An Update on Pediatric BLS and PALS Guidelines: The Current State of Affairs and Clinical Implications
An Update on Pediatric BLS and PALS Guidelines: The Current State of Affairs and Clinical Implications
Authors: Stephanie Doniger, MD, FAAP, Pediatric Emergency Medicine Fellow, Children's Hospital and Health Center/University of California, San Diego; Ghazala Q. Sharieff, MD, FACEP, FAAEM, FAAP, Director of Pediatric Emergency Medicine, Palomar-Pomerado Health System/California Emergency Physicians; Associate Clinical Professor, Children's Hospital and Health Center/University of California, San Diego
Peer Reviewer: James E. Colletti, MD, Assistant Professor, University of Minnesota, Associate Residency Director, Regions Hospital, St. Paul, MN
The 2005 American Heart Association (AHA) guidelines for cardiopulmonary resuscitation and emergency cardiovascular care of the pediatric patient and neonate are based on the evidence evaluation from the 2005 International Consensus Conference on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations, hosted by the AHA in 2005.
The scientific evidence was evaluated, and the recommendations are designed to improve survival from sudden cardiac arrest and acute life-threatening problems. The recommendations confirm the safety and effectiveness of many approaches, stratify treatment options, and suggest new treatments. The health care provider in charge of a resuscitation must be familiar with the changes and the evidence behind the recommendations, and be able to skillfully apply this information to the individual patient. The authors carefully review changes in pediatric Basic Life Support (BLS), neonatal care, and Pediatric Advanced Life Support (PALS), focusing on the issues of importance to clinicians and target areas where outcome may be affected.
— The Editor
Introduction
The AHA guidelines for 2005 were first released in December 2005. Several significant changes have been made, and new evidence has been reviewed to guide the management of cardiopulmonary resuscitation. This article will review all of the updates in BLS, neonatal care, and PALS.
Basic Life Support for Health Care Providers
The majority of the AHA guidelines for 2005 BLS emphasize effective cardiopulmonary resuscitation (CPR) (Table 1). The goal of early, high quality CPR and defibrillation is to improve overall survival from arrest. The single most important guideline change is that basic CPR is important with a focus on adequate compressions. Only one-third of hospital cardiac arrest victims receive CPR prior to emergency medical services (EMS) arrival.1 Those who actually receive CPR do not receive enough effective CPR. When CPR is performed, it is often suboptimal, with too few, too shallow (37%), and too weak chest compressions, and ventilations are often excessive (61%), with too many interruptions in chest compressions.2 Additional AHA recommendations have been simplified to include universal guidelines across all age groups.
| Table 1. BLS for Health Care Providers: Key Points |
|
|
The pediatric age group is defined to include those children 1 year of age to the onset of puberty. Puberty is determined by the presence of secondary sexual characteristics, which usually occurs between 12 and 14 years of age. For the lay rescuer, pediatric guidelines are applied to children between 1 and 8 years of age, with adult Advanced Cardiac Life Support (ACLS) guidelines applied to those patients older than 8 years.
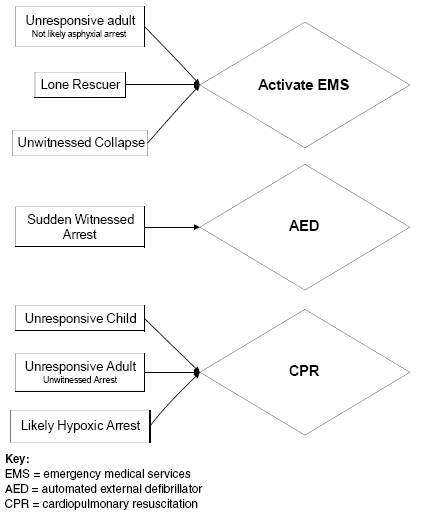
Priorities. The sequence of rescue actions depends upon the likely cause of arrest and the number of rescuers. Although described in previous AHA recommendations, this sequence is increasingly emphasized to improve patient survival. Figure 1 summarizes the management priorities in a patient with out-of-hospital cardiopulmonary arrest. It has been shown that CPR performed by a bystander at least doubles the survival rate.3
| Figure 1. Priorities in Out-of-Hospital Cardiac Arrest |
|
|
Airway. Assess the child for adequate breathing. In patients who do not exhibit adequate breathing, deliver rescue breaths rather than waiting for respiratory arrest. The guidelines recommend to try "a couple of times" to deliver 2 effective rescue breaths.4,5 In patients who are not breathing but who have a pulse, only respirations should be delivered, without compressions. Administer 12-20 breaths per minute (1 breath every 3-5 seconds) for infants and children, and 10-12 breaths per minute (1 breath every 5-6 seconds) for adults.
