Hypertension
Hypertension
Author:
Michael B. Holliday, MD, Assistant Professor, Department of Family and Community Medicine, University of Cincinnnati, OH.
Peer Reviewer:
Sula Mazimba, MD, MPH, Cardiology Fellow, Kettering Medical Center, Kettering, OH.
Introduction
The Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure is currently updating its work in JNC-7 in an effort to help physicians provide safe, effective, and patient-centered care for those with elevated blood pressure. Since JNC-7's release in 2003, hypertension continues to merit the attention of those in primary care. The estimated prevalence of hypertension in 2006 was 74,500,000. It is estimated that only 77.6% of these patients are aware of their condition, and of those who are under current treatment (67.9%), only 44.1% have their blood pressure under control. Hypertension increases the chance of cerebrovascular, cardiovascular, and renal disease and, at the age of 50, shortens one's lifespan by about 5 years.1 JNC-7 clarified the primary care provider's responsibilities when addressing this problem, which include: making an accurate diagnosis of hypertension; assessing cardiovascular risk; detecting target organ damage; identifying secondary causes of hypertension; and formulating a patient-centered treatment plan.2 This review will clarify these responsibilities in light of information available after JNC-7's release that may influence JNC-8's recommendations, which are anticipated in the fall of 2011.
First Things First: Accurate Blood Pressure Measurement
Despite the importance of hypertension, errors in blood pressure measurement are common. In fact, in a commentary on the JNC-6 report, Norman M. Kaplan, MD, lamented, "The measurement of blood pressure is likely the clinical procedure of greatest importance that is performed in the sloppiest manner."3 For example, one study noted that, of 114 physicians, 0% followed all of the American Heart Association recommendations. These providers followed individual recommendations anywhere from 3% (proper cuff size) to 38% (palpation to initially assess systolic blood pressure).4 These common errors result in significant inaccuracy. For example, smoking within 30 minutes of measurement can raise one's systolic blood pressure (SBP) 6-20 mm Hg. A patient who is talking or actively listening may have a 10 mm elevation of SBP. Placing the blood pressure cuff over the patient's clothing can increase the SBP by 5-20 mm Hg. Finally, an unsupported back can raise the SBP by 6-10 mm Hg.5 Such "sloppiness," if applied across large populations, has significant public health implications. As a case in point, a systematic overestimation of blood pressure by only 5 mm Hg could misclassify 27 million people as being hypertensive, resulting in excessive cost and potential harm to patients who might receive inappropriate treatment.6
Table 1: Blood Pressure Measurement
|
Reference: Williams JS, Brown SM, et al. Blood-pressure measurment. N Engl J Med 2009;360:e6. |
|
|
Contraindications |
Arterial–venous shunt, recent axillary node dissection |
|
Manometer |
Calibrated every six months |
|
Exam room |
Quiet, comfortable ambient temperature |
|
Patient preparation |
No recent food, caffeine, tobacco, physical activity for the past 30 minutes. Patient should not talk or listen to others talk while during measurement. |
|
Patient position |
Back and legs supported, feet on floor. |
|
Arm preparation |
Bare to the shoulder. Rolled-up sleeve should not constrict blood flow or interfere with cuff placement. |
|
Appropriately sized cuff |
Bladder encircled at least 80% of the arm circumference |
|
Cuff placement |
2 cm above the elbow crease, midline of bladder over the brachial artery. Snug but loose enough to allow two fingers to slide under the cuff |
|
Pulse obliteration pressure (POP) |
While palpating the radial artery, insufflate the cuff quickly to 80 mm. Then inflate the cuff about 10mm every 2-3 seconds noting the pressure when the radial pulse disappears. This is the POP. It is confirmed by slowly deflating the cuff, noting the point at which the pulse reappears. |
|
Measure the blood pressure |
Inflate the cuff to 20-30 mm above the POP, then deflate the cuff at a rate of 2 mm per second while listening for the appearance (SBP) and disappearance (DBP) of Korotkoff sounds. |
|
If sounds do not disappear ("persistent systole") |
May occur in elderly, pregnant patients and those with atherosclerotic disease. Muting of the pulsation sound (known as the fourth Korotkoff sound) estimates DBP |
Blood pressure measurement, though simple, demands some thought and preparation. Although one's use of the bell or the diaphragm has no statistical significance with respect to accuracy,7 many other steps are critical for effective blood pressure measurement.8 (See Table 1.)
Blood Pressure Measurement Outside the Office. As noted earlier, health care providers often make decisions based on pressures obtained in the wrong way. In addition, there is increasing consensus that some management decisions are based on data collected at the wrong time. Although office blood pressure measurement is an important tool to screen for elevated blood pressure and monitor those with hypertension, blood pressure is an inherently labile biomarker, with systolic variations up to 20 mm Hg being normal. Not surprisingly, there is increasing evidence that 24-hour ambulatory blood pressure monitoring (ABPM) and home blood pressure monitoring (HBPM) more closely approximate mean blood pressure than office blood pressure. As a result, several organizations have proposed more widespread use of home blood pressure monitoring within guidelines that increase its accuracy and ambulatory blood pressure monitoring when HBPM yields indeterminate results.9 In addition to approximating mean blood pressure, HBPM and ABPM provides additional insights. For example, these techniques can diagnose white-coat hypertension, a condition in which one has an elevated blood pressure in the office but normal mean ambulatory blood pressure. This condition, if managed as true hypertension, could result in the unnecessary treatment of 20% of hypertensive patients.10 Additionally, blood pressure measurement outside the office may detect unacceptably low pressures in the elderly, who are known to have higher rates of both white-coat hypertension and blood pressure variability. Between HBPM and 24-hour ABPM, 24-hour monitoring provides superior data. However, its use is not widespread due reimbursement issues.11 On the other hand, home blood pressure cuffs are affordable, widely available, and their use has been increasingly popular among patients and physicians alike.9 Due to the potential savings incurred by preventing unnecessary treatment, and its superior accuracy, some use of ABPM is certainly justified. Figure 1 shows the integration of office evaluation, HBPM and 24-hour monitoring in arriving at a diagnosis and treatment plan of those with elevated blood pressure.
Figure 1: Integration of Office and Home Blood Pressure Measurement
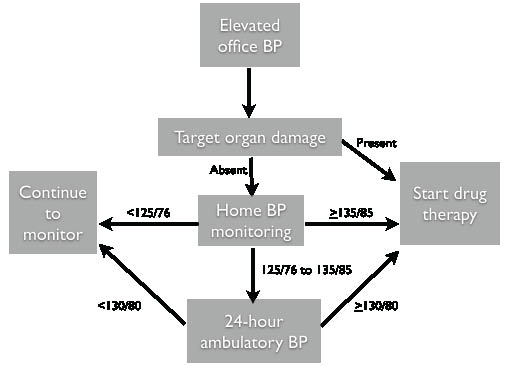
Note that the threshold blood pressures listed refer to the average of several readings obtained outside the office.
Reprinted with permission from: Pickering TG, Houston Miller N, et al. Call to action on use and reimbursement for home blood pressure monitoring: A joint scientific statement from the American Heart Association, American Society of Hypertension, and Preventative Cardiovascular Nurses Association. Hypertension 2008;52:10-29.
Hypertension: Diagnosis and Staging
In JNC-7, blood pressure is classified as normal (SBP < 120 and DBP < 80), prehypertension (SBP 120-139 or DBP 80-89), stage 1 hypertension (SBP 140-159 or DBP 90-99), or stage 2 hypertension (SBP ≥ 160 or DBP ≥ 100). This classification is based on the average of two or more properly measured readings obtained during two or more office visits. Since JNC-7, however, some have questioned applying strict definitions of hypertension based on blood pressure alone to large populations. In a newer classification proposed by the Writing Group of the American Society of Hypertension, blood pressure, along with other biomarkers such as target organ damage and the presence of cardiovascular disease, determine the stage of hypertension. In this model, the staging represents the degree to which the disease of hypertension has progressed, causing cardiovascular and other target organ disease.12 Some expect that such a patient-centered approach will be represented in JNC-8 by a greater emphasis on global cardiovascular risk when managing patients with elevated blood pressure.13
Cardiovascular Risk Assessment
Cardiovascular risk assessment is a fundamental role of the primary care provider. Because multiple cardiovascular risk factors tend to pool together in hypertensive patients, primary prevention potentially offers even greater benefit to this population than those without the disease. For example, in one study of 57,573 patients with hypertension but no CV disease, 56% had diabetes, hyperlipidemia, or a high body mass index (BMI).14 Assessing CV risk involves identifying CV risk factors (see Table 2) and estimating global cardiovascular risk with a tool such as the Framingham risk calculator (http://hp2010.nhlbihin.net/atpiii/calculator.asp). Once identified, modifiable risk factors are addressed in the context of one's CV risk and the risk/benefits of potential treatments. Despite the value of risk assessment, primary care providers are more likely to acknowledge CV risk factors when CV disease is already present, resulting in lost opportunities for primary prevention. For example, in a Dutch study of general practitioners, which analyzed the identification of smoking, diabetes, hypertension, and hyperlipidemia prior to the onset of a CV event, only 10% of patients had all four risk factors investigated prior to the onset of overt CV disease.15 The impact of cardiovascular risk on blood pressure targets should be noted. Particularly, in the high-risk patient such as those with diabetes mellitus, chronic kidney disease, known CAD or CAD equivalent (carotid artery disease, peripheral arterial disease, abdominal aortic aneurysm), or 10-year Framingham risk score >10%, the target blood pressure in current guidelines is less than 130/80.16
Identify and Address Target Organ Damage
Target organ damage (TOD) refers to disease of the eye, brain, kidney, heart, and blood vessels that results from hypertension. The search for TOD enables the primary care provider to better assess cardiovascular risk, often results in lower blood pressure treatment targets, and helps providers individualize treatment plans.
