Basic Emergency Ultrasound-guided Procedures
Basic Emergency Ultrasound-guided Procedures
Authors:
Teresa S. Wu, MD, FACEP, Director, EM Ultrasound Program, Co-director, Simulation Based Training Program, Associate Program Director, EM Residency Program, Maricopa Medical Center, Clinical Associate Professor, Emergency Medicine, University of Arizona School of Medicine, Phoenix.
Peter Stefanski, MD, Emergency Medicine Resident, Maricopa Medical Center, Department of Emergency Medicine, Phoenix, AZ.
Peer Reviewer:
Michael Blaivas, MD, Professor of Emergency Medicine, Associate Professor of Internal Medicine, Vice President, Society of Ultrasound Education, Department of Emergency Medicine, Northside Hospital Forsyth, Cumming, GA.
Introduction
Emergency physicians (EPs) often are called upon to perform a myriad of procedures. These procedures often are invasive and can carry a significant amount of risk. Recent studies have emphasized that EPs should routinely use ultrasound to help guide common procedures performed in the emergency department (ED). Ultrasound can be used to determine if certain procedures are necessary, and then utilized to help guide the procedural attempt. In general, ultrasound is readily available, easy to perform at the bedside, and poses no additional radiation risk to the patient. This article discusses the indications, contraindications, risks, and benefits of basic ultrasound-guided procedures, and describes how to perform each procedure, in addition to providing helpful pearls and pitfalls.
Ultrasound-guided Peripheral IV Placement
Indications. When patients are a "tough stick" and peripheral IV attempts have failed using the landmark technique alone, EPs often are called to the bedside to help obtain IV access. The external jugular vein (EJ) is an excellent option for peripheral venous access. Contraindications to utilizing the EJ include infection overlying the site of entry, fracture of the cervical spine, and anatomical obstructions at or around the access point. If the EJ is not accessible, the physician can attempt to secure peripheral IV (PIV) access using ultrasound guidance.
Although ultrasound is especially useful in patients without palpable or visible peripheral veins, it can also come in handy when searching for deeper target veins that are not identifiable on the surface.1 A bedside scan can help to localize a good target vein and map out its course for the highest chance of successful cannulation.
In many instances, peripheral venous cannulation via the landmark approach is impeded by obesity, venous degradation from chronic drug abuse, diabetes or chronic medical illness requiring multiple intravenous cannulations, overlying scar tissue, or hypovolemia. Two large-bore peripheral IVs can often deliver drugs and volume resuscitation at a higher volume and rate than smaller gauge triple- or double-lumen central venous lines. Placing an ultrasound-guided large-bore PIV is a good alternative in patients who have failed landmark-guided peripheral attempts, but do not necessarily need central venous access.
Ultrasound guidance also has been shown to be especially useful in the pediatric population. Using bedside ultrasound to place PIVs in pediatric patients with difficult peripheral access requires less overall time, fewer attempts, and fewer needle redirections than traditional approaches.2 Visualizing the target vein, and monitoring the needle trajectory during the procedure, leads to improved patient satisfaction, and higher rates of successful cannulation.
Contraindications. There are no absolute contraindications to performing an ultrasound-guided peripheral vein cannulation. Although ultrasound guidance may require a few extra minutes to gather and set up the ultrasound machine, most studies have shown that using ultrasound guidance will actually decrease the amount of time to successful venipuncture in patients at risk for difficult peripheral access.3-5 In addition, most studies demonstrate decreased number of attempts, faster time to successful catheterization, decreased number of complications, and higher patient satisfaction when ultrasound guidance is used.4
Although most studies have demonstrated clear benefits with ultrasound visualization, there are some studies that have produced opposing results. In 2009, Stein et al published a study of 59 patients suggesting that ultrasound-guided PIV cannulation did not decrease the number of attempts or time to successful catheterization, nor did it improve patient satisfaction compared with the landmark group.5
Risks. The risks of ultrasound-guided peripheral IV placement are similar to those of peripheral IV placement without ultrasound, including arterial puncture, hematoma formation, needle contact with major nerves and paresthesias, damage to surrounding structures, infiltration, infection, and pain.6,7 Ultrasound guidance can minimize these risks. When accessing a deeper peripheral vein, note that deeper IVs are at higher risk of dislodging and infiltrating the surrounding tissue. Standard-length catheters may be too short to access deeper peripheral veins, so it may be necessary to obtain longer catheters when attempting to cannulate deeper veins such as the brachial vein.8 Ensure that you can successfully aspirate venous blood, and flush the catheter smoothly before injecting any medications or bolus of fluid.
To minimize the risk for infection or contamination, attempt to use sterile ultrasound gel and follow standard sterilization techniques during ultrasound-guided PIV insertion. Studies have shown that both the landmark approach and ultrasound-guided approach to PIV access are associated with low infection rates and that there is no increased risk for infection by using ultrasound to assist with PIV access.9
Benefits. Ultrasound guidance for peripheral venous access improves patient safety by avoiding the unnecessary use of central lines, decreasing the number of PIV attempts, improving time to PIV cannulation, and reducing the number of complications, such as accidental puncture of adjacent structures. Often times, the deep brachial vein or the basilic vein is an ideal vessel for cannulation. These veins cannot be palpated and are not visible without ultrasound guidance. "Blind" attempts at puncturing these deeper veins can lead to accidental puncture of surrounding nerves and arteries. Ultrasound-guided cannulation of the deep brachial or basilic vein using longer catheters provides a rapid means for intravenous access and can be attempted in patients who need short-term vascular access.
How to Perform the Procedure.
Ultrasound guidance is typically used to access the antecubital, cephalic, basilic, and brachial veins in the arm, although ultrasound can be used to help cannulate any vein visible under sonographic guidance in the body.
You can use ultrasound to access the external jugular vein in the neck, but it is often difficult to visualize because it is so superficial and collapses easily under pressure.
Preparation and Positioning
Place the patient in a position of comfort.
Extend and externally rotate the patient's arm.
Use a tourniquet to increase venous filling and help prevent venous collapse during cannulation attempts.
If you are attempting to cannulate a very superficial, peripheral vein (< 5 mm from the skin's surface), you can typically use the standard catheters that are < 2.5 inches in length.
If you will be cannulating a vein > 5 mm below the skin's surface, secure a longer catheter (> 2.5 inches in length).
Recent studies have shown that ultrasound guidance can be used to insert a 15 cm catheter into the deep brachial or basilic vein via the Seldinger technique.8
Use the highest frequency linear array transducer available. Most linear array transducers span a range from 5-13MHz. (See Figure 1.)
Figure 1: High Frequency Linear Array Transducer

Prep and clean the patient and the ultrasound probe in a standard fashion.
Use sterile ultrasound gel during the scan. Single-use Surgilube packets work well for this application.
The procedure can be performed in the static or dynamic fashion in either the short-axis approach or the long-axis approach.
Static Approach
With the static approach, the target vessel is mapped out using ultrasound guidance.
In the short-axis, veins will appear as thin-walled, oval-shaped, anechoic structures on ultrasound.
Unlike arteries, veins should collapse when gentle pressure is applied with the transducer.
When in doubt, spectral Doppler and color Doppler can be used to distinguish an artery from a vein.
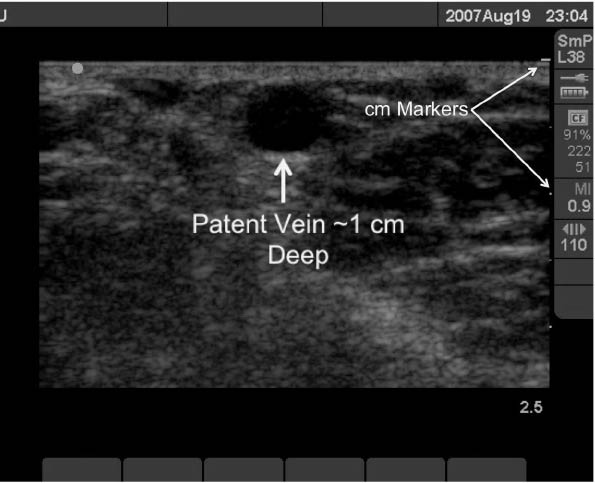
Using the depth markers on the side of the ultrasound screen, note the distance from the skin to the center of the target vein and choose the appropriate catheter for this depth.
