Influenza and Influenza Vaccination 2006-2007
Influenza and Influenza Vaccination 2006-2007
Authors: Sharon G. Humiston, MD, MPH, FAAP, Associate Professor of Emergency Medicine and Pediatrics, University of Rochester, NY; and Sandra M. Schneider, MD, FACEP, Professor and Chair, Department of Emergency Medicine, University of Rochester, NY.
Peer Reviewers: Frederick M. Abrahamian, DO, FACEP, Assistant Professor of Medicine, University of California—Los Angeles School of Medicine; Director of Education, Department of Emergency Medicine, Olive View—UCLA Medical Center, Sylmar, CA; and Jonathan Glauser, MD, FACEP, Acting Director, Department of Emergency Medicine, Cleveland Clinic Foundation; Faculty, Emergency Medicine Residency Program, MetroHealth Medical Center, Cleveland, OH; and Catherine D. Bacheller, MD, Assistant Professor, Infectious Diseases, Booneshoft School of Medicine, Wright State University, Director, Infection Prevention Center, Kettering Medical Center, Dayton, OH.
Everyone who provides care for patients is well aware of the annual winter-spring spike in patient volume due to influenza disease and its complications. In recent years, concern has grown about the potential for the rapid worldwide spread of a relatively newly evolved influenza strain to which humans are not immune. This article will review the important clinical aspects of prevention and treatment as they relate to both the expected annual epidemic and the potential for a pandemic from the avian influenza (H5N1) strain.
—The Editor
Influenza Disease
Epidemiology. Mortality and Morbidity. Influenza can lead to death from pneumonia as well as exacerbations of underlying diseases of the heart, lungs, or other organ systems. More than 90% of deaths attributed to influenza and pneumonia occur in the elderly, and in the United States the mortality rate has been rising. According to Thompson, et al, the average number of influenza-associated deaths was approximately 19,000 per year from 1976 to 1990, but 36,000 per year from 1990 to 1999.1 It is considered probable that this increase was due in part to the increasing number of seniors and because the relatively virulent H3N2 strain predominated in 90% of the more recent influenza seasons. Death from influenza is uncommon among children, even those with high-risk medical conditions. Overall, an estimated average of 92 deaths occur annually from influenza among children younger than 5 years of age1—a number that is dwarfed by the incidence of influenza death later in life, but is tragic nonetheless. During the 2003-2004 influenza season there were 153 laboratory-confirmed influenza-related pediatric deaths reported from 40 states; 63% of these deaths were among children aged younger than 5 years, and among the fatal pediatric cases who were not infants (i.e., aged 2-17 years) 70% had no underlying medical condition previously associated with an increased risk for influenza-related complications.2
Rates of hospitalization depend in part upon the predominant influenza virus strain. For example, based on National Hospital Discharge Data, the hospitalization rates were twice as high during influenza A(H3N2) influenza seasons as during A(H1N1) or influenza B seasons. For example, for persons younger than 65 years of age the hospitalization rates were 42 per 100,000 during years when H3N2 predominated, but only 20 per 100,000 when less virulent strains predominated.3
Rates of hospitalization also depend on host factors such as age, underlying medical conditions, and socioeconomic status. Infants and seniors both have been shown to have high rates of influenza-associated hospitalization. In a study of healthy infants and young children with Medicaid insurance from 1973-1993 during influenza season, the mean annual excess number of hospitalizations per 10,000 for cardiopulmonary conditions was 104 (birth to 5 months of age), 50 (6 to less than 12 months), 19 (1 year to less than 3 years), 9 (3 to less than 5 years), and 4 (5 to less than 15 years). In the same study, children with high-risk conditions had higher hospitalization rates than age-matched healthy children.4 In a more recent study (1992-1997) of healthy infants and young children enrolled in a health maintenance organization (an indicator of relatively high socioeconomic status), the influenza-associated hospitalization rate per 100,000 was 144 to 187 in those from birth to 23 months of age.5
Influenza commonly causes less severe clinical illness in both children and adults. Influenza infection is a major cause of fevers and ear infections in children, and upper respiratory infections in both children and adults. These minor illnesses often lead to medical visits with primary care, urgent care, and even emergency care providers. Unfortunately, because illnesses caused by influenza viruses are difficult to distinguish from illnesses caused by other respiratory pathogens solely on the basis of signs and symptoms, these visits often culminate in antibiotic prescriptions and, it can be speculated, a rising incidence of antibiotic resistance. The visits, the prescriptions, and the antibiotic resistance all contribute to healthcare costs.
Epidemic Behavior. Influenza epidemics are an annual event and usually one or two influenza viruses predominate. It was previously believed that only one influenza A subtype could circulate at a time, but since 1978 both H1N1 and H3N2 viruses have circulated concurrently. Also, it appears that influenza viruses do not continue to spread within a population between epidemics,6 but must be reintroduced each season.
Geographic Distribution. Influenza has worldwide distribution, including tropical regions where influenza can occur throughout the year. In the temperate regions of the southern hemisphere, the majority of influenza activity occurs during April to September. It should be remembered that travelers can be exposed to influenza at any time of the year, especially when traveling as part of large tourist groups (e.g., cruise ships) that include persons from areas of the world where influenza viruses are circulating7,8 and that airplane travel makes circulation of the infectious agent rapid.
