Omega-3 Fatty Acids for the Treatment of Rheumatoid Arthritis
Omega-3 Fatty Acids for the Treatment of Rheumatoid Arthritis
By Susan T. Marcolina, MD, FACP Dr. Marcolina is an internist and geriatrician in Issaquah, WA. Dr. Marcolina reports no consultant, stockholder, speaker's bureau, research, or other financial relationships with companies having ties to this field of study.
The use of omega-3 dietary fats combined with the standard medication therapy for management of rheumatoid arthritis (RA) has the potential to directly improve quality of life, decrease side effects, and collaterally decrease mortality from cardiovascular disease. Patients with RA generally do not have the traditional cardiovascular risk factors of hypertension, diabetes, and hyperlipidemia. Yet they do have an increased risk of death from cardiovascular disease relative to age- and sex-matched controls in the general population.1,2 This is particularly true for women younger than age 65 and for seropositive (rheumatoid factor-positive) individuals.3
Inflammation is a root pathophysiologic process for both RA and coronary heart disease. Since the chemical mediators of the inflammatory process are lipid derivatives, manipulation of the composition of dietary fats has become an important therapeutic tool that can be used adjunctively with the standard pharmacologicssuch as nonsteroidal anti-inflammatory drugs (NSAIDs), steroids, slow-acting antirheumatic drugs (SAARDs), disease-modifying anti-rheumatic drugs (DMARDs), and biologicsto mitigate the production of the inflammatory milieu in the joint space and the endothelium of blood vessels.
The dietary fats studied for this purpose include the omega-3 polyunsaturated fatty acids (PUFAs), specifically eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) found in fish oil, as well as alpha-linolenic acid (ALA) from plant sources.
Categories of Fat
Fatty acids are compounds of carbon, hydrogen, and oxygen with a chemical structure that renders them hydrophobic (water insoluble). They are categorized as saturated (no double bonds in the carbon chain), monounsaturated (one double bond), or polyunsaturated (more than one double bond). These chemical structures are responsible for their physical properties. Saturated fats, such as butter and marbling in animal meats, form solids at room temperatures. Monounsaturated fats, such as olive oil, are liquid at room temperature but form solids when refrigerated. Polyunsaturated fats, found in grain and seed oils, are liquid both at room temperature and when refrigerated.
Polyunsaturated fatty acids are further classified as omega-3 and omega-6 PUFAs. The biochemical numbering system for the carbon atoms of fats depends on the location of the first double bond with respect to the methyl (omega) end of the fatty acid molecule. For instance, the omega-3 PUFAs have their first double bond located at the third carbon atom; the omega-6 PUFAs have the first double bond at the sixth carbon position; and oleic acid, an omega-9 fat contained in olive oil, has its only double bond located at the ninth carbon position. Linolenic acid (LA) and ALA are considered essential fatty acids because they cannot be synthesized by humans and must be ingested through the diet.4,5
PUFA Dietary Composition
Today's typical Western diet contains a preponderance of omega-6 fatty acids relative to omega-3 fatty acids. In the early 1900s, the ratio of omega-6 to omega-3 fatty acids was 4:1. This has increased dramatically to greater than 25:1 due to the partially hydrogenated fatty acids used to extend the shelf life in snack and processed convenience foods. These trans fats are a major source of omega-6 fatty acids; other sources include beef, poultry, nuts, grains, and vegetable and seed oils. Since omega-6 PUFAs are subunits of arachidonic acid, the body's primary inflammatory precursor, this dietary change may play a role in the high incidence of chronic inflammatory conditions seen today.6
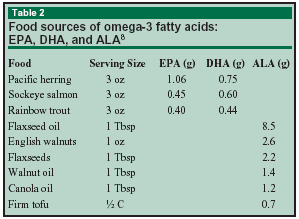
Table 1 lists the major types of oils used in food preparation and the primary type of PUFA they supply.7 Soybean oil is included in both categories because it is higher in omega-6 fatty acids than most omega-3 oils. Table 2 lists food sources of omega- 3 fatty acids.8
 |
 |
Pharmacologic Actions of Omega-3 Fatty Acids
