Selenium and Cancer
Selenium and Cancer
By Alexandra C. Frost, PhD Dr. Frost is a member of the Research Faculty, Dickson Institute for Health Studies, Carolinas Medical Center, Charlotte, NC; she reports no consultant, stockholder, speaker's bureau, research, or other financial relationships with companies having ties to this field of study.
Selenium was discovered in 1817 by the Swedish chemist, Jons Jacob Berzelius. He named this element "selene," the Greek word for "moon," because of its light conductive properties.1 In the 19th century it was used primarily as red glass coloring (CdSe), but later became a part of modern technology in photocopy machines and solar cells.1 Selenium is now recognized as a trace element.
Selenium is a naturally occurring element found in rocks, shale, sandstone, limestone, coal, soil, surface water, and plants. Selenium enters the food chain through plants; therefore, humans get selenium by either ingesting the products of these plants (e.g., wheat) or eating animals that feed on these plants (i.e., meat).2
Dietary sources of selenium include cereals, wheat, dairy products, meat, fish, and Brazil nuts. Selenium is less bioavailable in dairy products and meat than it is in grains. Selenium also occurs as selenate in foods such as beet leaves, garlic, and cabbage.1 The recommended daily allowance (RDA) of selenium is 55 mcg/d for women and 70 mcg/d for men. (One ounce of Brazil nuts supplies nearly 10 times the RDA.) The tolerable upper limit is 400 mcg/d and selenium deficiency is defined as less than 30 mcg/d.
Typical intakes range from 20 mcg/d in China, 35 mcg/d in Finland, to 50-200 mcg/d in North America. In the United States, plasma selenium levels generally range from 80 to 250 mcg/L,3 with an average consumption of 123 mcg/d.4 Unlike many minerals, blood levels are roughly equivalent to intake. Selenium is excreted through urine, feces, sweat, and skin loss. When intake is high, excretion is high; when intake is reduced, excretion is reduced.5
Role of Selenium in Health
While the importance of selenium in the diets of laboratory animals and livestock was recognized by the 1960s, it was not until 1979 that clear evidence established selenium as an essential element of human nutrition. The importance of selenium in the human diet was confirmed when a hospitalized woman on total parenteral nutrition (TPN) developed symptoms similar to white muscle disease (muscle pain, tenderness, atrophy) and was cured of her condition by an infusion of 100 mcg/d of selenium for a week.6
More evidence of the essential role of selenium in humans followed with investigations into the endemic cardiomyopathy (Keshan disease) that occurred until the 1980s in China. Supplementation of sodium selenite tablets dramatically reduced the incidence of this disease and continued supplementation in the selenium-poor region of China has virtually eliminated it.1,7 Sodium selenite tablets also remedied Kaschin-Beck disease (chronic, degenerative osteoarthritis), which was prevalent among children in the same area of China.8
As in animals, selenium toxicity (selenosis) is a problem in humans. Symptoms of selenosis include gastrointestinal upset, hair loss, white blotchy nails, and mild nerve damage.9 The most notable example of selenium toxicity was in the Republic of China from 1960 to 1964 during a period of drought.10 Residents of Enshi County ate more corn and high-selenium vegetables in lieu of rice (because of the drought) and began experiencing hair and nail loss and developed skin lesions on back of their hands and feet. Half of the residents of this area experienced symptoms. Analysis of urine from residents revealed average dietary consumption of 750 mcg/d.
Selenium and Cancer
Selenium emerged as a possible chemopreventive agent when it was observed that people living in areas with lower cancer mortality rates had higher blood selenium levels11,12 or higher diet/forage selenium.12-15 Strong data from carcinogenic studies supported this hypothesis. Combs and Combs reviewed more than 100 studies in a variety of experimental models and two-thirds of these studies reported that selenium compounds retarded or inhibited tumorigenesis.16 These findings laid the groundwork for what has become an extensive literature on selenium and cancer.
