The Lethargic Child
The Lethargic Child
Authors:
Kiran V. Raman, MD, Pediatric Emergency Medicine Fellow, Pediatric Emergency Medicine, University of Rochester Medical Center, NY.
Colleen O. Davis, MD, MPH, FAAP, FACEP, Associate Professor, Chief, Pediatric Emergency Medicine, University of Rochester Medical Center, Rochester, NY.
Peer Reviewer:
Christopher J. Haines, DO, FAAP, FACEP, Assistant Professor of Emergency Medicine, Drexel University College of Medicine, Philadelphia, PA; Director, Department of Emergency Medicine, St. Christopher's Hospital for Children, Philadelphia, PA.
Emergency Medicine Reports Received a Second Place Award for Best Instructional Reporting in the Specialty Information Publisher's Association Awards.
Introduction
Stedman's medical dictionary defines lethargy as a "relatively mild impairment of consciousness resulting in reduced alertness and awareness; this condition has many causes but is ultimately due to generalized brain dysfunction." Lethargy as described by parents, however, may mean listlessness in a young child who has a bad URI, drowsiness of a teenager in the morning, or altered mental status in a toddler who had access to grandma's medicine cabinet.
This article will review the most common causes of lethargy in pediatric emergency patients as they present in the infant, toddler, school age, and adolescent age groups. Several causes of lethargy span more than one age group and, when applicable, this is noted.
Infancy/Toddlerhood
Sepsis. Sepsis is a common reason for lethargy in infants. Lethargy with refusal to feed has been reported in as many as 28% of neonates with proven sepsis.1 Septic infants often will present with non-specific signs and symptoms, which in addition to lethargy, include apnea, hypotonia, and temperature instability. Traditionally, neonatal sepsis has been divided into two categories: early and late onset. Early-onset neonatal sepsis occurs in the first week of life and may be due to maternal vertical transmission. Late-onset sepsis occurs after the first week of age to three months. Early-onset sepsis is typically due to bacterial pathogens, including Group B Streptococcus, Staphylococcus aureus, Klebsiella species, and Escherichia coli.2 These pathogens, as well as additional gram negatives, such as Pseudomonas, Acinetobacter, and Citrobacter species, cause late-onset sepsis, especially in the preterm infant. Enterovirus and herpes simplex virus (HSV) central nervous system (CNS) infection can also present as neonatal lethargy. HSV CNS infection presents in the first month, usually between 2-3 weeks of age.3
Bacterial meningitis occurs in approximately 15% of neonates with sepsis.4 Lethargy has been reported to be about 40% sensitive but 80% specific for meningitis across all pediatric age groups.5 In neonates, signs of CNS irritation include seizures, vomiting, and irritability. Nuchal rigidity is uncommon in this age group, occurring in only 15% of infants with meningitis.4
Infants with lethargy or other signs concerning for sepsis or meningitis should undergo a workup, including blood and urine cultures, lumbar puncture for cerebrospinal fluid (CSF) cell counts, gram stain, and culture. In cases in which the infant is unstable, a lumbar puncture may be delayed after antibiotic administration.
Empiric therapy with IV antibiotics should be started. A common treatment regimen is dual therapy with ampicillin and gentamicin. Acyclovir should be administered for suspected HSV CNS infection.6 Risk factors for HSV meningitis include a positive maternal history, fetal scalp monitoring, and prematurity.
Metabolic Disorders. Inborn errors of metabolism can present with acute encephalopathy and lethargy in infants hours to months old. Metabolic derangements often are mistaken for sepsis, or, alternatively, are a contributing cause to sepsis. Lethargy in the infant occurs when metabolites toxic to the CNS (e.g., ammonia) accumulate due to enzyme or pathway abnormalities. Since the placenta protects against this in-utero, infants are usually are born appearing normal.