The preferred method of opening the airway for the lay rescuer is the head tilt-chin lift maneuver for both injured and non-injured victims. This method also is recommended for the health care provider in nontrauma settings. In trauma situations in which a cervical spine injury is suspected, a jaw thrust maneuver without a head tilt is recommended to open the airway and maintain manual stabilization of the head and neck. The jaw thrust is no longer recommended for lay rescuers because it is difficult to learn and perform, may be ineffective, and may cause spinal movement.6
Updated guidelines focus on maintaining a proper airway depending upon the skill level of the provider. Rescue breaths should be given over 1 second, with enough volume to create visible chest rise. There are no indications stating specific tidal volumes because it is difficult to estimate tidal volumes delivered during rescue breaths. In fact, much less tidal volume is required during resuscitation than in normal healthy individuals. During CPR, there is 24%-33% less blood flow to the lungs. Therefore, fewer breaths with smaller volumes are needed for oxygenation and ventilation.4,5
Foreign Body/Airway Obstruction. Additional recommendations include simplified guidelines for airway obstruction caused by a foreign body. Action must be taken in those cases of severe airway obstruction. Clinical signs of an airway obstruction include breathing difficulty, the inability to speak or breathe, poor air exchange, a silent cough, or poor air exchange. If the individual is unresponsive, it is recommended to activate EMS and perform CPR. The responder should inspect the mouth before administering rescue breaths. If a foreign body is seen, it should be removed. As stated in previous guidelines, it is unadvisable to perform blind finger sweeps. Further, it is no longer recommended to perform a jaw thrust maneuver. This technique is difficult, especially for inexperienced providers. The preferred method of opening the airway for the lay rescuer is the head tilt-chin lift maneuver. The recommendation still is to perform 5 back blows and 5 chest thrusts for infants and to perform the Heimlich maneuver in older children.
Circulation. Effective chest compressions are crucial in improving survival. Compressions provide blood flow to vital organs, such as the heart and brain during resuscitation. The AHA now recommends, "push hard and push fast". Interruptions in compressions should be limited to less than 10 seconds for interventions such as placing an advanced airway or defibrillation. Interruptions in compressions have been shown to decrease the rate of return to spontaneous circulation. Rhythm checks should be performed every 2 minutes, or 5 cycles of CPR. Once an advanced airway is in place, compressions and breaths should be performed continuously without interruption.
In those patients without a pulse, or newborns and children with a heart rate less than 60 beats per minute, compressions should be initiated (Figure 2). Because bradycardia is often a terminal rhythm in children, it is not necessary to wait for pulseless arrest to initiate compressions. Compressions should be performed at a rate of 100 per minute for all ages, except newborns, in whom compressions should occur at a rate of 120 per minute. The compression-to-ventilation ratio is 30:2 for single rescuers, while the ratio is 15:2 for two-rescuer health provider resuscitations in children. These universal rates simplify guidelines for providers and are best for all victims of cardiac arrest, including hypoxic arrests. Furthermore, it allows for sufficient time for adequate chest recoil, to allow for adequate cardiac filling and venous return.7
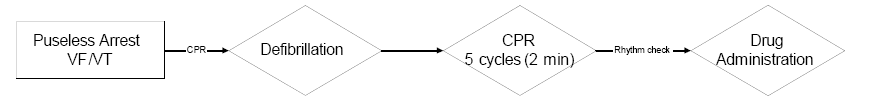
| Figure 2. Sequence of Resuscitation in Pulseless Arrest with Ventricular Fibrillation (VF) and Ventricular Tachycardia (VT) |
|
|
To perform adequate compressions for children, the heel of 1 or 2 hands can be used to compress the lower half of the sternum to a depth of one half to one-third of the chest diameter.8 For infants, the AHA now recommends that 2 thumbs press on the sternum, with the hands encircling the chest. In addition to compressing the sternum, the hands should squeeze the thorax. This improves coronary artery perfusion pressure and may generate higher systolic and diastolic blood pressures.9 When performing chest compressions, rescuers should be changed after 5 cycles of CPR or every 2 minutes to decrease rescuer fatigue. This switch should be performed in less than 5 seconds to minimize interruptions in CPR.10
The 2005 AHA algorithms focus on cardiac arrest secondary to ventricular fibrillation or rapid ventricular tachycardia. A single shock should be administered at a dose of 2 joules/kg, followed by immediate CPR. In at least 90% of cases, ventricular fibrillation is eliminated by the first shock.11 In those cases in which the first shock does not terminate ventricular fibrillation, CPR is of greater value. These algorithms will be discussed in more detail later.
