Updates in ENT Emergencies: Tales from the Trenches
Updates in ENT Emergencies: Tales from the Trenches
Authors:
John Bailitz, MD, Assistant Program Director, Cook County Emergency Medicine Residency, Assistant Professor of Emergency Medicine, Rush University Medical Center, Chicago, IL.
Vikas Patel, MD, Resident, Cook County Hospital, Chicago, IL.
Ron Kim, MD, Chief Resident, Cook County Hospital, Chicago, IL.
Peer Reviewer:
Steven M. Winograd, MD, FACEP, New York Community Hospital, Emergency Department Attending Physician, New York Presbyterian Healthcare System, Weill Medical College of Cornell University, Brooklyn, NY.
Life-threatening ear, nose, and throat (ENT) emergencies can present to any emergency department (ED) at any hour of the day or night.
The following article is a review of four cases from the ENT teaching files of the Stroger Hospital of Cook County, Illinois. Each case discussion illustrates the new and old challenges of these ENT emergencies and provides new insights and reminders crucial to life-saving ED management.
J. Stephan Stapczynski, MD, FACEP, Editor
Angioedema
Case. A 27-year-old female presents to the ED with an anxiety attack after her child's asthma acted up in a nearby store. She has a psychiatrist who prescribes alprazolam for these attacks, but she has not needed to take any medications in more than a year. As you prepare her discharge papers, she mentions in a now somewhat hoarse voice that she also has a history of "angioed...," which has once before resulted in an intubation after a stressful event. She bursts into tears over her husband, who recently left her, as her stridor begins.
Relevant Epidemiology and Pathophysiology. Angioedema (AE) is the abrupt onset of non-puritic, non-pitting edema of the deep dermis and subcutaneous tissue layer often involving the face, mouth, lips, tongue, extremities, genitalia, and/or abdominal viscera and lasting 24-96 hours. In contrast, urticaria affects the more superficial epidermis containing mast cells and nerves, resulting in edema with intense itching that lasts less than 24 hours. However, the distinction often may be more academic than clinical, since up to one half of AE cases are accompanied by urticaria.1
Traditionally, angioedema has been classified as allergic, hereditary, or acquired. Allergic angioedema involves an IgE-mediated reaction to insect bites, foods, or latex. Recently, subdividing non-allergic angioedema into 5 types has been proposed: hereditary angioedema (HAE), acquired angioedema, renin-angiotensin-aldosterone system blocker-induced, pseudo-allergic, and idiopathic.2
HAE and acquired AE most commonly result from abnormalities in C1 esterase inhibitor (C1-INH). HAE is an autosomal dominant disorder affecting approximately 1 in 50,000 people in the United States, resulting in up to 30,000 ED visits annually. Three types of HAE have been described. Type I results from a quantitative deficiency, type II results from a qualitative deficiency, and type III results from genetic mutations that lead to increased bradykinin production.3 Attacks most commonly involve the skin or the gastrointestinal (GI) tract, but sudden isolated laryngeal swelling remains a significant cause of mortality.4
With acquired angioedema, auto-antibodies deactivate C1-INH, resulting in a qualitative deficiency. Acquired AE has been reported in patients with lymphoproliferative diseases, lupus, and thyroid and kidney disease. Both HAE and acquired AE result in unopposed activity of the first component of the complement cascade and ultimately increased production of the potent vasodilator bradykinin.
Angiotensin-converting enzyme inhibitor-induced (ACEI) angioedema affects less than 1% of the patients taking ACEIs each year. However, 40 million Americans currently take ACEIs daily, which results in 400,000 cases of ACEI-induced AE annually.5 In fact, ACEI-induced AE accounts for 30-68% of cases of isolated AE each year.6,7 Compared to HAE, ACEI-induced AE often results in more slowly progressive swelling of the tongue, lips, and laryngeal soft tissue. Up to 13% of patients may require intubation.6,8 Although the highest incidence is seen within the first month of therapy, AE may occur at any time during ACEI therapy. Risk factors include African-American race, smoking, advanced age, and female gender. The pathophysiology may involve a decrease in the metabolism of bradykinin by inhibited ACE. However, the relatively low incidence, often delayed onset, and reccurrence with angiotensin receptor blockers (ARBs) suggests alternative mechanisms.9 Other common ED medications reported to result in AE include nonsteroidal anti-inflammatory drugs (NSAIDs), aspirin, penicillins, and even thrombolytics.
Key Clinical Features. HAE most commonly presents with recurrent unexplained swelling of the extremities or genitals, or diffuse abdominal tenderness accompanied by nausea and vomiting. However, half of HAE patients will have at least one laryngeal attack, with 85% of laryngeal attacks occurring without other swelling.10 Commonly reported attack triggers include minor trauma, infections, oral contraceptive pills, and even emotional stress. Signs of impending airway compromise include dyspnea, hoarseness, odynophagia, stridor, and inability to handle oral secretions.
Effective Diagnostic Strategies. Patient assessment is determined by the rapidity of symptom progression. Rapidly progressive symptoms of airway obstruction require emergent airway management as described below. Less acute symptoms allow for a more comprehensive assessment. AE patients with any vocal cord symptoms require visualization of the vocal cords to determine the need for intubation and disposition. Nasopharyngoscopy is the diagnostic modality of choice. When not available, consider traditional mirror laryngoscopy or direct laryngoscopy in patients able to lie flat and tolerate the procedure. Laboratory studies to consider include serum C1-INH level, C1-INH activity, C4 complement level, and CRP.3 However, results do not influence ED management.
Emergent Management. Airway protection is the first priority in any ENT emergency. Up to 13-35% of patients with moderate to severe angioedema will require airway management.6,11-13 Emergent intubation is required in any patient with respiratory distress, stridor, or inability to handle secretions. Urgent intubation is indicated for patients with marked edema of the floor of the mouth or tongue.6
All ENT emergencies requiring intubation must be considered difficult airways. The initial consideration is to choose the best location for airway management based on the patient's condition, operator experience, and available resources. For the relatively stable patient with urgent indications for airway management, the easiest intubation may occur via fiberoptic laryngoscopy in the ED or operating room. For unstable patients, airway management usually is required in the ED.
Thoughtful preparation for managing the difficult airway in the ED includes having equipment available and preparations made for the primary approach, a backup plan, and a rescue technique. (See Figure 1 and also refer to "Management of the Difficult Airway," Emergency Medicine Reports Aug. 8, 2005.)