Brain. Target organ damage in the brain includes stroke (hemorrhagic or ischemic) and transient ischemic attack (TIA). The search for TOD in the brain includes history to elicit past temporary or permanent neurological deficits, focused physical examination, and further diagnostic evaluation such as MRI, echocardiography, and carotid ultrasound if not already done. With respect to secondary prevention, the management of hypertension surpasses other measures such as anti-platelet and lipid-lowering therapy in importance, because it is the most significant modifiable risk factor for stroke.17 A distinction should be made, however, between blood pressure management in acute stroke and that for secondary prevention. The reader is referred to another review regarding the acute setting, in which it is noted that antihypertensive management for secondary prevention of stroke may start 24-72 hours after the onset of stroke symptoms, provided that the neurologic deficits are not worsening during this period.18 Trials indicate that patients with a history of stroke and TIA are likely to benefit from blood pressure reduction even without a prior history of hypertension. Although an exact target is not clear, benefit has been associated with an average reduction of 10/5 mm Hg, and one should consider a goal of bringing the blood pressure to normal levels (120/80 mm Hg). When choosing a specific agent to lower one's blood pressure after a stroke, there are multiple classes to choose from. Angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics, and calcium channel blockers all appear to lower the risk of stroke. This reduction seems to be more related to the extent of blood pressure lowering than the choice of a specific drug. One exception to this rule is the beta-blocker class, which has been represented by atenolol in many studies. Beta-blockers exert a relatively weak effect in reducing stroke compared to placebo or no treatment, and, compared to calcium channel blockers, ACE inhibitors or thiazides show evidence of worse outcome with respect to stroke.19 This trend may be explained by higher central aortic pressures seen in patients treated with beta-blockers compared to those treated with other agents.
Heart. Target damage in the heart includes congestive heart failure (CHF), coronary artery disease (CAD), and left ventricular hypertrophy (LVH). Hypertension is the most common condition contributing to CHF in the general population.20 History consistent with CHF includes dyspnea on exertion, cough, orthopnea, paroxysmal nocturnal dyspnea, peripheral edema, nocturia, and fatigue. Physical examination may reveal tachycardia, rales, jugular venous distension, hepatomegaly, third or fourth heart sounds, and displacement of the point of maximal impulse.21 Initial diagnostic testing includes the resting ECG, transthoracic two-dimensional echocardiography with Doppler, and additional testing and consultation if indicated. Once a diagnosis of CHF is confirmed, one must choose an antihypertensive regimen that not only lowers blood pressure, but also slows the progression of cardiac disease. In patients with reduced systolic function, the foundation of this effort consists of neurohormonal blockade by ACE inhibitors (or angiotensin receptor blockers if ACE inhibitors are not tolerated) and beta-blockers titrated to evidence-based dosing targets without causing symptomatic hypotension. Although the benefit from ACEIs and ARBs seem to be class-specific, evidence points to a drug-specific benefits with metoprolol succinate, bisoprolol, and carvediolol within the beta-blocker class. In those with dyspnea with minimal activity or at rest (New York Heart Association Class III and IV, respectively), aldosterone antagonists such as spironolactone and eplerenone are also recommended. The combination of hydralazine and nitrates, although proven to improve survival in those with heart failure, provides less benefit than ACEIs or ARBs and should be used only in those who remain hypertensive with neurohormal blockade or are unable to take ACEIs or ARBs. One should also keep in mind that non-dihydropyridine calcium channel blockers such as diltiazem and verapamil should be avoided in patients with systolic dysfunction.22,23 In patients who have heart failure with preserved systolic function, therapeutic recommendations are less well-defined. Multiple antihypertensive drugs potentially can be used to accomplish the following objectives: reduce venous congestion with venodilators and diuretics in cases of pulmonary edema; control the heart rate, particularly in atrial fibrillation with agents such as beta-blockers or non-dihydropyridine calcium channel blockers; and treat hypertension. Two of these efforts deserve additional explanation. First, preload reduction with venodilating agents and diuretics can be overdone in those with reduced left ventricular compliance, resulting in hypotension and syncope. As for the third objective, there does not seem to be consensus on how to accomplish it. However, given that diastolic dysfunction without overt failure is often associated with LVH, one should consider treatments that effectively lower blood pressure and cause LVH regression (see below).24
In addition to CHF, coronary artery disease also affects hypertension management and should be screened for in patients with elevated blood pressure. Historical clues to CAD include substernal discomfort or even breathlessness, fatigue, and/or nausea (so-called "anginal equivalents") in response to exertion or emotional upset.25 The resting electrocardiogram, recommended for every patient with elevated blood pressure, may reveal significant Q waves, poor R wave progression, ST segment depression, and T wave flattening or inversion. In addition to pointing to the need for further testing for definitive diagnosis, many of these changes have significant prognostic value. For example, one study found ST segment depression to have a risk ratio (RR) of 4.71 for cardiovascular disease death.26 In patients with angina pectoris or previous myocardial infarction, treatment consists of lowering blood pressure to less than 130/80 utilizing a beta-blocker and an ACEI or an ARB. If the blood pressure target is not reached with these medications alone, dihydropyridine calcium channel blockers and thiazide diuretics can also be used. For those patients with LV dysfunction and CAD, guidelines for systolic dysfunction should be used, including using beta-blockers known to be effective in these patients and avoiding negatively inotropic calcium channel blockers such as diltiazem and verapamil.