Temporal Distribution. In temperate climates, influenza is a cold weather disease. In the United States between 1976 and 2006, peak influenza activity was documented from November through April, with the most frequent peaks occurring in January (20%) and February (43%). Because a large proportion of the peaks were documented later in the season, the Advisory Committee on Immunization Practices (ACIP) has emphasized "that influenza vaccine should continue to be offered throughout the influenza season even after influenza activity has been documented in a community."9
Host Factors. Rates of serious morbidity and mortality from influenza are highest in persons aged 65 years and older, but school-aged children (ages 5-14 years) are the group most often infected. These young people may be the major source of infection for older people. Settings in which there is daily mixing of large numbers of susceptible people—such as day care, schools and colleges, military barracks, and nursing homes—create a fertile setting for the spread of influenza infection. There is no special susceptibility for any racial group or gender. The children of low-income families are at greater risk for influenza infection than are those of middle-income families10,11 presumably because of greater crowding. However, this intense exposure in childhood may lead to protection that persists into adulthood as suggested by lesser risk for influenza infection among low-income as compared to middle-income adults.12
Nosocomial Infections. Because large numbers of community members are infected during influenza season (including healthcare workers, hospital staff, patients, and visitors) and because influenza is transmitted easily from person to person, the potential for spread of influenza within a hospital is considerable. The occurrence and serious consequences of nosocomial influenza outbreaks are well documented. Vaccination of healthcare workers has been shown to be associated with fewer deaths among nursing home patients.13,14
Influenza Viruses. Influenza virus was the first human virus to be isolated and characterized. Influenza viruses are single-stranded RNA viruses, categorized as orthomyxoviruses. There are three types of influenza viruses: A, B, and C. Influenza A and B are the two types that cause epidemics in humans; influenza C tends not to cause significant disease. Both influenza A and B have two surface glycoproteins that are important for immune recognition: hemagglutinin and neuraminidase. Although these are not used for categorization of influenza B, the hemagglutinin and neuraminidase are key to categorization of influenza A. For example, A/Hong Kong/1/68 (H3N2) represents an influenza A virus first isolated in a laboratory in Hong Kong (culture number 1) in 1968 and determined to have a specific combination of hemagglutinin and neuraminidase antigens.15
The nucleus of influenza A contains eight gene segments that code for 10 proteins. The segmented nature of the genome allows for the frequent genetic reassortment that is considered the basis for the emergence of new subtypes of type A viruses. (Influenza B undergoes these changes less rapidly than influenza A.) When cells are simultaneously infected with two influenza A viruses with different genetic properties (e.g., a human and an avian virus), RNA assembly may incorporate gene segments from either parent. Minor antigenic changes (antigenic drift) occur when there are progressive alterations in antigenic sites that react with human antibodies. The constant antigenic flux of influenza viruses leads to annual epidemics. Major antigenic changes (antigenic shift) have been the basis for pandemics.
Mechanism and Route of Transmission. The virus is shed in respiratory secretions of an infected person for 5-10 days.16 The virus is highly contagious and generally spread from person to person via inhalation of airborne droplets elaborated during coughing or sneezing. Less commonly it is spread by contact with an infected person's secretions, for example, from hand to mouth via a doorknob or other inanimate object that has been recently handled by an influenza-infected person. Because influenza passes from person to person, it spreads easily during seasons when people stay indoors, such as winter in northern climates and the rainy season in the tropics.
Pathophysiology. Mechanism of Disease Process.12 Influenza is an acute infection of the respiratory tract. Influenza virus inhalation can lead to viral deposition in the upper or lower respiratory tract, with the latter probably being more susceptible. The influenza hemagglutinin attaches to influenza-specific receptors on mucoproteins in the airway's mucous coating. The influenza neuraminadase probably liquefies mucosal secretions and promotes influenza's access to the epithelial cells of the mucosa.
The incubation phase from exposure to shedding is 1-5 days. Shedding may precede symptoms by up to 1 day. Viral concentrations in respiratory secretions increase over the next 1-2 days and peak during the symptomatic peak of illness. Viral concentrations and disease severity correlate directly.
Disease Course. A simple case of influenza starts suddenly, causing fever, headache, muscle aches, and exhaustion, as well as respiratory tract symptoms such as sore throat, runny or stuffy nose, and dry cough. Very young children might also experience nausea and vomiting,17 but these symptoms are uncommon in adults with true influenza. (The term "stomach flu" is a misnomer; other viruses and bacteria cause nausea and diarrhea in adults.) Influenza infection usually is short-lived, averaging 2-3 days, but in some cases it can persist for weeks, especially among the elderly.
It should be noted that influenza B, which primarily affects children, generally causes milder disease than influenza A. This is, in large part, because the Type B virus shows more immunologic stability than Type A.
Potential Complications. Pneumonia is the chief complication of influenza. Pneumonia may be related primarily to influenza or to a bacterial complication from S. pneumoniae, S. aureus, or H. influenzae. Infection of cells by influenza A requires cleavage of the virus hemagglutinin by proteases. Some strains of S. aureus produce such proteases, possibly accounting for the frequency with which S. aureus pneumonia complicates influenza infections.18 Influenza may induce an exacerbation of chronic obstructive pulmonary disease or chronic bronchitis. In young infants, influenza infection may lead to croup or bronchiolitis. Upper respiratory infections such as sinusitis and otitis media may complicate the course.
Influenza has been implicated in cardiac disease such as myositis and myocarditis. Although the mechanism is not understood, the virus may induce neurologic symptoms including acute viral encephalitis, Reye's syndrome, and Guillain-Barré syndrome. Miscellaneous other complications attributed to influenza infection include a sepsis picture in infants, toxic shock syndrome, and myoglobinuria.
Immunity. Immunity to influenza depends upon immunity to surface antigens, including hemagglutinin. Immunity reduces both the likelihood of being infected and, if infected, the severity of clinical symptoms.19 Immunity to one antigenic variant does not guarantee protection against others and immunity to one influenza virus type confers limited or no protection against other types.20
Clinical Issues. Clinical Features. Patients may present with high fever, chills, malaise, myalgia, and headache. The chief complaint typically is respiratory, such as nasal congestion, rhinitis, sore throat, conjunctivitis, and nonproductive cough. Photophobia and shivering may be present. Gastrointestinal symptoms are not common in adults, but are reported more frequently in children. Cervical and generalized lymphadenopathy may be present.
Diagnostic Studies.21 The purpose of making an early diagnosis of influenza is to prevent unnecessary use of antibacterial antibiotics and maintain the opportunity for more directed antiviral therapy. From a public health perspective, laboratory diagnosis makes surveillance feasible, announcing the presence of influenza viruses in the community and identifying the predominant circulating types, subtypes, and strains.22
Tests for influenza include viral culture, rapid antigen testing, serology, polymerase chain reaction (PCR), and immunofluorescence (IF).23-25 There is a wide range of eligible specimens for viral isolation, including nasopharyngeal swab, throat swab, nasal wash, nasal aspirate, bronchial wash, and sputum. It should be kept in mind that bronchial wash and sputum should not be used for rapid diagnostic testing and that the nasopharyngeal specimens are more effective than throat swab specimens. Samples should be collected within the first four days of illness.
Viral cultures remain the gold standard. They are important adjuncts to rapid tests because they provide information on subtypes and strains and help monitor the emergence of antiviral resistance and novel influenza A subtypes that may become pandemic threats. Results typically take 5-10 days and, thus, are not useful in the acute situation.