Figure 1: Airway Management Algorithm for ENT Emergencies
Fiberoptic Intubation: Time-, Patient-, and Physician-Dependent.
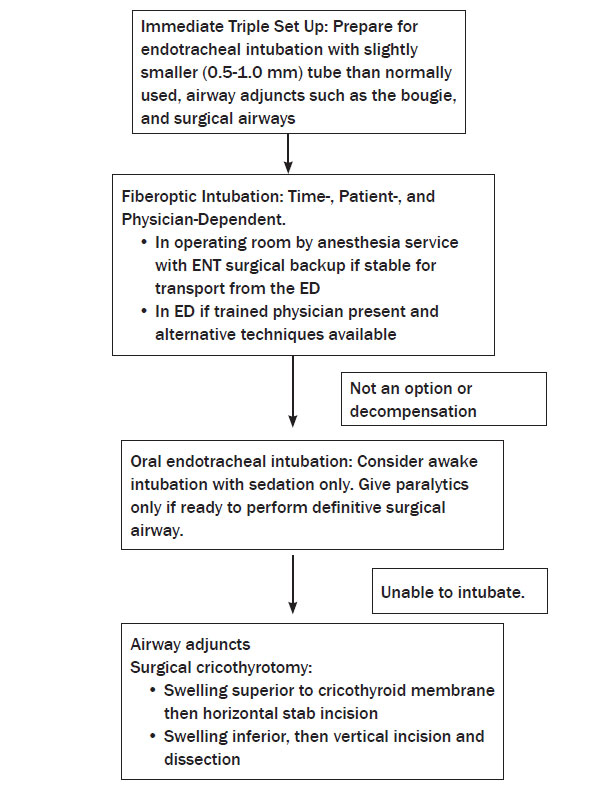
With a patent airway ensured, the mainstay of initial angioedema treatment includes antihistamines, epinephrine (nebulized, IV, or IM), and corticosteroids. Since pathophysiology and presentations overlap, these typical allergic urticaria treatments are commonly used in AE.
Disposition depends on clinical findings and ED course. One approach is to use a staging system.14 Any patients with laryngeal or oropharyngeal edema should be monitored closely in the ICU. Stage 1 patients may be discharged after at least 4 hours of observation with clinical improvement. (See Table 1.)
Table 1: Disposition for Patients with Angioedema
|
Stage |
Findings |
% Requiring Airway |
Disposition |
|
I |
Facial rash, swelling, limited lip edema |
0 |
Outpatient or ward |
|
II |
Soft palate edema |
0 |
Ward |
|
III |
Lingual edema |
7 |
ICU |
|
IV |
Laryngeal edema |
24 |
ICU |
Discharge patients with an Epipen® and explicit instructions on avoiding triggers, early warning signs, and when to return to the ED. Additional outpatient HAE therapies may include attenuated androgens that stimulate the synthesis of C1-INH, such as stanozolol or danazol.15 The fibrinolytic drug tranexamic acid may also be used for short- and/or long-term prophylaxis. ACE inhibitor-induced angioedema requires cessation of the offending drug. For outpatient blood pressure control, avoid angiotensin receptor blockers since patients with prior ACE inhibitor angioedema are more susceptible to ARB-induced angioedema.
Complications, Controversies, and Cutting Edge. Several case series have suggested that fresh frozen plasma (FFP) is an effective treatment for HAE by providing functional C1 esterase inhibitor.16,17 However, other experts have suggested that FFP may actually worsen HAE by providing high concentrations of complement proteins. Furthermore, the time required to type and cross and thaw prevents any role of FFP in the management of an impending airway obstruction in the ED.
Recently, the FDA approved C1-INH concentrate, available in Europe since 1974, for the treatment of both HAE and ACEI-induced AE. C1-INH concentrates for acute attacks include ecallantide (Kalbitor®, Dyax) and Berinert® (CSL Behring), while Cinryze (ViroPharma) is used for prophylaxis alone. In acute attacks, C1-INH concentrate should be initially administered at 10 to 30 units/kg IV (between 500-1000 units in total) and re-dosed 30-60 minutes later.2 A prospective study of ED patients with HAE found that a single 20-unit/kg dose of Berinert® resulted in symptom improvement within 15 minutes without significant side effects.18 Thus, in patients with slowly progressive urgent symptoms, C1-INH may prevent intubation.
Case Conclusion. In a matter of minutes, the patient became progressively stridorous despite conventional medications. As anesthesia was wheeling in the fiberoptic cart, the patient's pulse oximetry began dropping. Medications were administered after prepping the neck for cricothyrotomy. Edematous vocal cords and a small glottic opening were visualized. The initial 6.5 ET tube was too large to pass through the cords. A second attempt was successful with a size 6.0 ET tube. The patient recovered well.
Supraglottitis
Case. A 37-year-old male presents to the fast track of the ED complaining of 4-5 days of a runny nose, dry cough, and worsening sore throat. Vital signs reveal a slight fever with a heart rate in the 130s. He is sitting upright and appears somewhat ill. However, there is only mild trismus, reassuring bilateral tonsilar exudates, and posterior pharyngeal erythema on physical examination. With worsening respiratory distress, the patient was moved to a resuscitation bed. Following a nebulizer treatment with racemic epinephrine, a bedside soft-tissue neck radiograph was obtained.
Relevant Epidemiology and Pathophysiology. Epiglottitis, better described as supraglottitis (SG), is a localized cellulitis of the supraglottic structures. Above the vocal cords, the submucosal layer is only loosely adherent to the mucosa, allowing for rapid accumulation of edema and narrowing of the airway diameter. Inflammation readily spreads to involve not only the epiglottis, but also the aryepiglottic folds, arytenoid soft tissues, vallecula, base of the tongue, and lingual tonsils. Below the vocal cords, the submucosal layer is densely adherent to the mucosa, preventing involvement of infraglottic tissues. Hence, cricothyrotomy is typically successful as an emergent airway with complete airway obstruction in supraglottitis.