High levels of serum ammonia are caused by both urea cycle defects and organic acidemias, and result in acute lethargy and severe vomiting. These symptoms may be accompanied by hypotonia, feeding intolerance, or seizures.7
Urea Cycle Defects. Infants with urea cycle defects typically present in the first 24-48 hours of life after the infant has ingested breast milk or formula and has accumulated a protein load. Congenital enzyme deficiencies preclude normal metabolism of the milk proteins, causing serum ammonia levels to rise above 1000 µmol/L. Importantly, there is no metabolic acidosis.8
Sending samples to measure both plasma and urine amino acid levels will help those involved in the later diagnostic workup to identify the specific metabolic defects. Citrullinemia is characterized by high levels of citrulline and absent argininosuccinic acid. In contrast, infants with argininosuccinic aciduria have elevated argininosuccinic acid and somewhat elevated levels of citrulline.8 Other urea cycle defects include carbamyl phosphate synthetase deficiency and ornithine transcarbamylase deficiency. Citrulline is not found in the blood with either of these, and measuring urine orotic acid can differentiate between the two.
Infants with severe hyperammonemia should be restricted from further protein intake and aggressively treated with hemodialysis, if critically ill, in order to mitigate neurological sequelae. Intravenous fluids containing dextrose (D10 ¼ NS) will provide calories to the infant. If there will be a delay in transfer of care to a pediatric intensive care unit (ICU), a 10% arginine HCL infused at 6 mL/kg over 90 minutes will result in serum ammonia level reductions in those infants with citrullinemia or argininosuccinic aciduria.8
Organic Acidemias. Elevated levels of serum ammonia with metabolic acidosis (with or without anion gap) and hypoglycemia should raise concerns for an organic academia. These include conditions such as propionic acidemia, methylmalonic acidemia, and isovaleric acidemia. Ammonia levels in these conditions are typically less elevated than those in patients with urea cycle deficits. The lethargic infant with these conditions will usually have elevated ketone levels and may have elevated lactate levels.
If organic acidemia is suspected, vitamin B12 may improve the course of certain types of methylmalonic acidemia, while biotin is effective in multiple carboxylase deficiency.8 As in all hypoglycemic infants, glucose should be added to intravenous fluids (D10 ¼ NS). Infants with severe metabolic acidosis may need sodium bicarbonate (2-3 mEq/kg in 6 hours) to correct the pH.9 The parents' report of the smell of the urine may provide a clue to the type of acidemia. Isovaleric acidemia causes foul, sweat-smelling urine, while maple syrup urine disease causes urine to be sweet-smelling.8
Hypoglycemia. Hypoglycemia, loosely defined as serum glucose less than 40-60 mg/dL, regularly presents as lethargy in the infant. It may be the sole cause of lethargy or may be a result of other conditions, such as sepsis or many inborn errors of metabolism. It is also an axiom in emergency medicine that hypoglycemia should be assessed first as a potential cause for lethargy or coma regardless of patient age. Most cases of neonatal hypoglycemia occur in the first 48-72 hours of life. These often present in premature infants or infants of diabetic mothers. In the past, infants with hypoglycemia were typically recognized and treated before the infant was discharged from the hospital; however, after early discharge, many of these infants may present to the ED with lethargy. Newborn infants delivered at home may also present to the ED with undiagnosed hypoglycemia.
In infants older than 72 hours who present with hypoglycemia, the low glucose can be a non-specific sign of sepsis, or other physiological stress, or, alternatively, a marker of hyperinsulinism or an inborn error of metabolism. Infants presenting to the ED with lethargy, regardless of etiology, need both a bedside finger or heel stick blood glucose and confirmatory laboratory blood glucose test. Rapid dextrose infusion is most effective at alleviating acute hypoglycemia. An IV bolus of 0.25-1 g/kg of dextrose (about 5-10 mL/kg of 10% dextrose or 3-4 mL/kg of 25% dextrose) should suffice.10 Initially, repeated checks of blood glucose levels every half-hour are needed to determine if the infant is responding appropriately.