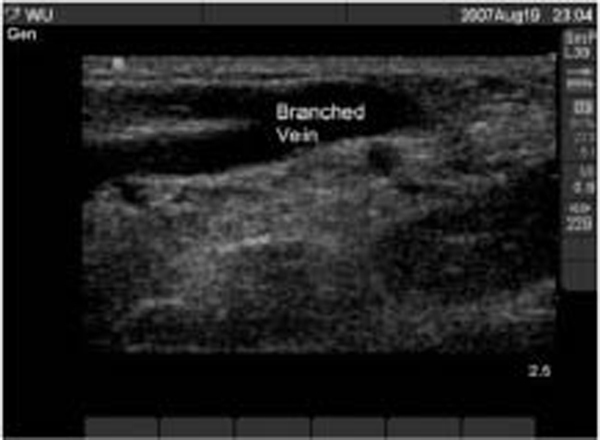
Scan proximally and distally along the vessel to delineate any bifurcations or changes in depth.
Mark the skin above the target vein with a skin marker or make a slight indentation in the skin with the cap of the peripheral IV catheter.
Remove the ultrasound probe and attempt to cannulate the vein under the marked skin.
Dynamic Approach
With the dynamic approach, the target vessel is mapped out using ultrasound guidance and then cannulated under direct ultrasound visualization.
Using the depth markers on the side of the ultrasound screen, note the distance from the skin to the center of the target vessel and choose the appropriate catheter for this depth.
Scan proximally and distally along the vessel to delineate any bifurcations or changes in depth.
Once the entry site has been chosen, cannulation can be performed under direct ultrasound guidance via the short-axis approach or long-axis approach.
Transverse Approach (Short-axis Approach)
Place the probe perpendicular to the long axis of the extremity. (See Figure 2.)
Figure 2: Ultrasound-guided Peripheral IV Placement Via the Short-axis Approach
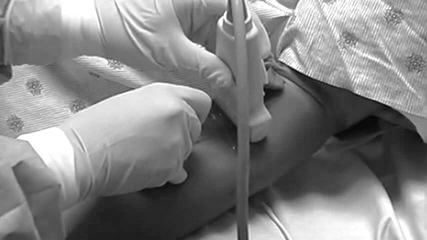
Note where the orientation marker is on the screen. If the orientation marker is on the left side of the screen, ensure that the orientation marker on the probe is facing the same direction.
In the short axis, veins will appear as thin-walled, oval-shaped, anechoic structures on the screen (See Figure 3.)
Figure 3: Oval Shaped, Anechoic Peripheral Vein
Unlike arteries, veins should collapse when gentle pressure is applied with the transducer.
When in doubt, spectral Doppler and color Doppler can be used to distinguish an artery from a vein.
When the target vein is centered in the middle of the screen, it is located directly under the middle of your transducer.
Insert your needle at a 45° angle and aim for the vessel directly under the middle of your probe.
When the vessel is being cannulated under dynamic ultrasound guidance, remember that direct visualization of the needle will only be noted when the ultrasound beam is angled directly at the needle. Because the field visualized by ultrasound is so narrow, needle position may need to be inferred by visualizing ring down artifact, soft-tissue changes, and tenting of the wall of the target structure.
To maintain visualization of the needle tip, you may need to fan the transducer or slide the probe proximally up the extremity as you advance your needle.
The short-axis approach is easier to perform and requires less hand-eye coordination.
The main disadvantage of the short-axis approach is not being able to visualize the needle tip throughout the duration of the procedure without manipulating the transducer. You may not visualize the needle tip puncturing through the posterior wall of the vessel in the short-axis approach.
Longitudinal Approach (Long-axis Approach)
Place the probe parallel to the long axis of the extremity. (See Figure 4.)
Figure 4: Ultrasound-guided Peripheral IV Placement Via the Long-axis Approach
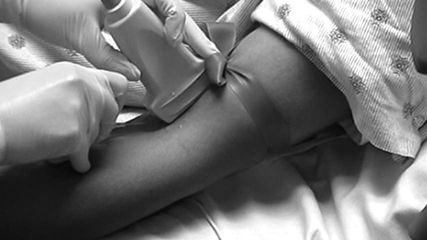
Note where the orientation marker is on the screen. Ensure that the orientation marker on the probe is pointing toward the patient's head.
In the long axis, veins will appear as thin-walled, tubular, anechoic structures on the screen. (See Figure 5.)
Figure 5: Anechoic, Tubular Peripheral Vein in the Long Axis
Scan around until you capture as much of the vein as you can on the screen in the long axis.
Insert the needle at a 45° angle just under the distal end of the probe.
On the ultrasound screen, you will visualize your needle tip entering the right corner of the image.
When the vessel is being cannulated under dynamic ultrasound guidance, remember that direct visualization of the needle will only be noted when the ultrasound beam is angled directly at the needle. Because the field visualized by ultrasound is so narrow, needle position may need to be inferred by visualizing ring down artifact, soft-tissue changes, and tenting of the wall of the target structure.
The long-axis approach requires a steady hand and good hand-eye coordination. It is easy to slip off to the medial or lateral side of the vessel and lose direct ultrasound visualization during the actual cannulation.
The advantage of performing the cannulation under long-axis guidance is the ability to visualize the needle tip entering the vessel lumen without having to manipulate the transducer during the procedure. In the long-axis view, you can see if the needle tip has accidentally punctured through the posterior wall of the target vein.
Pearls and Pitfalls.
Veins are typically oval-shaped, compressible anechoic structures on ultrasound. In contrast, arteries have thicker walls and usually are round, anechoic, non-compressible, pulsatile structures on ultrasound. Note that it may be difficult to distinguish between an artery and a vein in a hypotensive patient. With decreased intravascular volume, both arteries and veins will appear compressible on ultrasound.
Application of color Doppler over the vessel of interest can help distinguish an artery from a vein. Apply color Doppler over the target vessel. Flow within an arterial lumen will appear pulsatile. In contrast, a vein will demonstrate a more constant rumble of color flow.
The color corresponds to the direction of flow (toward or away from the transducer). Be sure to take note of the color scale displayed on your ultrasound screen to determine which color corresponds to flow toward the transducer and which color is shown with flow away from the transducer.
During the scan, minimize the amount of pressure applied with the probe. You may be missing superficial veins that have collapsed completely under the weight of the transducer.
Follow the Pythagorean theorem when determining the angle and trajectory of the needle. If the vessel lies 1 cm below the skin's surface, insert the needle 1 cm distal to the probe and follow the hypotenuse of the triangle distally toward the target vein that lies directly under the probe.
Ultrasound-guided Central Venous Access
Indications. There are many reasons why patients may require central venous access over peripheral venous access. The most common include requirements for longer-term vascular access, larger lumens, multiple lumens for rapid administration of drugs, fluid, or blood products, and central venous pressure monitoring.
The three most common sites for central venous catheter placement are the femoral, subclavian, and internal jugular veins. All three sites provide direct access to the central circulatory system, but each site comes with its own advantages and disadvantages. In all three sites, access to the vein can be obtained using either the landmark-guided approach or under direct ultrasound guidance. Studies have shown that central lines placed using the landmark technique alone can lead to complications such as inadvertent arterial puncture, multiple punctures through the target vessel, extensive bleeding, or accidental puncture of adjacent structures such as the lung, lymphatic channels, arteries, nerves, or the urinary bladder. The ultrasound-guided approach to central line placement has been recommended by various specialties, and the consensus is that ultrasound guidance can improve time to successful cannulation while minimizing complications.10
Contraindications. There are no absolute contraindications to performing an ultrasound-guided central vein cannulation. It is important to note that ultrasound guidance may require more time than a landmark-guided cannulation attempt. In time-sensitive, emergent situations, place the central line in the quickest manner possible. The absolute and relative contraindications for performing any central line are applicable with both the ultrasound-guided and landmark-guided approaches.