Although cases of pediatric supraglottitis have dramatically decreased since the introduction of the Haemaphilus influenzae type B (Hib) vaccine in 1992, cases of adult supraglottitis surprisingly have increased. By the late 1990s, the rate of pediatric supraglottitis had fallen to 0.02 per 100,000 population, while the rate of adult cases had risen to 3.1 per 100,000.19-21
Fortunately, mortality rates are low in adult cases, likely due to the larger diameter of the adult airway that better tolerates the narrowing effects of inflammation, as well as the availability of better airway management techniques.22-24 Post Hib studies further reveal a male-to-female predominance of 2.5:1, and an increased incidence in smokers in the 4th-6th decades of life.19,21,23-25 Immunocompromised patients typically have more severe or rapid presentations. In recreational drug users, inhalation of pieces of metal from crack-cocaine pipes and even ends of marijuana cigarettes have been reported to cause thermal injury to the supraglottic structures, mimicking acute infectious supraglottitis and more often resulting in abscess formation.21,26
In the post-vaccination era, the etiology of supraglottitis has likewise changed. Oropharyngeal cultures are typically negative, suggesting a viral etiology. Cultures from epiglottic abscesses most often reveal Staphylococcus, Streptococcus, Klebsiella, E. coli, Enterobacter, or normal oral flora. Immunocompromised patients are also susceptible to fungal infections, most commonly Candida.27
Key Clinical Features. A minority of adult patients present to the ED with the classic symptoms of stridor, respiratory difficulty, drooling, and tripoding. The typical patient reports an upper respiratory infection prodrome lasting from one to seven days. Many patients may have already seen a physician and received outpatient antibiotics.28 Worsening sore throat, odynophagia, inability to handle secretions, and hoarse voice are the most common complaints. Tachycardia is frequent, while fever is present in only 28-57% of patients.29 (See Table 2.)
Table 2: Supraglottitis
|
Children |
Adults |
|
Pre-Hib or unvaccinated 3-7 year olds Acute illness Tripod position Misdiagnosed as croup, viral syndrome, or viral pharyngitis |
Today Male smokers, immunocompromised 1-2 day prodrome Dysphagia, odynophagia, sore throat, disproportionate tachycardia Tenderness with movement of hyoid and larynx Concomitant oral disease does not exclude Misdiagnosed as Strep. pharyngitis |
The physician should consider supraglottitis not only in the patient with a killer sore throat and "normal physical examination" but also in the high-risk, ill-appearing patient with other upper respiratory tract infections, including tonsillitis or peritonsillar abscess.30 Significant tenderness on movement of the hyoid suggests supraglottitis.
Effective Diagnostic Strategies. Positive neck radiographs help confirm the diagnosis. However, the value of negative radiographs remains controversial. While initial studies indicated that the finding of an enlarged epiglottis > 7 mm on lateral neck radiographs had a sensitivity that approached 100%31,32, subsequent researchers recently have reported sensitivities ranging from 63-84%.25,28,29 Other positive findings on X-ray include edema of the prevertebral/retropharyngeal space and edema of the arytenoids and aryepiglottic folds.
In retrospective studies, CT and MRI have been reported to be useful in the diagnosis of supraglottitis.33,34 However, there have been no prospective clinical trials. Additionally, both modalities require that a patient with a potentially unstable airway be transported out of the ED and lie in a supine position.
To summarize, radiographs are helpful when positive and when negative in low-risk cases with an alternative diagnosis. In all other cases, direct visualization of the supraglottic structures should follow negative radiographs. CT may be helpful in the identification of epiglottic abscess and other adjacent infections in the patient able to lie flat during a 15-minute trip away from the ED. Likewise, routine blood work is of limited clinical utility.
The definitive method to reliably exclude supraglottitis is direct visualization. In stable patients without respiratory distress, direct laryngoscopy or indirect mirror laryngoscopy provides a fast method to visualize the glottic structures when preformed by experienced hands. Today, such approaches have largely been replaced by the better patient-tolerated and improved field of view provided by flexible fiberoptic nasopharyngoscopy.23,28 Additionally, endoscopes of adequate length facilitate fiberoptic intubation. Using either method, edematous, red, or occasionally even pale epiglottis or supraglottic structures confirms the diagnosis.
Emergent Management. Airway intervention is more likely with rapid progression of symptoms. For patients with significant respiratory distress, complete airway obstruction may be only moments away.
For patients without significant respiratory distress, direct visualization determines the need for airway protection. In patients with 50% or greater narrowing of the airway, a definitive airway is recommended.21,22,25,30 Other findings predictive of airway intervention include drooling, presence of diabetes, and concurrent epiglottic abscess.21
Antibiotic regimens should cover common pathogens including gram positives, gram negatives, and anaerobes. Appropriate agents include third-generation cephalosporins or ampicillin-sulbactam. For penicillin-allergic patients, administer trimethoprim-sulfamethoxazole, moxifloxacin, or levofloxacin. Retrospective analysis of steroid use has shown no benefit, but steroids are typically given to reduce inflammation.21,35
All patients with supraglottitis require admission for IV antibiotics, hydration, and airway observation. Patients with rapidly progressive symptoms, diabetes, drooling, shortness of breath, and/or abscess formation should go to the ICU.21,23,25 Patients who are intubated generally are discharged within one week.24 Patients without respiratory distress and less than 50% airway narrowing can be admitted to non-ICU beds for close observation and are often discharged within 72 hours.
Complications, Controversies, and Cutting Edge. After ensuring a patent airway, perform a CT scan on patients displaying asymmetry of the supraglottic structures, pronounced swelling, or thermal injury to evaluate for abscess formation,21,26 which can be found in up to 12% of patients diagnosed with supraglottitis.36
Case Conclusion. Soft-tissue neck radiographs demonstrated significant supraglottic edema. Furthermore, the patient admitted to smoking crack cocaine 4 days prior. After decompensating, the patient was emergently intubated with a size 6.0 ET tube. Laryngoscopy demonstrated an edematous and asymmetric epiglottis confirmed to be a supraglottic abscess on subsequent CT. (See Figure 2.)
Figure 2: CT of Epiglottic Abscess

Epistaxis
Case. A 67-year-old man presents to the ED at 3 a.m. with the sudden onset of profuse bleeding from both nares for 20 minutes. Medications include both aspirin and clopidogrel. He is now vomiting blood. BP is 220/110, HR is 135, RR 30, and pulse oximetry is 99%. The patient appears anxious, with a towel pressed tightly against his face. The charge nurse requests an order for IV medication to control the blood pressure and stop the bleeding.
Relevant Epidemiology and Pathophysiology. Epistaxis is common, affecting 60% of the general population.37 Anterior epistaxis accounts for about 90% of epistaxis cases. Patients are typically younger, presenting with mild to moderate bleeding from one naris originating from Kiesselbach's plexus on the anterior inferior nasal septum. This vascular watershed is supplied by both main branches of the carotid artery. The internal carotid artery gives rise to the anterior and posterior ethmoidal arteries. The external carotid artery gives rise to the superior labial branch of the facial artery and terminal branches of the sphenopalatine artery.