Rapid antigen tests can detect the virus within 30 minutes to 1 hour, but have a lower sensitivity than culture. Some brands of rapid tests only detect influenza A and none of the rapid tests provide any information regarding influenza A subtypes. Because the sensitivity of rapid tests is lower than for viral culture, physicians should consider confirming negative tests with viral culture or other means; that is, the possibility of a false-negative rapid test results is considerable especially during periods of peak community influenza activity.26,27
Serology requires two samples—one obtained from the patient during the first week of illness and another 2-4 weeks later. The length of time necessary to note an antibody level rise excludes this test from helping in the decision to start antiviral treatment.
PCR may be used to detect viral RNA in respiratory secretions. The full range of specimens used for culture may be used for PCR. Results are usually available in 1-2 days.
Immunofluorescence DFA antibody staining and enzyme immunoassays (EIA) can both detect both influenza A and B and take about two hours.
Differential Diagnosis. Influenza often is difficult to distinguish based solely on clinical symptoms because the early influenza symptoms are similar to those of other infectious agents such as Mycoplasma pneumoniae, adenovirus, respiratory syncytial virus, rhinovirus, parainfluenza viruses, and Legionella.
Prevention. The most efficacious means to combat influenza is prevention. As is often stated, good hand washing is one of the cornerstones of prevention. The influenza virus is heavier than tuberculosis and it does not stay airborne for prolonged periods; airborne/TB precautions are not necessary in hospitals for influenza. However, healthcare workers are advised not to touch their eyes or nose after contact with an infected patient. Avoidance of ill persons and crowds seems prudent as a means for the general public to prevent influenza, but such methods have not been shown to be effective12 and may not be practical in modern society. It has been shown, however, that epidemics do not spread extensively unless schools are in session,12 so school closings may be in order for severe outbreaks. Vaccination is an important and cost effective—although underutilized—preventive tool and is detailed below. A list of persons for whom annual influenza vaccination is recommended is included. (See Table 1.)
| Table 1. Persons for Whom Annual Vaccination Is Recommended |
| • Children aged 6-59 months • Women who will be pregnant during the influenza season • Persons older than 50 years • Children and adolescents (aged 6 months-18 years) who are receiving long-term aspirin therapy and, therefore, might be at risk for experiencing Reye's syndrome after influenza infection • Adults and children who have chronic disorders of the pulmonary or cardiovascular systems, including asthma (hypertension is not considered a high-risk condition) • Adults and children who have required regular medical follow-up or hospitalization during the preceding year because of chronic metabolic diseases (including diabetes mellitus), renal dysfunction, hemoglobinopathies, or immunodeficiency (including immunodeficiency caused by medications or by human immunodeficiency virus) • Adults and children who have any condition (e.g., cognitive dysfunction, spinal cord injuries, seizure disorders, or other neuromuscular disorders) that can compromise respiratory function or the handling of respiratory secretions, or that can increase the risk for aspiration • Residents of nursing homes and other chronic-care facilities that house persons of any age who have chronic medical conditions • Persons who live with or care for persons at high risk for influenza-related complications, including healthy house-hold contacts and caregivers of children aged 0-59 months • Healthcare workers |
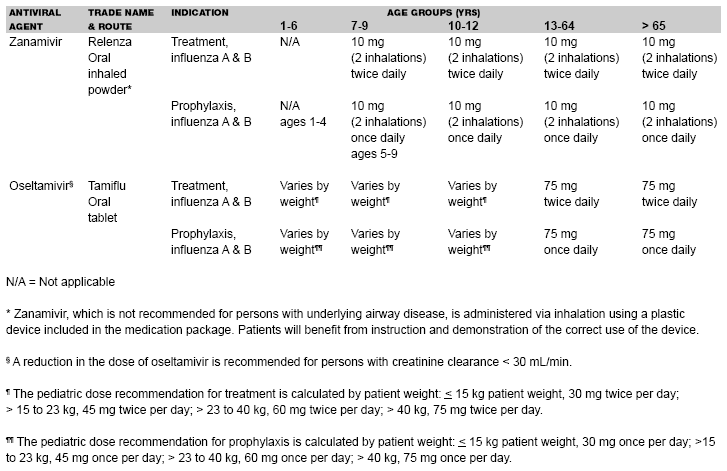
Use of Antiviral Medications. Four influenza antiviral agents are licensed for use in the United States: amantadine, rimantadine, zanamivir, and oseltamivir. Because of high levels of influenza A virus resistance to amantadine and rimantadine, ACIP recommends that neither be used until susceptibility has been re-established.28 Zanamivir and oseltamivir are neuraminidase inhibitors that are effective against both influenza A and B viruses. Both are approved for treatment of uncomplicated influenza virus infections and chemoprophylaxis of influenza in age groups as shown:
- Oseltamivir—Treatment older than 1 year of age, chemoprophylaxis older than 1 year of age;
- Zanamivir—Treatment older than 7 years of age, chemoprophylaxis older than 5 years of age.
The first table below shows the trade name, route of administration, indications, and recommended daily dosage of the two currently recommended influenza antiviral medications. The second table below shows the precautions and toxicities for these neuraminidase inhibitors.
| Table. Trade Name, Route of Administration, Indications, and Recommended Daily Dosage of Influenza Antiviral Medications |
|
| Table. Neuraminidase Inhibitors—Precautions and Toxicities |
|
Antiviral treatment should be given to any person experiencing a potentially life-threatening influenza-related illness and any person at high risk for serious complications of influenza who is within the first two days of illness. Additionally, treatment may be considered for other infected patients provided they meet the age requirements of the antiviral medications. Antiviral chemoprophylaxis should be given to all persons at high risk for serious complications of influenza if they are likely to exposed to others infected with influenza virus. In the event of an institutional outbreak, it should be given to those who live or work in institutions caring for people at high risk. Additionally, chemoprophylaxis can be considered for those who live in a community with circulating influenza if they:
- are at high risk and are not able to receive vaccine;
- are at high risk and received vaccine less than 2 weeks ago (less than 6 weeks ago if it was a first dose in a child younger than 9 years);
- have an immunosuppressive condition;
- are healthcare workers who have direct patient care responsibilities and are not able to receive vaccine.29
While these drugs may prevent symptoms and shorten the course of the illness by a day or two, they do not interfere with the development of immunity to the infecting influenza strain. It must be kept in mind that these are not panaceas. Their use may cause side effects and they are not licensed for use in infants before the first birthday. More resistant strains may develop, as occurred in 18% of Japanese children who received oseltamivir.30
The FDA has recently added warnings regarding some reported cases of delirium, hallucinations, and other unusual psychiatric behavior in patients treated with oseltamivir (Tamiflu). It is unclear at this time whether the behavioral changes are a result of influenza or the medication. The previous FDA-approved label mentioned only that "seizure and confusion" had been seen in some patients.