Risks. Placing a central line puts the patient at risk for accidental puncture of adjacent structures. When accessing the internal jugular vein or the subclavian vein, the risks include inadvertent injury or catheterization of the surrounding arteries, hematoma formation, air embolism, hemothorax, pneumothorax, chylothorax, nerve injury, dysrhythmias, cardiac perforation, cardiac tamponade, pseudoaneurysm formation, or line misplacement. During attempts to access the femoral vein, the patient is at risk for accidental puncture of the femoral artery, pseudoaneurysm formation, hematoma formation, bowel or bladder puncture, psoas abscess, puncture of the hip capsule, or nerve injury. Using ultrasound guidance can help guide needle entry and catheter placement, thereby reducing many of these risks.
Benefits. Use of ultrasound for central venous access improves patient safety by decreasing number of attempts, time to cannulation, and number of complications.11 In 2001, the Agency for Healthcare Research and Quality (AHRQ) listed use of ultrasound to achieve central venous access as one of the top practices to improve patient care.10 With direct ultrasound guidance, you can monitor the needle trajectory during the entire procedure, thereby minimizing the risk of injury to adjacent structures. Visualizing the path of the needle allows you to know when you have entered the target vessel lumen, and you can monitor vessel cannulation under dynamic ultrasound guidance.
How to Perform the Procedure.
Femoral Vein Access Under Ultrasound Guidance
Apply a large amount of ultrasound gel to improve the acoustic interface.
Gently place the 7.5 or 10 MHz linear transducer in a transverse orientation inferior to the middle segment of the inguinal ligament.
Ensure that the orientation marker on the transducer is pointing toward the patient's right.
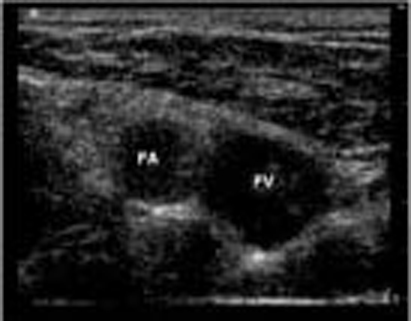
Under ultrasound, the femoral vein appears as an oval, thin-walled, anechoic structure that collapses easily under pressure. The adjacent femoral artery will be the round, anechoic, thicker-walled, pulsatile structure lying just lateral to the femoral vein (to the left of the screen). (See Figure 6.) The femoral artery should not collapse under pressure unless the patient is profoundly hypovolemic.
Figure 6: Ultrasound Image of the Femoral Artery Lying Lateral to the Femoral Vein
Application of color Doppler over the vessel of interest can help distinguish an artery from a vein. Apply color Doppler over the target vessel. Flow within an arterial lumen will appear pulsatile. In contrast, a vein will demonstrate a more constant rumble of color flow.
The color corresponds to the direction of flow (toward or away from the transducer). Be sure to take note of the color scale displayed on your ultrasound screen to determine which color corresponds to flow toward the transducer and which color is shown with flow away from the transducer.
Assess for anatomical variations or abnormal bifurcations by scanning in both a transverse and longitudinal fashion along the femoral vein.
In approximately 25% of patients, the femoral vein may course directly posterior to the femoral artery just below the inguinal ligament.
Scan caudally until the femoral vein can be visualized coursing distinctly away from the femoral artery.
Patient positioning may alter the relationship of the femoral vein in relationship to the femoral artery.12 Attempt rotating and abducting the femur in different angles to achieve the ideal spatial relationship between the femoral vein and femoral artery. (See Figure 6.)
Access the femoral vein in a sterile fashion, utilizing either the static or dynamic ultrasound-guided approach, via the short-axis or long-axis approach.
If the procedure is performed in a dynamic manner, remember to use sterile ultrasound gel and prep the ultrasound probe in a sterile fashion.
When the vessel is being cannulated under dynamic ultrasound guidance, remember that direct visualization of the needle will only be noted when the ultrasound beam is angled directly at the needle. Because the field visualized by ultrasound can be narrow, needle position may need to be inferred by visualizing ring down artifact, soft-tissue changes, and tenting of the wall of the target structure. (See Figures 7 and 8.)
Figure 7: Hyperechoic Ring Down Artifact from the Needle Advancing Toward the Vein
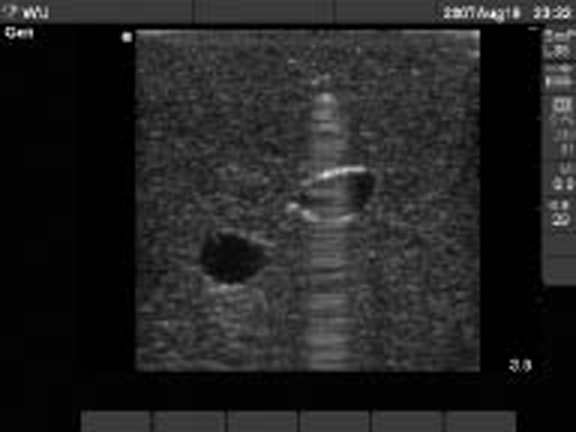
Figure 8: Tenting of the Vessel Wall as the Needle Is About to Enter the Lumen of the Vein

Under dynamic ultrasound guidance, the procedure can be performed via the short-axis or long-axis approach. (See Figure 8.)
Figure 9: Transducer Position for Visualization of the Right Internal Jugular Vein

Remember that the short-axis approach is easier to perform and requires less hand-eye coordination for novice users.13
The main disadvantage of the short-axis approach is not being able to visualize the needle tip throughout the duration of the procedure without manipulating the transducer. You may not visualize the needle tip puncturing through the posterior wall of the vessel in the short-axis approach unless you change the angle of the ultrasound beams.
The long-axis approach requires a steady hand and good hand-eye coordination. It is easy to slip off to the medial or lateral side of the vessel and lose direct ultrasound visualization during the actual cannulation.
The advantage of performing the cannulation under long-axis guidance is the ability to visualize the needle tip entering the vessel lumen without having to manipulate the transducer during the procedure. In the long-axis view, you can see if the needle tip has accidentally punctured through the posterior wall of the target vein. This is especially useful in hypotensive patients.14 Most experts recommend cannulating veins under long-axis guidance to maintain direct visualization of the needle at all times and avoid accidental puncture of adjacent vessels or structures.15
Recently, an oblique approach has been described in the literature. Obtaining an oblique view of the target vessel during dynamic cannulation enables the user to visualize the needle as it enters the vessel while providing the operator with direct visualization of surrounding structures.16
Internal Jugular Vein Access Under Ultrasound Guidance
Place the patient in Trendelenburg position and turn the patient's head gently toward the contralateral side, away from the site you are planning to access.
Turning the patient's head more than 30° past midline may cause compression of the internal jugular vein by the sternocleidomastoid muscle.
If the patient is alert and cooperative, have the patient use his or her sternocleidomastoid muscle to elevate the head momentarily to help visualize anatomic landmarks.
In the majority of patients, the internal jugular vein lies deep to the sternocleidomastoid muscle, lateral and superficial to the carotid artery. Note that anatomic variations exist, so use the superficial landmarks solely to guide where you place the transducer to begin the scan. With ultrasound, you will be able to visualize and identify all requisite landmarks and vessels and track the needle as it enters the target vein.
Apply a large amount of ultrasound gel to improve the acoustic interface.
Gently place the 7.5 or 10 MHz linear transducer at the bifurcation of the sternal and clavicular heads of the sternocleidomastoid muscle for the standard central approach. (See Figure 9.)
Under ultrasound, the internal jugular vein appears as an oval, thin-walled, anechoic structure that collapses easily under pressure. The adjacent carotid artery will be the round, anechoic, thicker-walled, pulsatile structure lying just medial to the internal jugular vein. The carotid artery should not collapse under pressure unless the patient is profoundly hypovolemic. (See Figure 10.)
Figure 10: Transverse (Short-axis) View of the Right Internal Jugular Vein (IJ) and Right Carotid Artery (CA)

Don't be overzealous with your compression test. Perform the compression test once or twice to ensure that the target vessel is a patent vein that does not house any intraluminal clot. Multiple compression attempts near the carotid sinus can stimulate baroreceptors causing bradycardia and hypotension if too much or prolonged pressure is applied.
Application of pulse wave Doppler over the vessel of interest can help distinguish an artery from a vein. Flow within an arterial lumen will appear pulsatile. In contrast, a vein will demonstrate a more constant rumble of color flow.
Scan along the internal jugular vein in a transverse and longitudinal fashion to assess for any anatomic or pathologic variations.