Posterior epistaxis accounts for the remaining 10%. Patients typically are older, with moderate to severe bleeding from both nares originating from the posterior septum or lateral nasal wall supplied by the posterior branch of the sphenopalatine artery. (See Table 3.)
Table 3: Anterior vs. Posterior Epistaxis
|
Anterior |
Posterior |
|
|
Who, Why, Where |
90% Children, cold, URIs Kiesselbach's plexus on anterior nasal septum: anterior ethmoidal from internal carotid artery and others from external carotid |
10% Elderly, atherosclerosis Sphenopalatine artery off the external carotid artery |
|
Sxs |
Mild to moderate, unilateral, minimal blood in oropharynx |
Moderate to severe, bilateral, blood in oropharynx |
Local and systemic factors lead to the development of epistaxis. Local factors include trauma, self-induced digital trauma, mucosal trauma from topical nasal drugs or other irritants, dryness of mucosa, rhinosinusitis, and neoplasms. Systemic factors include atherosclerosis, hypertension, barotrauma, coagulopathy, drug-induced, and hereditary hemorrhagic telangiectasia (HHT). In a retrospective study, 45% of patients admitted for epistaxis possessed one or more systemic factors, such as genetic disorders, acquired coagulopathies, anticoagulant use, or hematologic cancers.38
Key Clinical Findings. Minor anterior bleeding is common in otherwise healthy children and adolescents, while significant bleeding typically occurs in adults older than 50 years with multiple complicating co-morbidities. With heavy anterior or posterior bleeding, swallowed blood often results in hematemesis. Patients are anxious, often resulting in significant elevations in blood pressure above their already elevated baseline. Ask directly about the intensity and initial site of bleeding, prior episodes, known tumors or coagulopathies, and systemic diseases.
Effective Diagnostic Strategies. The history and physical examination are the most important diagnostic tests in any patient with epistaxis. As with all ENT emergencies, assessment begins by ensuring a patent airway. Next, control the bleeding to facilitate visualization of the bleeding site. Then establish definitive bleeding control.
A bloody towel pressed up against the nose with the head tilted back is the most common patient position at triage, yet the least effective initial treatment for active bleeding. Begin by instructing the patient to clear ineffective clots by blowing the nose. Next, apply pressure to the nasal septum by pinching the cartilaginous portion just below the nasal bones for 15 minutes. Position the patient upright with the neck slightly flexed in a dental chair or stretcher so that the floor of the nasal cavity is at or tilted down just below parallel to the floor. This upright and forward position causes any continued bleeding to flow anteriorly out of the nose instead of collecting posteriorly in the nasopharynx, potentially resulting in the swallowing or aspiration of blood. A basin may be held with the other hand for the patient to spit out any blood from the oropharynx or collect any blood from the nares.
When bleeding persists or recurs despite application of vasocontrictors and adequate packing, the patient may have an acquired or congenital coagulopathy or other underlying anatomic abnormalities such as HHT or neoplasm. In this case, obtain a complete blood count, coagulation panel, type and cross-match, and metabolic profile including renal function, although the results are normal in more than 80% of patients.38 Repeated episodes, especially if unilateral or associated with other nasal symptoms, warrant further radiographic studies to rule out neoplastic processes.
Emergent Management. Management varies with bleeding severity, presumed location, and predisposing factors.
Persistent bleeding requires the application of vasoactive agents to the nasal mucosa, followed by topical anesthesia via spray or soaked pledgets. Topical vasoconstrictors alone stop bleeding in 65% of cases.39 Common combinations include epinephrine, oxymetolazine, or phenylephrine with lidocaine. Phenylephrine with 4% lidocaine (maximum dose of 300 mg) is similarly efficacious to the combined vacoconstrictor and anesthetic effects of 4% cocaine (maximum dose of 2-3 mg/kg).40 Avoid cocaine in patients with coronary artery disease or poorly controlled hypertension. Another alternative is 0.05% oxymetazoline with 1% tetracaine (maximum dose of 50 mg). This provides anesthesia for up to 60 minutes.41 (See Figure 3.) However, there are no warning neurologic side effects, only cardiovascular collapse at high tetracaine doses.
Figure 3: ED Management of Epistaxis
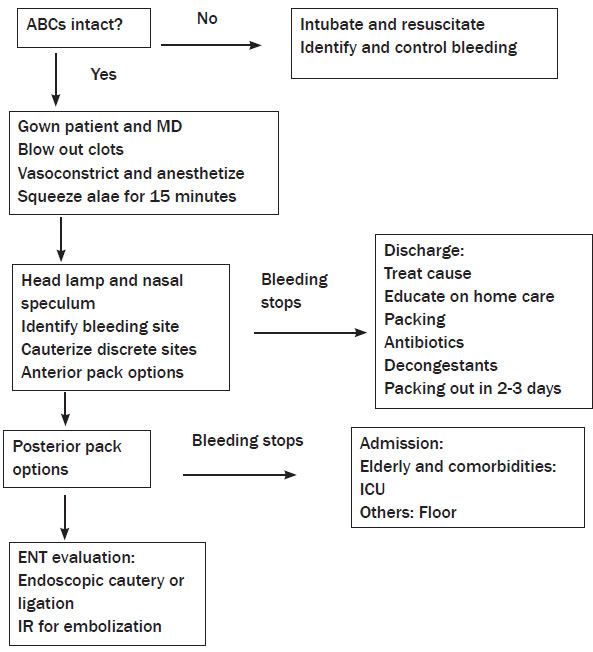
Anterior Bleeding. Many anterior bleeds are self-limited and do not require any intervention other than direct pressure and vasoconstriction. Consider silver nitrate to chemically cauterize a focal and slow bleeding source. Apply concentrically from outside to inside, keeping the area less than 0.75 cm and apply for no more than 5-10 seconds to avoid irreversible septal damage. Likewise, avoid bilateral silver nitrate as well as electric cautery without prior training. Cautery will be less effective in coagulopathic patients.
With refractory anterior epistaxis, nasal packing is required to provide effective mechanical tamponade. Non-absorbable options available in most EDs include nasal tampons (Merocel® [Medtronic ENT]) and inflatable balloon devices (Rapid Rhino® [Applied Therapeutics]).
