48 The most common organisms are S. aureus and S. pyogenes.48,49 The inciting factor for felon formation is usually inoculation of the tissues by penetrating trauma to the fingertip, with the index finger and the thumb being most commonly affected.48 A felon also may extend from a preexisting paronychia.48 Patients present with pain, erythema, and edema of the distal fingertip, which does not extend past the DIP joint.48 The pain usually is out of proportion to the infection because the fibrous septa limit the extension of the infection and subsequently result in high pressures in the tissue, which can lead to necrosis.48 If left untreated, the infection has been shown to cause osteomyelitis by penetrating the periosteum.48,49 An early felon can be treated with oral antibiotics and warm water soaks, but most require incision and drainage.39,48,49 An x-ray should be obtained to rule out a foreign body or evidence of osteomyelitis.48 The incision should be made over the area of maximal fluctuance and may include a single volar long incision, high lateral incision, or a through-and-through incision.39,48 (See Figure 7.) If the infection is complicated, a hand surgeon should be consulted for possible admission and debridement in the operating room.39,48 The finger should be splinted, elevated, and soaked in warm water twice daily.48,49 Empiric oral antibiotics, which include cephalexin or antistaphylococcal penicillin, should be started and the patient should be seen in follow-up with a hand surgeon in 24-48 hours.39,48,49
| Figure 7. Felon |
|
|
Herpetic Whitlow. A herpetic whitlow may mimic a felon, but it is treated differently.48 It is caused by the herpes virus 1 or 2 and usually results from autoinoculation of the virus via broken skin.48 Nurses and others who give mouth care are particularly at risk. The patient presents with erythema of the finger and pain out of proportion to the infection.48 The initial lesion is vesicular, and the vesicles becomes cloudy and can appear pyogenic.48 Fever and lymphadenitis are more common with herpetic whitlow than with a felon.39,48 Incision and drainage is contraindicated in herpetic whitlow; therefore, it is important to differentiate this from a felon.48 An oral antiviral such as acyclovir may help if initiated in the first 48 hours of the infection. Otherwise, treatment is supportive care.48
Flexor Tenosynovitis. Flexor tenosynovitis is an infection of the flexor tendon sheaths of the hand.48 The infection usually arises from trauma to the flexor crease where the tendon is the most superficial.48 The infection spreads rapidly through the sheath and becomes pyogenic.39,48 The patient usually presents with Kanavel's four signs, which include uniform symmetric digit swelling, digit held in partial flexion at rest, tenderness along the course of the tendon sheath, and pain with passive extension.48 It is difficult to distinguish a subcutaneous abscess from flexor tenosynovitis.48 The most common organisms are S. aureus, Streptococcus, and anaerobes, and empiric treatment includes a parenteral cephalosporin or ampicillin-sulbactam.48 The diagnosis and treatment of flexor tenosynovitis must be made early to prevent tendon necrosis, scarring, and spread of infection.48 A hand surgeon should be involved as soon as flexor tenosynovitis is suspected. If the infection is in the early stages (within 12-24 hours of symptoms), medical management including splinting, elevation, and antibiotics may be adequate.48 The definitive treatment, if medical therapy fails, is surgical irrigation and debridement.48
Breast Abscess
An abscess of the breast can occur as a consequence of mastitis in the puerperium, but most breast abscesses are nonpuerperal.39 Ten to thirty percent of breast abscesses are lactational and result from milk stasis and bacterial infection leading to mastitis and an abscess.50 Mastitis occurs postpartum in 1-3% of nursing mothers within the first 2-6 weeks after delivery.39 Mastitis is inflammation of the breast tissue that results from bacterial invasion through the nursing mother's cracked or abraded nipple.39 The bacteria are introduced from the mouth of the nursing baby and most commonly are staph or strep.39 The most common organism in lactational and peripheral skin abscesses is S. aureus, whereas nonlactational, periareolar, or deep abscesses often are polymicrobial with anaerobic bacteria.39,50