Influenza Vaccine
The Role of the Emergency Department in Vaccinating Patients. Although not a standard practice, there have been emergency departments (EDs) that have vaccinated patients against influenza since 1992. A sample of ED visits obtained from National Hospital Ambulatory Medical Care Survey data indicated that during the nine-year period from 1992-2000, approximately 247,000 influenza vaccinations were administered in the ED setting. In 77% of these cases, patients requested vaccination as their chief complaint. Clearly, because of the large number of chronically ill patients who seek care in the emergency setting, there is room for expansion of vaccination programs if resources permit.31 The new pediatric recommendation raises the possibility of offering influenza vaccine within the context of pediatric emergency medicine settings as well.32 Interestingly, paramedics have implemented influenza immunization programs in a host of settings such as retail stores, community events, EMS stations, churches, and senior citizen complexes.33
Two Types of Vaccines. There are two main types of influenza vaccine: the injectable inactivated and live intranasal spray vaccines. In both, the influenza viruses are grown in eggs so anaphylactic-type allergic reaction to chicken eggs is a true contraindication to all influenza vaccines produced currently. Both types of vaccine must be given annually. (See Table 2.)
| Table 2. Influenza Vaccines |
|
Inactivated Influenza Vaccine. Inactivated injectable vaccines against influenza have been in use since 1945. The inactivated influenza production process has improved with time and there are three products currently licensed in the United States. Description.The trivalent inactivated vaccine (TIV) is an injectable vaccine made from three strains of inactivated or killed influenza viruses along with added antibiotics to prevent bacterial contamination. Because of heated controversy around the use of thimerosal (a mercury-based preservative) in vaccines, a pediatric formulation that does not contain thimerosal as a preservative has been licensed.34
Dose. An intramuscular dose of 0.5 mL of TIV is recommended for children 3 years of age of age or older and for adults (irrespective of body weight). Children 6-35 months of age should receive only 0.25 mL. Children younger than 9 years of age who have never been immunized should receive two doses at least four weeks apart.
Vaccine Efficacy. The effectiveness of TIV depends on two main factors: host characteristics and the degree of similarity between circulating influenza virus and the vaccine virus. If there is a good match, the vaccine prevents influenza in 70-90% of healthy adults younger than 65.35,36 Among children younger than 16 years, a five-year study found TIV to be 77-91% efficacious in preventing influenza symptomatic illness culture positive for H1N1 and H3N2, respectively, but considerably less efficacious against influenza seroconversion.37 Conflicting results regarding the reduction of otitis media have been reported.38,39 Among non-institutionalized persons 60 years or older, influenza vaccine efficacy may be as low as 58% against influenza respiratory illness and even lower among that portion of the population older than 70 years of age.40 Among non-institutionalized persons 65 or older, inactivated influenza vaccine is 30-70% effective in preventing hospitalization for influenza and pneumonia41 and as much as 80% effective in preventing influenza-related death.
Adverse Reactions. Allergic reactions may occur after any vaccine or medicine. These are the other risks and side effects that have been known to occur after influenza vaccines:
- Minor pain or tenderness, swelling, or warmth at the injection site may begin soon after the shot and usually resolve within two days. Between 10% and 64% of people will experience these mild reactions.
- General side effects, including fever, muscle aches, or malaise may begin 6-12 hours after the shot and can persist for a day or two. More children than adults experience these symptoms. Overall, fewer than 1% of vaccine recipients report these side effects.
- Guillain-Barré syndrome, or GBS, causes potentially severe motor peripheral neuropathy. GBS affects 10-20 per million adults in the general population. In 1976, the swine influenza vaccine was associated with an increase in the basal incidence of GBS. Since then according to the CDC, "…if there is a risk of GBS from current influenza vaccines, it is estimated at 1 or 2 cases per million persons vaccinated."42 If a patient has a history of GBS and is at high risk of influenza complications, the CDC does not consider use of the inactivated vaccine as contraindicated.
- Bell's palsy was found to have a possible association with TIV in one study. The authors concluded that a population-based controlled study is needed to determine whether this association could be causal and to quantify the risk.43
Live Attenuated Influenza Vaccine. An influenza vaccine in the form of a nasal spray was licensed in the United States in 2003 for use by people between 5 and 49 years of age who are free of chronic illness. No needle is used so there is no risk of sharps injury, there is no pain at the injection site, and immunity develops in the lining of the nose as well as systemically.
Description. The live attenuated influenza vaccine (LAIV) is an intranasal spray comprised of live influenza virus that is, through serial culturing, cold-adapted. That is, it replicates in the cooler temperatures of the upper respiratory tract, but not in the warmer lower airways.15 There is only one intranasal influenza vaccine currently licensed in the United States and it has the trade name FluMist.44
Dose. Each syringe-like sprayer contains a 0.5 mL dose, with 0.25 mL to be sprayed into each nostril. Previously unimmunized children 5-8 years old should receive two doses at least six weeks apart.
Vaccine Efficacy. In one pre-licensure study of the LAIV during a season in which the circulating virus strains were well matched with those in the vaccine, efficacy was 93% among children receiving two doses.45 Among adults 18-49 years of age, when compared to placebo recipients, LAIV recipients had fewer upper respiratory infection episodes (26%) and consequently fewer lost days of work (27%), as well as fewer days of healthcare visits for febrile illness and antibiotic use.46
Adverse Reactions. Clinical signs and symptoms that occurred more frequently among children receiving LAIV than among those receiving placebo included nasal congestion or runny nose, headache, fever, vomiting, abdominal pain, and myalgias.47-49 In contrast, signs and symptoms that occurred more frequently among adults receiving LAIV than among those receiving placebo included nasal congestion or runny nose, headache, and sore throat. Serious adverse events were not associated with U.S.-licensed LAIV use.
Composition. The virus strains used for vaccine manufacture vary from year to year depending on a scientific prediction of what influenza viruses will circulate in the following winter. If there is a poor match between the influenza viruses chosen for the vaccine and those that actually make their way to North America, the vaccine may not be very effective. Fortunately, the influenza experts from WHO and CDC usually have been successful in predicting which strain will come each year.