In the community, automated external defibrillators (AEDs) have been shown to increase survival rates. There has been sufficient evidence to show that AEDs can be used safely for those patients older than 1 year.12 In a sudden witnessed collapse, the AED should be used as soon as it becomes available. However, if the collapse is unwitnessed, CPR should be performed for 5 cycles or 2 minutes, prior to the use of the AED. Pediatric AED pads and energy levels should be used in those patients 1-8 years of age. If the pediatric dose is unavailable, the adult dose is a reasonable alternative.
Neonatal Resuscitation
The major priority for neonatal resuscitation is to establish effective ventilation and oxygenation. The 2005 AHA guidelines focus on the use of oxygen during resuscitation, the clearing of meconium, and advanced airway techniques (Table 2).
| Table 2. Neonatal Resuscitation: Key Points |
|
|
Airway. Whenever positive pressure is indicated for resuscitation, supplemental oxygen is recommended. For those babies who are breathing but who have central cyanosis, free-flow oxygen is indicated. The standard is to use 100% FiO2. However, it is reasonable to begin with an oxygen concentration less than 100% or room air. However, if there is no improvement after 90 seconds, oxygen should be administered. This updated recommendation reflects the possible adverse effects that high-concentration oxygen has on the respiratory physiology and cerebral circulation of newborns.13 In the same token, oxygen deprivation and asphyxia cause further tissue damage. Therefore, the goal is to provide adequate oxygenation, which is a balance between oxygen delivery and tissue demand.
It is no longer recommended to perform oropharyngeal and nasopharyngeal suctioning of meconium-stained amniotic fluid at the perineum in a vigorous infant. A large multicenter trial showed that suctioning is ineffective in preventing meconium aspiration in this group of patients.14 Those infants who are not vigorous warrant endotracheal suctioning immediately after birth.
To ventilate a newborn, it is often necessary to provide positive pressure. This can be achieved with the use of a self-inflating bag, a flow-initiating bag, or a T-piece device. The T-piece is a valved device that regulates pressure and limits flow.15 The best indicator of successful ventilation is an increase in the heart rate. Once an advanced airway is placed, the preferred primary technique for placement confirmation is exhaled CO2 detection.16
Circulation. Compressions and ventilations should be given in a 3:1 ratio of compressions to ventilations, with 90 compressions and 30 breaths in 1 minute for a total of 120 events per minute. When compressions are given continuously, the rate should be 120 compressions per minute.
In newborn resuscitation, drug therapy is rarely indicated.17 However, the 2005 AHA recommendations for drug therapy in the newborn focus on the indications for the use of epinephrine and naloxone. High-dose epinephrine is no longer recommended but a dosage of 0.01-0.03 mg/kg/dose is appropriate. The intravenous (IV) concentration is 1:10,000 and the IV route for epinephrine administration is preferred. Although the endotracheal route for drug administration is unpredictable for drug delivery, it may be used when there is difficulty obtaining access. The endotracheal concentration of epinephrine is 1:1000. Naloxone should not be given endotracheally due to the lack of clinical data in newborns. Furthermore, naloxone is no longer recommended in primary resuscitative efforts. Heart rate and color must be restored first through ventilatory support prior to its administration.6
Post-Resuscitative Care. In previous years, there has been much controversy regarding temperature control in post-resuscitation care. Recent evidence is insufficient to recommend the routine use of systemic or selective cerebral hypothermia after resuscitation. However, it is important to avoid hyperthermia, especially in hypoxic-ischemic events and very low-weight infants. Currently, the recommendation is to maintain a normal body temperature. To help maintain normal body temperatures, polyethylene bags may be used.18
Lastly, neonatal recommendations include guidelines regarding withholding and withdrawing resuscitative efforts. Optimally, such decisions are best reached when there are opportunities for parental agreement. It may be reasonable to withhold resuscitation in those patients with conditions associated with an unacceptably high mortality. Such situations include extremely low birth weights (< 400 grams), young gestational age (< 23 weeks), and certain congenital anomalies (e.g., anencephaly, trisomy 13).6 Alternatively, resuscitation is indicated in those cases that have a high survival rate and an acceptable mortality. In other situations, the parental desires would dictate resuscitative efforts. Examples include babies with an uncertain prognosis, borderline survival, a relatively high morbidity rate, and a high anticipated burden to the child. Furthermore, after 10 minutes of adequate continuous resuscitation if there are still no signs of life (i.e., no heartbeat or respiratory effort), it is reasonable to discontinue resuscitation. This is due to evidence that shows that there is a high mortality rate and chance for severe neurodevelopmental disability.19