Possible Mechanisms of Anti-Carcinogenesis
Unlike other trace elements, the biological effects of selenium result from properties of its various compounds rather than properties intrinsic to the element.17 Selenium functions largely through an association with proteins, known as selenoproteins.3 Of the selenoproteins, the ones that have been studied most are the selenium-dependent glutathione peroxidases (GSH-Px1-4). Thirty-six percent of total selenium in the body is associated with GPX, which catalyzes the reduction of various organic peroxides as well as hydrogen peroxide.5 Increased selenium intake also boosts the production of selenium metabolites thought to have an anti-carcinogenic effect: hydrogen selenide, methylselenol, and selenodiglutathione (often called SDG).18 Difficulties in studying the effects of selenium stem from its multiple forms and complex metabolic and biochemical pathways.3
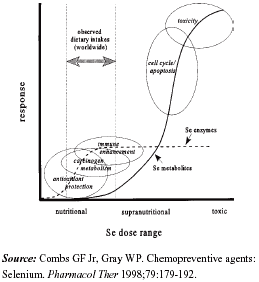
Results from a randomized clinical trial of selenium and skin cancer highlight the paucity of knowledge of selenium's biochemistry relevant to carcinogenesis.19 Basic scientists are now attempting to decipher the pathways through which selenium might influence cancer risk. Anti-carcinogenic pathways of selenium, hypothesized from laboratory studies, include the repair and prevention of oxidative damage, alteration of metabolism of carcinogenic agents, regulation of immune response, p53-independent apoptosis, and repair of DNA damage.20-23 It is likely that selenium acts as an anti-carcinogen through several mechanisms, which vary in importance based on nutritional and disease status of the individual (see Figure). The most commonly hypothesized mechanisms are discussed below.
| Figure. Potential anti-carcinogenic pathways of selenium |
|
|
Prevention of oxidative damage. Selenium-dependent GPX (glutathione peroxidase) is a selenoenzyme that protects cells from oxidative stress, genetic damage, and UVB irradiation by removing DNA-damaging hydrogen peroxide. The role of selenium enzymes (GPXs) in the antioxidant pathway was the initial hypothesis for selenium's role in cancer prevention24 and has received much attention. Evidence against anti-carcinogenic action through selenium's usual role as an essential nutrient (i.e., via GPX) is mounting. Two epidemiological studies, one of selenium and colorectal cancer and the other of selenium and colorectal adenomas found that GPX activity (selenium's action point on the antioxidant pathway) was not related to selenium status.25,26 Another found that GPX activity was only a good indicator of selenium status in populations with below-normal selenium consumption.27 Furthermore, forms of selenium that lack nutritional functions (synthesis of selenoproteins) show promise as chemopreventive agents.28 It is currently believed that selenium may decrease cancer risk in selenium-deficient humans by increasing expression of selenoproteins or enhancing immune surveillance. However, in selenium-adequate populations (e.g., most of the United States), it appears that GPX is not a major pathway through which selenium might reduce the risk of cancer.
Repair of DNA damage. A recent report suggested a strong candidate for selenium's mechanism of action: better DNA repair.23 High levels of selenomethionine, the primary organic form of selenium, triggered cells in culture to initiate DNA repair through activation of p53. After cells were exposed to selenomethionine the p53 activity was three times higher and the excision of base pairs (sign of DNA repair) was twice as high. This is an exciting observation because DNA repair is a key mechanism for preventing cancer. It might be possible, then, for people (with functional p53) to increase capacity for DNA repair simply by consuming more selenium.23 Unfortunately, the fact that p53 is mutated in most cancers may diminish the utility of this finding, in terms of cancer prevention.
Apoptosis. Apoptosis, or programmed cell death, has evolved in multi-cellular organisms to remodel tissue during development, maintain tissue homeostasis (proliferation-apoptosis balance), remove senescent cells, and delete cells with irreparable genetic damage. It has been suggested that certain diseases, such as cancer, evolve because of the inhibition of normal apoptosis.29-31 In fact, evasion of apoptosis has been described as one of the "six capabilities" of the cancer cell because the chances of neoplastic growth increase if mutated cells do not self-terminate.32 Apoptosis enhances the elimination of injured and dysfunctional cells that may arise because of oxidative stress and DNA damage.32
The possibility that selenium may increase anticancer apoptotic activity has been suggested by a number of carcinogenesis studies.33-41 Literature in this area is complicated by the fact that selenium is involved in many biochemical pathways, can exist in multiple forms, and can create a number of metabolites. Lanfear et al found that SDG induced apoptosis in addition to the accumulation of p53 protein in cells that contain normal p53.33 Stewart et al found that selenite (> 10 microM) induced cell differentiation and apoptosis in human colonic carcinoma cells.38 Researchers have been focused, recently, on determining the actions of selenium metabolites such as SDG and methylselenol.