EPA and DHA are precursors to a group of eicos-anoids (prostaglandins, leukotrienes, and thromboxanes) which have anti-inflammatory, antithrombotic, and vasodilatory effects. Although EPA and DHA also can be produced endogenously from dietary ALA, the conversion process is inefficient because ALA and omega-6 fatty acids are competitive substrates for the rate-limiting enzyme delta-6 desaturase, which controls the metabolic conversion of the omega-6 and omega-3 PUFAs into their respective eicosanoids. The greater concentration of the dietary omega-6 fatty acids, especially arachidonic acid, favors its formation into a group of eicosanoids that have proinflammatory and prothrombic effects such as tumor necrosis factor-alpha (TNF-alpha), thromboxane A2, prostaglandin E2, and leukotriene B4.9 The EPA found in fish oil competitively inhibits arachidonic acid metabolism, which results in the synthesis of less thrombogenic and less inflammatory eicosanoids.10
Eicosanoids are short-lived, lipid-derived mediators responsible for the profound biological responses that occur in acute inflammation. In general, those derived from the omega-3 fatty acids have biological potencies for the induction of cellular inflammatory responses that are 1/10 to 1/100 of those derived from the omega-6 fatty acids.11
Pathophysiology of Eicosanoid Immunomodulatory Functions in RA and Cardiovascular Disease
The arachidonic acid-derived eicosanoids, such as TNF-alpha and interleukin-1 beta (IL-1 beta), contribute to the tissue destruction and pain of RA. The presence of both cytokines has been documented in rheumatoid synovial fluid and synovium as well as in human atherosclerotic lesions but not in normal vessels.12,13 The biological actions of these cytokines cause synovial hyperplasia, leukocyte infiltration, and cartilage degradation, as well as the medial hyperplasia and leukocyte infiltration seen in atheromatous blood vessels. Inflammatory cytokines such as TNF-alpha and platelet-derived growth factor activate subendothelial and endothelial myofibroblast proliferation, which facilitates plaque growth and proliferation.14,15
As the levels of C-reactive protein (CRP), an acute phase reactant, rise during periods of RA activity, it also plays a role in atherogenesis. Elevated CRP levels have been shown in several studies to increase the risk of cardiovascular events by a number of mechanisms.16 CRP is directly related to interleukin-6 levels, a cytokine that promotes leukocyte adherence to the blood vessel walls.17 CRP also stimulates macrophages to produce tissue factor, an important procoagulant found in atherosclerotic plaques.18
Medications used to treat RA may contribute to the atherosclerotic process. Corticosteroids, though used in low dosage, have a recognized atherogenic effect that is mediated through their effects on plasma low-density lipoproteins. Two studies, however, failed to show an association between cardiovascular mortality and steroid use in RA.19,20 Methotrexate and sulfasalazine may cause atherogenesis due to their antifolate effects with subsequent hyperhomocysteinemia. Elevated homocysteine levels are a known risk factor for atherothrombosis since homocysteine is directly toxic to endothelial cells because it potentiates LDL oxidation and has prothrombic effects. The standard use of folate supplementation with methotrexate, however, mitigates this risk.21
Cardiovascular Mortality and RA
One study examined the mortality of an inception cohort of 1,236 patients who were in the primary care-based Norfolk Arthritis Register in the United Kingdom and were followed over a nine-year period.2 All patients in the study group had RA duration of fewer than two years at study entry. The researchers found that women who were seropositive at baseline were twice as likely to die of cardiovascular causes as were women in the general population. When stratified by age at death, women younger than 65 had a threefold increased risk of dying from cardiovascular disease compared with women in the general population.2 In another study, 3,501 male and female RA patients from four centers in the United States and Canada were followed prospectively over 35 years.1 The overall standardized mortality ratio (the ratio of observed deaths in the group under study to expected deaths in the general population) was 2.26 with the majority of deaths attributed to cardiovascular disease.