Pediatric Advanced Life Support (PALS)
The new 2005 AHA recommendations emphasize that good PALS begins with good BLS (Table 3).20
| Table 3. Pediatric Advanced Life Support: Key Points |
|
|
Airway. For those untrained in advanced airway, one must focus on effective bag-valve-mask technique. The use of laryngeal mask airways (LMA) during cardiac arrest does not have sufficient evidence to guide a recommendation for or against routine use. However, for those trained in endotracheal tube placement, it is the preferred route of securing an airway. Successful endotracheal intubation and minimal complications is associated with length of training, supervised experience in the operating room, ongoing experience, and the use of rapid sequence intubation (RSI). Because insertion of an advanced airway may cause a prolonged interruption in compressions, one must weigh the risks and benefits. Recommendations now include the option of using a cuffed endotracheal tube in all ages except neonates. A cuffed endotracheal tube is especially useful for those patients with poor lung compliance, increased airway resistance, or in those patients with a large glottic air leak.21 Attention must be paid to tube size, position, and pressures. For children 1-10 years of age, the endotracheal size can be calculated by the following formulas:22
The size of a cuffed endotracheal tube is determined by:
Size (mm ID) = (age in years/4) + 3
The size of an uncuffed endotracheal tube is determined by:
Size (mm ID) = (age in years/4) + 4
Once placed, the endotracheal tube cuff pressure should be maintained at less than 20 cm H2O.23 When endotracheal intubation is not possible, a reasonable alternative is the placement of an LMA. However, the placement of LMAs are associated with a higher incidence of complications in children.24
Confirmation of tube placement should include clinical assessment and auscultation of breath sounds. New recommendations include measuring the exhaled CO2 level. This can be measured by a calorimetric detector or by capnography; however, their use is limited to those patients exhibiting a perfusing rhythm.25 In those patients weighing more than 20 kg, one may consider esophageal detector devices for confirmation of tube placement.26 It is important to repeatedly verify endotracheal tube placement after the tube is inserted, during transport, and after the movement of the patient. A color change or the presence of a capnography wave form is used to confirm position in the trachea, but does not exclude right mainstem placement.
Once an advanced airway is in place, respirations should be administered simultaneously with chest compressions, at a rate of 8-10 per minute. Note that this rate is markedly lower than previous recommendations. Hyperventilation is not recommended because it can actually be harmful to the patient. Increased respiratory rates cause an increased intrathoracic pressure, thereby, decreasing venous return and coronary perfusion pressure. This has been shown to decrease survival rates.27 In addition, hyperventilation may cause air trapping and barotrauma in children with small-airway obstruction (e.g., bronchiolitis) and increases the risk of regurgitation and aspiration.
Circulation. IV or intraosseous (IO) routes are preferred for vascular access and for the administration of all drugs. Drug administration via the endotracheal tube is not recommended because drug delivery via it is unpredictable and results in lower blood concentrations than the same dose given intravascularly. In addition, recent animal studies suggest that the lower epinephrine levels achieved when the drug is given by the endotracheal route may produce detrimental adrenergic effects.28 However, if vascular access is unavailable, lipophilic drugs may be administered at higher doses through the endotracheal tube. These drugs include "LEAN" (lidocaine, epinephrine, atropine, and narcan).29 Flush with a minimum of 5mL normal saline followed by 5 assisted manual ventilations.
The recommendation for the first-line fluid for resuscitation of shock continues to be isotonic crystalloid solution (e.g., lactated Ringer's solution or normal saline) with no documented benefit to the use of colloids. Bolus therapy with a glucose-containing solution should be reserved for patients with documented hypoglycemia.
