Glucosamine Sulfate for Osteoarthritis
Glucosamine Sulfate for Osteoarthritis
By Dónal P. O'Mathùna, PhD. Dr. O'Mathùna is Senior Lecturer in Ethics, Decision-Making & Evidence, School of Nursing, Dublin City University, Ireland; he reports no financial relationship to this field of study.
Glucosamine sulfate continues to grow in popularity as a treatment for osteoarthritis. Global sales reached almost $2 billion in 2008, an increase of 60 percent over the previous five years.1 This update will present the results of research published since this topic was last reviewed here two years ago. Further results from the NIH-funded Glucosamine/chondroitin Arthritis Intervention Trial (GAIT) have since been published, along with some clinical guidelines.2 Given glucosamine's widespread use, clinicians should be aware of recent research findings and their impact on osteoarthritis management.
Glucosamine occurs naturally in the body as one component of the cartilage and synovial fluid found within joints.3 Glucosamine forms part of the structure of some compounds whose levels are reduced with osteoarthritis. A number of salts of glucosamine are available, with some debate over their relative effectiveness. While questions have been raised about the bioavailability of oral glucosamine, recent studies have shown that it is absorbed and can accumulate in synovial fluid.3 However, its precise mechanism of action remains unclear, although a number of mechanisms have been proposed.
Background
Osteoarthritis is the most common form of arthritis and a leading cause of disability in the United States and Europe.3 Prevalence increases with age, and is higher in women. However, actual values are difficult to estimate due to different diagnostic criteria and because about half the people with X-ray evidence of the disease report no symptoms.4 The knees, hips, and small joints of the hands are most commonly affected, with pain and stiffness the most common symptoms.3
The etiology of osteoarthritis remains unclear. Once viewed as a consequence of aging, it is now known to develop due to complex interactions involving genetics, age, gender, obesity, joint injury, and muscle weakness.5 With the disease, cartilage inside joints gradually degenerates, exposing bone inside the joint and narrowing the joint space. This leads to stiffness and pain, and eventually the joint may become so immobile and painful that joint replacement surgery is the best alternative. (See Figures 1 and 2.) Osteoarthritis is only one of a number of arthritic diseases, resulting in the term arthritis being used commonly to refer to any pain and stiffness in the muscles and joints. For this reason, some who have osteoarthritis refer to it as arthritis. Technically speaking, arthritis involves inflammation in the joints and osteoarthritis is a non-inflammatory degenerative disease. All of the research reviewed here was conducted with older adults diagnosed with osteoarthritis.
Figures 1 and 2. A Healthy Joint and a Joint with Severe Osteoarthritis
Figure 1. In a healthy joint, the ends of bones are encased Figure 2. With osteoarthritis, the cartilage becomes wornin smooth cartilage. Together, they are protected by a joint away. Spurs grow out from the edge of the bone, andcapsule lined with a synovial membrane that produces synovial fluid increases. Altogether, the joint feels stiff andsynovial fluid. The capsule and fluid protect the cartilage, sore.muscles, and connective tissues.
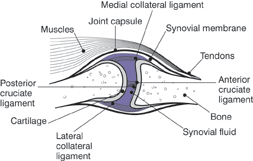
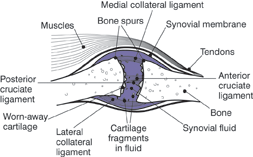
Source: National Institute of Arthritis and Musculoskeletal and Skin Diseases. Handout on Health: Osteoarthritis. NIH Publication No. 10-4617. Available at: http://www.niams.nih.gov/Health_Info/Osteoarthritis/#pic_2. Accessed Jan. 15, 2011.
Currently, osteoarthritis does not have a cure.6 The Osteoarthritis Research Society International (OARSI) recommends that patients should be directed toward pharmacological and non-pharmacological treatments.7 Conventional drugs for chronic pain, especially non-steroidal anti-inflammatory drugs (NSAIDs), are commonly used. However, long-term use of NSAIDs raises concerns about adverse effects, especially in combination with the other drugs many older people take.2 Non-pharmacological modalities include education, lifestyle management such as exercise and weight reduction, and complementary therapies like acupuncture.7
Although all of these are important for managing symptoms, they do not treat the underlying pathology. Claims that dietary supplements can reverse cartilage damage, and that they have fewer adverse effects than pharmaceutical drugs, have fueled interest in complementary approaches to managing osteoarthritis symptoms. Several dietary supplements are being used for osteoarthritis.8 Foremost among these are glucosamine and chondroitin, which are some of the most popular dietary supplements sold in the United States.9 Although the two supplements are sometimes taken together, most studies have tested them separately.5 This review will focus primarily on glucosamine sulfate.