Prior to nasal tampon insertion, estimate the correct size and length of the nasal tampon. Trim nasal tampons when needed to fit the patient, being sure to round off edges. Lightly coat the nasal tampons with antibiotic ointment. Although the prophylactic effects are likely insignificant, the lubrication eases placement. Next, instruct the patient to keep the face relaxed and the floor of the nasal cavity parallel to the room's floor. Then insert the tampon along the floor of the nasal cavity between 4.5 to 6 cm. Often, persistent bleeding will expand the tampon. Otherwise, insert a long 20-gauge IV catheter attached to a 10 mL syringe filled with water or saline along the floor of the nasal cavity alongside the tampon so that the tip of the catheter is at the posterior aspect of the tampon. Then slowly inject until the tampon gradually expands from the back. Posterior expansion first prevents the tampon from being expelled from the nares, as can occur with anterior-first expansion.
Balloon devices are likewise lightly coated with antibiotic ointment or lidocaine gel, inserted along the nasal floor, and then inflated with enough saline to tamponade the bleeding without significant discomfort. Moistened, gel-coated, balloon-inflated tampons have been demonstrated to be more comfortable on insertion and removal and associated with less recurrent bleeding than dry hydrophilic tampons, although there is no significant difference in bleeding control.43,44 Occasionally an additional nasal packing is needed in the opposite naris to provide effective tamponade. Bleeding may worsen with traumatic insertion of any nasal packing, especially in coagulopathic patients. All non-absorbable packs remain in place for 2-3 days.
Absorbable nasal packing agents provide an alternative for patients unable to tolerate non-absorbable packing or patients with mild persistent bleeding despite packing. Commonly available options include gelatin sponges such as Gelfoam® (Pfizer) or knitted fabric of oxidized cellulose Surgicel® (Johnson & Johnson). Prior to insertion, the degradable material should be lightly coated with antibiotic ointment or thrombin in coagulopathic patients. Apply to the site of discrete bleeding or oozing, onto clot, after cautery, before a tampon, or as a tampon wrap.
Instruct the patient to use a moisturizing saline spray for 24-48 hours, which will slowly dissolve absorbable packing.
Posterior Bleeding. Posterior bleeding is the likely source in a older patient with the sudden onset of heavy bilateral bleeding or the patient with persistent bleeding despite adequate bilateral anterior packing. Similar nasal packing options include longer nasal tampons as well as dual anterior and posterior balloon devices such as the Epistat® (Medtronic). With dual balloon devices, insert the device along the nasal floor until the tip is visualized in superior posterior pharynx. Then inflate slowly with 5-10 mL of saline as needed to tamponade the bleeding without causing significant patient discomfort. Rapid or overzealous inflation ensures septal and palate deviation, marked patient discomfort, and occasionally vagal reactions including diaphoresis and bradycardia. Gently pull the device anteriorly 1-2 cm to be sure that the device is appropriately lodged in the posterior nasopharynx. Next, slowly inflate the anterior balloon.
Substitute a 14 French Foley catheter when a dual-balloon device is not available. Trim the distal tip off the Foley catheter prior to insertion to prevent gagging triggered by continued stimulation of the posterior pharynx.45 Follow Foley insertion with anterior packing.
Thrombogenic gels such as Floseal® (Baxter Healthcare) have also been reported to be effective in persistent epistaxis. Although more expensive, Floseal® may be more effective, better tolerated, and require no second visit for removal versus nonabsorbable packing.46 Consult an ENT specialist first, since further examination and operative cautery for persistent bleeding is much more challenging after filling the nasal cavity with a thick gel. Evicel® (Omrix), a fibrin gel made from human cryoprecipitate, has been shown to be safe and effective with anterior and posterior epistaxis, especially in HHT, and requires only a small puff of medication.47
Patients with posterior packing traditionally have been admitted for 2-3 days of observation. It is becoming more common for patients to be packed and then taken to the operating room for endoscopic ligation.48 A recent randomized prospective trial comparing endoscopic sphenopalatine artery ligation with packing revealed a significant reduction in hospital stay and treatment costs in the operative group.49 With persistent bleeding after ENT evaluation, interventional radiology (IR) may be needed for embolization of terminal branches of the external carotid artery.
Complications, Controversies, and Cutting Edge. Complications of epistaxis management include pressure necrosis, nasal trauma, bradycardia, nasopulmonary reflex, aspiration, infection, and hypoxia. Toxic shock syndrome after placement of nasal packing remains extremely rare.50 No data exist demonstrating a risk reduction with use of prophylactic antibiotics. However, the medico-legal risk-benefit ratio may still favor prescribing a prophylactic antibiotic such as cephalexin or trimethoprim-sulfamethoxazole.
The role of hypertension in epistaxis remains controversial.50 Hypertension is a risk factor for initial and recurrent epistaxis.51,52 Nevertheless, there seems to be no clear relationship between the degree of hypertension and the prevalence of epistaxis among hypertensive patients.53 Significant elevations in blood pressure may be due in large part to patient anxiety. Avoid treating elevated blood pressure in patients with active epistaxis until the bleeding is controlled and anxiety reduced.
Recombinant factor VIIa (rFVIIa) has been reported to help control severe bleeding in patients with both inherited and acquired coagulopathies.54,55 Typical reported dosing for non-hemophiliac bleeding ranges from 90 to 120 μg/kg intravenously every 2-3 hours until the bleeding stops.55 Even a single dose of rFVIIa as low as 30 μg/kg has been reported to be effective.56 Despite this initial data, currently the Food and Drug Administration has only approved rFVIIa for the treatment and prophylaxis of bleeding in patients with a known hemophilia A or B inhibitor or deficiency.
Historically, posterior packing has been reported to elicit the nasopulmonary reflex, potentially resulting in bradycardia, bronchoconstriction, and hypoxia, and necessitating admission to the ICU. However, recent studies demonstrate no significant changes in hemodynamics or respiratory status in otherwise healthy patients.57,58 But patients with active comorbidities or significant blood loss should be admitted to the ICU.
Case Conclusion. Anterior, bilateral, and posterior packing failed to stop the bleeding. However, overzealous inflation of the posterior pack did briefly result in bradycardia and diaphoresis followed by syncope. After 2 hours of bleeding and 3 units of blood, the patient's bleeding was effectively controlled by ENT ligation of the distal sphenopalatine artery in the operating room followed by admission to the ICU for observation.
Peritonsillar Abscess
Case. A 25-year-old man presents to the ED complaining of 10 days of a progressive sore throat despite antibiotics prescribed by his primary doctor 7 days ago. During the past 48 hours, the patient has developed progressive trismus as well as left-sided neck swelling. (See Figure 4.)