High-dose epinephrine is not recommended in any age group and is actually associated with a worse outcome, especially in cases of asphyxia.30 Therefore, the standard recommended dose is 0.01 mg/kg IV/IO for all doses, which correlates to 0.1 mL/kg. Although the preferred routes of administration are IV or IO, it may be given via the endotracheal tube when such access is unable to be obtained (0.1 mg/kg ETT). In exceptional cases, such as patients with beta-blocker overdoses, high-dose epinephrine may be considered.
The majority of cardiac arrest in children results from a progression of shock and respiratory failure to cardiac arrest. However, in situations of sudden witnessed collapse, immediate defibrillation is warranted, followed by CPR, followed by drug administration. CPR provides some blood flow, delivering oxygen and substrate to the heart muscle, thereby, making it more likely to abort ventricular fibrillation. CPR is beneficial immediately post-defibrillation. This 'primes' the heart for the next defibrillation attempt and also treats pulseless electrical activity (PEA). In cases of prolonged ventricular fibrillation, it has been shown that giving CPR prior to defibrillation increased survival rates from 4% to 22%.31 The dosages of defibrillation are now 2 joules/kg followed by 4 joules/kg for subsequent dosages, regardless of the type of defibrillator. It is important to note that stacked shocks are no longer recommended. A single shock is recommended, followed by CPR, largely due to the prolonged period of time to administer 3 shocks. Do not interrupt CPR until 5 cycles or 2 minutes for a pulse/rhythm check. In cases of asystole or PEA, one should initiate immediate CPR.
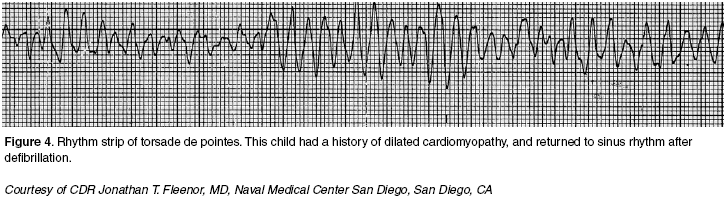
More specifically, the treatment of each rhythm disturbance can be classified according to the tachycardia algorithm (Figure 3). Of note, sinus tachycardia with adequate perfusion is no longer included in the algorithm. In addition, polymorphic ventricular tachycardia now is considered most likely to be an unstable rhythm (Figure 4). Therefore, it is recommended to use unsynchronized shocks rather than synchronized shocks. In contrast to previous recommendations, low energy synchronized shocks have a high likelihood of provoking ventricular fibrillation.1
| Figure 4. Rhythm Strip of Torsade de Pointes |
|
|
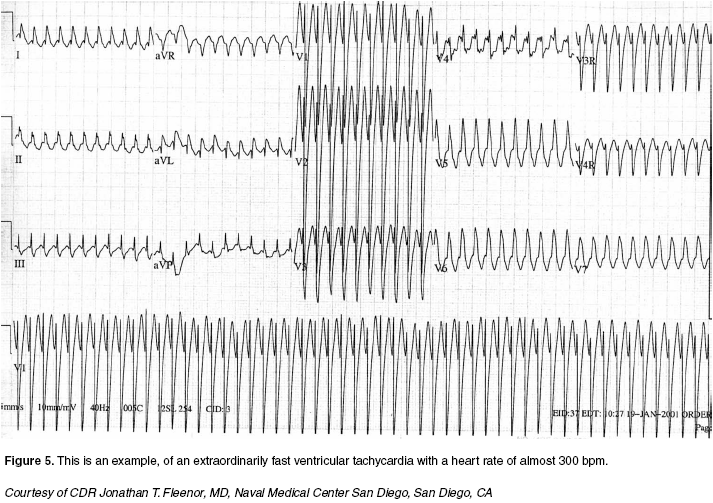
Medications. Generally, the algorithm drug dosages remain the same in the updated 2005 AHA recommendations. Drug delivery should not interrupt CPR. Amiodarone is the preferred drug for treatment for pulseless arrest (e.g., ventricular fibrillation, ventricular tachycardia, Torsade de pointes) because adult studies have shown it to be more effective. Amiodarone may cause hypotension, the severity of which is related to the infusion rate. The ECG needs to be monitored very carefully for complications that may include bradycardia, heart block, and torsade de pointes ventricular tachycardia. Lidocaine is only recommended when amiodarone is unavailable.32 Lidocaine has not been shown to be as effective as amiodarone for improving intermediate outcome measures (return of spontaneous circulation or survival to hospital admission) among adult patients with ventricular fibrillation refractory to a shock and epinephrine. Neither lidocaine nor amiodarone improves survival to hospital discharge in patients with ventricular fibrillation cardiac arrest. Additionally, lidocaine is no longer listed on the stable ventricular tachycardia algorithm. It has been replaced by amiodarone and procainamide. It is important to note that amiodarone and procainamide should not be administered together because they can lead to severe hypotension and prolongation of the QT interval (Figure 5).