Access the internal jugular vein in a sterile fashion, utilizing either the static or dynamic ultrasound-guided approach.
If the procedure is performed in a dynamic manner, remember to use sterile ultrasound gel and prep the ultrasound probe in a sterile fashion.
When the vessel is being cannulated under dynamic ultrasound guidance, remember that direct visualization of the needle will only be noted when the ultrasound beam is angled directly at the needle. Because the field visualized by ultrasound is so narrow, needle position may need to be inferred by visualizing ring down artifact, soft-tissue changes, and tenting of the wall of the target structure. (See Figures 7 and 8.)
Under dynamic ultrasound guidance, the procedure can be performed via the short-axis or long-axis approach.
Remember that the short-axis approach is easier to perform and requires less hand-eye coordination for novice users.
The main disadvantage of the short-axis approach is not being able to visualize the needle tip throughout the duration of the procedure without manipulating the transducer. You may not visualize the needle tip puncturing through the posterior wall of the vessel in the short-axis approach unless you change the angle of your ultrasound beams.
The long-axis approach requires a steady hand and good hand-eye coordination. It is easy to slip off to the medial or lateral side of the vessel and lose direct ultrasound visualization during the actual cannulation.
The advantage of performing the cannulation under long-axis guidance is the ability to visualize the needle tip entering the vessel lumen without having to manipulate the transducer during the procedure. In the long-axis view, you can see if the needle tip has accidentally punctured through the posterior wall of the target vein. This is especially useful in hypotensive patients.14 Most experts recommend cannulating veins under long-axis guidance in order to maintain direct visualization of the needle at all times and avoid accidental puncture of adjacent vessels or structures.15
Recently, an oblique approach has been described in the literature. Obtaining an oblique view of the target vessel during dynamic cannulation enables the user to visualize the needle as it enters the vessel while providing the operator with direct visualization of surrounding structures as well.16
Subclavian Vein Access Under Ultrasound Guidance
Ultrasound can be used to visualize the subclavian vein as it connects to the caudal portion of the internal jugular vein.
The subclavian vein is difficult to visualize with ultrasound if an infraclavicular approach is utilized because the sonographic window can be obscured by the clavicle. To circumvent this limitation, visualize the subclavian vein from a supraclavicular approach.
Apply a large amount of ultrasound gel to improve the acoustic interface.
Place the 7.5 to 10 MHz linear transducer in a transverse orientation 1 cm lateral to the sternal notch and just superior to the clavicle. (See Figure 11.)
Figure 11: Transducer Position to Visualize the Subclavian Vein Using a Supraclavicular Approach

Angle the probe caudally to visualize the subclavian vein lying just lateral to the internal jugular vein.
In this plane, the subclavian vein appears as an anechoic, thin-walled structure lying just below the hyperechoic clavicle. The thicker-walled, pulsatile subclavian artery may be seen coursing just below the subclavian vein. (See Figure 12.)
Figure 12: Ultrasound Image of the Subclavian Vein Lying on Top of the Subclavian Artery Via the Supraclavicular Approach

Fanning the probe in a cranial fashion may provide you with an image of the internal jugular vein joining the subclavian vein. (See Figure 13.)
Figure 13: Ultrasound Image of the Internal Jugular Vein (IJ) Joining the Subclavian Vein from a Supraclavicular Approach

Scan along the subclavian vein to assess for any anatomic or pathologic variations.
Access the subclavian vein in a sterile fashion, utilizing either the static or dynamic ultrasound-guided approach.
If the procedure is performed in a dynamic manner, remember to use sterile ultrasound gel and prep the ultrasound probe in a sterile fashion.
When the vessel is being cannulated under dynamic ultrasound guidance, remember that direct visualization of the needle will only be noted when the ultrasound beam is angled directly at the needle. Because the field visualized by ultrasound is so narrow, needle position may need to be inferred by visualizing ring down artifact, soft-tissue changes, and tenting of the wall of the target structure.
Under dynamic ultrasound guidance, the procedure can be performed via the short-axis or long-axis approach.
Because ultrasound visualization of the subclavian vein can be impeded by the overlying clavicular bone, the ease of performing the cannulation via the short-axis approach versus the long-axis approach is largely patient-dependent. Image the vessel in both the long and short axis and determine which view offers the best visualization of the vein and anticipated needle path.
The advantage of performing the cannulation under long-axis guidance is the ability to visualize the needle tip entering the vessel lumen without having to manipulate the transducer during the procedure. In the long-axis view, you can see if the needle tip has accidentally punctured through the posterior wall of the target vein. This is especially useful in hypotensive patients.14 Most experts recommend cannulating veins under long-axis guidance to maintain direct visualization of the needle at all times and avoid accidental puncture of adjacent vessels or structures.15
Recently, an oblique approach has been described in the literature. Obtaining an oblique view of the target vessel during dynamic cannulation enables the user to visualize the needle as it enters the vessel, meanwhile providing the operator with direct visualization of surrounding structures as well.16
Pearls and Pitfalls
Ultrasound-guided placement of a central venous catheter requires good hand-eye coordination and a basic understanding of the principles of ultrasound. Performing the procedure becomes quicker and easier with practice.
When evaluating the target vein for cannulation, use ultrasound to evaluate for the presence of intraluminal clot, also. Because veins are thin-walled vessels, they should collapse easily under pressure. Apply gentle pressure over the target vein with the ultrasound transducer. If the walls of the vein do not collapse completely and "wink" at you on the ultrasound screen, an intraluminal clot may be present.
Although you will be able to visualize most sub-acute, hyperechoic intravascular clots on ultrasound, know that acute clots may possess the same echogenicity as the surrounding blood in the vessel lumen, and are therefore very difficult to see. Always perform a compressibility test over the vessel you are aiming to cannulate.
During the procedure, you can use ultrasound guidance to monitor guide-wire placement and ensure that it is feeding in the intended direction and not through the posterior wall of the vessel.
Ultrasound-guided Suprapubic Aspiration
Indications. Prolonged urinary retention can lead to obstructive nephropathy and renal cell ischemia. A suprapubic aspiration and catheterization may be required when urinary retention occurs secondary to prostatic obstruction, gynecologic or other malignancy, spinal cord injury, and other processes that impede emptying of the bladder. It is also indicated in urethral trauma, urethral stricture, chronic urethral infection, phimosis, and to obtain sterile urine studies in children younger than 2 years of age.17 Traditionally, suprapubic aspirations are performed using landmark guidance alone. A quick bedside scan of the bladder can help confirm the diagnosis of urinary retention and guide catheterization attempts.
Contraindications.Contraindications to suprapubic aspiration include an empty bladder, recent abdominal surgery, lower abdominal wound or cellulitis, previous pelvic radiation with resultant scarring, and known bladder cancer. There may be increased risk of bowel or peritoneal penetration in patients with previous abdominal surgeries or scarring.18,19
Risks. Use of ultrasound confers no additional risk to the patient. With the landmark approach, the patient is at risk for accidental puncture of the bowel or peritoneum. Perforation of the uterus, rectum, and vagina is also possible. Using ultrasound to guide the procedure greatly diminishes these risks. As with the landmark-guided approach, ultrasound-guided suprapubic aspirations can still put the patient at risk for post-procedure infection, abscess formation, bleeding, hematoma formation, hematuria, tube dislodgement, persistent urine extravasation, ileus, and catheter malfunction.20 Using a blind approach, the overall rate of complications with suprapubic cystostomy is only 1-2%, but up to 5% in complicated patients such as those with a large body habitus, abdominal adhesions, or in uncooperative patients such as children.20,21
Benefits. Using ultrasound guidance to perform a suprapubic aspiration or placement of a cystostomy tube is safer and easier to perform than using the landmark technique alone. In complicated patients, using ultrasound confers a higher rate of success and decreases the risk of complications. Studies have shown that physicians have twice the success with suprapubic bladder needle aspiration when using ultrasound guidance. It is easy to learn and allows clear visualization of a full bladder. Ultrasound is sensitive in confirming bladder distention, which bears clinical importance. Ensuring that the bladder is full and abutting the abdominal wall is paramount in preventing penetration of bowel and other procedural complications. Bedside ultrasound facilitates catheter placement in complicated patients by confirming a distended bladder and defining its limits. Experts recommend the use of ultrasound guidance in all cases where there is any question of bladder volume, anatomical limits, or position.22,23 If ultrasound is used to guide cystostomy tube placement, it can not only help confirm proper tube placement, but also can be used to monitor bladder decompression and successful drainage.