Clinical Studies
Clinical studies have been conducted on glucosamine sulfate for osteoarthritis for more than 20 years. Many of the early studies were conducted in Europe using a product available by prescription in many European countries. These 1,500 mg sachets are to be dissolved in water and are manufactured by Rotta Pharmaceuticals of Italy. Studies with this product were sponsored by its manufacturer and generally had favorable results. Later independent studies tended to have negative results, raising concerns about bias. However, the differences among various trials are more complicated, involving different glucosamine formulations and study designs. Independent studies have used products available in the United States as dietary supplements, many of which are formulated as capsules, which may explain some of the differences with the Rotta product, which is dissolved before being taken.4
Over the last decade, a number of systematic reviews and meta-analyses have been published on glucosamine. These generally have been critical of the quality of earlier studies, finding they were small and of short duration. More recent meta-analyses have included only high-quality randomized controlled trials (RCTs). A Cochrane Collaboration systematic review, originally published in 2005, was updated in 2009.10 The review now includes 25 trials with almost 5,000 osteoarthritis patients. Its conclusions remain unchanged from the original review. Overall, glucosamine was significantly better than placebo in producing a 22% reduction (from baseline) in pain, and an 11% improvement in function using the Lequesne Index (LI). However, significant differences were not found when outcomes were measured using the Western Ontario and McMaster Universities (WOMAC) osteoarthritis indices of pain, function, and stiffness. Studies comparing glucosamine to NSAIDs found similar benefits.
Sub-group analyses were performed to identify possible reasons for the variability between studies. The highest quality studies (having adequate allocation concealment) showed no benefit in pain and WOMAC scores, but were significantly better than placebo using the LI for a measure of function. Further differences were seen when studies with the Rotta product were compared to those using any other product. None of the latter produced significant improvements, while the Rotta product was significantly better than placebo for all pain measures, LI, and WOMAC indices. Two of these RCTs also measured joint space reduction within the knee by X-ray. Both studies found significant improvements for those using the Rotta product. Another review limited to RCTs lasting 1 year or longer similarly found good evidence of benefit using the Rotta product.3 However, most of the Rotta studies were funded by the manufacturer and include some older ones of lower quality.
The most recent meta-analysis was published in 2010 and came to very different conclusions.1 These reviewers limited their review to studies involving at least 200 patients. This was chosen as an indicator of methodological quality. They used a somewhat different method of analysis called a network meta-analysis, which uses more complex Bayesian statistical methods. Their review included 10 RCTs and concluded that "glucosamine, chondroitin, and their combination do not result in a relevant reduction of joint pain nor affect joint space narrowing compared with placebo."1 This review since has been criticized for statistical methods and choice of various cut-off values for outcome measures.11 These published letters were primarily from the authors of earlier systematic reviews and researchers at Rotta.
Meanwhile, findings from the NIH-funded GAIT trial continue to appear.12 This study randomized more than 1,500 patients with symptomatic knee osteoarthritis to daily doses of either 1,500 mg glucosamine hydrochloride, 1,200 mg chondroitin sulfate, both glucosamine and chondroitin, 200 mg celecoxib, or placebo. Two-thirds of the participants were women. The primary outcome measure was a 20% reduction in knee pain measured by the WOMAC scale after six months of treatment. No significant reduction in pain occurred for the groups taking glucosamine or chondroitin, alone or together, compared to placebo, while pain was significantly reduced in the celecoxib group. Patients with moderate-to-severe joint damage as revealed by X-ray were asked to continue in a follow-up study focused on joint space width.5 Patients continued to take the same treatment to which they had been assigned for a total of two years. No statistically significant differences in joint space width were observed between any of the groups.2 The most recent publication examined pain and function in these participants who continued in the study for two years. No significant differences were found in WOMAC pain and function indices.