Clinical and Community Trial Research
A number of clinical and community trials have been conducted to evaluate the association between selenium and cancer. Many studies, however, combined selenium with other nutrients such as vitamin E and beta-carotene, making it hard to evaluate the impact of selenium alone. The relationship between several micronutrients and esophageal cancer was studied in a series of large trials in Linxian, Henan Province, China, from 1984 to 1991. A 13% reduction in cancer mortality from lung, esophageal, and stomach cancer was noted in the group supplemented with a combination of 50 mcg of selenium, vitamin E, and beta carotene.42 In India, a trial was conducted among smokers with a combination of 50 mcg of selenium, vitamin A, riboflavin, and zinc.43 After one year of treatment, subjects with precancerous lesions had fewer precancerous lesions than those in the control group. Again, selenium was combined with other nutrients making it difficult to discern the effect of selenium alone.
In the Qidong Province of China a community intervention trial was performed from 1984 to 1992 to investigate the impact of selenium on the rate of hepatitis B virus (HBV) and primary liver cancer (PLC).44 This selenium-deprived community had problems with extremely high rates of both PLC and HBV. Table salt was fortified with 15 ppm of sodium selenite (30-50 mcg/d) for the duration of the study. The incidence of PLC was reduced from 50.4 to 27.2 per 100,000 and the rate of HBV was also significantly reduced.
Perhaps the strongest data supporting a reduction in cancer risk by selenium supplementation were produced in a multicenter, double-blind, randomized, controlled clinical trial of selenium and carcinoma of the skin from 1983 to 1996.19 The Nutritional Prevention of Cancer Trial (NPC Trial) recruited patients with a history of skin cancer from seven dermatology clinics in the eastern United States, and randomized them to 200 mcg/d of selenium (selenized yeast) or placebo. Participants were followed for an average of five years. Although there was no reduction in incidence of basal or squamous cell carcinomas of the skin, there was a substantial reduction in incidence of several other cancers. Strongest associations were seen with prostate cancer (relative risk [RR] = 0.4), colorectal cancer (RR = 0.4), and lung cancer (RR = 0.5).19 Results were recently published reflecting three additional years of follow-up to this study. The risk of both prostate and colon cancer remained lower in the selenium-supplemented group; however, the association between selenium and lung cancer was attenuated after the additional years of follow-up.45
Epidemiologic Studies
Highlighted below are key research results for the cancers that have received the most attention in the research community in terms of chemoprevention by selenium: prostate cancer, lung cancer, and colon cancer.
Prostate cancer. Results from studies on prostate cancer are very consistent46 and have been statistically combined in a recent meta-analysis. This analysis of 16 studies (11 cohort studies and five case-control studies) was conducted to determine the consensus from the literature. The pooled relative risk for prostate cancer of moderate intake (between second and third quartile or third and fourth quintile of distribution) was 0.7 (0.6-0.9) for cohort studies and 0.7 (0.4-1.4) for case-control studies.47 These results indicate that selenium is a promising chemopreventive agent for prostate cancer.
The Selenium and Vitamin E Cancer Prevention Trial (SELECT Trial) with a target recruitment of 32,400 participants is being conducted to confirm the results from epidemiologic studies.48 Participants are being randomized to one of four treatment arms: selenium, selenium and vitamin E, vitamin E, or placebo. Participants will receive the intervention for 7-12 years and will be followed for clinical incidence of prostate cancer. It is hoped that this simple dietary intervention (200 mcg l-selenomethionine) will reduce the risk of this very common cancer among men. Results are expected in 2013 and will also provide information on many other diseases potentially affected by selenium and vitamin E supplementation.