How to Perform the Procedure.
Assemble the required equipment for a suprapubic catheterization. If a pre-packaged catheterization kit is not available, secure the following items:
– Antiseptic (typically povidone-iodine)
– Sterile gloves and drapes
– Gauze pads (4x4)
– 1% lidocaine
– 10 mL syringe
– 25-gauge standard needle
– 22-gauge spinal needle (1.5 inches for children; 3 inches for adults)
– J-tip guide-wire
– 11 blade scalpel
– Dilator
– Introducer sheath
– Foley catheter (should be 1F smaller than diameter of introducer sheath)
– Sterile, closed system urinary drainage bag
– Sterile dressing.
Have the patient lie supine in a position of comfort.
Prep and drape the patient and the ultrasound transducer in a sterile fashion.
You can use a low- or high-frequency transducer for this procedure. If the patient has an abundance of subcutaneous fat and tissue, use a low-frequency transducer (5-1MHz curvilinear or phased array). If the patient is thin and devoid of much subcutaneous fat, use the high-frequency transducer (13-5MHz linear array).
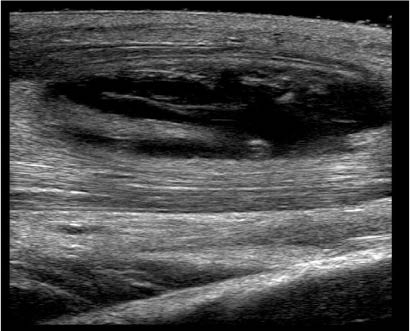
Visualize the urinary bladder via bedside ultrasound. A distended bladder will appear as a circular or square-shaped anechoic structure on ultrasound. (See Figure 14.)
Figure 14: Ultrasound of a Normal Bladder
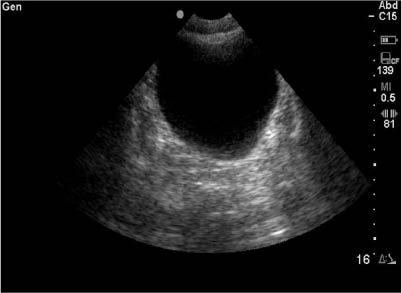
The bladder volume can be estimated using the equation: volume = length x width x height x 0.52. The 3 measurements (length, width, and height) must be taken in two separate planes (longitudinal and transverse).24
Examine the bladder boundaries in both the transverse and longitudinal planes. Take note of any adjacent bowel, vascular structures, and organs.
Once the bladder has been visualized and noted to be full, administer local anesthesia to the skin and subcutaneous tissues superficial to the middle of the bladder.
Attach a 10 mL syringe to the back of the spinal needle.
Using ultrasound guidance, insert the needle and aim toward the urinary bladder directly under the ultrasound probe. Gently aspirate back on the syringe as you slowly advance the needle. Monitor the needle trajectory as you aim toward the middle of the bladder.
Look for tenting of the anterior bladder wall as your needle is about to penetrate it.
Continue applying negative pressure on the syringe until the anterior bladder wall is penetrated and urine is noted in the syringe. (See Figure 15.)
Figure 15: Ultrasound of Needle Tip Inside the Bladder for a Suprapubic Aspiration
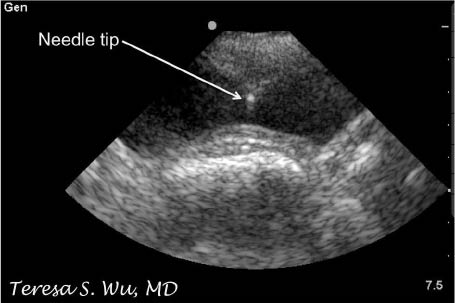
Once the needle tip is clearly in the urinary bladder, attach a 20-60 cc syringe to a 3-way stopcock and continue urine aspiration until the patient's bladder is emptied.
If the needle becomes clogged during the aspiration attempt, inject 1-2 mL of normal saline as needed to flush the needle and confirm the needle tip location under direct ultrasound visualization.
If a cystostomy tube is to be placed, it can be fed into the bladder through the spinal needle using the Seldinger technique.
Remove the syringe from the spinal needle and pass the guide-wire through the spinal needle into the bladder. Ultrasound can be utilized to visualize the guide-wire in the urinary bladder.
Remove the needle over the guide-wire, leaving the guide-wire in place.
Make a small stab incision adjacent to the guide-wire with an 11-blade scalpel.
Advance the dilator and introducer sheath into the bladder over the guide-wire.
Remove the dilator and guide-wire, and secure the sheath.
Advance the cystostomy catheter through the sheath.
Visualize the urinary catheter within the bladder on ultrasound.
Inflate the balloon and attach the catheter to the collection reservoir.
Remove the peel-away sheath and pull back on the catheter until resistance is met.
Secure the catheter to the patient's abdomen and/or leg.
Pearls and Pitfalls
Avoid unsuccessful catheterization attempts by determining if the bladder is full of urine before performing the procedure.
Always fan through the urinary bladder and obtain both longitudinal and transverse views. Note any bladder diverticula, clots, thickened walls, or masses that may interfere with the aspiration attempt.
Aim the ultrasound beams so they bisect the needle tip as it punctures the skin and enters the subcutaneous tissue. Utilize tissue movement and artifacts, such as ring down, to help visualize the needle trajectory. You may need to fan or slide the probe away from the puncture site to maintain visualization of the needle tip as you advance the needle deeper into the patient.
In emergent situations, a central line kit can be used in lieu of finding a suprapubic catheter kit. The triple or double-lumen central line can serve as a conduit for urinary drainage until a cystostomy tube can be secured. Note that patients with a large number of blood clots or large blood clots present should have large diameter catheters inserted, or have their smaller-diameter conduits changed out to larger-diameter drainage systems once they become available.
Ultrasound-guided Abscess Localization and Incision & Drainage
Indications. Diagnosing an abscess versus cellulitis can be difficult based on palpation alone. Ultrasound can be used at the bedside to determine if an abscess is present, to delineate if there are multiple fluid pockets present, to demonstrate the depth of the fluid collection, and to help direct incision and drainage attempts.25 In patients who are considered to have a high probability of an abscess, ultrasound can be used to ensure that all pockets are successfully drained. Ultrasound is most useful in patients who are low to moderate probability of having an underlying abscess. In these patients, ultrasound has been shown to detect subcutaneous fluid collections that may otherwise have been missed based on physical exam findings alone. Before performing an incision and drainage, use bedside ultrasound to augment the clinical exam findings and help localize the best site(s) for drainage.26
Contraindications. There are no absolute contraindications to using ultrasound for the diagnosis and management of an abscess. Prior to performing an incision and drainage of an abscess, please note that some abscesses should be managed in the operating suite, such as perianal abscess with fistula in ano, an abscess in a location where drainage presents a major risk of damage to adjacent organs or vessels, deep abscesses that cannot be accessed easily in the emergency department, abscesses that may cause systemic bacteremia when incised, and abscesses in patients who have a coagulopathy.27
Risks. There is very little risk to using ultrasound to find and drain an abscess. Note that the use of bedside ultrasound is operator dependent, so in rare cases, it can yield false-positive results.28 Cellulitic tissue can often be surrounded by a large amount of edema, which can be mistaken for an abscess pocket if not analyzed thoroughly. Also, blood vessels and pseudoaneurysms can appear very similar to abscess pockets on ultrasound evaluation. Before performing an abscess incision and drainage, note the risks of the procedure itself. These include damage to surrounding structures, systemic infection, pain, bleeding, hematoma formation, osteomyelitis, septic arthritis, painful neuroma, skin necrosis, and cosmetic flaws or deformities.29,30
Benefits. Ultrasound can be used to reliably identify the presence of a drainable fluid pocket and determine the exact location and depth of the abscess. The benefit of identifying a fluid collection with bedside ultrasound outweighs the small risk of a false positive. The definitive management for soft-tissue abscesses requires incision and drainage of all fluid pockets. Bedside ultrasound can be used to identify multiple pockets and deeper fluid collections that may not be identifiable on physical examination alone.31 In addition, an ultrasound-guided incision and drainage minimizes the risk of accidental puncture to adjacent organs, nerves, vessels, or tendons. With the aid of color Doppler or spectral Doppler, ultrasound can be used to identify abscess mimics such as a pseudoaneurysm, enlarged lymph node, or large, tortuous vessels.