Patients with a breast abscess may present early in the course of mastitis with erythema, warmth, fever, and chills, or late in the course with a painful, fluctuant mass.39,50 Breast abscesses are classified anatomically as peripheral, periareolar, and retromammary.39 Peripheral breast abscesses are skin abscesses that evolve spontaneously and present as a circumscribed, erythematous collection of pus.39 Periareolar abscesses are deeper and more extensive and present as a swollen, tender, inflamed breast.39 Periareolar abscesses arise from occluded and inflamed mammary ducts and recur after treatment in 38% of patients.39 Retromammary abscesses occur between the breast tissue and the chest wall, and in these cases the fluctuance may be difficult to appreciate.39
The treatment of a breast abscess is incision and drainage.39 Peripheral breast abscesses can be drained in the ED with an incision that radiates from the nipple.39 Periareolar and retromammary abscesses require incision and drainage in the operating room. Obstetrics and gynecology should be consulted for assessment and drainage in this population.39,50 If the abscess is lactational, an antistaphylococcal penicillin such as nafcillin or oxacillin should be added.50
Pilonidal Abscess
A pilonidal abscess is an abscess that occurs in the gluteal fold overlying the coccyx.29 The abscess is a result of minor disruption of the epithelium, which forms a small pit. The pit that is formed is gradually lined with squamous epithelia and eventually plugs with hair and keratin, preventing drainage and promoting abscess formation.29,51 The average age at presentation is 21 years old, and males are twice as likely to be affected.51 Risk factors associated with pilonidal abscess formation include obesity, sedentary lifestyle, hirsutism, poor hygiene, and increased sweat activity.29,51 The organisms involved include fecal flora and anaerobes.39
A pilonidal abscess presents as a warm, tender, fluctuant swelling lateral to midline over the sacrum.51 Pus, which usually is malodorous, may be draining through the midline pit.51
Treatment includes incision and drainage with a full-length incision to prevent recurrences.39 All visible hair and debris must be removed, and the wound should be packed with iodoform gauze.39 Antibiotics are not necessary for uncomplicated pilonidal abscess.29,39,51 Patients should follow up with their regular physician for ultimate referral to a surgeon for eventual sinus tract removal. The recurrence rate is 10-90% and is due to the formation of new pits and remaining granulation tissue.29 Definitive treatment includes excision of the sinus after the inflammatory process has resolved.39
Bartholin Abscess
A Bartholin abscess arises from infection of the Bartholin glands at the introitus of the vagina.52 The glands are located bilaterally at the posterior introitus and drain through ducts that empty into the vestibule at the 4 and 8 o'clock positions.52 The glands are palpable only if the duct becomes cystic or if a gland abscess is present.52 Bartholin abscesses are common in women of reproductive age, with the highest incidence in women 20-29 years of age.52 Up to 2% of women will develop a duct cyst or gland abscess in their life with abscesses being three times as common as cysts.52
The infection usually is polymicrobial with a mixture of aerobic and anaerobic flora from the vagina.29,52 Gonorrhea or Chlamydia are present in up to 10% of the cases.29 Patients present with pain worsened by walking, sitting, or sexual intercourse.52 Erythema and induration are present around the gland, which usually does not drain spontaneously.52
Incision and drainage is not the treatment of choice because of the high rate of recurrence.52 Definitive treatment consists of placement of a Word catheter (a 10 French sized rubber stem attached to an inflatable balloon).29,39,52,53 Placement of a Word catheter involves grasping the skin overlying the abscess with small forceps and making a 5 mm stab incision on the mucosal surface of the cyst within the introitus, but external to the hymenal ring.29,52 The abscess cavity often is deeper than most subcutaneous abscesses, so care must be taken to ensure entry into the abscess.39 Once in place, the catheter balloon is inflated with 2-3 mL of saline.52 The free end of the catheter can be placed into the vagina and the patient should be instructed to perform sitz baths 2-3 times daily until the infection clears.52 The catheter should be left in place for 4-6 weeks to allow for epithelialization of the tract, and the patient may have sexual intercourse with the catheter in place.52 In one study comparing a Word catheter to a Jacobi ring, neither was found to be superior.53 If significant cellulitis is present at the time of treatment, broad-spectrum antibiotics may be started. Patients should follow up with their obstetrician/gynecologist for monitoring of the Word catheter.52 Refractory cases require surgical treatment.