The influenza viruses for the 2006-2007 vaccine include:9
- A/New Caledonia/20/1999 (H1N1)-like;
- A/Wisconsin/67/2005 (H3N2)-like (for this, manufacturers may use the antigenically equivalent A/Hiroshima/52/2005 virus); and
- B/Malaysia/2506/2004-like antigens (for this, manufacturers may use the antigenically equivalent B/Ohio/1/2005 virus).
Timing of Vaccination. Antibodies reach protective levels approximately two weeks after vaccination.50,51 For optimal protection during influenza season, vaccination has been recommended in October or November. For travelers to the southern hemisphere, it may be advisable to protect patients during April through September. There are two chief reasons that annual revaccination is necessary for protection. Firstly, immunity from the vaccine wanes after vaccination and may fall below the protective level within a year, or even within a few months in the elderly.52 Secondly, the predominant virus usually changes from year to year.
Who Should Receive Influenza Vaccine? The current list of individuals for whom the vaccine is recommended (as of July 2006) is detailed in Table 1.9 Healthcare workers with direct patient contact remain on the list of individuals for whom the influenza vaccine is recommended. Of course, most emergency personnel fit this description. Influenza vaccination is important for EMS and ED personnel not only because it limits absenteeism during a very busy season, but also to avoid putting patients, families, and others at risk.53
Who Should Not Receive the Influenza Vaccine?15 Several contraindications and precautions to vaccination are common to both the injectable and the intranasal influenza vaccines. These vaccines should not be given to a person if he or she:
- had a hypersensitivity reaction to a prior dose of influenza vaccine or to any vaccine component, including eggs. Appropriate allergy evaluation and desensitization is in order if the person is at risk for influenza complications.
- has a moderate to severe illness. If this is the case, the person may receive the vaccine when feeling better.
- is outside the age range for which the vaccine is licensed for use. The appropriate ages for the injectable influenza vaccine brands are 6 months of age or older for Fluzone, 4 years of age or older for Fluvirin, and 18 or older for Fluarix. The intranasal influenza vaccine, FluMist, is licensed for persons 5-49 years of age.
The live intranasal influenza vaccine has several more contraindications to its use. Additional groups of people who should not be given the spray include:
- all patients with a chronic illness.
- pediatric patients on chronic aspirin therapy. Since the nasal spray influenza vaccine is an attenuated live virus, children who take aspirin should not receive this vaccine because of the potential for inducing Reye's syndrome.
- persons with a personal past medical history of GBS.
- pregnant women.
- healthcare workers and other close contacts of patients who are in a protected environment because of severe immunosuppression (e.g., receiving a bone marrow transplant). This limitation is because of the remote risk for transmission of the vaccine-strain virus.
Note that pregnancy is not a precaution to injectable influenza vaccination. In fact, because of high rates of influenza complications in women late in pregnancy, influenza vaccine is recommended for women who will be pregnant during influenza season. In previous years, only a minority of pregnant women have been vaccinated.54
Can Influenza Vaccine Cause Infection? Many patients and healthcare providers believe that influenza vaccine causes influenza disease. There are no live viruses in the injectable influenza vaccine, so influenza infection resulting from the injectable vaccine is not possible. Recipients may mistake minor side effects such as fever and muscle aches as influenza. Patients should also be aware that, because the vaccine does not work for about two weeks after it is given, people who are vaccinated may get influenza infection if infected before being vaccinated. Finally, the vaccine does not protect against many other viruses that prompt influenza-like symptoms (e.g., adenovirus, respiratory syncytial virus, rhinovirus, and parainfluenza viruses).
Avian Influenza (H5N1)
Pandemic influenza is cyclic, occurring in 1918-1919 (25 million deaths in the United States), 1957-1958 (70,000 deaths in the United States), and in 1968 (34,000 deaths in the United States). There is great concern that the current influenza epidemic in the bird population will spread to humans and produce a pandemic of the magnitude of the 1918 experience.
Influenza A is common among birds. Of the 16 subtypes of hemagluttins, only H5 and H7 are highly pathogenic. In addition there are 9 neuraminidase subtypes. Only four of the subtypes cause human disease—H5N1, H7N7, H9N2, and H7N3. There is little to no human immunity to H5N1 and infection in humans has resulted in a high mortality rate.55
The first outbreak of H5N1 in birds was in 1996 in China.56 In 1997 an outbreak in Hong Kong led to 18 human cases, all from close contact with ill birds.57 An epidemic was avoided by the killing of the entire poultry population (1.5 million birds) within a three-day period.57 The current epidemic began in China and Southeast Asia in 2003 and is genetically distinct from the Hong Kong outbreak. The spread of the disease is thought to occur from the trade of ill birds and the migration of wild birds infected with the virus. Spread of the disease can be tracked on the website www.fao.org along with maps of the migration of wild birds.
H5N1 can be highly virulent or a low virulent form. Much of the disease in the wild bird population is less virulent, allowing the disease to spread with the migration of flocks. In the domestic bird population, the virus is highly virulent and very contagious. The virus is shed in respiratory and fecal materials, and infects water supplies, especially those shared between domestic and wild populations. There is evidence that the virus has increased in virulence in birds. It now survives for longer periods outside its host (35 days at 4°C, 48 hours on non-porous surfaces), and it is spreading into mammals.56,57 (See Table 3.)
| Table 3. Species in which H5N1 Has Been Isolated |
| • Domestic fowl including ducks, geese, chickens, turkeys • Wild fowl including ducks, geese, swans, gulls, storks, herons, doves, pigeons, buzzards, falcons, eagles, vultures, partridges, quail, pheasants, turkeys, finches, robins, crows, orioles, sparrows, magpies, starlings, flamingos, owls • Cats, civets, tigers, leopards • Martins, ferrets, rabbits, rats, pigs, emus • Humans |
Human Disease
To date human cases have been rare, with fewer than 200 worldwide, but with a mortality rate of 50%.55 Nearly all cases have been from direct, close contact with diseased birds. Human-to-human spread may have occurred recently in Indonesia (May 2006) where there was close contact within a household.58,59 However the virus is constantly mutating and there is concern that a mutation will make human to human spread more likely. Such change could occur either by spontaneous mutation or reassortment in which the virus accumulates some human genetic material that facilitates human infection.57 Reaasortment would make human-to-human transmission more likely. To date no reassortment has been detected.