How to Perform the Procedure.
Place the patient in a position of comfort.
Prep and clean the patient and the ultrasound probe in a standard fashion.
You will be using a high-frequency linear array transducer (13-5MHz). For superficial abscesses, an acoustic standoff pad can be used to enhance image resolution if a higher frequency transducer is not available. If commercial acoustic standoff pads are not available, use a large amount of ultrasound gel or scan through a water-filled glove sandwiched between two layers of gel.
To minimize the risk of contamination and further infection, try to use sterile ultrasound gel during the scan. Single-use Surgilube packets work well for this application.
Start by scanning the normal tissue margins surrounding the area of interest. Note that normal subcutaneous tissue will have dark, hypoechoic regions of fat mixed in with brightly hyperechoic muscle, fascial, and tendon planes. (See Figure 16.)
Figure 16: Ultrasound of Normal Soft Tissue
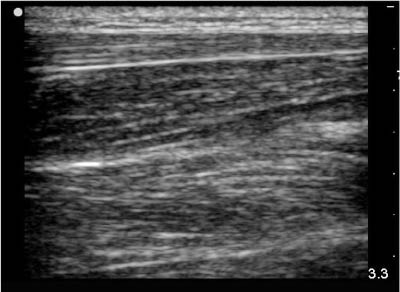
As you scan toward the area of interest, look for a spherical or elliptical collection of hypoechoic material indicating abscess formation. The hypoechoic or anechoic fluid collection may have ring enhancement indicated by a rim of hyperechoic surrounding tissue.
Abscesses may appear hypoechoic or anechoic during the initial stages of formation. As the inflammatory process progresses, the pus may begin to appear more heterogeneous with a mix of hypoechoic and hyperechoic material swirled together. (See Figure 17.)
Figure 17: Ultrasound of an Abscess Pocket
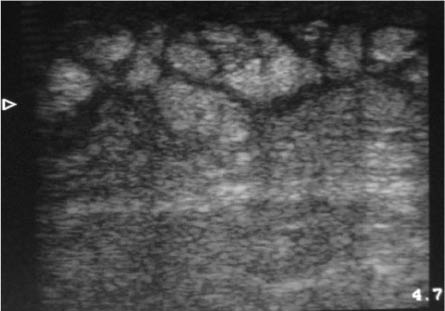
Differentiating between cellulitis and small abscess pockets may be difficult. Cellulitic soft tissue will have a "cobblestone appearance" on ultrasound. (See Figure 18.) Large collections of subcutaneous edema can appear similar to small abscess pockets. Apply a small amount of pressure over the area of interest. Fluid collections secondary to cellulitis will redistribute with pressure. Abscess pockets will not diffuse under pressure.
Figure 18: Ultrasound of Soft Tissue Cellulitis
Always obtain images in multiple planes (longitudinal, transverse, oblique) to help define the borders of the abscess pocket(s).
Identify surrounding nerves, lymphatic channels, vessels, and organs to help prevent accidental puncture during the incision and drainage.
The procedure can be performed in the static or dynamic fashion once the abscess pockets and surrounding structures have been identified.
Pearls and Pitfalls
Minimize your chances of accidentally incising a large vessel or pseudoaneurysm or necrotic lymph node by obtaining Doppler enhanced images of the target structure. Apply pulse wave Doppler or spectral Doppler over the anechoic area of interest.
Utilize contralateral limbs and adjacent areas of normal-appearing tissue for comparison.
Use the depth markers on the side of the ultrasound screen to help you estimate the required depth of your incision for adequate dissection and drainage.
Scan through the entire area of interest to determine if there are satellite pockets of fluid or multiple loculated collections that also require drainage.
Ultrasound can be used to determine if a foreign body is present within the abscess pocket before incision and drainage is attempted.
Re-evaluate the abscess site with ultrasound after drainage to determine if an adequate amount of purulent drainage was expressed during the procedure. Ensure that all loculated collections were incised.
Ultrasound-guided Foreign Body Localization
Indications. It is often difficult to determine, by history and physical alone, whether a patient's wound contains a foreign body (FB). Occult foreign bodies are common, and only radiopaque objects such as metal, leaded glass, and stone are discovered by conventional X-rays. Radiolucent objects (i.e., thorns, wood, plastic) are easily missed on standard plain films.32 Although CT and MRI have more sensitivity and specificity in detecting foreign bodies, their routine use for this purpose is not practical. With the portability and ease of using bedside ultrasound, many clinicians have turned to this modality when there is a question as to whether a foreign body may be present.33 Initial studies to determine efficacy of ultrasound in identifying radiolucent foreign bodies employed various tissue models. These studies yielded conflicting results and found that accuracy of ultrasound depends on the physical model used, operator experience, and size of foreign body.34-36 Clinical studies were more favorable, as the value of ultrasound to locate foreign bodies was repeatedly demonstrated in various clinical settings.37-39 In situations where a potential radiolucent foreign body is suspected, bedside ultrasound can be used to make the diagnosis and to guide attempts for foreign body removal.
Contraindications. There are no absolute contraindications to using bedside ultrasound to detect the presence of a foreign body or for its use to guide removal attempts. Before attempting a foreign body removal, note there are several relative contraindications to the procedure itself. These include coagulopathy or bleeding diathesis, chronic medical problems that delay healing (diabetes, uremia, immunocompromised state), involvement of the abdomen, pelvis, or thorax, proximity to major vascular structures that are difficult to visualize, uncooperative patients, failure to localize the foreign body, allergy to anesthetic, and joint involvement.40,41
Risks. The only risk of using ultrasound to detect a foreign body is the risk of obtaining a false-positive or false-negative result. This risk is reduced with practice. The decision to retrieve a foreign body does come with certain risks. The procedure can cause pain or aggravate local bleeding or injury, and even damage neurovascular structures, if not performed carefully. Patients who undergo foreign body removal are also at risk for localized or systemic infection, cosmetic defects or flaws, and further wound contamination.
Benefits. Ultrasound allows identification and removal of foreign bodies that are not visible on conventional X-rays or visual inspection alone. Using bedside ultrasound for the evaluation of a suspected foreign body minimizes the risks associated with blind exploration attempts. Ultrasound can be used in real-time to localize and guide retrieval attempts of foreign bodies under direct visualization.
How to Perform the Procedure.
Place the patient in a position of comfort.
Prep and clean the patient and the ultrasound probe in a standard fashion.
For most superficial foreign bodies, use a high-frequency linear array transducer (13-5MHz).
An acoustic standoff pad can be used to enhance image resolution of superficial wounds. If commercial acoustic standoff pads are not available, scan through a water-filled glove sandwiched between two layers of gel.
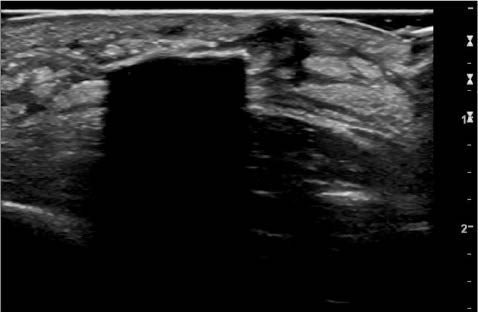
Figure 19: Ultrasound of a Foreign Body in the Arm
To minimize the risk of contamination and further infection, try to use sterile ultrasound gel during your scan. Single-use Surgilube packets work well for this application.
Start by scanning the normal tissue margins surrounding the area of interest. Note that normal subcutaneous tissue will have dark, hypoechoic regions of fat mixed in with brightly hyperechoic muscle, fascial, and tendon planes. (See Figure 16.)
In general, in the acute phase, foreign bodies will appear as bright white, hyperechoic structures on ultrasound. (See Figure 19.) Note the acoustic shadowing behind the foreign body.
Some foreign bodies will demonstrate unique sonographic artifacts. Metal objects will demonstrate a comet-tail artifact secondary to sound wave reverberation. (See Figure 20.)