Dietary Studies
One study showed that ingestion of encapsulated fish oil supplements resulted in suppression of mononuclear cell TNF-alpha and IL-1 beta synthesis.22 However, the cost and inconvenience of ingesting large numbers of pills daily affected patient compliance. In an eight-week dietary intervention study with a parallel, crossover design, two groups of 15 healthy males received an omega-3 (flaxseed oil) enriched diet or an omega-6 (sunflower oil) diet. The flaxseed group's diet was low in omega-6 PUFA content, while the sunflower group maintained their usual omega-6 PUFA diet and used sunflower oil, spreads, and dressings. Each group maintained its diet for eight weeks; after the first four weeks, both groups received fish oil capsules that provided 1.62 g/d EPA and 1.08 g/d DHA. Four weeks after the initial dietary intervention, the flaxseed oil group showed a decrease in production of TNF-alpha, IL-1 beta, thromboxane B2, and prostaglandin E2 of 30%, 31%, 29%, and 39%, respectively, whereas there was no change in the production of these inflammatory mediators in the sunflower group. After four weeks of fish oil supplementation, the flaxseed and sunflower groups showed respective inhibitions of 77% and 70% in TNF-alpha, 81% and 78% in IL-1 beta, 48% and 52% in thromboxane B2, and 28% and 55% in prostaglandin E2. This dietary intervention study showed that the increased intake of omega-3 fatty acids coupled with dietary restriction of omega-6 PUFAs resulted in decreased levels of inflammatory mediator production whether the ingested omega-3 fatty acid was EPA from fish oil or its biosynthetic progenitor ALA.23
Clinical Studies
Two epidemiologic studies showed a decreased prevalence and milder course of RA in Eskimos and Faroe Islanders compared with Europeans.24,25 Both populations consume diets high in marine-source omega-3 fatty acids. In a population-based case-control study of women in western Washington, 324 incident RA patients were compared with 1,245 controls using a validated food-frequency questionnaire. The researchers found that consumption of more than two servings per week of baked or broiled fish (other than tuna) was associated with a decreased risk of RA (adjusted odds ratio = 0.57, P for trend = 0.01). Although a retrospective, recall study, when the researchers restricted the analysis to cases seropositive for rheumatoid factor, the association with intake of baked/broiled fish was stronger with an adjusted odds ratio of 0.32.26
In a placebo-controlled, double-blind, randomized 15-week study of 50 RA patients whose dietary omega-6 fatty acid intake was restricted to < 10 g/d, researchers found that patients given 40 mg/kg/d of fish oil experienced a modest, but statistically significant, improvement in measurement of clinical disease activity including duration of morning stiffness, the patient and physician global assessment of arthritis activity, and the Health Assessment Questionnaire score.27 However, these clinical improvements did not occur until the patients had taken the supplement for 15 weeks. This time interval to clinical improvement after initiation of omega-3 fatty acid supplementation is in agreement with other findings28-30 of fish oil supplementation in RA patients. All patients in the study were maintained on stable antirheumatic and anti-inflammatory medication regimes.27 This may have modulated the beneficial effects of the fish oil, since the medications and the omega-3 fats have overlapping effects with regard to the reduction of inflammation.
In a meta-analysis of 10 studies in which fish oil supplementation effects on RA had been reported, researchers chose a 12-week time as the common time in all the studies for a pooled analysis.31 The results showed statistically significant improvement for tender joint number and morning stiffness in the fish oil group as compared to placebo. The researchers then examined the primary data sets from each study in the meta-analysis to identify potential cofounders such as age, gender, rheumatoid factor status, disease duration, and medications. Both analyses supported the significant reduction in the number of tender joints and the duration of morning stiffness with the use of omega-3 fatty acids but no other outcome measures were changed significantly. The fact that NSAIDs and SAARDs were continued throughout the trial period that fish oil was ingested most likely moderated the anti-inflammatory effects of the omega-3 fatty aids.31,32
Drug/Diet Treatment Combinations for RA
Antirheumatoid drugs, which require the suppression of omega-6 eicosanoid production or action for their therapeutic effect, may show greater efficacy if the drug treatment is combined with a dietary regimen that is composed of a LA/ALA ratio closer to the more favorable 4:1 ratio. One study found an additive interaction between dietary EPA and the DMARD auranofin for the inhibition of LTB4 synthesis.33