Human cases present with lower respiratory symptoms, such as cough and fever and rapidly progress to respiratory failure, often requiring ventilator support. Most patients develop multi-organ failure. Antiviral medications appear to shorten the duration of disease but mortality remains very high.
The two main classes of antiviral medications are the M2 ion channel blockers—amantidine and ranitidine, and neuraminidase inhibitors—oseltamivir and zanimavir. Resistance to M2 drugs requires only a single amino acid substitution and therefore these medications are not considered appropriate for mass prophylaxis. The neuramidase inhibitors require several mutations before resistance is seen.60 Using complex epidemiological modeling, there is concern that widespread prophylaxis with neuramidase inhibitors will lead to the emergence of a resistant strain in a matter of weeks.60
Worldwide surveillance for both human and animal disease is ongoing, but clearly it is difficult in rural areas in developing nations. Cultures require 2-3 days, however a new PCR for the virus has been developed that requires only a few hours.61
To contain/eliminate the current pandemic it is recommended that flocks be isolated from potential exposure. Domestic birds should be kept isolated from wild birds and from watering systems used by wild birds. Purchased fowl and birds taken to market should be kept separate. Thorough cleaning of cages and fecal material is essential. Once disease enters a flock, the entire flock must be culled. The economic implication of culling flocks to date (150 million birds) is estimated to be in excess of $10 billion.56 Current avian vaccine is costly, requires multiple injections, and is not 100% effective. Improved vaccines are being created as well as vaccines that can be distributed by inhalation (fuming an entire building).
The current strategy to prevent disease in humans is to eliminate close contact between birds and humans. In areas where small flocks are allowed free range (including into households), such measures have proven unsuccessful. Eating poultry is safe, provided it is cooked properly.
Public Health Measures
We are currently at Pandemic Alert Level 3 with limited human to human transmission. This alert level can be monitored at www.who.int. This site also displays the cumulative human cases and their country. In a limited human outbreak, steps would be taken to isolate the infection. Vaccine, if available, would be used to form a ring of immunity around the infected area. Treatment with antiviral medications would be given to the ill and to exposed individuals.
Should the virus develop the ability to spread from human to human, a pandemic could occur rapidly. Using computer modeling, epidemiologists estimate the disease would spread throughout the United States within a period of 2.2 weeks.62 Based on previous pandemics, it is estimated that up to 20% of the population could be infected, with a mortality rate of 15-25%.55 Of note is that the current mortality rate is higher than this prediction and higher than in 1918. There is a fascinating calculator available on the Internet (www.cdc.gov/flu/flusurge.htm) to assess the impact to one's community.
Links to each state's plan can be found on www.pandemicflu.gov. Some states are concentrating on preparedness for mass illness (i.e., ventilators, beds, etc.), while others are concentrating on distribution systems for vaccine and/or antiviral medications. There is no available human vaccine, although extensive research and development is ongoing.
At a local level of a pandemic, the sick and exposed must be isolated from those who are well. Emergency departments should have a separate entrance for individuals with symptoms of influenza, and should screen patients before entry. In the first few weeks of the disease, healthcare workers who care for patients with flu should be quarantined from nonexposed individuals. All healthcare workers must wear masks and other personal protection, and will have high priority for antiviral prophylaxis and immunizations. Later, those workers who have recovered from the disease (and therefore developed immunity) may become the primary caregivers for the sick population.
There will be a significant attempt to isolate populations at home. Schools and other public meeting places will be closed. Nonessential businesses will be asked to close. Those who are sick will be asked to stay home unless their case requires hospital care.63
Unanswered Questions
Will healthcare workers respond to such a pandemic in appropriate numbers to care for a massive need? A recent survey suggested the response might be less than anticipated because of conflicting duties (multiple jobs, family responsibilities, etc.) or concern for personal safety. Hypothetical response to an epidemic ranged from 80% of healthcare workers for a mild disease with treatment to 18% for a potentially fatal disease with only experimental treatment.64 Will our crowded healthcare system be able to handle a surge of ill patients? Response to short duration or isolated surge events is feasible, but longer duration or more widespread events will clearly stretch resources. Isolation of the sick seems impossible in the crowded EDs and inpatient units. Another pandemic influenza may significantly stretch our healthcare system that is already challenged. (See Table 4.) The ability of the economy to sustain itself for a continuous 12-week epidemic has been speculated to be dismal. How will the owner of a "nonessential" business pay his employees? Who will compensate him for lost business? Who will provide care to schoolchildren or to the homebound sick?
| Table 4. Clinical Preparation for H5N1 |
| • Review your state plan at www.pandemicflu.gov • Review your departmental plan for mass casualty and infectious disease and verify that it contains: -Triage for influenza symptoms BEFORE entry into the department -Masks for symptomatic patients on entry into department (assure an adequate number) -Personal protection for healthcare workers (including masks, goggles, gowns, and gloves) -Separate treatment areas for influenza victims and well -List of healthcare workers who might be available (retired physicians, nurses, techs) -Surge plan for a sustained mass casualty event • Personal preparation (consider) -Home supplies for 3-12 weeks -Monetary access (liquid assets) |
Finally, will a human vaccine be available? There is extensive work to develop a human vaccine, but there are significant challenges. Clearly the virus may mutate before becoming pandemic in humans. Vaccines to date have shown relatively low antibody titers after injection. The vaccines are costly to manufacture and there are questions as to whether enough vaccine could be produced to immunize a significant portion of the world's population.65,66
Conclusions
Influenza viruses cause influenza, a highly communicable infectious disease. These viruses will never be eradicated because humans become susceptible again each time the viruses undergo an antigenic change. Influenza takes an enormous toll on humanity with respect to mortality, hospitalization, and medically attended illness. Despite the licensure of antiviral medications, immunization is the best strategy for controlling influenza. Because patients eligible for influenza vaccine frequent EDs, healthcare providers working in this arena should either offer the vaccine or refer patients following current guidelines to sites where the vaccine is obtainable.
Many experts believe another pandemic is quite possible if antigenic shift occurs and entire populations are without immunity to the new strain. A concerted worldwide effort to detect new strains as soon as they arise is in place, but at present there is no mechanism for the rapid mass production of relevant vaccine. Until difficulties in vaccine development and manufacturing can be solved, pandemics of influenza will be a threat so long as there are humans to serve as virus incubators and launch pads. When such a pandemic does occur, primary care and emergency department physicians will certainly be the first interface of the healthcare system with those infected. It is essential that physicians be familiar with influenza illness, modes of transmission, means of prevention, and therapeutic options. Even during typical yearly epidemics, the impact of influenza is not inconsequential. The drama of pandemics should not lead us to underestimate the destructive power influenza exhibits year after year in this country and throughout the world.