Adverse Effects
The GAIT study reported no significant differences in adverse effects between the groups, with a similar number of participants withdrawing from each group because of adverse events.12 The follow-up study similarly reported no additional adverse effects in the glucosamine group.2 Meta-analyses have found a similar safety profile between placebo and glucosamine sulfate, with it comparing more favorably to NSAIDs.10 Its safety profile is thus considered excellent.
Concerns have been raised that glucosamine might interfere with glycemic control in diabetic patients since animal models have demonstrated such effects. However, evidence from studies lasting up to 2 and 3 years have not substantiated these concerns.2
Glucosamine sulfate is obtained from chitin extracted from marine exoskeletons, which has raised concerns about allergic reactions. Those with seafood allergies probably should avoid glucosamine, depending on the severity of their allergy, or be carefully monitored if they try it.6 Glucosamine also can be obtained from chitin in fungi, which may provide an alternative for people with a shellfish allergy.3
Conclusion
The GAIT study and a small number of others would appear to demonstrate than glucosamine hydrochloride is not effective in reducing pain or improving function for osteoarthritis patients. Only the Rotta brand of glucosamine sulfate has consistently demonstrated effectiveness in reducing symptoms and slowing joint deterioration. However, as the most recent meta-analysis demonstrates, when only the largest and highest-quality RCTs are included, even those benefits fade, although this meta-analysis may have been overly restrictive in how it measured effectiveness.
Product quality has been shown to be problematic with glucosamine dietary supplements available in the United States. One study found that products contained between 0%–115% of the mg stated on the label, while another contained between 59%–138% of labeled sulfate.13 This may explain some of the lack of efficacy found with non-Rotta products.
Recommendation
For those with osteoarthritis, a trial of glucosamine sulfate may be warranted, especially when they do not respond well to conventional treatments. Symptoms should be evaluated carefully over a few months. However, even in those trials where glucosamine sulfate has been found to be effective, participants continued to use other analgesics when pain or discomfort were particularly problematic. In addition, the vast majority of trials have been conducted with knee osteoarthritis. Glucosamine sulfate may not be effective with osteoarthritis in other sites, as was recently found in a RCT of patients with degenerative lumbar osteoarthritis.14 As with many chronic conditions, control of weight, exercise, and other symptom-relieving strategies will remain important in the overall management of osteoarthritis.
References
1. Wandel S, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: Network meta-analysis. BMJ 2010;341:c4675.
2. Sawitzke AD, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis 2010;69:1459-1464.
3. Black C, et al. The clinical effectiveness of glucosamine and chondroitin supplements in slowing or arresting progression of osteoarthritis of the knee: A systematic review and economic evaluation. Health Technol Assess 2009;13:1-148.
4. McAlindon T. Why are clinical trials of glucosamine no longer uniformly positive? Rheum Dis Clin North Am 2003;29:789-801
5. Sawitzke AD, et al. The effect of glucosamine and/or chondroitin sulfate on the progression of knee osteoarthritis: A report from the glucosamine/chondroitin arthritis intervention trial. Arthritis Rheum 2008;58:3183-3191.
6. Grainger R, Cicuttini FM. Medical management of osteoarthritis of the knee and hip joints. Med J Aust 2004;180:232-236.
7. Zhang W, et al. OARSI recommendations for the management of hip and knee osteoarthritis, part I: Critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage 2007;15:981-1000.
8. De Silva V, et al. Evidence for the efficacy of complementary and alternative medicines in the management of osteoarthritis: A systematic review. Rheumatol epub 17 Dec 2010.
9. Dahmer S, Schiller RM. Glucosamine. Am Fam Physician 2008;78:471-476.
10. Towheed T, et al. Glucosamine therapy for treating osteoarthritis. Cochrane Database Syst Rev 2005(2): CD002946.
11. Pelletier J-P, et al. Glucosamine and osteoarthritis [letters]. BMJ 2010;341:c4675.
12. Clegg DO, et al. Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med 2006;354:795-808.
13. Russell AS, et al. Active ingredient consistency of commercially available glucosamine sulfate products. J Rheumatol 2002;29:2407-2409.
14. Wilkens P, et al. Effect of glucosamine on pain-related disability in patients with chronic low back pain and degenerative lumbar osteoarthritis: A randomized controlled trial. J Am Med Assoc 2010;304:45-52.
Glucosamine sulfate continues to grow in popularity as a treatment for osteoarthritis. Global sales reached almost $2 billion in 2008, an increase of 60 percent over the previous five years.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