The possibility of a drug-sparing effect of a dietary supplement is an important aspect of treatment for RA patients because, although therapeutic effects of pharmaceuticals frequently are documented in drug trials in the short term, long-term observational studies indicate that most patients have chronic progressive disease despite therapy34 and frequently undergo changes in their drug regimens, either due to lack of efficacy or toxicity. Prashker et al estimated that the toxicities of RA pharmacotherapies contribute about 60% of the total cost of treating RA patients in the United States.35
As the dietary ratio of LA/ALA approaches the more optimum 4:1 ratio, increased fruit and vegetable consumption are recommended to replenish antioxidant stores that may be depleted with long-term fish oil supplementation. Meydani et al noted significant reductions in plasma alpha-tocopherol levels with an increase in lipid peroxides in a group of 15 young healthy women and 10 healthy older women after three months of EPA (1.68 g/d) and DHA (720 mg/d) supplementation.36 This indicates that fish oil consumption may require a graded increase in vitamins E and C intake to ensure the dietary intake does not increase free radical production, which ultimately could worsen cardiovascular outcomes.37
Dosage
Dosage of fish oil supplements should be based upon the amount of EPA and DHA in a product rather than the total amount of fish oil. A common amount of omega-3 fatty acids in fish oil capsules is 0.18 g (180 mg) EPA and 0.12 g (120 mg) DHA. The source should be wild rather than farm-raised fish.38 Clinical trials have used a range of doses, most commonly between 3-5 g/d of EPA and DPA. The duration of the supplementation has ranged from 12-52 weeks for RA. When choosing fish oil supplements, it is important to have third-party verification of product quality and safety, such as the Consumer Lab seal of approval, which means the manufacturer's claimed EPA and DHA content has been evaluated and verified, tested for purity (i.e., the presence of methylmercury, polychlorinated biphenyls, and dioxin), and tested for freshness as oxidation of the fish oil generates free radicals and renders them unsuitable for ingestion. Since neither the FDA, the Environmental Protection Agency (EPA), nor any other federal or state agency routinely tests fish or marine oil supplements for quality prior to sale, third-party verification of product quality and safety is important.39
Adverse Effects
Persons allergic or hypersensitive to fish should avoid products containing fish oil or omega-3 fatty acid products derived from fish. Persons with nut allergies should avoid ALA or omega-3 fatty acid products derived from the types of nuts to which they are reactive.
Gastrointestinal symptoms are common with the use of fish oil supplements, with up to 5% of patients in clinical trials affected. The most common symptoms are nausea, increased eructation, reflux, indigestion, and a fishy aftertaste. Diarrhea may occur and can become severe at very high doses. Such side effects can be minimized if the fish oils are taken with meals and if low doses are increased gradually.38
FDA has classified intake of up to 3 g/d of omega-3 fatty acids from fish as generally recognized as safe. The Department of Health and Human Services and the EPA have recommended that pregnant and breast-feeding females and young children avoid eating shark, swordfish, king mackerel, and tile fish because these large predatory fish contain elevated levels of methylmercury. An on-line brochure prepared by the FDA on this subject can be found at www.cfsan.fda.gov/~dms/admehg3b.html. Since methylmercury accumulates in fish meat and not in the oil, commercial fish oil supplements appear to contain almost no mercury.38
Persons on any type of antiplatelet or anticoagulant medication should take omega fatty acid supplementation with caution and have their INRs and bleeding times checked regularly, as concomitant use can prolong bleeding times unpredictably.40,41
Fish oil taken for several months may result in a vitamin E deficiency and depletion of antioxidant levels. It is important to incorporate the recommended five servings of fresh fruit and vegetables into a heart- and joint-healthy diet when taking these supplements. Vitamin E (as d-alpha-tocopherol) and often vitamin C are added to encapsulated fish oils to prevent lipid peroxidation, but the amounts added may not be adequate to replenish depleted levels in individual patients.37 A recommended regimen would include 400 IU/d vitamin E, 200 mg/d vitamin C, and 200 mcg/d selenium.6
Storage Considerations
Omega-3 fatty acid supplements are subject to denaturation by light and heat. Check cooking temperatures for all oils to be certain the smoke point is not exceeded during the cooking or baking process. They must be maintained in dark containers and in cool dry places for maximum shelf life. Some require refrigeration after opening.
