Can Nebulized Heparin Reduce Ventilator-Induced Lung Injury?
Can Nebulized Heparin Reduce Ventilator-Induced Lung Injury?
By David J. Pierson, MD, Editor, Professor, Pulmonary and Critical Care Medicine, Harborview Medical Center, University of Washington, Seattle, is Editor for Critical Care Alert.
Synopsis: In this single-center, placebo-controlled clinical trial of 50 patients with acute respiratory failure, nebulized heparin was associated with fewer days of mechanical ventilation, although measures intended to reflect its assumed mechanism of action in reducing microvascular fibrin deposition showed no differences.
Source: Dixon B, et al. Nebulized heparin is associated with fewer days of mechanical ventilation in critically ill patients: A randomized controlled trial. Critical Care 2010;14:R180..
Laboratory studies and clinical data support the concept that prolonged mechanical ventilation (PMV) can induce or worsen lung injury, via activation of inflammatory mediators and/or microvascular fibrin deposition, processes that may be ameliorated by heparin. This study from a tertiary referral center in Melbourne, Australia, sought to determine whether heparin delivered via aerosol would prevent or reduce ventilator-induced lung injury, as indicated by decreased indices of inflammation in alveolar lavage fluid, improved arterial oxygenation, shortened duration of mechanical ventilation, reduced need for tracheostomy, and decreased ICU and hospital lengths of stay, among adult patients requiring PMV for acute respiratory failure.
For this study PMV was defined as the requirement for invasive ventilatory support for > 48 hours. Patients predicted to require PMV were entered into the study within 24 hours of intubation; exclusion criteria included the use of high-frequency oscillatory ventilation, extracorporeal membrane oxygenation, inhaled nitric oxide, renal replacement therapy, or systemic heparin in therapeutic doses. By means of an Aeroneb Pro® nebulizer, enrolled patients received either heparin (25,000 units in 5 mL saline) or plain saline every 4 or 6 hours depending on body size, for 14 days or until weaned from mechanical ventilation. The primary outcome variable was the PaO2/FIO2 ratio, measured once daily at 4:00 am. A variety of inflammatory mediators were measured in bronchoalveolar lavage fluid obtained non-bronchoscopically on study days 1, 2, 4, 8, and 14. Other outcomes measured were ventilator-free days as of 28 days after enrollment among survivors, development of acute lung injury by international consensus criteria following enrollment, tracheostomy, vasopressor- and renal failure-free days, ICU and hospital lengths of stay, and mortality.
The investigators screened 1537 patients admitted to their ICU during the 17 months of the study, of whom 219 met the inclusion criteria and 50 were ultimately enrolled, with 25 patients randomized to each study arm. Patients in the heparin and placebo groups were well matched with respect to age, sex, primary diagnosis, severity of illness by APACHE II score (about 20), initial PaO2/FIO2 ratio (approximately 170), and presence of acute lung injury on admission (16% in each group). Results showed that the average PaO2/FIO2 ratio while ventilated remained the same in both groups. There were no significant differences in any of the eight inflammatory mediators measured, nor in the new acute lung injury or tracheostomy rates, vasopressor- or renal failure-free days, ICU or hospital length of stay, or mortality (18% overall). However, surviving patients who received nebulized heparin had significantly more ventilator-free days as of day 28 than did those who received placebo: 22.6 ± 4.0 vs 18.0 ± 7.1 days (P = 0.02).
Commentary
In the last few years there has been a great deal of interest in mechanical ventilation per se as a contributor to and even as an initiator of acute lung injury. Injury has been ascribed to the use of excessive distending volumes and pressures ("barotrauma"), shear stresses involved in the repetitive opening and closing of collapsed lung areas ("atelectrauma"), and the release of injurious mediator substances by mechanical lung distension ("biotrauma"). Prolonged mechanical ventilation has been associated with the deposition of fibrin in the pulmonary microvasculature and alveoli, both in animal models and in patients. This fibrin deposition in turn has been hypothesized to play a role in lung inflammation, hyaline membrane formation, gas exchange abnormalities, and the need for PMV, as well as in clinical outcomes such as mortality and length of stay. These proposed relationships are shown schematically in the Figure (below). By interfering with fibrin deposition, heparin might block or at least attenuate the adverse effects listed on the right side of the figure. The present study is the latest in a series of investigations by these authors in pursuit of this hypothesis.
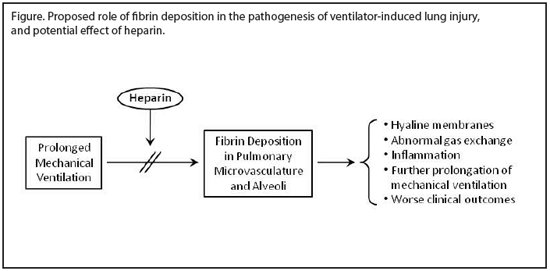
In this relatively small, single-center clinical trial, the administration of nebulized heparin was associated with more ventilator-free days among surviving patients than was placebo. This is an encouraging finding although as the authors state it needs to be confirmed by further studies. Heparin is readily available and cheap, but before we all start nebulizing it in our ventilated patients to prevent acute lung injury (an off-label use of the drug), several cautionary points should be made about this study.
Given the rationale for using heparin during PMV, it seems odd that no significant differences were found in any of the other measures reflecting its putative effects as shown in the figure. One would expect that a favorable effect of heparin on ventilator-free days would somehow reflect a reduction in lung injury, facilitating more rapid recovery. There were no differences between the heparin and placebo groups with respect to gas exchange (PaO2/FIO2 ratio or PaCO2), peak inspiratory pressure, end-expiratory pressure, delivered tidal volume, or minute ventilation. Nothing is mentioned in the paper about ventilator liberation criteria, other than the use of pressure support for weaning, so in the absence of differences in illness severity, inflammation, or ventilatory status, the reason for the substantial difference in ventilator-free days (mean, 4.6 days) is not apparent.
Laboratory studies and clinical data support the concept that prolonged mechanical ventilation (PMV) can induce or worsen lung injury, via activation of inflammatory mediators and/or microvascular fibrin deposition, processes that may be ameliorated by heparin.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