References
1. Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003;289:179-186.
2. Bhat N, Wright JG, Broder KR, et al. Influenza-associated deaths among children in the United States, 2003-2004. N Engl J Med 353;24:2559-2567.
3. Simonsen L, Fukuda K, Schonberger LB, et al. The impact of influenza epidemics on hospitalizations. J Infect Dis 2000;181:831-837.
4. Neuzil KM, Mellen BG, Wright PF, Et al. Effect of influenza on hospitalizations, outpatient visits, and courses of antibiotics in children. N Engl J Med 2000;342:225-231.
5. Izurieta HS, Thompson WW, Kramarz P, et al. Influenza and the rates of hospitalization for respiratory disease among infants and young children. N Engl J Med 2000;342:232-239.
6. Yorke JA, Nathanson N, Pianigiani G, et al Seasonality and the requirement for perpetuation and eradication of viruses in populations. Am J Epidemiol 1979;109:103-123.
7. Miller JM, Tam TW, Maloney S, et al. Cruise ships: High-risk passengers and the global spread of new influenza viruses. Clin Infect Dis 2000;31:433-438.
8. Uyeki TM, Zane SB, Bodnar UR, et al. Large summertime influenza A outbreak among tourists in Alaska and the Yukon Territory. Clin Infect Dis 2003;36:1095-1102.
9. Bresee JS, Shay DK, Uyeki TM, et al. Prevention and Control of Influenza. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Motal Wkly Rep July 28, 2006/55(RR10);1-42. Available at www.cdc.gov/mmwr/preview/mmwrhtml/rr5510a1.htm?s_cid=rr5510a1_e. Accessed September 1, 2006.
10. Glezen WP, Frank AL, Taber LH, et al. Influenza in childhood. Pediatr Res 1983;17:1029-1032.
11. Glezen WP, Paredes A, Taber LH. Influenza in children: Relationship to other respiratory agents. JAMA 1980;243:1345-1349.
12. Viral Infections Of Humans. In: Evans S, Kaslow RA, eds. 4th ed. New York: Plenum Publishing Corporation; 1997.
13. Carman WF, Elder AG, Wallace LA, et al. Effects of influenza vaccination of health-care personnel on mortality of elderly people in long-term care: A randomised controlled trial. Lancet 2000;355:93-97.
14. Potter J, Stott DJ, Roberts MA, et al. Influenza vaccination of health care workers in long-term-care hospitals reduces the mortality of elderly patients. J Infect Dis 1997;175:1-6.
15. Centers for Disease Control and Prevention. Epidemiology and Prevention of Vaccine-Preventable Diseases. Atkinson W, Hamborsky J, McIntyre L, Wolfe S, eds. 9th ed. Washington DC: Public Health Foundation, 2006.
16. Leblebicioglu H, Brook I. Influenza. eMedicine Updated May 18, 2006, at www.emedicine.com/ped/topic3006.htm. Accessed September 1, 2006.
17. American Academy of Pediatrics. Influenza. In: Pickering LK, ed. Red Book: 2006 Report of the Committee on Infectious Diseases. 27th ed. Elk Grove Village: American Academy of Pediatrics; 2006: 401.
18. Wong D. Influenza Viruses. www.virology-online.com/viruses/Influenza.htm. Accessed September 1, 2006.
19. Clements ML, Betts RF, Tierney EL, et al. Serum and nasal wash antibodies associated with resistance to experimental challenge with influenza A wild-type virus. J Clin Microbiol 1986;24:157-160.
20. Couch RB, Kasel JA. Immunity to influenza in man. Annu Rev Microbio.l 1983;37:529-549.
21. Centers for Disease Control and Prevention. Lab Diagnosis. www.cdc.gov/flu/professionals/labdiagnosis.htm. Accessed September 1, 2006.
22. Poehling KA, Zhu Y, Tang YW, Edwards K. Accuracy and impact of a point-of-care rapid influenza test in young children with respiratory illnesses. Arch Pediatr Adolesc Med 2006;160:713-718.
23. Bridges CB, Harper SA, Fukuda K, et al. Prevention and control of influenza. Recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep 2003;52:1-33.
24. Cox NJ, Subbarao K. Influenza. Lancet 1999;354:1277-1282.
25. Preboth M. ACIP releases guidelines on the prevention and control of influenza. Advisory Committee on Immunization Practices. Am Fam Phys 2001;64:1270-1275.
26. Storch GA. Rapid diagnostic tests for influenza. Curr Opin Pediatr 2003;15:77-84.
27. Uyeki TM. Influenza diagnosis and treatment in children: A review of studies on clinically useful tests and antiviral treatment for influenza. Pediatr Infect Dis J 2003;22:164-177.
28. CDC. CDC Recommends Against the Use of Amantadine and Rimantadine for the Treatment Or Prophylaxis of Influenza In the United States During the 2005-06 Influenza Season. Available at http://www.cdc.gov/flu/han011406.htm. Accessed on August 31, 2006.
29. CDC. Influenza Antiviral Medications: 2005-06 Interim Chemoprophylaxis and Treatment Guidelines. Available at www.cdc.gov/flu/professionals/treatment/0506antiviralguide.htm. Accessed on August 31, 2006.
30. Kiso M, Mitamura K, Sakai-Tagawa Y, et al. Resistance influenza A viruses in children treated with oseltamivir: Descriptive study. Lancet 2004;364:759-765.
31. Pallin DJ, Kim S, Emond JA et al. National study of pneumococcal and influenza vaccinations among adult emergency department patients, 1992 to 2000. [Abstract] Ann Emerg Med 2003;42:S70.
32. Pappano D, Humiston S, Goepp J. Efficacy of a pediatric emergency department-based influenza vaccination program. Arch Pediatr Adolesc Med 2004;158:1077-1083.
33. Mosesso VN, Packer CR, McMahon J, et al. Influenza immunizations provided by EMS agencies: The MEDICVAX Project. Prehosp Emerg Care 2003;7:74-78.
34. Food and Drug Administration. Product Approval Information—Licensing Action. www.fda.gov/cber/products/inflave090402.htm. Accessed September 1, 2006.