LVH should also be searched for and addressed when evaluating high BP. Within the primary care setting, LVH is found in approximately one-third of patients with hypertension.27 It is associated with an up to a five- to ten-fold increased risk of cardiovascular events. Obesity and elevated blood pressure are the two strongest predictors for the presence of LVH. However, LVH is a potential predictor of cardiovascular events even in those with normal blood pressure, carrying an independent risk ratio of 2.5.28 Once found, specific therapies can be employed to lower blood pressure while maximally reducing left ventricular mass. Although there is obvious utility in detecting LVH, screening for this disorder poses challenges. Presently, the resting ECG is the most widely available test to screen for LVH. Although the ECG has a specificity of up to 96%, its sensitivity of only 15-21% renders it unreliable as a screening tool to rule out this important condition.29 Echocardiography, on the other hand, is both sensitive and specific and often gives additional diagnostic information, including valvular function. Some have suggested the echocardiogram provides higher diagnostic accuracy at a price that precludes its widespread use in hypertension evaluation and excludes its presence from current hypertension guidelines.30 Potential ways to overcome this barrier include keeping in mind other indications for echocardiography such as a murmur requiring additional evaluation, suspected pulmonary hypertension, or symptoms attributed to a potential cardiac etiology such as dyspnea on exertion.31 Additionally, one can order a limited echocardiographic study at a cost of $255 (2009 Medicare Ambulatory Payment Classification) in order to estimate the left ventricular mass if there are no other indications for a complete study ($431). Once accomplished, the detection of LVH provides an opportunity to individualize therapy in an effort to reverse this damage while lowering blood pressure. Studies show this strategy reduces cardiovascular outcomes independent of blood pressure reduction. The agents shown to be effective at facilitating regression of LVH are, in descending order of LV mass reduced, ARBs (13%), calcium channel blockers (11%), ACEIs (10%), diuretics (8%), and beta-blockers (6%).29
Kidney. In the United States, chronic kidney disease, defined as kidney damage and/or reduction in glomular filtration rate (GFR), has increased from 10% of the general population during the years of 1988-1994 to 13% during 1999-2004.32 Since 1995, hypertension has been second only to diabetes as the leading cause of chronic kidney disease in the United States. Target organ damage in the kidney occurs via eventual nephron loss due to a process known as nephrosclerosis.33 The search for TOD in the kidney facilitates several objectives. Appropriate evaluation enables the provider to initiate more aggressive, targeted treatment aimed to reduce blood pressure and further nephron loss. Additionally, if chronic kidney disease is found, one can follow through with guidelines from the National Kidney Foundation, which stress classifying the severity of kidney disease after estimating the GFR (Stage 1 = kidney damage and GFR ≥ 90, stage 2 = kidney damage and GFR 60-89, stage 3 = GFR of 30-59, stage 4 = GFR of 15-29, and stage 5 = GFR of < 15), diagnosing the cause of CKD (which may encompass etiologies other than HTN), initiating definitive treatment, addressing complications of CKD, and referring to a nephrologist when needed.34,35 In patients with elevated blood pressure, kidney disease is found after estimating GFR and measuring proteinuria. GFR is estimated by calculating the creatinine clearance, which is done by inputting serum creatinine into either the MDRD study equation or the Cockcroft–Gaultan equation. The MDRD study equation has the advantage of not requiring a weight for calculation and is often used to report an estimation of GFR on laboratory results. Although not without flaws, these equations are widely accepted and more practical than 24-hour urine collections for the estimation of GFR. One should note, however, the factors other than reduced renal function that can change SCr (increased by African race, ingestion of cooked meats, muscular habitus, trimethoprim, cimetidine, fibric acid derivatives other than gemfibrozil; decreased by advanced age, vegetarian diet, muscle wasting), thus making GFR more difficult to estimate.36 In contrast to GFR, proteinuria is measured directly. In 2003, JNC-7 recommended a urinalysis with urine microalbumin measurement being optional.2 However, given that renal damage, including the presence of microalbuminuria (defined as an albumin-to-creatinine ratio of 30-300 mcg/mg), is associated with a 3.3 times higher risk of having cardiovascular abnormalities,37 one should consider universal measurement of microalbumin in patients with elevated blood pressure. If microalbuminuria is found, a confirmatory test should be done at a later time due to potential confounding factors such as fever and recent strenous exercise. In cases of macroalbuminuria (> 300 mcg/mg), one can also use the protein-to-creatinine ratio from a spot urine sample to approximate the grams of protein excreted during 24 hours.38 If this value is greater than 1, corresponding to greater than 1 gram of protein excreted in 24 hours, and the patient has only mild or moderate hypertension, one should search for causes of proteinuria other than hypertension that may be clinically significant.33 Once the diagnoses of hyptertension and CKD are established, one should choose pharmacotherapy that not only lowers blood pressure, but also slows the progression of renal disease. Drugs that block the renin-angiotensin system (RAS) such as ACEIs and ARBs, are first-line in this regard. Although it is known that ACEIs and ARBs are synergistic at reducing the surrogate outcome, proteinuria, there is ongoing investigation (the VA NEPHRON-D study) to determine if synergistic inhibition of the RAS (such as with an ACEI and an ARB) is safe and results in favorable hard outcomes such as reduced progression to end-stage renal disease. Because patients with CKD may require 3 or more drugs to control blood pressure,39 ACEIs or ARBs will often have to be combined with other medications. Although many other drugs can act synergistically with RAS blockers to lower blood pressure, diuretics and calcium channel blockers are well studied and will be reviewed. Diuretics should be used in most CKD patients who require additional blood pressure reduction. Because thiazides are ineffective in patients with severely reduced GFR, they should be reserved for those with stages 1-3 disease (GFR > 30 mL/min), whereas loop diuretics are used for those with stages 4-5 disease (GFR < 30 mL/min).40 If furosemide is used, its short half-life requires twice-daily dosing, while torsemide can be used once daily.41 Calcium channel blockers are effective at lowering blood pressure. However, their use in CKD requires caution. Calcium channel blockers impair renal autoregulation of blood flow, allowing systemic pressures to be transmitted to the glomerulus. This is more pronounced with dihydropyridine agents such as amlodipine and felodipine and less so with non-dihydropyridine agents such as diltiazem and verapamil, which seem to have a renal-protective effect.42 Because lowering blood pressure is a cardinal goal of chronic kidney disease treatment, CCBs can be used in this setting but should be done so with concurrent ACEI or ARB use.43
Table 2: Cardiovascular Risk Factors
- Hypertension
- Cigarette smoking
- Obesity (body mass index ≥ 30 kg/m2)
- Physical inactivity
- Dyslipidemia
- Diabetes mellitus
- Microalbuminuria or estimated GFR < 60 mL/min
- Age (older than 55 for men, 65 for women)
- Family history of premature cardiovascular disease (men younger than age 55 or women younger than age 65)
Reference: Chobanian AV, Bakris GL, Black HR, et al. Seventh report of the joint national committee on prevention, detection, evaluation and treatment of high blood pressure. Hypertension 2003;42;1206-1252.
Table 3: Causes of Secondary Hypertension45-55
|
Cause |
Epidemiology |
Initial presentation |
Initial diagnostic workup |
Coordination of care as needed for definitive diagnosis and treatment |
Definitive treatment |
Antihypertensive Pharmacotherapy |
|
Primary aldosteronism |
Up to 20% with resistant HTN |
Resistant hypertension, unprovoked hypokalemia (only 1/3 patients) |
ARR >20-30, 24h urinary aldosterone > 12 mcg/24h, thin-cut CT of the adrenals |
Nephrology, endocrine surgery |
Surgery |
Aldosterone antagonists |
|
Pheochromo-cytoma |
0.2-0.4% of hypertensive patients |
Labile or continuous HTN; symptoms of sympathetic overload |
Plasma free metanephrines, urine metanephrines and catecholamines |
Nephrology or endocrinology, endocrine surgery |
Surgery |
Adequate alpha blockade followed by beta1 blockade. |
|
Cushings syndrome |
<1.0% of general population |
Variable manifestations of glucocorticoid excess (see text) |
24h urine free cortisol |
Endocrinology endocrine surgery |
Surgery |
Aldosterone antagonists |
|
Hyperthyroidism |
HTN in 20-26% of hyperthyroid patients |
Wide pulse pressure, tachycardia |
Thyroid function studies |
Endocrinology, nuclear medicine, endocrine surgery |
Antithyroid drugs, radioiodine ablation, surgery |
Beta-blockers |
|
Hypothyroidism |
HTN in 14.8% of hypothyroid patients |
Diastolic hypertension, bradycardia |
Thyroid function studies |
Endocrinology |
Thyroid hormone replacement |
Usually not needed after thyroid replacement |
|
Hyperpara- thyroidism |
HTN in up to 50% of hyperparathyroid patients |
Hypercalcemia with no other symptoms is the most common presentation |
iPTH, 25-OH D level |
Endocrinology, endocrine surgery |
Surgery |
HTN often does not resolve after surgery; avoid thiazides |
|
Renal parenchymal disease |
2.5-5.0% of hypertensive patients |
Micro or macroalbuminuria, decreased GFR |
Urinalysis, laboratory testing, renal ultrasound |
Nephrology as needed for diagnosis, management of complications and renal replacement therapy |
Treat underlying cause if possible |
RAS blockade, additional medications to lower blood pressure. CCBs with caution (continued) |
|
Renal artery stenosis |
3.0-4.0% of hypertensive patients |
New onset resistant HTN in young female, the old or previously controlled patient, abdominal bruit, coexisting atherosclerotic disease |
Imaging |
Nephrology, interventional cardiology, vascular surgery, interventional radiology |
Angioplasty for FMD, stent for astherosclerotic RAS |
RAS blockade (worsening HTN may result in patients with bilateral stenosis); additional treatment as needed |
|
Coarctation of the aorta |
<1% of hypertensive patients |
Differential in upper and lower extremity pulse |
Imaging |
Cardiothoracic surgery, interventional cardiology |
Balloon angioplasty or surgery |
N/A |
|
Obstructive sleep apnea |
30-40% of hypertensive patients |
Obesity, hypersomnolence, snoring |
Polysomnogram |
Sleep medicine, otolaryngology |
CPAP |
Aldosterone antagonists |
Secondary Hypertension
Secondary hypertension (SH), a condition in which a specific etiology is the cause of elevated blood pressure, comprises 5-10% of all HTN cases. The search for SH can result in definitive treatment of HTN and decrease one's dependence on pharmacotherapy, if not eliminate it. Additionally, some causes of SH such as pheochromocytoma, which has a malignancy rate of 10%, may result in morbidity and even mortality independent of hypertension if not discovered.44 Table 3 lists several causes of SH, their prevalence, initial work-up, and treatment. As one can see, some secondary causes are readily diagnosed by history, physical examination, or limited diagnostic studies. On the other hand, many forms of SH pose more of a diagnostic challenge, including renal artery stenosis, Cushing's syndrome, primary hyperaldosteronism, and pheochromocytoma. Primary care providers play a key role in the definitive treatment of these conditions through identifying patients at increased risk of SH, initiating appropriate diagnostic evaluations, referring to other specialists when needed for further diagnosis and treatment, and choosing appropriate antihypertensive medication.