Figure 20: Ultrasound Image of a Metal Foreign Body and Reverberation Artifact

Glass and plastic will demonstrate a more diffuse, scattered acoustic shadow farfield, whereas sand, gravel, and wood will create a dark, dense acoustic shadow deep to the object. (See Figure 21.)
Figure 21: Ultrasound Image of a Rock with Dense Acoustic Shadowing Farfield

With time, the sonographic appearance of foreign bodies can change. Plant substances and wood can absorb water and appear less echogenic with time. In the subacute or chronic phase, look for acoustic shadowing to help localize the foreign body.
One the foreign body has been visualized, use the depth markers on the side of the ultrasound screen to determine the depth of exploration attempts.
Foreign body retrieval can be performed in either the static or dynamic fashion.
Under dynamic ultrasound guidance, metal finder needles and forceps can be seen and monitored during the procedure. Use ultrasound to guide the trajectory of your forceps or finder needle.
Scan the area of interest after the procedure has been completed to determine if there are any retained pieces of the foreign body.
Pearls and Pitfalls
Often times, the foreign body may not be able to be visualized directly on ultrasound. Look for acoustic artifacts such as shadowing or reverberation artifact to help localize the object.
Some foreign bodies are surrounded by layers of hypoechoic fluid due to inflammatory changes in the surrounding soft tissue. The fluid can help enhance visualization of the foreign body.
In contrast, if there is air surrounding the foreign body, it will be more difficult to visualize on ultrasound. To circumvent the air interface, gently compress the overlying tissue with the transducer to minimize the air-tissue interface.
Even under direct ultrasound guidance, note that it may be impossible to detect and remove all foreign bodies. Inform and educate the patient about the risks and benefits of the procedure.
Aim the ultrasound beams so they bisect the needle tip or forceps tips as they enter the wound above the foreign body. The best approach is to obtain a long-axis view of the needle and its trajectory toward the foreign body. Follow the needle in the long axis as you advance it deeper toward the target structure.
Ultrasound-guided Arthrocentesis
Indications. An arthrocentesis is typically performed for diagnostic or therapeutic purposes. Traditionally, this is performed by palpating external anatomic landmarks and directing a needle into the joint space. Care must be taken to avoid accidental puncture of surrounding tendons, nerves, and vascular structures. When there is prominent overlying edema, subcutaneous fat, or scar tissue, identification of the external landmarks and the target joint space is often rather difficult. Bedside ultrasound can be utilized to map out the vital structures surrounding the target joint space and used to guide needle trajectory during the arthrocentesis.42 Although ultrasound can be used to guide an arthrocentesis of any joint, the most common joints evaluated include the shoulder, elbow, wrist, hand, hip, knee, ankle, and foot.43
Contraindications. There are no absolute contraindications to performing an ultrasound-guided arthrocentesis. Bear in mind, the relative contraindications to performing a joint arthrocentesis include cellulitis or an abscess overlying the joint, skin lesions or dermatitis overlying the puncture site, bacteremia, adjacent osteomyelitis, uncontrolled coagulopathy, or presence of an artificial joint.
Risks. The biggest risk of performing a landmark-guided arthrocentesis is the chance of lacerating adjacent nerves, vessels, cartilage, or tendons. Patients are also at risk of a dry tap, developing a hemarthrosis, and introduction of organisms into the joint with a subsequent joint infection.
Benefits. Ultrasound can be used at the bedside to clearly identify the bony structures surrounding the joint of interest. In patients where the landmark technique can be unreliable, ultrasound can not only define the joint space, but can also be used to determine if there is intra-articular fluid present.44,45 Using ultrasound guidance to evaluate the joint and to map out overlying structures can minimize the risk of a dry tap and prevent inadvertent injury to surrounding nerves, vessels, cartilage, and tendons.
How to Perform the Procedure.
Place the patient in a position of comfort where the target joint is easily accessible.
Slight flexion of the target joint will increase space at the entry site.
Prep and drape the ultrasound transducer and the patient in a sterile fashion.
You will be using a high-frequency, linear array transducer for most superficial joint aspirations (13-5MHz). If you are accessing a deeper joint, or if there is an abundance of overlying edema or subcutaneous tissue, use the lower-frequency curvilinear or phased array transducer (5-1MHz).
If a commercially packaged arthrocentesis kit is unavailable, gather the following supplies:
– Sterile gloves and drapes
– Sterile ultrasound probe covers
– Sterile ultrasound gel
– Gauze pads (4x4 inch)
– Skin sterilization solution (Betadine or povidone-iodine)
– Lidocaine 1-2% with epinephrine
– 5 mL, 10 mL, and 20 mL syringe
– 18-gauge and 25-gauge standard needles (larger patients may require the use of a 20- to 22-gauge spinal needle)
– Hemostat
– Specimen tubes
– Occlusive dressing, bandage, or gauze and tape.
In a sterile fashion, scan around the target joint to identify the bony structures that make up the joint.
Bones will appear as bright white, hyperechoic structures on the ultrasound screen. Bones will typically cast dark acoustic shadows behind them farfield on the screen. The space in between them is the joint space. (See Figure 22.)
Figure 22: Ultrasound Image of Bones and a Joint Space

Fluid in and around the joint will appear dark and hypoechoic. (See Figure 23.) If a purulent infection has formed or if the patient has a hemarthrosis, the fluid may be speckled with hyperechoic heterogeneous clot or pus.
Figure 23: Ultrasound Image of a Joint Effusion

Align the joint space in the middle of the ultrasound screen.
Map out any overlying or adjacent nerves, tendons, cartilage, and vessels that must be avoided during the arthrocentesis.
Inject local anesthetic to anesthetize the skin and subcutaneous tissue overlying the joint.
Attach a 10 mL syringe to the back of the aspiration needle and insert the needle directly under the center of the ultrasound probe.
Using ultrasound guidance, insert the needle and aim toward the space between the two hyperechoic bones. Gently aspirate back on the syringe as you slowly advance the needle. Monitor the needle trajectory as you aim toward the middle of the joint.
Continue applying negative pressure on the syringe until joint fluid is noted in the syringe.
If fluid stops flowing into the syringe during the procedure, use ultrasound guidance to confirm needle tip placement first, then attempt to milk more fluid from the joint by squeezing the dependent fluid up toward the needle tip.
Pearls and Pitfalls
Ultrasound is especially useful in identifying smaller joint spaces such as those in the hand, wrist, and foot.
Flex and extend the joint under direct ultrasound visualization to determine the best position to maximize the amount of joint space available for aspiration.
Under ultrasound guidance, you can determine if the fluid you are aspirating is actually from the target joint and not from fluid external to the bursa.
If the needle is dislodged from the joint during the procedure, attempt to localize the tip via direct ultrasound visualization. If the tip is difficult to see, inject a small amount of normal saline and monitor where the saline bubbles originate on ultrasound.
Aim the ultrasound beams so they bisect the needle tip as it enters the skin, subcutaneous tissue, and into the joint. Utilize tissue movement and artifacts, such as ring down, to help you visualize the needle trajectory. You may need to fan or slide the probe away from the entry site to maintain visualization of the needle tip as you advance it slowly into the joint space.
An acoustic standoff pad can be used to help improve image resolution of superficial joints. If acoustic standoff pads are not readily available, use an abundance of gel or scan through a water-filled glove sandwiched between two layers of gel.
Conclusion
Bedside ultrasound provides a safe, inexpensive, and efficient way of analyzing patient anatomy and mapping out the best course to proceed with various procedures. In well-trained hands, ultrasound can be used to maximize the chances of success while minimizing the inherent risks involved with any procedure. Prior to performing any invasive procedure, utilize bedside ultrasound to map out the target structure and all of the adjacent organs and vessels that should be avoided. With ultrasound guidance, you can improve patient satisfaction, decrease the time required to successfully perform a procedure, and minimize complications.
References
1. Abboud PA, Kendall JL. Ultrasound guidance for vascular access. Emerg Med Clin North Am 2004;22(3):749-773.
2. Chinnock B, Thornton S, Hendey GW. Predictors of success in nurse-performed ultrasound-guided cannulation. J Emerg Med 2007;33(4):401-405.