Necrotizing Fasciitis
Necrotizing fasciitis is a severe cellulitis with extensive involvement of the subcutaneous tissues and was first described in 1871 by Joseph Jones.25,54 There are many different terms for the pathophysiologic characteristics of necrotizing fasciitis, but all involve the same process of necrosis of underlying subcutaneous tissues.25 The tissue destruction of necrotizing fasciitis is caused by the release of bacterial exotoxins, which induce cytokine release from T-cells and macrophages.25 This cytokine release increases vascular permeability and causes edema, which results in tissue hypoxia and necrosis.25 Risk factors for necrotizing fasciitis include age over 50 years, male sex, peripheral vascular disease, diabetes, malnutrition, obesity, alcohol abuse, and IV drug use.55,56 The most commonly affected areas of the body are the perineum (20%), extremities (53%), and abdomen (18%).25,55 Most infections of necrotizing fasciitis are polymicrobial, with 15-29% involving a single organism.25 Monomicrobial infections commonly involve Group A beta hemolytic streptococcus (GABHS), S. aureus, and C. perfringens.55 There are two categories of bacterial involvement. Type 1 involves mixed anaerobic and facultative bacteria, and type 2 is a monomicrobial source and usually involves S. aureus or GABHS.55 The most common anaerobes involved are Peptostreptococcus, B. fragilis, and Prevotella.25 Enterococci also are present in 16-34% of cases.54 The most commonly involved gram-negative organisms include E. coli, Enterobacter, and Pseudomonas.54
Patients usually present a few days after the initiation of treatment for cellulitis.54 Necrotizing fasciitis usually presents as cellulitis with pain out of proportion to exam, and the pain often extends beyond the visible limits of the erythema.25,55 As the infection progresses, anesthesia over the affected skin develops secondary to infarction of the superficial nerves.28 Bullae, which are filled with clear fluid, develop initially and may progress to hemorrhagic fluid as a result of dermal necrosis.28 The infection spreads rapidly along fascial planes beyond the margins of the erythema.28 These patients often show signs of systemic toxicity and may be hemodynamically unstable.25,54,55 Plain radiographs may show gas in the soft tissues, but the absence of subcutaneous gas does not exclude the diagnosis.25 The imaging study of choice is a contrast enhanced T1 weighted MRI, which can aid in diagnosis and assessment of the extent of disease.25,57 Contrast enhanced fascia or hypointense signals in the muscles are characteristic MRI findings.57
The treatment for necrotizing fasciitis is early and aggressive surgical debridement, which can reduce mortality.25 A surgical consult should be called as soon as necrotizing fasciitis is suspected.25 Fluid resuscitation and broad-spectrum antibiotics are important to initiate as soon as possible.25,28,55 Antibiotics of choice include ampicillin-sulbactam, pipercillin-tazobactam, imipenem-cilastatin, penicillin with clindamycin, and ampicillin plus gentamycin plus clindamycin or flagyl.28,55 Clindamycin inhibits toxin production and therefore is a good adjunct to treatment.28 A tetanus booster should be administered if indicated.55 Hyperbaric oxygen treatment still remains controversial and is not recommended as a replacement for surgical debridement or IV antibiotics.55
Fournier's Gangrene
Fournier's gangrene is a term for necrotizing fasciitis involving the scrotal soft tissues.54,58 Diabetics, alcoholics, and patients with poor immune function are at increased risk for the development of Fournier's gangrene, and most patients have a primary source of infection such as a rectal abscess, a urinary source, or a testicular or skin infection.58 The mortality has been 15-52%, although over the last few decades mortality has been decreasing secondary to prompt and aggressive treatment.54 Surgical debridement within 24 hours of presentation can reduce mortality by up to 71%.54 Fournier's gangrene is most commonly polymicrobial with E. coli, Proteus, Pseudomonas, Streptococcus, S. aureus, Bacteroides, Clostridium, Salmonella, Klebsiella, Enterococcus, and anaerobes being common pathogens.58