In order to recognize these conditions, one must be able to identify patients with an elevated pretest probability of both SH and a specific condition before embarking on potentially costly work-ups. One should suspect SH in general when there is resistant hypertension (not meeting treatment goal despite adherence to full doses of three antihypertensive medications, one of which is a diuretic), worsening control in a previously controlled patient, blood pressure over 180/110, onset of HTN prior to age 20 or after age 50, significant hypertensive TOD, or lack of a family history of HTN.56 Additionally, SH is more likely if other explanations for apparently resistant HTN are ruled out, such as incorrect measurement, white-coat hypertension, nonadherence to the drug regimen, interfering substances (such as NSAIDs, oral contraceptives, sympathomimetics, stimulants, and licorice), and lifestyle factors (such as obesity, high salt diet, and excessive alcohol intake).57 If SH is a consideration after this initial appraisal, a specific secondary cause or causes can be investigated.
Aldosteronism. Primary hyperaldosteronism (PA) is the most common form of SH and may be present in 20% of patients with resistant HTN. It is classically suspected when one encounters unprovoked hypokalemia. However, this is present in only one-third of cases, potentially causing many patients with this condition to be missed. Because of its prevalence, it is reasonable to work up this condition when resistant hypertension is the only presenting problem. Screening for PA begins with calculating an aldosterone-to-renin ratio (ARR), where the numerator is the plasma aldosterone concentration (ng/dL) and the denominator is the plasma renin activity (ng/mL/h). This test is best done in the morning, and 4 weeks after both correction of hypokalemia and withdrawal of potentially interfering medications such as aldosterone antagonists, potassium-sparing diuretics, and the synthetic progestin drospirenone. Alpha antagonists and nondihydropyridine calcium channel blockers are the least likely blood pressure medications to interfere with testing.
If the ARR is greater than 20-30, confirmatory testing follows, which includes measuring 24-hour urine aldosterone excretion after 3-4 days of salt supplementation (4-6g/d). If the value is greater than 12 mcg/24-h, this is considered positive for PA. Once PA is confirmed, thin-cut CT of the adrenal glands is used to identify the source. The CT may reveal a unilateral nodule, unilateral or bilateral enlargement, multinodular enlargement, or even a completely normal scan (such as with a microadenoma). Surgical management, based largely on imaging results, is outlined in other reviews.45 Medical management of patients with PA includes the use of aldosterone antagonists such as spironolactone or the newer agent, eplerenone, which is less likely to cause endocrine side effects such as gynecomastia.46
Pheochromocytoma. Much less common than PA, pheochromocytoma is found in only 0.2-0.4% of hypertensive patients. One should suspect pheochromocytoma in patients with sustained or paroxysmal hypertension, headaches, palpitations and sweating, although up to 21% may be normotensive.48,49 Additional presentations include orthostatic hypotension, flushing or pallor, anxiety, fatigue, vomiting, and visual disturbances. Further, pheochromocytomas may also present as an adrenal incidentaloma during abdominal imaging. For example, one study found that 4.2% of these adrenal lesions were pheochromocytomas.49
Once suspected, pheochromocytomas are usually diagnosed by detecting excess catecholamine or catecholamine metabolites in the urine and blood. Urine testing consists of measuring catecholamines and metanephrines in two separate 24-h urine collections, accounting for the sporadic nature of the condition. Plasma testing, on the other hand, can detect metanephrines, which are continuously elevated in pheochromocytoma. Urine testing has both a sensitivity and specificity of 98%. Plasma testing of metanephrines has a sensitivity and specificity of 99% and 85-89%, respectively. It should be noted that false-positive results may occur with use of tricyclic antidepressants, high-dose diuretics, levodopa, theophylline, caffeine, and nicotine.46
Once diagnosed biochemically, imaging is done, which may include CT, MRI, positron emission tomography, and nuclear scintigraphy studies to localize the lesion and, in some cases, metastatic disease. In 10% of cases, pheochromocytoma can be extra-adrenal, increasing the challenge of diagnosis. Although surgical treatment is definitive, these patients need to be treated medically as well. One should initiate preoperative pharmacotherapy, consisting of alpha blockade with phenoxybenzamine to prevent a hypertensive crisis and beta-blockade with beta-1-selective agents such as atenolol to control tachycardia. One must remember, however, not to use beta-blockers until there is adequate alpha-blockade, in order to prevent worsening hypertension.48 Finally, because approximately one-fourth of pheopchromocytoma patients have a genetic mutation associated with their disease, a genetics evaluation is recommended for those with pheochromocytoma.58
Cushing's Syndrome. Another endocrine cause of hypertension is Cushing's syndrome. Glucocorticoid excess, assuming it is not iatrogenic, occurs as a result of either ACTH-dependent or ACTH-independent overproduction of cortisol. ACTH-dependent causes typically result from either ACTH secretion from a pituitary adenoma (also known as Cushing's disease), or ectopic ACTH secretion from oat cell, small-cell lung tumors, or carcinoid tumors of the bronchus or thymus. ACTH-independent cortisol release is found with adrenal adenomas, carcinoma, or hyperplasia.46 Cushing's syndrome can present with either classic findings such as moon facies, bruising, purple striae, truncal obesity, buffalo hump and hypertension, or may present with just a few nonspecific isolated findings such as fatigue. Laboratory clues to diagnosis include neutrophilia, hyperglycemia, hyperlipidemia, and hypokalemia.
Once suspected, laboratory diagnosis of Cushing's syndrome begins with 24-h measurement of urinary free cortisol, which is only accurate in those with an estimated GFR greater than 30 mL/min/1.73 m2. Levels greater than 100 mcg/d are suggestive of the disease, with a sensitivity and specificity of 95-100% and 94-98%, respectively. However, due to potential errors in collection and day-to-day changes in hormone level, one should consider more than one collection.59 Additionally, an overnight dexamethasone suppression test can be done, in which the patient takes 1 mg of dexamethasone at midnight and plasma cortisol is measured the next morning at 6 a.m. Values < 2 mcg/dL are considered normal. However, studies have shown that a significant number of Cushing's patients can also have levels in this range, making this approach an unreliable screening tool if used alone.60
Further workup, including differentiation of ACTH-dependent and -independent disease and localization of the disease process to the pituitary, the adrenal gland, or elsewhere, must be done prior to definitive surgical treatment.61 When treating hypertension in the patient with Cushing's syndrome, potassium-sparing diuretics such as spironolactone and amiloride can be useful to correct hypertension and hypokalemia in these patients.46
Renal artery stenosis (RAS) also should be in the differential when SH is considered. Although it is present in only 0.5% of the Medicare population, its prevalence rises in patients who also have conditions such as peripheral vascular disease, hypertension, coronary artery disease, and chronic kidney disease.62 Likewise, several clinical scenarios increase the possibility of RAS. For example, new-onset stage 2 HTN (>160/100) after age 50 or in the absence of a family history of hypertension, azotemia in response to an ACEI, hypertensive crises such as flash pulmonary edema, hypertension in a young female, and resistant hypertension all point to the possibility of RAS.63
Once RAS is suspected, one must use imaging to make the diagnosis. The test of choice depends on available modalities and expertise as well as patient factors such as renal function and body habitus. The available imaging techniques include conventional angiography, CT angiography, contrast-enhanced MR angiography (CE-MRA), renal duplex ultrasonography, and ACEI-enhanced nuclear renography. Conventional angiography provides accurate anatomic detail and enables percutaneous intervention. However, due to radiation and contrast dye exposure, it is not an ideal screening test. CT angiography also provides great anatomic detail and may be more accurate in the detection of fibromuscular dysplasia (FMD) than other noninvasive studies.64 However, contrast exposure also limits its use, particular in those with chronic kidney disease. CE-MRA has the advantage of good anatomic detail and no radiation exposure.