3. Bauman M, Braude D, Crandall C. Ultrasound-guidance vs. standard technique in difficult vascular access patients by ED technicians. Am J Emerg Med 2009;27(2):135-140.
4. Doniger SJ, Ishimine P, Fox JC, et al. Randomized controlled trial of ultrasound-guided peripheral intravenous catheter placement versus traditional techniques in difficult-access pediatric patients. Pediatr Emerg Care 2009;25(3):154-159.
5. Stein J, George B, River G, et al. Ultrasonographically guided peripheral intravenous cannulation in emergency department patients with difficult intravenous access: A randomized trial. Ann Emerg Med 2009;54(1):33-40.
6. Keyes LE, Frazzee BW, Snoey ER. Ultrasound-guided brachial and basilica vein cannulation in emergency department patients with difficult intravenous access. Ann Emerg Med 1999;34:711-714.
7. Costantino T.G., Fojtik J.P. Success rate of peripheral IV catheter insertion by emergency physicians using ultrasound guidance [abstract]. Acad Emerg Med 2003;10:487.
8. Mills CN, Liebmann O, Stone MB, et al. Ultrasonographically guided insertion of a 15-cm catheter into the deep brachial or basilic vein in patients with difficult intravenous access. Ann Emerg Med 2007;50(1):68-72.
9. Adihikari S, Blaivas M, Morrison D, et al. Comparison of infection rates among ultrasound-guided versus traditionally placed peripheral intravenous lines. J Ultrasound Med 2010;29:741-747.
10. Shojania KG, Duncan BW, Mc Donald DM, et al. Making health care safer: A critical analysis of patient safety practices. Evid Rep Technol Assess 2001;43:1-668.
11. Leung J, Duffy M, Finckh A. Real-time ultrasonographically-guided internal jugular vein catheterization in the emergency department increases success rates and reduces complications: A randomized prospective study. Ann Emerg Med 2006;48:540-547.
12. Werner SL, Jones RA, Emerman CL. Effect of hip abduction and external rotation on femoral vein exposure for possible cannulation. J Emerg Med 2008;35(1):73-75.
13. Mahler SA, Wang H, Lester C, et al. Short vs. long-axis approach to ultrasound-guided peripheral intravenous access: A prospective randomized study. Am J Emerg Med 2010; Oct 14 [Epub ahead of print].
14. Blaivas M. Video analysis of accidental arterial cannulation with dynamic ultrasound guidance for central venous access. J Ultrasound Med 2009;28(9):1239-1244.
15. Stone MB, Moon C, Sutijono D, et al. Needle tip visualization during ultrasound-guided vascular access: Short-axis vs. long-axis approach. Am J Emerg Med 2010;28(3):343-347.
16. Phelan M, Hagerty D. The oblique view: An alternative approach for ultrasound-guided central line placement. J Emerg Med 2009;37(4):403-408.
17. Patel N, Kaji AH. Suprapubic catheterization. In: Shah, Mason, eds. Essential Emergency Procedures. Philadelphia, PA: LWW; 2008.
18. Aguilera PA, Choi T, Durham BA. Ultrasound-guided suprapubic cystostomy catheter placement in the emergency department. J Emerg Med 2004;26(3):319-321.
19. Gochman RF, Karasic RB, Heller MB. Use of portable ultrasound to assist urine collection by suprapubic aspiration. Ann Emerg Med 1991;20:631–635.
20. Mond DJ, Lee WJ. Fluoroscopically guided suprapubic cystostomy in complex urological cases. J Vasc Interv Radiol 1994;5:911–914.
21. Flock WD, Litvak AS, McRoberts JW. Evaluation of closed suprapubic cystostomy. Urology 1978;11:40–42.
22. Tibbles CD, Porcaro W. Procedural applications of ultrasound. Emerg Med Clin North Am 2004;22(3):797-815.
23. Promes S. Miscellaneous ultrasound applications. In: B. Simon, editor. Ultrasound in Emergency and Ambulatory Medicine. Mosby; St. Louis, MO; 1997: 256–260.
24. Simforoosh N, Dadkhah F, Hosseini SY, et al. Accuracy of residual urine measurement in men: Comparison between real-time ultrasonography and catheterization. J Urol 1997;158:59-61.
25. Craig JG. Infection: Ultrasound-guided procedures. Radiol Clin North Am 1999;37(4):669-678.
26. Loyer EM, DuBrow RA, David CL, et al. Imaging of superficial soft-tissue infections: Sonographic findings in cases of cellulitis and abscess. AJR Am J Roentgenol 1996;166(1):149-152.
27. Page-Wills C, Simon B, Christy D, et al. Utility of ultrasound on emergency department management of suspected cutaneous abscess. Acad Emerg Med 2000;7:493.
28. Squire BT, Fox JC, Anderson C. ABSCESS: Applied bedside sonography for convenient evaluation of superficial soft tissue infections. Acad Emerg Med 2005;12(7):601-606.
29. Dewitz A, Frazee B. Soft tissue applications. In: Ma O, Mateer J, editors. Emergency Ultrasound. New York: Mcgraw-Hill; 2003;361-391.
30. Loyer EM, Kaur H, David CL, et al. Importance of dynamic assessment of the soft tissues in the sonographic diagnosis of echogenic superficial abscesses. J Ultrasound Med 1995;14(9):669-671.
31. Yiengpruksawan A, Ganepola GA, Freeman HP. Acute soft tissue infection in intravenous drug abusers: Its differential diagnosis by ultrasonography. J Natl Med Assoc 1986;78(12):1193-1196.
32. Flom LL, Ellis GL. Radiologic evaluation of foreign bodies. Emerg Med Clin North Am 1992;10(1):163-177.
33. Ginsburg MJ, Ellis GL, Flom LL. Detection of soft-tissue foreign bodies by plain radiography, xerography, computed tomography, and ultrasonography. Ann Emerg Med 1990;19(6):701-703.
34. Schlager D, Sanders AB, Wiggins D, et al. Ultrasound for the detection of foreign bodies. Ann Emerg Med 1991;20(2):189-191.
35. Turner J, Wilde CH, Hughes KC, et al. Ultrasound-guided retrieval of small foreign objects in subcutaneous tissue. Ann Emerg Med 1997;29(6):731-734.
36. Schlager D. Ultrasound detection of foreign bodies and procedure guidance. Emerg Med Clin North Am 1997;15(4):895-912.
37. Bray PW, Mahoney JL, Campbell JP. Sensitivity and specificity of ultrasound in the diagnosis of foreign bodies in the hand. J Hand Surg (Am) 1995;20(4):661-666.
38. Manthey DE, Storrow AB, Milbourn JM, et al. Ultrasound versus radiography in the detection of soft-tissue foreign bodies. Ann Emerg Med 1996;28(1):7-9.
39. Crawford R, Matheson AB. Clinical value of ultrasonography in the detection and removal of radiolucent foreign bodies. Injury 1989;20(6):341-343.
40. Vermeiren B, De Maeseneer M. Medicolegal aspects of penetrating hand and foot trauma: Ultrasound of soft tissue foreign bodies. JBR-BTR 2004;87(4):205-206.
41. Fornage BD, Schernberg FL. Sonographic diagnosis of foreign bodies of the distal extremities. AJR Am J Roentgenol 1986;147(3):567-569.
42. Wiler JL, Costantino TG, Filippone L, et al. Comparison of ultrasound-guided and standard landmark techniques for knee arthrocentesis. J Emerg Med 2010;39(1):76-82.
43. Del Cura JL. Ultrasound-guided therapeutic procedures in the musculoskeletal l system. Curr Probl Diagn Radio 2008;37(5):203-218.
44. Wiler JL, Costantino TG, Fillippone L, et al. Comparison of ultrasound-guided and standard landmark techniques for knee arthrocentesis. J Emerg Med 2010;39(1):76-82.
45. Tsung JW, Blaivas M. Emergency department diagnosis of pediatric hip effusion and guided arthrocentesis using point-of-care ultrasound. J Emerg Med 2008;25(4):393-399.
Emergency physicians (EPs) often are called upon to perform a myriad of procedures. These procedures often are invasive and can carry a significant amount of risk. Recent studies have emphasized that EPs should routinely use ultrasound to help guide common procedures performed in the emergency department (ED).Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