Figure 4: PTA Patient with Neck Swelling, Open Mouth View

Relevant Epidemiology and Pathophysiology. Quinsy, or peritonsillar abscess (PTA), is often considered to be the most common deep space infection of the head and neck. The abscess develops in a space bounded anteriorly and posteriorly by the tonsillar pillars, the lateral pharyngeal space, the hard palate superiorly, and the pyriformis fossa inferiorly.
PTAs affect 10-30/100,000 patients per year without seasonal variability.59,60 Smoking and prior treatment with antibiotics are common to both supraglottitis and PTAs. In fact, smoking is an independent risk factor for PTA that is seen in up to 45% of patients.61,62 Up to two-thirds of patients have been treated in the previous several days with appropriate antibiotics before presenting to the ED with worsening symptoms.
Historically, PTAs were considered to be a progression of disease that began with pharyngitis. However, between 20% and 66% of patients do not have a preceding pharyngeal infection.61,63 The tonsillar crypts and Weber's glands instead may be the initial source of infection. Fibrous septations direct infection superiorly and anteriorly. The bacteria most commonly implicated are Group A beta hemolytic Streptococci followed by Bacteriodes. Less than 2% of PTAs are associated with mononucleosis.60
Key Clinical Features. Patients usually present complaining of up to one week of odynophagia, dysphagia, unilateral symptoms, and fever, often accompanied by drooling and a "hot potato" voice.61 Physical examination at times is initially difficult due to trismus. The clinical signs most predictive of a PTA are trismus, uvular deviation, and inferior displacement of the superior pole of the tonsil.62
Effective Diagnostic Strategies. Although many clinicians may disagree, the literature demonstrates that diagnosing PTAs often is difficult. In a prospective trial, clinical findings were only 78% sensitive and 50% specific for diagnosing PTA.64 Needle aspiration may confirm the diagnosis but likewise has a 10-24% false-negative rate.65
Intra-oral ultrasound provides an easy-to-learn diagnostic and procedural adjunct to distinguish PTA from hypertrophic tonsillitis or peritonsillar cellulitis. A covered endocavitary probe is placed in the oral cavity against the tonsillar pillar to view the abscess cavity. When only an ill-defined cavity is seen, a subtle swirl of fluid resulting from gentle posterior probe pressure helps to confirm a fluid-filled abscess. In experienced hands, the sensitivities and specificities of intra-oral ultrasound range from 89-100% and 88-100%, respectively, without the radiation or IV dye associated with a CT scan.64,66 Furthermore, ultrasound localizes the internal carotid artery and safely guides needle aspiration.
Even after a negative aspiration and ultrasound, in the high-risk ill-appearing patient, a CT of the neck to evaluate for the presence of other life-threatening head and neck infections is useful, since physical examination alone underestimates disease severity in up to 70% of patients.67,68 Laboratory testing is not typically helpful, although patients will often have an elevated WBC count.
Emergent Management. For the majority PTA patients, IV fluids, steroids, and analgesics provide enough relief from pain and trismus to allow for a thorough examination followed by drainage. Patients receiving a single high dose of steroids have decreased hospital stay, trismus, pain, and fever over antibiotics alone.69 Antibiotics for PTAs should cover aerobic and anaerobic flora. Generally, penicillin has been the standard, with the addition of metronidazole for anaerobic coverage with significant infections. For penicillin-allergic patients, use clindamycin since macrolide resistance is increasing.60 Definitive management requires drainage via needle aspiration or incision and drainage.
Needle aspiration is the less painful, easier-to-perform technique. A meta-analysis of three prospective trials found that needle aspiration had similar success rates to incision and drainage: 93.7% and 91.6%, respectively.70 More recent case series have described novel techniques to simplify drainage and improve patient comfort. Braude described using the bottom half of a plastic vaginal speculum in order to depress the tongue and provide illumination concurrently.71 The speculum's light source obviates the need for a separate tongue depressor and light source. Chang described using a Trendelenberg position for patients during drainage rather than the traditional upright or semi-upright positioning. The physician is at the head of the bed and approaches the drainage site in the same manner as if placing a laryngoscope for intubation.72 Leong described a new technique for identifying the drainage site. The intersection of an imaginary horizontal line from the base of the uvula and a vertical line from the base of the anterior palatal arch determines where to perform drainage. The point of intersection corresponds to the superior peritonsillar bulge where the abscess cavity is most often easily drained.73
Aiming directly posterior, insert a 16- or 18-gauge needle with syringe attached no more than one centimeter deep, remembering that the internal carotid artery can track just 1.5 cm laterally. With a large or inadequately draining abscess, consider incision and drainage. A simple vertical incision approximately a half centimeter in length and no more than one centimeter deep is sufficient.74 Bite blocks created from tongue blades taped together, a piece of tape to mark the needle or blade 1 cm from the tip, and ultrasound guidance all help make drainage easier and safer.
The majority of patients can be successfully managed as outpatients following ED needle aspiration.75 Admit patients with active immunosuppressive disease such as diabetes or with any signs of airway compromise.75 Discharged patients should return for reassessment within 48 hours, since repeat drainage may be necessary if re-accumulation occurs. Patients who return with re-accumulation of pus often require admission for tonsillectomy.
Complications, Controversies, and Cutting Edge. In deep space head and neck infections, pus does not respect tissue planes. Complications related to PTAs include spread of the infection into the nearby lateral pharyngeal, retropharyngeal, or submandibular space. From the lateral pharyngeal space, pus may spread into the carotid sheath or other facial planes of the neck. Ludwig's angina, Lemierre's syndrome (septic thrombophlebitis of the internal jugular vein), and necrotizing fasciitis have all been reported. With the help of gravity and time, infection may spread to the mediastinum or lungs.
Figure 5: CT of Necrotizing Fasciitis

Case Conclusion. A CT scan revealed necrotizing fasciitis of the neck extending from the lateral pharyngeal space to the level of the cricoid cartilage. The patient was immediately taken to the operating room for neck debridement and tracheostomy. (See Figure 5.)
References
1. Winters M. Evidence-based diagnosis and management of ENT emergencies. Medscape Review 2007.
2. Bas M, Adams V, Suvorava T, et al. Nonallergic angioedema: Role of bradykinin. Allergy 2007;62:842-856.
3. Zuraw BL. Clinical practice. Hereditary angioedema. N Engl J Med 2008;359:1027-1036.
4. Bork K, Hardt J, Schicketanz KH, et al. Clinical studies of sudden upper airway obstruction in patients with hereditary angioedema due to C1 esterase inhibitor deficiency. Arch Intern Med 2003;163:1229-1235.