Although the risk of complications from contrast dye with CE-MRA is a consideration, it appears to be less risky than CTA. For example, nephrogenic interstitial fibrosis occurs in < 5% of patients with a GFR of < 30mL/min while contrast-induced nephrotoxicity occurs in 10% of patients who undergo contrast-enhanced CT with GFR < 60 mL/min.65 Therefore, CTA is not ideal in patients with even mild to moderate renal dysfunction, while CE-MRA is a safer option in these patients, as long as the GFR is > 30 mL/min, renal dysfunction is not acute, and there has not been an antecedent pro-inflammatory state such as sepsis or trauma.66
ACEI-enhanced nuclear renography appears to have reasonable sensitivity and specificity and may provide prognostic information; however, it appears to be inaccurate if used in those with diminished renal function. Given the above attributes and limitations, some guidelines favor the following approach: avoiding imaging in those with stable renal function and good blood pressure control where there appears to be little value in diagnosing this condition; using CTA, CE-MRA nuclear renography, or renal duplex ultrasound depending on the technical expertise available and the patient's renal function and limiting invasive renal arteriography to those cases where confirmation of results are needed or percutaneous intervention is likely.67
Once this condition is confirmed, RAS is managed with medical therapy and procedures that correct the stenosis, which are reserved for carefully selected patients. Medical therapy includes blockers of the RAS and additional medications as needed. When definitive treatment is attempted, endovascular procedures are preferred over open procedures. Those with FMD are typically treated with balloon angioplasty, and those with atherosclerotic disease are are treated with stenting.63 Because there is poor evidence of the benefit of procedural interventions for atherosclerotic RAS compared to medical therapy, one should consider enlisting a hypertensive specialist to select patients most likely to experience reduction in blood pressure and preservation of renal function from these procedures.68
Patient-centered Treatment of Hypertension
Treating patients with hypertension in a patient-centered fashion includes recommending appropriate lifestyle changes and pharmacotherapy.
Recommended lifestyle changes for hypertensive patients include dietary changes, increased physical activity, smoking cessation, and weight loss.2 Healthful lifestyle behaviors are not only necessary for effective hypertension management, but also appear to prevent age-related increase in blood pressure that is associated with industrialized nations.69 Dietary recommendations are summarized in the Dietary Approaches to Stop Hypertension (DASH) eating plan. These include increasing fruits, vegetables, low-fat dairy products, and whole grains while reducing saturated and total fat, sodium to less than 2400 mg/d and less than 1500 mg/d if possible.70 In addition, alcohol intake of no greater than 1 drink/d in women and 2 drinks/d in men is recommended, given that 5-7% of hypertension is attributed to heavy alcohol use.71
Increasing physical activity to 30 minutes on most days, which can result in a 4 mm Hg reduction in systolic blood pressure, is also recommended.72 Weight reduction can result in blood pressure reduction. Although maintenance of a BMI of 25 or less is ideal, weight loss of as little as 10 pounds reduces blood pressure significantly.2 Finally, even though smoking is not associated with sustained elevations of blood pressure, smoking cessation should be encouraged for all hypertensive patients who smoke to lower the risk of cardiovascular events.73
Pharmacotherapy
Once the diagnosis of hypertension has been made, target organ damage has been diagnosed, secondary causes of hypertension have been ruled in or out, the patient's history is more fully known, and lifestyle changes have failed to lower blood pressure to target levels, one must formulate a pharmacologic treatment plan. The goals of this plan include slowing progression or preventing target organ damage, achieving target blood pressure levels that are dictated by patient characteristics, and treating secondary causes of hypertension. Further, the specific agents chosen to meet these goals must take into account factors such as cost, comorbid conditions, race, sex, and likelihood of adherence. Secondary hypertension and target organ damage, including implications for drug selection, have already been addressed. What follows is an overview of blood pressure targets in at-risk populations and the major pharmacologic classes of antihypertensive medication.
How Low Should One Go? When choosing a target blood pressure for treatment, especially in groups at risk such as diabetics, and those with CKD or cardiovascular disease, the answer to this question is not clear. The uncertainty arises in part from the way in which blood pressure targets have been selected in the past. When one examines the epidemiology of blood pressure, there is a clear correlation between rising blood pressure and cardiovascular risk starting with a systolic blood pressure as low as 115 mm of Hg. Congruent with this risk and balance by the risk and cost of treatment, the patient with hypertension but no target organ damage or diabetes has a recommended blood pressure goal of < 140/90, and those with a history of diabetes or target organ damage such as chronic kidney disease have an understandably more aggressive target of < 130/80.2
Recently released trials looking at patient-oriented outcomes in selected groups at increased risk have called some of these guidelines into question. In particular, targets for those with diabetes, cardiovascular disease, and chronic kidney disease will be addressed here. Recently, the Action to Control Cardiovascular Risk in Diabetes (ACCORD) blood pressure trial (ACCORD BP) compared cardiovascular outcomes in patients randomized to either intensive blood pressure control with an SBP target of < 120 or standard therapy with an SBP target of < 140. Surprisingly, the intensive therapy group had composite outcomes of fatal and nonfatal cardiovascular events comparable to the standard therapy group, with more adverse effects as a result of treatment. It should be noted, however, that one outcome, stroke, was lower in the intensive treatment group, with the number needed to treat to prevent one stroke over five years being 89.
In light of these results, some have argued that study times longer than ACCORD's five years and randomized trials comparing less aggressive goals that are still lower than standard therapy (such as SBP <130) are needed to clarify the ideal blood pressure target in diabetics. For now, one approach may be increased flexibility in assigning blood pressure targets in diabetics. For example, as long as one is not experiencing symptomatic hypotension, the provider may choose intensive blood pressure treatment in a patient where secondary prevention of stroke is the principle desired goal.
The desired blood pressure target for those with CKD is another area of controversy. For example, in a recent trial comparing the progression of CKD between an intensive treatment group (mean blood pressure = 130/78) and a standard treatment group (mean blood pressure = 141/86), there did not appear to be a difference in disease progression. However, if baseline proteinuria was taken into account, there did seem to be a benefit from intensive treatment for those starting the study with a urine protein to creatinine ratio of > 0.22, which corresponds to a 24h urinary protein excretion of 220 mg/d.74 It is hoped that the Systolic Blood Pressure INTervention (SPRINT) trial, a study of more than 10,000 patients assigned to either a BP goal of < 120/80 or < 140/90, will provide more guidance on the treatment of blood pressure in CKD.
Also up for debate is the best target blood pressure for those with cardiovascular disease. Part of this debate is fueled by the so-called "J-curve" concept, in which both high and low blood pressures, particularly low diastolic pressures, are associated with worse outcomes. However, some feel that many with a lower diastolic pressure who have poor outcomes have that low pressure due to their disease, and that attributing harm to more aggressive treatment of blood pressure may be an error in assigning causation where there is only correlation. Nevertheless, some proponents of the more aggressive target of <130/80 for those with high cardiovascular risk (diabetes, CKD, coronary artery disease, peripheral vascular disease, 10-year Framingham risk score of 10% or higher), concede that rapid or too severe a reduction of diastolic pressure (such as <65 mm Hg) may not be safe.
Pulse pressure (the difference between the systolic and diastolic pressures) may be a major roadblock to balancing adequate reduction of systolic pressure, which is known to be beneficial, and avoiding too low of a diastolic pressure, which may pose harm. Many agents such as renin-angiotensin system blockers, calcium channel blockers, and diuretics improve vascular compliance and thus lower SBP greater than DBP, which reduces pulse pressure. On the other hand, beta-blockers often increase pulse pressure. This is particularly problematic in the elderly, where pulse pressure is a greater determinant of risk than DBP or SBP.75
Major Antihypertensive Drug Classes. When choosing drug therapy for hypertension, it is imperative to be aware of that medication's evidence-based benefits and potential safety issues that may require monitoring or preclude the use of a medication. RAS blockers, diuretics, calcium channel blockers, and beta-blockers will be reviewed in this light.
ACE inhibitors (or angiotensin receptor blockers if ACE inhibitors are not tolerated) have proven to be effective at lowering blood pressure and preventing adverse cardiovascular events in those with uncomplicated hypertension.76 Additionally, these medications reduce the incidence of negative outcomes in those with left ventricular systolic dysfunction, diabetes, previous MI or stroke, high coronary disease risk,77 and chronic kidney disease.78 Additionally, a recent study reveals that these medications, especially in comparison to thiazides and conventional beta-blockers, may decrease the onset of diabetes with a number needed to treat to prevent one case of diabetes being 100 for ACE inhibitors and 50 for ARBs.