Lung cancer. Overall, the epidemiological data on selenium and lung cancer are mixed and are seen as less compelling since the recent publication of the updated NPC Trial data. These data show that the relationship between selenium and lung cancer incidence was attenuated and was no longer statistically significant after three additional years of follow-up.45 A recent meta-analysis, however, combined the results of 13 studies on selenium and lung cancer. Pooled results suggested that protective effects of selenium occur primarily in populations where overall selenium levels are low (RR = 0.7; 95% confidence interval [CI], 0.41-1.08).49 Because people in the United States generally do not have a problem with low selenium, other avenues of lung cancer prevention are probably more promising in this country.
Colorectal cancer. Mixed results have been reported from observational studies of selenium and colorectal cancer. Several studies reported a reduced risk of colon cancer for those with higher levels of selenium. In a study of men of Japanese ancestry in Hawaii, Nomura et al reported that those with high serum selenium (> 133 mcg/L) had a 45% reduced risk of having colorectal cancer in comparison with those with low serum selenium.50 Ghadirian et al found that participants with high toenail selenium (> 1 ppm) had a 58% reduced risk of colorectal cancer in comparison to those with low toenail selenium (odds ratio [OR] = 0.4; 95% CI 0.2-0.9).51 Knekt et al observed a decrease in relative risk of colorectal cancer for those in the highest fourth of serum selenium (> 49 mcg/L) vs. low serum selenium (< 49 mcg/L) in their male population (OR = 0.7), but reported a slight increase in risk for their female participants (OR = 1.3).52 Van den Brant et al did not find a statistically significant relation between toenail selenium and incidence of colon cancer; however, their results suggested a trend of reduced risk of colon cancer with increasing levels of toenail selenium (P = 0.10 for trend).53
A few studies reported increased risk with higher selenium levels. Nelson et al reported an increased risk of colorectal cancer with high selenium (> 144 mcg/L, OR = 1.7; 95% CI 0.5-5.9).54 Garland et al, using the Nurses Health Study data, reported that selenium in the upper third of selenium distribution (> 0.94 mcg/g) was associated with an increased risk of colon cancer (OR = 2.0; 95% CI 0.9-4.8).55 There were also a few studies with null or inconclusive results.56,57
The relation between selenium and colon cancer is debatable. The investigators of the SELECT trial plan to evaluate the incidence of colorectal cancer as a secondary endpoint. It will be interesting to see whether this large trial supports an association.
Conclusion
Research suggests that supplementing the diet with a moderate amount of selenium may reduce the risk of several cancers. More randomized clinical trials need to be conducted, optimal amounts of selenium need to be determined, and effective selenium compounds must be identified.
Recommendation
A great deal more research is needed before a solid recommendation for cancer risk reduction by selenium supplementation can be made. Selenium holds particular promise with respect to prostate cancer prevention; however, the future results from the SELECT Trial are a necessary part of the body of evidence. Even then, there is no magic supplement or pill that can take the place of a healthy, well-balanced diet. That said, a few Brazil nuts would be a nice addition to the diet. Two average sized brazil nuts provide 200 mcg of selenium, contain less than 50 calories, and provide poly- and mono-unsaturated fats that help reduce LDL cholesterol.
References
1. Sunde RA. Selenium. In: Stipanuk MH, ed. Biochemical and Physiological Aspects of Human Nutrition. London: W.B. Saunders Company; 2000:782-809.
2. Fan AM, Kizer KW. Selenium. Nutritional, toxicologic, and clinical aspects. West J Med 1990;153:160-167.
3. Food and Nutrition Board of the Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids: A report of the Panel on Dietary Antioxidants and Related Compounds, Subcommittees on Upper Reference Levels of Nutrients and the Interpretation and Use of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Washington, DC: National Academy Press; 2000.
4. Niskar AS, et al. Serum selenium levels in the US population: Third National Health and Nutrition Examination Survey, 1988-1994. Biol Trace Elem Res 2003;91:1-10.
5. Berdanier C.D. Trace Minerals. Advanced Nutrition: Micronutrients. New York: CRC Press; 1998:184-208.
6. van Rij AM, et al. Selenium deficiency in total parenteral nutrition. Am J Clin Nutr 1979;32:2076-2085.