35. Bridges CB, Thompson WW, Meltzer MI, et al. Effectiveness and cost-benefit of influenza vaccination of healthy working adults: A randomized controlled trial. JAMA 2000;284:1655-1663.
36. Demicheli V, Jefferson T, Rivetti D, et al. Prevention and early treatment of influenza in healthy adults. Vaccine 2000;18:957-1030.
37. Neuzil KM, Dupont WD, Wright PF, et al. Efficacy of inactivated and cold-adapted vaccines against influenza A infection, 1985 to 1990: The pediatric experience. Pediatr Infect Dis J 2001;20:733-740.
38. Heikkinen T, Ruuskanen O, Waris M, et al. Influenza vaccination in the prevention of acute otitis media in children. Am J Dis Child 1991;145:445-448.
39. Hoberman A, Greenberg DP, Paradise JL, et al. Effectiveness of inactivated influenza vaccine in preventing acute otitis media in young children: A randomized controlled trial. JAMA 2003;290:1608-1616.
40. Govaert TM, Thijs CT, Masurel N, et al. The efficacy of influenza vaccination in elderly individuals. A randomized double-blind placebo-controlled trial. JAMA 1994;272:1661-1665.
41. Nichol KL, Wuorenma J, von Sternberg T. Benefits of influenza vaccination for low-, intermediate-, and high-risk senior citizens. Arch Intern Med 1998;158:1769-1776.
42. Centers for Disease Control and Prevention. Inactivated Influenza Vaccine Information Statement. www.cdc.gov/nip/publications/VIS/vis-flu.pdf. Accessed September 1, 2006.
43. Zhou W, Pool V, DeStefano F. A potential signal of Bell's palsy after parenteral inactivated influenza vaccines: Reports to the Vaccine Adverse Event Reporting System (VAERS)—United States, 1991-2001. Pharmacoepidemiol Drug Saf 2004;13:505-510.
44. Food and Drug Administration. CBER—Questions and Answers on FluMist (Influenza Virus Vaccine). www.fda.gov/cber/flu/flumistqa.htm. Accessed September 1, 2006.
45. Belshe RB, Mendelman PM, Treanor J, et al. The efficacy of live attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine in children. N Engl J Med 1998;338:1405-1412.
46. Nichol KL, Mendelman PM, Mallon KP, et al. Effectiveness of live, attenuated intranasal influenza virus vaccine in healthy, working adults: A randomized controlled trial. JAMA 1999;282:137-144.
47. Zangwill KM, Droge J, Mendelman P, et al. Prospective, randomized, placebo-controlled evaluation of the safety and immunogenicity of three lots of intranasal trivalent influenza vaccine among young children. Pediatr Infect Dis J 2001;20:740-746.
48. Redding G, Walker RE, Hessel C, et al. Safety and tolerability of cold-adapted influenza virus vaccine in children and adolescents with asthma. Pediatr Infect Dis J 2002;21:44-48.
49. Piedra PA, Yan L, Kotloff K, et al. Safety of the trivalent, cold-adapted influenza vaccine in preschool-aged children. Pediatrics 2002;110:662-672.
50. Gross PA, Russo C, Dran S, et al. Time to earliest peak serum antibody response to influenza vaccine in the elderly. Clin Diagn Lab Immunol 1997;4:491-492.
51. Brokstad KA, Cox RJ, Olofsson J, et al. Parenteral influenza vaccination induces a rapid systemic and local immune response. J Infect Dis 1995;171:198-203.
52. McElhaney JE, Gravenstein S, Upshaw CM, et al. Immune response to influenza vaccination in institutionalized elderly: Effect on different T-cell subsets. Vaccine 1998;16:403-409.
53. The National Foundation for Infectious Diseases (NFID). Influenza: Unimmunized health care workers put patients at risk. www.nfid.org/influenza/professionals_workersflu.html. Accessed September 1, 2006.
54. Wallis DH, Chin JL, Sur DK. Influenza vaccination in pregnancy: Current practices in a suburban community. J Am Board Fam Pract 2004;17:287-291.
55. World Health Organization web site. Avian influenza frequently asked questions. www.who.int/csr/disease/avian_influenza/avian_faqs. Accessed 10/20/2006.
56. Center for Infectious Disease Research and Policy. Avian influenza (bird flu): Agricultural and wildlife considerations. www.cidrap.umn.edu/cidrap/content/influenza/avianflu/biofacts/avflu.html. Accessed 10/20/2006.
57. World Health Organization. Avian influenza (bird flu) fact sheet. February 2006. www.who.int/mediacentre/factsheets/avian_influenza/en/print.html. Accessed 10/20/2006.
58. Ungchusak K, Auewarakul P, Dowell SF, et al. Probable person-to-person transmission of avian influenza A (H5N1). N Engl J Med 2005;352:333-340.
59. Center for Infectious Disease Research and Policy. Case for human spread of avian flu in Indonesia grows. www.cidrap.umn.edu/cidrap/content/influenza/avianflu/news/may2306indo.html. Accessed 10/20/2006.
60. Regoes RR, Bonhoeffer S. Emergence of drug-resistant influenza virus: Population dynamical considerations. Science 2006;312:389-391.
61. United States Department of Health and Human Services. FDA approves new laboratory test to detect human infections with avian influenza A/H5 Viruses. Press Release. Feb. 3, 2006. www.hhs.gov/news/press/2006pres/20060203.html.
62. Viboud C, Bjornstad ON, Smith DL, et al. Synchrony, waves and spatial hierarchies in the spread of influenza. Science 2006;312:447-451.
63. Bartlett JG. Planning for avian influenza. Ann Intern Med 2006:145:141-144.
64. Syrett JI, Benitez JG, Livingston WH III, et al. Will emergency healthcare providers respond to mass casualty incidents? Prehospital Disaster Medicine 2007 (in press).
65. Treanor JJ, Campbell JD, Zangwill KM, et al. Safety and immunogenicity of an inactivated subvirion influenza A (H5N1) vaccine. N Engl J Med 2006;354:1343-1351.
66. Poland GA. Vaccines against avian influenza—a race against time. N Engl J Med 2006;354:1411-1413.
Influenza can lead to death from pneumonia as well as exacerbations of underlying diseases of the heart, lungs, or other organ systems. More than 90% of deaths attributed to influenza and pneumonia occur in the elderly, and in the United States the mortality rate has been rising.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.