5. Vleeming W, van Amsterdam JG, Stricker BH, et al. ACE inhibitor-induced angioedema. Incidence, prevention and management. Drug Saf 1998;18:171-188.
6. Chiu AG, Newkirk KA, Davidson BJ, et al. Angiotensin-converting enzyme inhibitor-induced angioedema: A multicenter review and an algorithm for airway management. Ann Otol Rhinol Laryngol 2001;110:834-840.
7. Banerji A, Clark S, Blanda M, et al. Multicenter study of patients with angiotensin-converting enzyme inhibitor-induced angioedema who present to the emergency department. Ann Allergy Asthma Immunol 2008;100:327-332.
8. Grant NN, Deeb ZE, Chia SH. Clinical experience with angiotensin-converting enzyme inhibitor-induced angioedema. Otolaryngol Head Neck Surg 2007;137:931-935.
9. Chiu AG, Krowiak EJ, Deeb ZE. Angioedema associated with angiotensin II receptor antagonists: Challenging our knowledge of angioedema and its etiology. Laryngoscope 2001;111:1729-1731.
10. van Amsterdam JG, te Biesebeek JD, van de Kuil T, et al. Repeated administration of whole-cell and acellular pertussis vaccines affects haemodynamics and autonomic responsiveness. Vaccine 1998;16:1668-1674.
11. Cohen EG, Soliman AM. Changing trends in angioedema. Ann Otol Rhinol Laryngol 2001;110:701.
12. Banerji A, Oren E, Hesterberg P, et al. Ten-year study of causes of moderate to severe angioedema seen by an inpatient allergy/immunology consult service. Allergy Asthma Proc 2008;29:88-92.
13. Zirkle M, Bhattacharyya N. Predictors of airway intervention in angioedema of the head and neck. Otolaryngol Head Neck Surg 2000;123:240-245.
14. Ishoo E, Shah UK, Grillone GA, et al. Predicting airway risk in angioedema: Staging system based on presentation. OtolaryngolHead Neck Surg 1999;121:263.
15. Banerji A, Sloane DE, Sheffer AL. Hereditary angioedema: A current state-of-the-art review, V: Attenuated androgens for the treatment of hereditary angioedema. Ann Allergy Asthma Immunol 2008;100:S19-22.
16. Hill BJ, Thomas SH, McCabe C. Fresh frozen plasma for acute exacerbations of hereditary angioedema. Am J Emerg Med 2004;22:633.
17. Pekdemir M, Ersel M, Aksay E, et al. Effective treatment of hereditary angioedema with fresh frozen plasma in an emergency department. J Emerg Med 2007;33:137-139.
18. Craig TJ, Wasserman RL, Levy RJ, et al. Prospective study of rapid relief provided by c1 esterase inhibitor in emergency treatment of acute laryngeal attacks in hereditary angioedema. J Clin Immunol 2010;30:823-829.
19. Nakamura H, Tanaka H, Matsuda A, et al. Acute epiglottitis: A review of 80 patients. J Laryngol Otol 2001;115:31-34.
20. Alcaide ML, Bisno AL. Pharyngitis and epiglottitis. Infect Dis Clin North Am 2007;21:449-469.
21. Berger G, Landau T, Berger S, et al. The rising incidence of adult acute epiglottitis and epiglottic abscess. Am J Otolaryngol 2003;24:374-383.
22. Mayo-Smith MF, Spinale JW, Donskey CJ, et al. Acute epiglottitis. An 18-year experience in Rhode Island. Chest 1995;108:1640.
23. Cheung CS, Man SY, Graham CA, et al. Adult epiglottitis: 6 years experience in a university teaching hospital in Hong Kong. Eur J Emerg Med 2009;16:221-226.
24. Briem B, Thorvardsson O, Petersen H. Acute epiglottitis in Iceland 1983-2005. Auris Nasus Larynx 2009;36:46-52.
25. Ng HL, Sin LM, Li MF, et al. Acute epiglottitis in adults: A retrospective review of 106 patients in Hong Kong. Emerg Med J 2008;25:253-255.
26. Mayo-Smith MF, Spinale J. Thermal epiglottitis in adults: A new complication of illicit drug use. J Emerg Med 1997;15:483.
27. Carey MJ. Epiglottitis in adults. Am J Emerg Med 1996;14:421.
28. Wong EY, Berkowitz RG. Acute epiglottitis in adults: The Royal Melbourne Hospital experience. ANZ J Surg 2001;71:740-743.
29. Price IM, Preyra I, Fernandes CM, et al. Adult epiglottitis: A five-year retrospective chart review in a major urban centre. CJEM 2005;7:387-390.
30. Hafidh MA, Sheahan P, Keogh I, et al. Acute epiglottitis in adults: A recent experience with 10 cases. J Laryngol Otol 2006;120:310-313.
31. Yong MG, Choo MJ, Yum CS, et al. Radiologic laryngeal parameters in acute supraglottitis in Korean adults. Yonsei Med J 2001;42:367.
32. Ducic Y, Hebert PC, MacLachlan L, et al. Description and evaluation of the vallecula sign: A new radiologic sign in the diagnosis of adult epiglottitis. Ann Emerg Med 1997;30:1.
33. Smith MM, Mukherji SK, Thompson JE, et al. CT in adult supraglottitis. AJNR Am J Neuroradiol 1996;17:1355-1358.
34. Ozanne A, Marsot-Dupuch K, Ducreux D, et al. Acute epiglottitis: MRI. Neuroradiology 2004;46:153-155.
35. Katori H, Tsukuda M. Acute epiglottitis: Analysis of factors associated with airway intervention. J Laryngol Otol 2005;119:967-972.
36. Kim SG, Lee JH, Park DJ, et al. Efficacy of spinal needle aspiration for epiglottic abscess in 90 patients with acute epiglottitis. Acta Otolaryngol 2009;129:760-767.
37. Viehweg TL, Roberson JB, Hudson JW. Epistaxis: Diagnosis and treatment. J Oral Maxillofac Surg 2006;64:511-518.
38. Awan MS, Iqbal M, Imam SZ. Epistaxis: When are coagulation studies justified? Emerg Med J 2008;25:156-157.
39. Krempl GA, Noorily AD. Use of oxymetazoline in the management of epistaxis. Ann Otol Rhinol Laryngol 1995;104:704.
40. Cara DM, Norris AM, Neale LJ. Pain during awake nasal intubation after topical cocaine or phenylephrine/lidocaine spray. Anaesthesia 2003;58:777.