Patients present with scrotal pain, erythema, and tenderness.58 Crepitus is present in up to 64% of patients, and plain radiography shows subcutaneous air in 89% of cases.58 Ultrasound may help differentiate scrotal cellulitis from early Fournier's gangrene by demonstrating gas in the soft tissue and thickening of the scrotal skin.58 Patients appear systemically ill and often have abnormal lab values.54 In one study a serum sodium less than 135, a BUN greater than 15, and a white blood cell count greater than 15,000 were predictive of necrotizing infection.54
Treatment of Fournier's gangrene consists of aggressive fluid resuscitation, empiric broad-spectrum antibiotics, aggressive control of blood sugar in diabetics, and wide debridement of tissues.54 A surgical consult should be called as soon as possible if Fournier's gangrene is suspected because early, wide debridement improves outcomes.54 The necrotizing infection is associated with decreased tissue oxygen tension, ischemia, and defective host cellular immunity. Hyperbaric oxygen treatment is controversial, but the increased oxygen tension is thought to reverse ischemia and improve host defense mechanisms.54 Hyperbaric oxygen therapy consists of wound exposure at 2.5-3 atms while breathing 100% O2 for 90 minutes every 8 hours for 24 hours, then twice per day for a minimum of 5 days. Hyperbaric oxygen is utilized in conjunction with IV antibiotics and surgical debridement.54 Elevated oxygen in hyperbaric oxygen treatment also enhances antibiotic agents by facilitating transport across the bacterial cell wall.54 Research involving the morbidity and mortality reduction associated with hyperbaric oxygen use is currently under way.54
Conclusion
Skin and soft-tissue infections continue to be common complaints of patients presenting to emergency departments. Many of these infections are superficial and can be managed with outpatient therapy. More extensive or deep infections, or those complicated by significant comorbidities, may require parenteral antibiotics and admission. Except for the situations discussed here, most cutaneous abscesses require only incision and drainage. With the increasing prevalence of MRSA and the increased utilization of treatment modalities such as hyperbaric oxygen, it is important for the clinician to remain current with the local pathogens and treatment options. Some serious soft-tissue infections can progress rapidly and lead to significant morbidity or death. The clinician must recognize these infections early and immediately initiate aggressive treatment and surgical intervention.59
References
1. Mancini AJ. Bacterial skin infections in children: The common and the not so common. Pediatr Ann 2000;29:26-35.
2. Brown J, Shriner DL, Schwatrz RA, et al. Impetigo: An update. Internat J Derm 2003;42:251-255.
3. Swartz MN. Cellulitis and subcutaneous tissue infections. Mandell, Douglas, and Bennett's Principles and Practices of Infectious Diseases. Orlando, FL: W.B. Saunders; 1995:909-928.
4. Schachner LA. Treatment of uncomplicated skin and skin infections in the pediatric and adolescent patient populations. J Drugs in Dermatology 2005;4(6 Supppl):s30-33.
5. Hedrick J. Acute bacterial skin infections in pediatric medicine: Current issues in presentation and treatment. Paediatr Drugs 2003;5 Suppl 1:35-46.
6. Rhody C. Bacterial infections of the skin. Primary Care: Clinics in Office Practice 2000;27:459-473.
7. Habif TP. Bacterial infections. In: Habif TP, ed. Clinical Dermatology—A Color Guide to Diagnosis and Therapy, 4th ed. New York: Mosby; 2004:267-278.
8. Watkins P. Impetigo:aetiology, complications and treatment options. Nursing Standard 2005;19:50-54.
9. Taylor JS. Interventions for impetigo. Am Fam Physician 2004;70:1680-1681.
10. Gilbert DN, Moellering RC, Eliopoulus GM, et al. The Sanford Guide to Antimicrobial Therapy 2005, 35th ed. Digital electronic version 5.0. Hyde Park, VT: Antimicrobial Therapy, Inc.; 2005.