Conclusion
Diets with increased ratios of omega-3 to omega-6 fatty acids can decrease the production of TNF-alpha and IL-1 beta, thus providing a more favorable background environment for the effective application of drug therapies for RA. In addition, dietary supplementation of 2-3 g/d of DHA and EPA as fish oil supplements or fatty fish meals provides a direct source of omega-3 fatty acids, which has the potential for sparing pharmaceuticals and decreasing the side effect profile and mortality from cardiovascular diseases in RA patients. Omega-3 fatty acid oils and supplements have finite shelf lives and requirements with regard to temperatures at which they are used for cooking. Since omega-3 fatty acids can prolong bleeding times, their use in patients on anticoagulants should be monitored carefully.
Recommendations
For patients with RA, dietary supplementation of up to 3 g/d EPA + DHA fish oil or 2-4 g/d ALA oil for vegetarian patients and dietary counseling to limit omega-6 fatty acid intake, are important adjunctive measures. The duration of time the therapy should be continued has not been established, but beneficial effects may take at least three months to occur. Patients on chronic fatty acid supplementation may run the risk of depletion of antioxidants. Therefore, it is also important to follow a heart- and joint-health diet which includes five servings of fruit and vegetables daily.
References
1. Wolfe F, et al. The mortality of rheumatoid arthritis. Arthritis Rheum 1994;37:481-494.
2. Goodson NJ, et al. Mortality in early inflammatory polyarthritis: Cardiovascular mortality is increased in seropositive patients. Arthritis Rheum 2002;46:2010-2019.
3. del Rincon ID, et al. High incidence of cardiovascular events in a rheumatoid arthritis cohort not explained by traditional cardiac risk factors. Arthritis Rheum 2001;44:2737-2745.
4. Harper CR, Jacobson TA. The fats of life: The role of omega-3 fatty acids in the prevention of coronary heart disease. Arch Intern Med 2001;161:2185-2192.
5. Roche HM. Unsaturated fatty acids. Proc Nutr Soc 1999;58:397-401.
6. Rakel D, ed. Integrative Medicine. St. Louis, MO: Saunders; 2003:668-676.
7. Covington MB. Omega-3 fatty acids. Am Fam Physician 2004;70:133-140.
8. U.S. Department of Agriculture. Agricultural Research Service. Food sources of omega-3 fatty acids: EPA, DHA and ALA. In: USDA Nutrient Database for Standard Reference, Release 18, 2005. Available at: www.nal.usda.gov/fnic/foodcomp. Accessed Feb. 8, 2006.
9. Gerster H. Can adults adequately convert alpha-linolenic acid (18:3n-3) to eicosapentaenoic acid (20:5n-3) and docosahexaenoic acid (22:6n-3)? Int J Vitam Nutr Res 1998;68:159-173.
10. Lands WE. Biochemistry and physiology of n-3 fatty acids. FASEB J 1992;6:2530-2536.
11. Alexander JW. Immunonutrition: The role of omega-3 fatty acids. Nutrition 1998;14:627-633.
12. Fontana A, et al. Interleukin 1 activity in the synovial fluid of patients with rheumatoid arthritis. Rheumatol Int 1982;2:49-53.
13. Di Giovine FS, et al. Tumour necrosis factor in synovial exudates. Ann Rheum Dis 1988;47:768-772.
14. Arend WP, Dayer JM. Inhibition of the production and effects of interleukin-1 and tumor necrosis factor alpha in rheumatoid arthritis. Arthritis Rheum 1995;38:151-160.
15. Barath P, et al. Tumor necrosis factor gene expression in human vascular intimal smooth muscle cells determined by in situ hybridization. Am J Pathol 1990;137:503-509.
16. Ridker PM, et al. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med 2000;342:836-843.
17. Wolbink GJ, et al. CRP-mediated activation of complement in vivo: Assessment by measuring circulating complement-C-reactive protein complexes. J Immunol 1996;157:473-479.
18. Cermak J, et al. C-reactive protein induces human peripheral blood monocytes to synthesize tissue factor. Blood 1993;82:513-520.
19. Wallberg-Jonsson S, et al. Cardiovascular morbidity and mortality in patients with seropositive rheumatoid arthritis in Northern Sweden. J Rheumatol 1997;24:445-451.