| Figure 5. Ventricular Tachycardia |
|
|
The experience with the use of vasopressin in pediatric patients is limited, and the results from the adult studies have been inconsistent. Insufficient evidence exists to make a recommendation for or against the routine use of vasopressin during cardiac arrest.
Torsade de Pointes. (See Figure 4.)Torsade de pointes may be congenital or may occur secondary to toxicity with type IA antiarrhythmics or type III antiarrythmics, tricyclic antidepressants, digitalis, or drug interactions. Regardless of the etiology, it should be treated with a rapid infusion of magnesium.
Supraventricular Tachycardia (SVT). SVT should be treated with attempted vagal stimulation unless the patient is very unstable and it does not unnecessarily delay electrical or chemical cardioversion (Figure 6). In infants and young children, place ice to the face without occluding the airway. In older children, use a Valsalva maneuver (i.e., have the child blow through an obstructed straw). Do not apply pressure to the eye because this has the potential to damage the retina.
Special Situations. Trauma resuscitations in children have several unique features. The cervical spine should be immobilized whenever there is a potential injury. Hyperventilation is not recommended, even in children with a head injury. If signs of impending herniation are present, then brief hyperventilation may be used as a temporary therapy. Any child with an abdominal injury should be evaluated for a thoracic injury, and the re-verse is also true. If signs of shock persist after administration of 40 to 60 mL/kg of isotonic crystalloid, administer 10 to 15 mL/kg of blood. If possible, warm the blood prior to administration.
Toxicologic emergencies that pose unique resuscitation problems in pediatrics include an overdose of cocaine, narcotics, tricyclic antidepressants, calcium channel blockers, and beta-adrenergic blockers. The most frequent cocaine-related reason for hospitalization in adults is acute coronary syndrome.33 Specific guidelines for cocaine-related resuscitation include:
- Provide aggressive cooling.
- Administer nitroglycerin, a benzodiazepine, and phentolamine for coronary spasm.
- Do not administer beta-adrenergic blockers.
- In addition to standard treatments, consider sodium bicarbonate for ventricular arrhythmias.
- To prevent an arrhythmia secondary to a myocardial infarction, consider a lidocaine bolus and drip.
Specific guidelines for tricyclic antidepressant overdose-related resuscitation include:
- Treat with sodium bicarbonate to maintain alkalosis.
- Do not administer class IA or class III (amiodarone and sotalol) medications.
- Specific guidelines for hypotension-related resuscitation: Give boluses of normal saline. If a vasopressor is needed, epinephrine and norepinephrine are more effective than dopamine in raising blood pressure.
Specific guidelines for calcium channel blocker overdose-related resuscitation include:
- Treat with small boluses of normal saline for mild hypotension.
- Calcium administration has been shown to have variable efficacy.
- If the patient develops bradycardia and hypotension, consider a high-dose vasopressor such as norepinephrine or epinephrine.
Specific guidelines for beta-adrenergic blocker overdose-related resuscitation include:
- May be treated with high-dose epinephrine infusion
- Glucagon and an infusion of glucose and insulin may be considered.