7. Chen X. Selenium and cardiomyopathy (Keshan disease). Acta Pharmacol Toxicol (Copenh) 1986;59(Suppl 7):325-330.
8. Peng A, Yang CL. Examination of the roles of selenium in the Kaschin-Beck disease. Cartilage cell test and model studies. Biol Trace Elem Res 1991;28:1-9.
9. Koller LD, Exon JH. The two faces of seleniumdeficiency and toxicityare similar in animals and man. Can J Vet Res 1986;50:297-306.
10. Yang GQ, et al. Endemic selenium intoxication of humans in China. Am J Clin Nutr 1983;37:872-881.
11. Shamberger RJ, Frost DV. Possible protective effect of selenium against human cancer. Can Med Assoc J 1969;100:682.
12. Yu SY, et al. Regional variation of cancer mortality incidence and its relation to selenium levels in China. Biol Trace Elem Res 1985;7:21-29.
13. Clark LC, et al. Selenium in forage crops and cancer mortality in U.S. counties. Arch Environ Health 1991;46:37-42.
14. Schrauzer GN, et al. Cancer mortality correlation studiesIII: Statistical associations with dietary selenium intakes. Bioinorg Chem 1977;7:23-31.
15. Shamberger RJ, Willis CE. Selenium distribution and human cancer mortality. CRC Crit Rev Clin Lab Sci 1971;2:211-221.
16. Combs GF, Combs S. Selenium and Cancer. In: Combs GF Jr, ed. The Role of Selenium in Nutrition. San Diego, CA: Academic Press; 1986:413-462.
17. Ganther H, Ip C. Thioredoxin reductase activity in rat liver is not affected by supranutritional levels of monomethylated selenium in vivo and is inhibited only by high levels of selenium in vitro. J Nutr 2001;131:301-304.
18. Combs GF, Jr., et al. An analysis of cancer prevention by selenium. Biofactors 2001;14:153-159.
19. Clark LC, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA 1996;276:1957-1963. Erratum in: JAMA 1997;277:1520.
20. Combs GF Jr, Gray WP. Chemopreventive agents: Selenium. Pharmacol Ther 1998;79:179-192.
21. Ganther HE. Selenium metabolism, selenoproteins and mechanisms of cancer prevention: Complexities with thioredoxin reductase. Carcinogenesis 1999;20:1657-1666.
22. Ip C. Lessons from basic research in selenium and cancer prevention. J Nutr 1998;128:1845-1854.
23. Longtin R. Selenium for prevention: Eating your way to better DNA repair? J Natl Cancer Inst 2003;95:98-100.
24. Shamberger RJ. Increase of peroxidation in carcinogenesis. J Natl Cancer Inst 1972;48:1491-1497.
25. Clark LC, et al. Plasma selenium concentration predicts the prevalence of colorectal adenomatous polyps. Cancer Epidemiol Biomarkers Prev 1993;2:41-46.
26. Psathakis D, et al. Blood selenium and glutathione peroxidase status in patients with colorectal cancer. Dis Colon Rectum 1998;41:328-335.
27. Whanger PD, et al. Blood selenium and glutathione peroxidase activity of populations in New Zealand, Oregon, and South Dakota. FASEB J 1988;2:2996-3002.
28. Ganther HE, Robert JR. Chemical transformations of selenium in living organisms: Improved forms of selenium for cancer prevention. Tetrahedron 1997;53:12299-12310.
29. Sjostrom J, Bergh J. How apoptosis is regulated, and what goes wrong in cancer. BMJ 2001;322:1538-1539.
30. Vander A, et al. Human Physiology: The Mechanisms of Body Function. 8th ed. New York, NY: McGraw-Hill; 2001.
31. Wong BC, et al. Aspirin induced apoptosis in gastric cancer cells. Biomed Pharmacother 1999;53:315-318.
32. Hanahan D, Weinberg RA. The hallmarks of cancer. Cell 2000;100:57-70.
33. Lanfear J, et al. The selenium metabolite selenodiglutathione induces p53 and apoptosis: Relevance to the chemopreventive effects of selenium? Carcinogenesis 1994;15:1387-1392.