41. Noorily AD, Noorily SH, Otto RA. Cocaine, lidocaine, tetracaine: Which is best for topical nasal anesthesia? Anesth Analg 1995;81:724.
42. Corbridge RJ, Djazaeri B, Hellier WP, et al. A prospective randomized controlled trial comparing the use of merocel nasal tampons and BIPP in the control of acute epistaxis. Clin Otolaryngol Allied Sci 1995;20:305-307.
43. Singer AJ, Blanda M, Cronin K, et al. Comparison of nasal tampons for the treatment of epistaxis in the emergency department: A randomized controlled trial. Ann Emerg Med 2005;45:134.
44. Badran K, Malik TH, Belloso A, et al. Randomized controlled trial comparing Merocel and Rapid Rhino packing in the management of anterior epistaxis. Clin Otolaryngol 2005;30:333-337.
45. Hartley C, Axon PR. The Foley catheter in epistaxis management a scientific appraisal. J Laryngol Otol 1994;108:399-402.
46. Mathiasen RA, Cruz RM. Prospective, randomized, controlled clinical trial of a novel matrix hemostatic sealant in patients with acute anterior epistaxis. Laryngoscope 2005;115:899-902.
47. Vaiman M, Martinovich U, Eviatar E, et al. Fibrin glue in initial treatment of epistaxis in hereditary haemorrhagic telangiectasia (Rendu-Osler-Weber disease). Blood Coagul Fibrinolysis 2004;15:359-363.
48. Douglas R, Wormald PJ. Update on epistaxis. Curr Opin Otolaryngol Head Neck Surg 2007;15:180-183.
49. Moshaver A, Harris JR, Liu R, et al. Early operative intervention versus conventional treatment in epistaxis: Randomized prospective trial. J Otolaryngol 2004;33:185-188.
50. Schlosser RJ. Clinical practice. Epistaxis. N Engl J Med 2009;360:784-789.
51. Herkner H, Laggner AN, Mullner M, et al. Hypertension in patients presenting with epistaxis. Ann Emerg Med 2000;35:126.
52. Herkner H, Havel C, Mullner M, et al. Active epistaxis at ED presentation is associated with arterial hypertension. Am J Emerg Med 2002;20:92.
53. Karras DJ, Ufberg JW, Harrigan RA, et al. Lack of relationship between hypertension-associated symptoms and blood pressure in hypertensive ED patients. Am J Emerg Med 2005;23:106.
54. Hacihanefioglu A, Tarkun P, Gonullu E. Use of recombinant factor VIIa in the management and prophylaxis of bleeding episodes in two patients with Bernard-Soulier syndrome. Thromb Res 2007;120:455-457.
55. Lieberman BL, Kennedy MK, Lorenzo DR, et al. Control of life-threatening head and neck hemorrhage after dental extractions: A case report. J Oral Maxillofac Surg 2010;68:2311-2319.
56. Midathada MV, Mehta P, Waner M, et al. Recombinant factor VIIa in the treatment of bleeding. Am J Clin Pathol 2004;121:124-137.
57. Jacobs JR, Levine LA, Davis H, et al. Posterior packs and the nasopulmonary reflex. Laryngoscope 1981;91:279.
58. Loftus BC, Blitzer A, Cozine K. Epistaxis, medical history, and the nasopulmonary reflex: What is clinically relevant? Otolaryngol Head Neck Surg 1994;110:363.
59. Sunnergren O, Swanberg J, Molstad S. Incidence, microbiology and clinical history of peritonsillar abscesses. Scand J Infect Dis 2008;40:752-755.
60. Hanna BC, McMullan R, Gallagher G, et al. The epidemiology of peritonsillar abscess disease in Northern Ireland. J Infect 2006;52:247-253.
61. Marom T, Cinamon U, Itskoviz D, et al. Changing trends of peritonsillar abscess. Am J Otolaryngol 2010;31:162-167.
62. Kilty SJ, Gaboury I. Clinical predictors of peritonsillar abscess in adults. J Otolaryngol Head Neck Surg 2008;37:165-168.
63. Dunn N, Lane D, Everitt H, et al. Use of antibiotics for sore throat and incidence of quinsy. Br J Gen Pract 2007;57:45-49.
64. Scott PM, Loftus WK, Kew J, et al. Diagnosis of peritonsillar infections: A prospective study of ultrasound, computerized tomography and clinical diagnosis. J Laryngol Otol 1999;113:229-232.
65. Ramirez-Schrempp D, Dorfman DH, Baker WE, et al. Ultrasound soft-tissue applications in the pediatric emergency department: To drain or not to drain? Pediatr Emerg Care 2009;25:44-48.
66. Blaivas M, Theodoro D, Duggal S. Ultrasound-guided drainage of peritonsillar abscess by the emergency physician. Am J Emerg Med 2003;21:155-158.
67. Crespo AN, Chone CT, Fonseca AS, et al. Clinical versus computed tomography evaluation in the diagnosis and management of deep neck infection. Sao Paulo Med J 2004;122:259-263.
68. Reynolds SC, Chow AW. Life-threatening infections of the peripharyngeal and deep fascial spaces of the head and neck. Infect Dis Clin North Am 2007;21:557-576.
69. Ozbek C, Aygenc E, Tuna EU, et al. Use of steroids in the treatment of peritonsillar abscess. J Laryngol Otol 2004;118:439-442.
70. Johnson RF, Stewart MG, Wright CC. An evidence-based review of the treatment of peritonsillar abscess. Otolaryngol Head Neck Surg 2003;128:332-343.
71. Braude DA, Shalit M. A novel approach to enhance visualization during drainage of peritonsillar abscess. J Emerg Med 2008;35:297-298.
72. Chang EH, Hamilton GS. Novel technique for peritonsillar abscess drainage. Ann Otol Rhinol Laryngol 2008;117:637-640.
73. Leong SC. Demystifying the technique of aspirating a quinsy. Clin Otolaryngol 2007;32:140-141.
74. Rahn R, Hutten-Czapski P. Quinsy (peritonsillar abscess). Can J Rural Med 2009;14:25-26.
75. Al Yaghchi A, Cruise A, Kapoor K, et al. Out-patient management of patients with a peritonsillar abscess. Clin Otolaryngol 2008;33:52-53.
Life-threatening ear, nose, and throat (ENT) emergencies can present to any emergency department (ED) at any hour of the day or night.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