Accordingly, one should consider using these medications in the patient with prediabetes and/or metabolic syndrome.79 Despite many potential beneficiaries of RAS blockers, it has been noted that blacks are less likely than whites to reach target blood pressure on these medications. Although this contention is supported by the literature, further study has revealed that failure to reach target blood pressure is in part due to higher baseline blood pressure in blacks in these studies, rather than a striking difference in response to ACE inhibitors between these two racial groups. As a result, some believe that race is superseded by factors such as target organ damage, comorbid conditions, and patient preference when considering use of an ACE inhibitor.80
Notwithstanding the many benefits of ACEIs and ARBs, there are some potential adverse effects that may affect compliance or even preclude use of these drugs in some patients. Due to accumulation of bradykinin caused by ACEIs, 15-30% of patients experience a non-productive cough. Much less common but potentially deadly, angioedema occurs in 0.1-0.7% of patients on ACEIs.81
This adverse effect usually occurs within weeks of initiating use but has been known to have a latency of up to several years after the start of therapy.82 If a patient has a history of angioedema, either idiopathic or hereditary, ACEIs are contraindicated and an ARB should be selected. Similarly, if a patient has had angioedema on an ACEI, and a RAS blocker is indicated, an ARB can be used. One must be mindful, however, that angioedema may still occur with ARB use, and patients should be counseled accordingly.83 Caution is also warranted in the woman of reproductive age, as these medications are associated with fetal harm in all three trimesters.84
Once an ACEI or ARB is initiated in an appropriate patient, serum creatinine and potassium, which may rise as a result of therapy, should be monitored. However, one may safely continue an ACEI or an ARB even if the creatinine rises by up to 20-30% or the potassium increases up to a level of 5.5 mEq/L, as long as these levels do not increase further. To ensure stability, a subsequent measurement should take place within a week.85
Diuretics. In patients with resistant blood pressure, lack of appropriate diuretic therapy has been cited as the most common pharmacologic cause. Thiazides such as hydrochlorothiazide and chlorthalidone are the most common diuretics used for hypertension. The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) demonstrated the superiority of chlorthalidone in preventing adverse cardiovascular events and lowering blood pressure, all at reduced cost at that time.86 Given these results, thiazides were considered first-line treatments for hypertension in JNC-7 and will likely continue to play a key role in JNC-8's recommendations.
However, there are some issues to keep in mind when using these medications. First, the thiazide most often used in the United States, hydrochlorothiazide, is less potent and has a shorter duration of action than chlorthalidone, the drug studied in ALLHAT. Given these differences, one may choose to use chlorthalidone or use HCTZ with total daily doses of 25-50 mg, which approximates the dosages of chlorthalidone studied. Additionally, if once-daily dosing results in elevated blood pressure just prior to the next dose, one might consider a BID dosing regimen. Additionally, hypokalemia from thiazides, although usually mild, is associated with an increased risk of diabetes and coronary events and should be corrected. Co-administration with ACEIs or potassium-sparing diuretics can be preventative and may be considered when the baseline potassium is less than 3.8 mEq/L. Sodium restriction, which is recommended for all hypertensives, also reduces thiazide-induced hypokalemia. Finally, supplemental potassium may be used if needed.
Another area of concern is an increased risk of diabetes with thiazides. A recent study noted that abdominal obesity as well as other features of the metabolic syndrome increases one's risk of developing diabetes while on thiazides. This risk increases with duration of exposure. Although the hypokalemic effect of thiazides has been associated with thiazide-related diabetes in other trials, such a link was not observed in this study.87 Additionally, HCTZ can cause subcutaneous to visceral fat redistribution and nonalcoholic fatty liver disease.88 In light of this information, one should reconsider using thiazide diuretics first line (and perhaps use an ACEI or an ARB as noted earlier) in those with increased risk of metabolic adverse events, including those with the metabolic syndrome, abdominal obesity, prediabetes, and fatty liver.
Another metabolic concern is thiazide-induced hyponatremia (TIH), which has been found in 4-11% of elderly patients studied. This complication is more common in elderly women, those with low body mass, and those taking other medications that can inhibit water excretion such as selective serotonin reuptake inhibitors (SSRIs). TIH can develop quickly and be fatal or cause permanent neurological damage. Accordingly, one should monitor those at risk within a day of starting therapy, stop the drug if the sodium falls greater than a few mEq/L, and confirm that normal or minimally decreased sodium is stable a couple of days later.89
Additional precautions include drug hypersensitivity and reduced excretion of both calcium and uric acid. Surprisingly, sulfonamide allergy is not a contraindication to thiazide use as there does not appear to be a cross-sensitivity risk between thiazides and sulfonamides. However, if one develops an allergy to HCTZ or chlorthalidone, ethacrynic acid, a non-sulfonamide, can be used. If one has hypercalcemia such as with hyperparathyroidism, thiazides should not be used. Also, if one develops an attack of gout during therapy and an elevated uric acid level is confirmed, thiazide therapy should be withheld.41
In addition to thiazides, loop diuretics and aldosterone antagonists have a place in hypertension treatment. As noted earlier, loop diuretics are best reserved for those hypertensive patients with a GFR <30 ml/min who require a diuretic to lower BP. Aldosterone antagonists also have a niche in hypertensive treatment, particularly in those with obesity and/or obstructive sleep apnea. In fact, at doses of only 25 mg daily, spironolactone can reduce SBP by 20-30 mm Hg in patients with both obstructive sleep apnea and obesity.90 One should monitor for hyperkalemia, however, and be mindful of endocrine side effects such as decreased libido and gynecomastia.41
Calcium channel blockers (CCB), particularly dihydropyridine CCBs such as amlodipine also have demonstrated effectiveness in hypertensive patients at preventing CV adverse outcomes comparable to other antihypertensive agents.86 In addition, when an agent is required to further lower blood pressure and risk of CV events in those on an ACE inhibitor, adding a dihydropyridine CCB provides a relative risk reduction of 19.6% compared with using a thiazide in this setting. As noted earlier, however, dihydropyridine calcium channel blockers may impair autoregulation in the kidney and are best used in conjunction with an RAS blocker along with monitoring for worsening of protein excretion and/or GFR in patients with CKD. Additionally, nondihydropyridine CCBs can be used for their antiproteinuric property in CKD patients91 and their negative chronotropic action in those atrial fibrillation patients who require rate control. Nevertheless, it should be noted that beta-blockers offer superior rate control in atrial fibrillation patients.92
Beta-blockers, although considered a first-line treatment for hypertension in JNC-7, may have an attenuated role in JNC-8. Specifically, there are concerns with both metabolic side effects such as glucose and lipid metabolism and hemodynamic side effects such as increased pulse pressure that should be considered when using these medications in those with impaired glucose metabolism and in the elderly, respectively. Nevertheless, beta-blockers have proven benefits in those with heart failure, atrial fibrillation with rapid ventricular response, and coronary artery disease. Also, newer agents with better metabolic and hemodynamic profiles (such as carvedilol and nebivolol) may prove to be good choices with more expanded roles and tolerability.
Despite these benefits, research shows that these agents are underutilized.93 One potential reason for underprescribing these drugs, the fear of adverse effects in those with COPD or asthma, may be overstated. Research shows that cardioselective beta-blockers in particular, such as metoprolol and atenolol, do not pose any risk in the setting of obstructive lung disease. Additionally, some data is emerging that even non-selective agents, such as carvediolol, have less pulmonary adverse reactions than once feared. Nevertheless, especially in those with little margin in their pulmonary function, one should use non-selective beta-blockers with some caution and monitor for either worsening lung function or reduced response to inhaled beta-agonists.94
Alpha-blockers. The ALLHAT trial revealed poorer CV outcomes (particularly increased heart failure) compared to chlorthalidone in those high-risk patients treated with alpha blockers, making this class a suboptimal choice for monotherapy. However, further study indicates some role for alpha-blockers such as doxazosin as add-on therapy to further lower blood pressure and CV risk in patients already on two or more drugs.95 This may be particularly attractive in men with BPH who require additional medications to control blood pressure.
Strategies for Uncontrolled Hypertension
As noted earlier, uncontrolled hypertension may improve by addressing adherence to drug therapy, eliminating interfering substances, and treating secondary causes of hypertension. Additionally, multi-drug therapy, plasma renin-directed therapy, and use of aldosterone antagonists can be effective.
To meet present BP targets, up to three-quarters of patients will require combination therapy. One should consider initiating therapy with two drugs in patients with a baseline BP that is 20/10 mm above target BP. One can choose from a variety of effective drug combinations when needed. ACEIs can be combined effectively with thiazides and dihydropyridine CCBs, although the latter combination is associated with better outcomes data. Also effective is the addition of a potassium-sparing agent to higher-dose thiazides such as chlorthalidone 25 mg or HCTZ 50 mg, which can prevent hypokalemia. However, hyperkalemia may result in the setting of CKD. Another potential combination is a diuretic and a beta-blocker to provide additive BP reduction, but with potential metabolic side effects. Finally, dihydropyridine CCBs can be added to beta-blockers to lower BP, but nondihydropyridine agents such as diltiazem may result in bradycardia and heart block when used with a beta-blocker.