34. Lu J, et al. Selenite induction of DNA strand breaks and apoptosis in mouse leukemic L1210 cells. Biochem Pharmacol 1994;47:1531-1535.
35. Redman C, et al. Involvement of polyamines in selenomethionine induced apoptosis and mitotic alterations in human tumor cells. Carcinogenesis 1997;18:1195-1202.
36. Redman C, et al. Inhibitory effect of selenomethionine on the growth of three selected human tumor cell lines. Cancer Lett 1998;125:103-110.
37. Shen HM, et al. Sodium selenite-induced oxidative stress and apoptosis in human hepatoma HepG2 cells. Int J Cancer 1999;81:820-828.
38. Stewart MS, et al. Induction of differentiation and apoptosis by sodium selenite in human colonic carcinoma cells (HT29). Cancer Lett 1997;117:35-40.
39. Stewart MS, et al. Selenium compounds have disparate abilities to impose oxidative stress and induce apoptosis. Free Radic Biol Med 1999;26:42-48.
40. Waters DJ, et al. Effects of dietary selenium supplementation on DNA damage and apoptosis in canine prostate. J Natl Cancer Inst 2003;95:237-241.
41. Zhong W, Oberley TD. Redox-mediated effects of selenium on apoptosis and cell cycle in the LNCaP human prostate cancer cell line. Cancer Res 2001;61:7071-7078.
42. Li JY, et al. Nutrition intervention trials in Linxian, China: Multiple vitamin/mineral supplementation, cancer incidence, and disease-specific mortality among adults with esophageal dysplasia. J Natl Cancer Inst 1993;85:1492-1498.
43. Krishnaswamy K, et al. A case study of nutrient intervention of oral precancerous lesions in India. Eur J Cancer B Oral Oncol 1995;31B:41-48.
44. Yu SY, et al. Protective role of selenium against hepatitis B virus and primary liver cancer in Qidong. Biol Trace Elem Res 1997;56:117-124.
45. Duffield-Lillico AJ, et al. Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial: A summary report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiol Biomarkers Prev 2002;11:630-639.
46. Chan JM, et al. Role of diet in prostate cancer development and progression. J Clin Oncol 2005;23:8152-8160.
47. Etminan M, et al. Intake of selenium in the prevention of prostate cancer: A systematic review and meta-analysis. Cancer Causes Control 2005;16:1125-1131.
48. Klein EA. Selenium and vitamin E cancer prevention trial. Ann N Y Acad Sci 2004;1031:234-241.
49. Zhuo H, et al. Selenium and lung cancer: A quantitative analysis of heterogeneity in the current epidemiological literature. Cancer Epidemiol Biomarkers Prev 2004;13:771-778.
50. Nomura A, et al. Serum selenium and the risk of cancer, by specific sites: Case-control analysis of prospective data. J Natl Cancer Inst 1987;79:103-108.
51. Ghadirian P, et al. A case-control study of toenail selenium and cancer of the breast, colon, and prostate. Cancer Detect Prev 2000;24:305-313.
52. Knekt P, et al. Serum vitamin E, serum selenium and the risk of gastrointestinal cancer. Int J Cancer 1988;42:846-850.
53. van den Brandt PA, et al. A prospective cohort study on toenail selenium levels and risk of gastrointestinal cancer. J Natl Cancer Inst 1993;85:224-229.
54. Nelson RL, et al. Serum selenium and colonic neoplastic risk. Dis Colon Rectum 1995;38:1306-1310.
55. Garland M, et al. Prospective study of toenail selenium levels and cancer among women. J Natl Cancer Inst 1995;87:497-505.
56. Poole C. High Dietary Selenium Intake and Incidence of Cancers of the Lung, Breast and Large Bowel. Cambridge, MA: Harvard School of Public Health; 1989.
57. Schober SE, et al. Serologic precursors of cancer. I. Prediagnostic serum nutrients and colon cancer risk. Am J Epidemiol 1987;126:1033-1041.
Frost AC. Selenium and cancer. Altern Med Alert 2006;9(3):30-35.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.