11. Stulberg DL, Penrod MA, Blatny RA. Common bacterial skin infections. Am Family Physician 2002;66:119-124.
12. Mossad S. Cellulitis and erysipelas. Cleve Clin J Med 2004;71:129-143.
13. Holdiness MR. Erythrasma and common bacterial skin infections. Am Family Physician 2003;67:254.
14. O'Dell ML. Skin and wound infections: An overview. Am Fam Physician 1998;57:2424-2432.
15. Holdiness MR. Management of cutaneous erythrasma. Drugs 2002;62:1131-1141.
16. Wharton JR, Wilson PL, Kincannon JM. Erythrasma treated with a single-dose clarithromycin. Arch Dermatology 1998;134:671-672.
17. Miller SD, David-Bajar K. Images in clinical medicine. A brilliant case of erythrasma. N Engl J Med 2004;531:1666.
18. Khan MO, Montecalvo MA, Davis I, et al. Ecthyma gangrenosum in patients with acquired immunodeficiency syndrome. Cutis 2000;66:121-123.
19. Patel GK, Finlay AY. Staphylococcal scalded skin syndrome, diagnosis and management. Am J Clin Dermatol 2003;4:165-175.
20. Yamasaki O, Yamaguchi T, Sugai M, et al. Clinical manifestations of staphylococcal scalded-skin syndrome depend on serotypes of exfoliative toxins. J Clin Micronbiol 2005;43:1890-1893.
21. Patel GK. Treatment of staphylococcal scalded skin syndrome. Expert Rev Anti-infect Ther 2004;2:575-587.
22. Dobson CM, King CM. Adult staphylococcal scalded skin syndrome: Histological pitfalls and new diagnostic perspectives. British J of Derm 2003;148:1058-1087.
23. Johnston GA. Treatment of bullous impetigo and the staphylococcal scalded skin syndrome in infants. Expert Rev Anti-infect Ther 2004;2:439-446.
24. Simpson C. The management of staphylococcal scalded skin syndrome in infants. Nursing Times 2003;99:59-61.
25. Allen CH, Patel B, Endom EE. Primary bacterial infections of the skin and soft tissue, changes in epidemiology and management. Clinical Pediatric Emergency Medicine 2004;5:246-255.
26. Bhumbra NA, McCullough SG. Skin and subcutaneous tissue infections. Primary Care; Clinics in Office Practice 2003;30:1-24.
27. Valeriano-Marcet J, Carter JD, Vasey FB. Soft tissue disease. Rheum Dis Clin North Am 2003;29:77-88.
28. Vinh DC, Embli JM. Rapidly progressive soft tissue infections. Lancet Infectious Diseases 2005;5:501-513.
29. Meislin HW, Guisto JA. Soft tissue infections. In: Marx, ed. Rosen's Emergency Medicine: Concepts and Clinical Practice, 5th edition St. Louis: Mosby; 2002: 1944-1956.
30. Mills AM, Chen EH. Are blood cultures necessary in adults with cellulitis. Ann Emerg Med 2005;45:548-549.
31. Gilbert DN, Moellering RC, Eliopoulos GM, et al. The Sanford Guide to Antimicrobial Therapy. Sperryvill, VA: Antimicrobial Therapy Inc.; 2006: 38-41.
32. Vu BL, Dick PT, Levin AV, et al. Development of a clinical severity score for preseptal cellulitis in children. Pediatric Emergency Care 2003; 19:302-307.
33. Pasternack A, Irish B. Ophthalmic infections in primary care. Clinics in Family Practice 2004;6:19-34.
34. Laurence BG. Periorbital versus orbital cellulitis. The Pediatric Infectious Disease Journal 2002;21:1157-1158.
35. Tovilla-Canales JL, Nava A, Tovilla y Pomar JL. Orbital and periorbital infections. Curr Opin Ophthalmol 2001;12:335-341.
36. Jain A, Rubin P. Orbital cellulitis in children. International Ophthalmology Clinics 2001;41:71-86.
37. Goodyear PWA, Firth AL, Strachan DR, et al. Periorbital swelling: The important distinction between allergy and infection. Emerg Med J 2004;21:240-242.