Post-resuscitative Care. In general, it is recommended to avoid hyperthermia in resuscitative efforts. There are possible benefits of induced hypothermia (32°-34° C) for 12 to 24 hours following successful resuscitation.34 In addition, new recommendations recognize the probable benefits of vasoactive medications, including ionodilators (e.g., inamrinone, milrinone) to treat post-resuscitation myocardial depression. However, there may be adverse effects on cerebral circulation and hyperventilation. Further studies are recommended prior to universal initiation of these agents.35
There are no reliable predictors of outcome during resuscitation to guide when efforts should be terminated. Witnessed collapse, bystander CPR, and a short interval from collapse to arrival at professionals improves the chances of a successful resuscitation. Previously it was believed that children who did not have a return of spontaneous circulation after 2 doses of epinephrine were not likely to survive. New evidence suggests that the length of resuscitation is not an adequate prognostic indicator of survival. Intact survival has been reported, even in those cases with prolonged resuscitation and where 2 doses of epinephrine have been administered.36
Family Presence during Resuscitation. The majority of family members would like to be present during resuscitation.37,38 Families report comfort from being present at a family member's side at the end of life, improved adjustment after the event, and a willingness to participate again.39 Family members should be offered the opportunity, whenever possible, to be present during the resuscitation.40,41
Summary
This article reviews the recent changes in the pediatric BLS, neonatal, and pediatric advanced life support guidelines. The most significant change has been the focus on adequate compressions. The 'push hard, push fast' sentiment is prevalent throughout the changes and is indicative of the growing body of literature supporting a return to the CPR basics.
References
1. Donoghue A, Nadkarni V, Bern RA, et al. Out-of-hospital pediatric cardiac arrest: an epidemiologic review and assessment of current knowledge. Ann Emerg Med 2005;46(6):512-522.
2. Abella B, Alvarado JP, Mykiebust H, et al. Quality of cardiopulmonary resuscitation during in-hospital cardiac arrest. JAMA 2005; 293(3):363-365.
3. Herlitz J, Svensson L, Holmberg S, et al. Efficacy of bystander CPR: intervention by lay people and by health care professionals. Resuscitation 2005;66:291-295.
4. ECC Committee. Highlights of the 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Currents 2006; 15(4).
5. Hazinski M, et al. PALS Provider Manual. Dallas: American Heart Association;2002.
6. ECC Committee. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: Part 13: Neonatal Resuscitation Guidelines. Circulation 2005; 112:188-195.
7. Aufderheide T, Pirallo RG, Yannopoulos D, et al. Incomplete chest wall decompression: a clinical evaluation of CPR performance by EMS personnel and assessment of alternative manual chest compression-decompression techniques. Resuscitation 2005 64:355-362.
8. Stevenson AG, McGowna J, Evans, AL, et al. CPR for children: one hand or two? Resuscitation 2005;64:205-208.
9. Ishimine P, Menegazzi J, Weinstein D. Evaluation of two-thumb chest compression with thoracic squeeze in a swine model of infant cardiac arrest. Acad Emerg Med 1998;5:397.
10. Ashton A, McCluskey A, Gwinnutt CL, et al. Effect of rescuer fatigue on performance of continuous external chest compressions over 3 min. Resuscitation 2002;55:151-155.
11. Martens PR, Russell JK, Wolcke B, et al. Optimal Response to Cardiac Arrest study: defibrillation waveform effects. Resuscitation 2001;49:233-243.
12. Atkinson E, Mikysa B, Conway JA, et al. Specificity and sensitivity of automated external defibrillator rhythm analysis in infants and children. Ann Emerg Med 2003;42:185-196.
13. Tan A, Schulze A, O'Donnell CP, et al. Air versus oxygen for resuscitation of infants at birth. Cochrane Database Syst Rev 2004;3: CD002273.
14. Vain N, Szyid EG, Prudent LM, et al. Oropharyngeal and nasopharyngeal suctioning of meconium-stained neonates before delivery of their shoulders: multicentre, randomised controlled trial. Lancet 2004;364:597-602.
15. Allwood AC, Madar RJ, Baumer JH, et al. Changes in resuscitation practice at birth. Arch Dis Child Fetal Neonatal Ed 2003;88:F375-F379.
16. Repetto JE, Donohue PA-C PK, Baker SF, et al. Use of capnography in the delivery room for assessment of endotracheal tube placement. J Perinatol 2001;21:284-287.
17. Perlman JM, Risser R. Cardiopulmonary resuscitation in the delivery room. Associated clinical events. Arch Pediatr Adolesc Med 1995; 149:20-25.
18. Vohra S, Frent G, Campbell V, et al. Effect of polyethylene occlusive skin wrapping on heat loss in very low birth weight infants at delivery: a randomized trial. J Pediatr 1999;134(5):547-551.
19. Haddad B, Mercer BM, Livingston JC, et al. Outcome after successful resuscitation of babies born with apgar scores of 0 at both 1 and 5 minutes. Am J Obstet Gynecol 2000;182:1210-1214.