On the other hand, there are other drug combinations that are less synergistic at lowering BP. ACEIs in combination with either ARBs or beta-blockers do not seem to result in significant lowering of blood pressure, although an ACEI or an ARB and a beta-blocker should be used together to improve CV outcomes in patients with CHF and CAD.96 Unlike the ACEI (or ARB) and beta-blocker combination, using an ACEI with an ARB is not associated with improved CV endpoints.97 Another less effective combination is beta-blockers with centrally-acting agents such as clonidine, which can cause severe bradycardia and may result in severe rebound hypertension if stopped suddenly.96
Although a topic of some controversy, treatment directed by plasma renin activity (PRA) has shown recent promise to individualize drug therapy. Those with a low PRA (< 0.65 ng/mL/h) have a sodium-water excess state that is more likely to respond to diuretics, spironolactone, and CCBs and less likely to respond to ACEIs, ARBs, and beta-blockers. In those with a higher PRA (≥ 0.65), indicating renin-angiotensin vasoconstriction excess, ARBs, ACEIs, and beta-blockers are more beneficial. Tailoring drug therapy based on PRA was found to be effective in a recent randomized controlled trial.98
One final strategy, the use of aldosterone antagonists such as spironolactone in resistant HTN, deserves mention. In a recent study of 175 patients, spironolactone was added to a drug regimen (consisting of a median of four drugs) and resulted in a median 16/9 mm Hg reduction in BP.99
Summary
Hypertension is a significant public health problem that requires primary care providers to execute a systematic, patient-centered approach to measurement, assessment of cardiovascular risk, detection of target organ damage, search for secondary causes, and initiation of nonpharmacologic and pharmacologic treatment. Since the release of JNC-7, new insights have become available with additional patient-oriented outcome data that help physicians to approach this problem in an increasingly patient-centered and evidence-based manner.
References
1. American Heart Association. Heart Disease and Stroke Statistics 2010 Update. Dallas, Texas: American Heart Association; 2010.
2. Chobanian AV, Bakris GL, Black HR, et al. Seventh report of the joint national committee on prevention, detection, evaluation and treatment of high blood pressure. Hypertension 2003;42;1206-1252.
3. Kaplan NM. Commentary on the sixth report of the Joint National Committee (JNC-6). Am J Hypertension 1998;11:134-6.
4. McKay DW, Campbell NR, Parab LS, et al. Clinical assessment of blood pressure. J Hum Hypertension 1990;4:639-645.
5. Handler J. The importance of accurate blood pressure measurement. The Permanente Journal 2009;13:51-54.
6. Jones DW, Appel LJ, et al. Measuring blood pressure accurately: New and persistent challenges. JAMA 2003;289:1027-1030.
7. Kantola I, Vesalainen R, et al. Blood pressure measurement: Bell or diaphragm? J Hypertension 2005;23:499-503.
8. Williams JS, Brown SM, et al. Blood pressure measurement. N Engl J Med 2009;360;e6.
9. Pickering TG, Houston Miller N, et, al. Call to action on use and reimbursement for home blood pressure monitoring: A joint scientific statement from the American Heart Association, American Society of Hypertension, and Preventative Cardiovascular Nurses Sssociation. Hypertension 2008;52:10-29.
10. Pierdomenico SD, Mezzetti A, et al. White-coat hypertension in patients with newly diagnosed hypertension: Evaluation of prevalence by ambulatory monitoring and impact on cost of health. Eur Heart J 1995;16: 692-69.
11. Wexler R. Ambulatory blood pressure monitoring in primary care. South Med J 2010;103:447-452.
12. Venkata C, Ram S, et al. The evolving definition of systemic arterial hypertension. Curr Atheroscler Rep 2010;12:155-158.
13. Germino FW. JNC8: Expectations, challenges, and wishes-a primary care perspective. J Clin Hypertension 2009;11:573-576.
14. Weycker D, Nichols GA, et al. Risk-factor clustering and cardiovascular disease risk in hypertensive patient. AJH 2007;20:599-607.
15. van Wyk JT, van Wijk MA, et al. Identification of the four conventional cardiovascular disease risk factors by Dutch general practitioners. Chest 2005;128:2521-2527.
16. Rosendorff C, Black HR, et al. Treatment of hypertension in the prevention and management of ischemic heart disease: a scientific statement from the American Heart Association Council for High Blood Pressure Research and the Councils on Clinical Cardiology and Epidemiology and Prevention. Circulation 2007;115:2761-2788.
17. Adams HP, Secondary prevention of atherothrombotic events after ischemic stroke. Mayo Clin Proc 2009;84:43-51.
18. Fisher M, Lees K. Management of blood pressure for acute and recurrent stroke. Stroke 2009;40:2251-2256.
19. De Caterina AR, Leone AM. Why beta blockers should not be used as first choice in uncomplicated hypertension. Am J Cardiol 2010;105:1433-1438.
20. Levy D, Larson M, et al. The progression from hypertension to congestive heart failure. JAMA 1996;275:1557-156.
21. Shamsham F, Mitchell J. Essentials of the diagnosis of heart failure. Am Fam Physician 2000;61:1319-28.
22. Ramani, GV, Uber PA, et al. Chronic heart failure: Contemporary diagnosis and management. Mayo Clin Proc 2010;85:180-195,
23. McConaghy JR, Smith SR. Outpatient tratment of systolic heart failure. Am Fam Physician 2004;70:2157-64,2171-2.
24. Janardhanan R, Akshay SD, et al. Therapeutic approaches to diastolic dysfunction. Curr Hypertens Reports 2009;11:283–291.
25. Abrams, J, Thadani U. Therapy of stable angina pectoris: The uncomplicated patient. Circulation 2005;112:e255-e259.
26. De Bacquer D, De Backer, et al. Prognostic value of ECG findings for total, cardiovascular disease, and cardiovascular disease death in men and women. Heart 1998;80:570-577.
27. Pewsner D, Juni P, et al. Accuracy of electrocardiography in diagnosis of left ventricular hypertrophy in arterial hypertension: Systematic review. BMJ 2007;335:771.
28. Simpson HJ, Gandy SJ, et al. Left ventricular hypertrophy: Reduction of blood pressure already in the normal range further regresses left ventricular mass. Heart 2010;96:148-152.
29. Bauml MA, Underwood DA. Left ventricular hypertrophy: An overlooked cardiovascular risk factor. Cleve Clin J Med 2010;77:381-7.
30. Cuspidi C, Negri F. Echocardiography in clinical practice: The burden of arterial hypertension. A multicenter Italian survey. J Hum Hypertens 2010;24:395-402.
31. Douglas PS, Khandheria B, et al. ACCF/ASE/ACEP/ASNC/SCAI/SCCT/SCMR 2007 Appropriateness criteria for transthoracic and transesophageal echocardiography. J Am Coll Cardiol 2005;46:1606-13.
32. Coresh J, Selvin E, et al. Prevalence of chronic kidney disease in the United States. JAMA 2007;298:2038-2047.
33. Mensah GA, Croft JB, et al. The heart, kidney, and brain as target organs in hypertension. Curr Probl Cardiol 2003;28:156-193.
34. Johnson CA, Levey AS, et al. Clinical practice guidelines for chronic kidney disease in adults: Part I. definition, disease stages, evaluation, treatment and risk factors. Am Fam Physician 2004;70:869-76.
35. Johnson CA, Levey AS, et al. Clinical practice guidelines for chronic kidney disease in adults: Part II. Glomerular filtration rate, proteinuria and other markers. Am Fam Physician 2004;70:1091-7.
36. Stevens LA, Coresh J, et al. Assessing Kidney Function measured and estimated glomerular filtration rate. N Engl J Med 2006;354:2473-83.
37. Leoncini G, Ratto E, et al. Global risk stratification in primary hypertension: the role of the kidney. J Hypertens 2008;26:427-432.
38. Rose BD, Post TW. Measurement of urinary protein excretion. In:UpToDate, Basow, DS (Ed), UpToDate, Waltham, MA, 2010.
39. Barri YMi. Hypertension and kidney disease: A deadly connection. Curr Hypertens Rep 2008;10:39-45.
40. Levey AS, Roco MV, et al. National Kidney Foundation K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis 2004;43(5 suppl 1):S1-290.
41. Ernst ME, Moser M. Use of diuretics in patients with hypertension. N Engl J Med 2009;361:2153-64.
42. Griffen KA, Bidani AK. Potential risks of calcium channel blockers in chronic kidney disease. Curr Cardiol Rep 2008;10:448-55.