38. Starkey CR, Steele RW. Medical management of orbital cellulitis. Pediatric Infectious Disease Journal 2001;20:1002-1005.
39. Butler KH. Incision and drainage. In: Roberts JR, Hedges JR, eds. Clinical Procedures in Emergency Medicine, 4th ed. Philadelphia: Saunders; 2004: 739-744.
40. Iyer S, Jones DH. Community acquired methicillin-resistant S. aureus skin infection: A retrospective analysis of clinical presentation and treatment of a local outbreak. J Am Acad Dermatol 2004;50:854-858.
41. Frazee BW, Charlebois LJ, Lambert L, et al. High prevalence of methicillin-resistant staphylococcus aureus in emergency department skin and soft tissue infections. Ann Emerg Med 2005;45:311-320.
42. Zetola N, Francis JS, Nuermberger EL, et al. Community acquired methicillin-resistant Staphylococcus aureus: An emerging threat. Lancet Infectious Disease 2005;5:275-286.
43. Schweiger ES, Weinberg JM. Novel antibacterial agents for skin and skin structure infections. J Am Acad Dermatol 2004;50:331-340.
44. Moran GJ, Talan DA. Community-associated methicillin resistant Staphylococcus aureus: Is it in your community and should it change practice? Ann Emerg Med 2005;45:321-322.
45. Calder KK, Severyn FA. Surgical emergencies in the intravenous drug user. Emerg Med Clin North Am 2003;21:1089-1116.
46. Ebright JR, Peiper B. Skin and soft tissue infections in injection drug users. Infect Dis Clin North Am 2002.;16:697-712.
47. Levine DP, Brown PD. Infections in injection drug users. In: Mandell GL, Bennett JE, and Dolin R, eds. Principles and Practice of Infectious Diseases, 6th ed. London: Churchill Livingstone; 2005:3463-3464.
48. Clark DC. Common acute hand infections. Am Fam Physician 2003;68:2167-2176.
49. Wright PE. Hand infections. In: Canale, ed. Campbell's Operative Orthopedics, 10th ed. St. Louis, MO: Mosby; 2003: 3811-3813.
50. Nemoto T. Abscess, breast. In: Ferri, ed. Ferri's Clinical Advisor: Instant Diagnosis and Treatment, 8th ed. St. Louis, MO: Mosby; 2006: 6.
51. Prasad AG. Pilonidal disease. In: Ferri, ed. Ferri's Clinical Advisor: Instant Diagnosis and Treatment, 8th ed. St. Louis, MO: Mosby; 2006: 644
52. Omole F, Simmons BJ, Hacker Y. Management of bartholin's duct cyst and gland abscess. Am Fam Physician 2003;68:135-140.
53. Gennis P, Li SF, Provataris J, et al. Jacobi ring catheter treatment of bartholin's abscess. Am J Emerg Med 2005: 414-415
54. Kuncir EJ, Tillou A, Hill CR, et al. Necrotizing soft tissue infections. Emerg Med Clin North Am 2003;21:1075-1087.
55. Headley AJ. Necrotizing soft tissue infections: A primary care review. Am Fam Physician 2003;68:323-328.
56. Hasham S, Metteucci P, Stanley PR, et al. Necrotizing fasciitis. BMJ 2005;330:830-833.
57. Soto T, Hagiwaro K, Matsuno H, et al. A case of necrotizing fasciitis caused by coagulase-negative staphylococcus: Utility of magetic resonance imaging for the preoperative diagnosis of necrotizing fasciitis. J Infect Chemother 2005;11:160-163.
58. Morrison D, Blaivas M, Lyon M. Emerging diagnosis of Fournier's gangrene with bedside ultrasound. Am J Emerg Med 2005;23:544-547.
59. Powers RD. Diagnosis and management of skin and soft tissue infections. Emerg Med Rep 1990;11:229-236.
This report begins with discussion of the most superficial lesions and progresses through the skin and soft tissue to the deeper and systemic disease processes.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.