20. Nadkarni V. Revised AHA Guidelines: Scope of the Problem. Presented at: American Academy of Pediatrics National Conference & Exhibition;October 7-10, 2006;Atlanta, GA.
21. Newth CJ, Rachman B, Patel N, et al. The use of cuffed versus uncuffed endotracheal tubes in pediatric intensive care. J Pediatr 2004;144(3):333-337.
22. Khine HH, Corddry DH, Kettrick RG, et al. Comparison of cuffed and uncuffed endotracheal tubes in young children during general anesthesia. Anesthesiology 1997;86:627-631.
23. Hoffman RJ, Parwani V, Haha IH. Experienced emergency medicine physicians cannot safely inflate or estimate endotracheal tube cuff pressure using standard techniques. Am J Emerg Med 2006;24(2): 139-143.
24. Park C, Bahk JH, Ahn WS, The laryngeal mask airway in infants and children. Can J Anaesth 2001;48(4):413-417.
25. Bhende MS, Thompson AE, Orr RA. Utility of an end-tidal carbon dioxide detector during stabilization and transport of critically ill children. Pediatrics 1992;89(6 Pt 1):1042-1044.
26. Sharieff GQ, Rodarte A, Wilton N, et al. The self-inflating bulb as an airway adjunct: Is it reliable in children weighing less than 20 kilograms? Acad Emerg Med 2003;41:623-629.
27. Aufderheide TP, Lurie KG. Death by hyperventilation: a common and life-threatening problem during cardiopulmonary resuscitation. Crit Care Med 2004;32(9 Supp):S345-351.
28. Efrati O, Ben-Abraham R, Barak A, et al. Endobronchial adrenaline: should it be reconsidered? Dose response and haemodynamic effect in dogs. Resuscitation 2003;59(1):117-122.
29. Johnston C. Endotracheal drug delivery. Pediatr Emerg Care 1992; 8:94-97.
30. Perondi MB, Rels AG, Palva EF. A comparison of high-dose and standard-dose epinephrine in children with cardiac arrest. N Engl J Med 2004;350(17):1708-1709.
31. Wik LL, Kramer-Johansen J, Myklebust H. Quality of cardiopulmonary resuscitation during out-of-hospital cardiac arrest. JAMA 2005;293:299-304.
32. Dorian P, Cass D, Schwartz B. Amiodarone as compared with lidocaine for shock-resistant ventricular fibrillation. N Engl J Med 2002; 346:884-890.
33. Hollander JE, Hoffman RS, Gennis P, et al. Prospective multicenter evaluation of cocaine-associated chest pain. Cocaine Associated Chest Pain (COCHPA) Study Group. Acad Emerg Med 1994; 2(4):331-333.
34. Holzer M, Bernard A, Hachimi-Idrissi S, et al. Hypothermia for neuroprotection after cardiac arrest: systematic review and individual patient data meta-analysis. Crit Care Med 2005;33(2):1449-1452.
35. Abdallah I, Shawky H. A randomised controlled trial comparing milrinone and epinephrine as ionotropes in paediatric patients undergoing total correction of tetralogy of Fallot. Egyptian H Anaesthesia 2003;19:323-329.
36. Lopez-Herce J, Garcia C, Dominquez P. Characteristics and outcome of cardiorespiratory arrest in children. Resuscitation 2004;63:311-320.
37. Barratt F, Wallis DN. Relatives in the resuscitation room: their point of view. J Accid Emerg Med 1998;15:109-111.
38. Boie ET, Moore GP, Brummett C, et al. Do parents want to be present during invasive procedures performed on their children in the emergency department? A survey of 400 parents. Ann Emerg Med 1999; 34:70-74.
39. Doyle CJ, Post H, Burney RE, et al. Family participation during resuscitation: an option. Ann Emerg Med 1987;16:673-675.
40. Meyers TA, Eichhorn DJ, Guzzetta CE. Do families want to be present during CPR? A retrospective survey. J Emerg Nurs 1998;24:400-405.
41. Boyd R. Witnessed resuscitation by relatives. Resuscitation 2000; 43:171-176.
The AHA guidelines for 2005 were first released in December 2005. Several significant changes have been made, and new evidence has been reviewed to guide the management of cardiopulmonary resuscitation. This article will review all of the updates in BLS, neonatal care, and PALS.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.