20. Stern MP, et al. Adrenocortical steroid treatment of rheumatic diseases: Effects on lipid metabolism. Arch Intern Med 1973;132:97-101.
21. Welch GN, Loscalzo J. Homocysteine and atherothrombosis. N Engl J Med 1998;338:1042-1050.
22. Endres S, et al. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N Engl J Med 1989;320:265-271.
23. Caughey GE, et al. The effect on human tumor necrosis factor alpha and interleukin 1 beta production of diets enriched in n-3 fatty acids from vegetable oil or fish oil. Am J Clin Nutr 1996;63:116-122.
24. Horrobin DF. Low prevalence of coronary heart disease (CHD), psoriasis, asthma and rheumatoid arthritis in Eskimos: Are they caused by high dietary intake of eicosapentaenoic acid (EPA), a genetic variation of essential fatty acid (EFA) metabolism or a combination of both? Med Hypotheses 1987;22:421-428.
25. Recht L, et al. Hand handicap and rheumatoid arthritis in a fish-eating society (the Faroe Islands). J Intern Med 1990;227:49-55.
26. Shapiro JA, et al. Diet and rheumatoid arthritis in women: A possible protective effect of fish consumption. Epidemiology 1996;7:256-263.
27. Volker D, et al. Efficacy of fish oil concentrate in the treatment of rheumatoid arthritis. J Rheumatol 2000;27:2343-2346.
28. Kremer JM, et al. Fish-oil fatty acid supplementation in active rheumatoid arthritis. A double-blinded, controlled, crossover study. Ann Intern Med 1987;106:497-503.
29. Cleland LG, et al. Clinical and biochemical effects of dietary fish oil supplements in rheumatoid arthritis. J Rheumatol 1988;15:1471-1475.
30. Geusens P, et al. Long-term effect of omega-3 fatty acid supplementation in active rheumatoid arthritis. A 12-month, double-blind controlled study. Arthritis Rheum 1994;37:824-829.
31. Fortin PR, et al. Validation of a meta-analysis: The effects of fish oil in rheumatoid arthritis. J Clin Epidemiol 1995;48:1379-1390.
32. Lau CS, et al. Effects of fish oil supplementation on non-steroidal anti-inflammatory drug requirements in patients with mild rheumatoid arthritisA double-blind, placebo controlled study. Br J Rheumatol 1993;32:982-989.
33. James MJ, et al. Inhibition of human neutrophil leukotriene B4 synthesis by combination auranofin and eicosapentaenoic acid. Biochem Pharmacol 1992;43:695-700.
34. Pincus T. The paradox of effective therapies but poor long-term outcomes in rheumatoid arthritis. Semin Arthritis Rheum 1992;21(6 Suppl 3):2-15.
35. Prashker MJ, Meenan RF. The total costs of drug therapy for rheumatoid arthritis: A model based on costs of drug, monitoring, and toxicity. Arthritis Rheum 1995;38:318-325.
36. Meydani M, et al. Effect of long-term fish oil supplementation on vitamin E status and lipid peroxidation in women. J Nutr 1991;121:484-491.
37. Darlington LG, Stone TW. Antioxidants and fatty acids in the amelioration of rheumatoid arthritis and related disorders. Br J Nutr 2001;85:251-269.
38. MayoClinic.com. Omega-3 fatty acids, fish oil, alpha linolenic acid. Available at: www.mayoclinic.com/health/fish-oil/NS_patient-fishoil. Accessed Jan. 10, 2006.
39. ConsumerLab.com. Product Review: Omega-3 fatty acids (EPA and DHA) from fish/marine oils Available at: www.consumerlab.com/results/omega3.asp. Accessed Jan. 15, 2006.
40. Bender NK, et al. Effects of marine fish oils on the anticoagulation status of patients receiving chronic warfarin therapy. J Thromb Thrombolysis 1998;5:257-261.
41. Buckley MS, et al. Fish oil interaction with warfarin. Ann Pharmacother 2004;38:50-52.
Marcolina ST. Omega-3 fatty acids for the treatment of rheumatoid arthritis. Altern Ther Womens Health 2006;8(3):17-23.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