43. Palmer BF. Renal dysfunction complicating the treatment of hypertension. N Engl J Med 2002;347:1256-61.
44. Emmanuel L, Brav EL, Gifford RW. Pheochromocytoma: Diagnosis, localization and management. N Engl J Med 1984; 311:1298-130.
45. Mattsson C, Young WF. Primary aldosteronism: Diagnostic and treatment strategies. Nat Clin Pract Nephrol 2006;2:198-208.
46. Sica DA. Endocrine causes of secondary hypertension. J Clin Hypertens 2008;10:534-540.
47. Boscaro M, Barzon L, Fallo F, et al. Cushing's syndrome. Lancet 2001;357:783-91.
48. Reisch N, Peczkowska M, et al. Pheochromocytoma: Presentation, diagnosis and treatment. J Hypertens 2006;24:2331-9.
49. Adler JT, Meyer-Rochow GY, et al. Pheochromocytoma: Current approaches and future directions. Oncologist 2008;13:779-93.
50. Preston RA, Singer I, Epstein, et al. Renal parenchymal hypertension: Current concepts of pathogenesis and management. Arch Intern Med 1996;156:602-11.
51. Scholz DA. Hypertension and hyperparathyroidism. Arch Intern Med 1977;137:1123-4.
52. Fuleihan GE, Silverberg SJ. Clinical manifestations of primary hyperparathyroidism. In UpToDate, Basow, DS (Ed), UpToDate, Waltham, MA, 2009.
53. Chiong JR, Aronow WS, et al. Secondary hypertension: Current diagnosis and treatment. Int J Cardiology 2008;124:6–21.
54. Bravo EL. Pheochromocytoma, thyroid disease, and hyperparathyroidism. J Clin Hypertens 2007;7;173-7.
55. Goodfriend TL. Obesity, sleep apnea, aldosterone and hypertension. Curr Hypertens Reports 2008;10:222-6.
56. Onusko E. Diagnosing secondary hypertension. Am Fam Physician 2003;67:67-74
57. Gonzaga CC, Calhoun DA. Resistant hypertension and hyperaldosteronism. Curr Hypertens Rep 2008;10:496-503.
58. Amar L, Bertherat J, et al. Genetic testing in pheochromocytoma or functional paraganglioma. J Clin Oncol 2005;23:8812-8.
59. Boscaro M, Barzon L, Fallo F, et al. Cushing's syndrome. Lancet 2001;357:783-91.
60. Findling JW, Raff H, Aron DC. The low-dosedexamethasone suppression test: A reevaluation in patients with Cushing's syndrome. J Clin Endocrinol Metab 2004;89:1222-6.
61. Newell-Price J, Bertagna X, Grossman AB, et al. Cushing's syndrome. Lancet 2006;367:1605-17.
62. Kalra PA, Guo H, Kausz AT, et al. Atherosclerotic renovascular disease in United States patients aged 67 years or older: Risk factors, revascularization, and prognosis. Kidney Int 2005;68:293-301
63. Dworkin LD, Cooper CJ. Renal-artery stenosis. N Engl J Med 2009;361:1972-8.
64. Mantha, M, Kumar, SK, MacGinley, R, et al. Screening tests for diagnosis of renal artery stenosis. Nephrology 2010;15:S218-226.
65. Morcos, SK. Chronic kidney disease: CT or MRI? Applied Radiology 2008;37:19-24.
66. Schlaudecker JD, Bernheisel CR. Gadolinium-associated nephrogenic systemic fibrosis. Am Fam Physician 2009;80:711-714.
67. Hartman RP, Kawashima A. Am Fam Physician 2009;80:273-279.
68. Simon JF. Stenting atherosclerotic renal arteries: Time to be less aggressive. Cleve Clin J Med 2010;77:178-89.
69. Sacks FM, Campos H. Dietary therapy in hypertension. N Engl J Med 2010;362:2102-12.
70. National Heart, Lung, and Blood Institute. The DASH eating plan. Bethesda, Md.: U.S. Department of Health and Human Services, 2006. Accessed online October 30, 2010, at: http://www.nhlbi.nih.gov/health/public/heart/hbp/dash.
71. Klatsky AL. Alcohol-associated hypertension. Hypertension 2004;44:805.
72. Wexler R, Aukerman G. Nonpharmacologic strategies for managing hypertension. Am Fam Physician 2006;73:1953-56.
73. Kaplan NM. Smoking and hypertension. In:UpToDate, Basow, DS (Ed), UpToDate, Waltham, MA, 2010.
74. Appel LJ, Wright JT, et al. Intensive blood-pressure control in hypertensive chronic kidney disease. N Engl J Med 2010;363:918-29.
75. Safar ME. Pulse pressure, heart rate and drug treatment of hypertension. Curr Hypertens Rep 2004;6:190-194.
76. Matterson BJ, Preston RA. Angiotensin-converting enzyme inhibitors in hypertension. A dozen years of experience. Arch Intern Med 1994;154:513-523.
77. López-Sendón J, Swedberg K, et al. Expert consensus document on angiotensin converting enzyme inhibitors in cardiovascular disease. The Task Force on ACE-inhibitors of the European Society of Cardiology. Eur Heart J 2004;25:1454-70.
78. K/DOQI Clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis 2004;43:5(Suppl1):S1.
79. Al-Mallah M, Khawaja O, et al. Do angiotensin converting enzyme inhibitors or angiotensin receptor blockers prevent diabetes mellitus? A meta-analysis. Cardiol J 2010;17: 448-456.
80. Sehgal A. Overlap between whites and blacks in response to antihypertensive drugs. Hypertension 2004;43:566-572.
81. Sánchez-Borges M, González-Aveledo LA. Angiotensin-converting enzyme inhibitors and angioedema. Allergy Asthma Immunol Res 2010;2:195–198.
82. Garcia-Pavia P, Tomas JM, et al. Late-onset angioedema due to an angiotensin-converting enzyme inhibitor. Can J Cardiol 2007;23:315–316.
83. Abdi R, Dong VM, Lee CJ, et al. Angiotensin II receptor blocker-associated angioedema: On the heels of ACE inhibitor angioedema. Pharmacotherapy 2002;22:1173-1175.
84. Cooper WO, Hernandez-Diaz S, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med 2006;354:2443-51.
85. Paige NM, Nagami GT. The top ten things nephrologists wish every primary care physician knew. Mayo Clin Proc 2009;84:180-6.
86. ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group; Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA 2002;288:2981-2997.
87. Cooper-Dehoff RM, Wen S, et al. Impact of abdominal obesity on incidence of adverse metabolic effects associated with antihypertensive medications. Hypertension 2010;55:61-68.
88. Eriksson JW, Jansson P, et al. Hydrochlorothiazide, but not candesartan, aggravates insulin resistance and causes visceral fat accumulation. Hypertension 2008;52:1030.
89. Hwang KS, Kim G. Thiazide-induced hyponatremia. Electrolytes Blood Press 2010;8:51-7.
90. Goodfriend TL. Obesity, sleep apnea, aldosterone and hypertension. Curr Hypertens Reports 2008;10:222-6.
91. Sandeep N, Pepine CJ. Calcium antagonists: Effects on cardio-renal risk in hypertensive patients. Hypertension 2005;46:637.
92. Olshansky B, Rosenfeld LE, et al. The atrial fibrillation follow-up investigation of rhythm management (AFFIRM) study: Approaches to control rate in atrial fibrillation. J Am Coll Cardiol 2004;43:1201-8.
93. Marique C, Giles TD, et al. Realities of newer beta-blockers for the management of hypertension. J Clin Hypertens 2009;11:369-375.
94. Salpeter SR. Cardioselective beta blocker use in patients with asthma and chronic obstructive lung disease: An evidence-based approach to standards of care. Posted: 11/14/2003; © 2003 Le Jacq Communications, Inc. Accessed 11/15/10.
95. Chapman, N. Time to re-appraise the role of alpha-1 adrenoceptor antagonists in the management of hypertension? J Hypertens 2010;28:1796-1803.
96. Gradman AH, Basile JN, et al. Combination therapy in hypertension. J Am Soc Hypertens 2010;4:42-50).
97. Yusuf S, Teo KK, et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Eng J Med 2008;358:1547-1559.
98. Egan BM, Basile JN, et al. Plasma renin test-guided drug treatment algorithm for correcting patients with treated but uncontrolled hypertension: A randomized controlled trial. Am J Hyperntens 2009;22:792-801.
99. de Souza, Muxfeldt E, et al. Efficacy of spironolactone therapy in patients with true resistant hypertension. Hypertension 2010;55:147.
The Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure is currently updating its work in JNC-7 in an effort to help physicians provide safe, effective, and patient-centered care for those with elevated blood pressure.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