Hypoglycemia is frequently a finding in fatty oxidation disorders and disorders of carbohydrate metabolism. Glycogen storage diseases (GSD) will present with hepatomegaly and lactic acidosis in addition to the hypoglycemia. Dysfunction in the fatty acid oxidation pathway is most typified by medium-chain acyl-CoA dehydrogenase deficiency (MCAD). Children are typically 6-18 months of age at onset, are hypoglycemic but non-ketotic, and may also have hyperammonemia, elevated AST and ALT, and metabolic acidosis.8,9 The diagnosis is made via serum carnitine levels and urine organic acid analysis. Acute management for both GSD and MCAD in the ED consists of dextrose infusion to correct the hypoglycemia, avoidance of fasting, and supportive care. Infants with MCAD should have their dietary fat restricted. L-carnitine supplementation should be started with guidance from a metabolic subspecialist.
Galactosemia and hereditary fructose intolerance are rare metabolic disorders that can present with hypoglycemia and lethargy. Infants with galactosemia usually present in their second week of life after several feedings of formula or breast milk. Symptoms include lethargy, hypoglycemia, vomiting, diarrhea, and poor weight gain.8 Jaundice and liver dysfunction are prominent. Infants with galactosemia initially may have elevated indirect bilirubin levels. These will then shift to elevated direct bilirubin levels. Diagnosis is made by testing for urine-reducing substances. An infant with galactosemia is at increased risk of E. coli infection, and sepsis may present first.8
In mobile infants, toddlers, and young children with hypoglycemia, ethanol, salicylate, or oral hypoglycemic agent ingestions should be considered.10
Methemoglobinemia. Infants are at risk for acquired methemoglobinemia after exposure to nitrates, antibiotics, or local anesthetics. Sepsis and severe diarrhea-causing metabolic acidosis have also been noted to cause secondary methemoglobinemia in infants.11 The skin of patients with methemoglobinemia has a bluish hue. Additional symptoms include lethargy, dyspnea, and tachycardia. Cyanosis does not improve with supplemental oxygen. Finger oxygen saturation probe measurements will not indicate hypoxia. Blood gases may indicate metabolic acidosis. Concurrent hypoglycemia is often severe, and should be treated with dextrose infusion. Typically treatment for acquired methemoglobinemia is 1% methylene blue at 1 mg/kg. However, affected infants with congenital enzyme deficiency causing methemoglobinemia usually only exhibit cyanosis and do not respond to treatment with methylene blue.11
Intussusception. Lethargy in infants and young children can be caused by gastrointestinal obstruction, the most common cause of which is intussusception.12 Intussusception, a telescoping of proximal bowel into another section of more distal bowel, usually occurs between the ages of 3 months to 5-6 years of age,12,13 with a peak incidence at 5-9 months.14,15 This invagination of bowel occurs most commonly around the ileocecal junction, and can be intermittent. Older children with intussusceptions often have a pathologic lead-point, such as Meckel's diverticulum, polyp, Henoch-Schönlein purpura, lymphoma, or a lymphoid patch. Cystic fibrosis, adenovirus, and celiac disease have all been associated with intussusception.14
Children with intussusception typically present with nonbilious vomiting and intermittent abdominal pain. Parents may describe inconsolable crying, with knees flexed to hold legs close to the torso. Any hemoccult positive or grossly bloody stools with abdominal pain should raise the suspicion for intussusception.16 Blood and mucous-filled or "currant jelly" stools are present in only 10% of patients with early intussusception; it is usually a late finding indicative of ischemic and necrotic bowel.17 The classic triad of intermittent abdominal pain, currant jelly stools, and abdominal mass has only been found in 15-40% of patients.14,18 Diarrhea occurs in approximately 25% of cases of intussusception and can lead to a misdiagnosis of gastroenteritis. Infants with intussusception may present with lethargy, poor tone, and poor feeding. In some cases, lethargy can be the only or initial presenting symptom of intussusception, with abdominal symptoms developing later.13
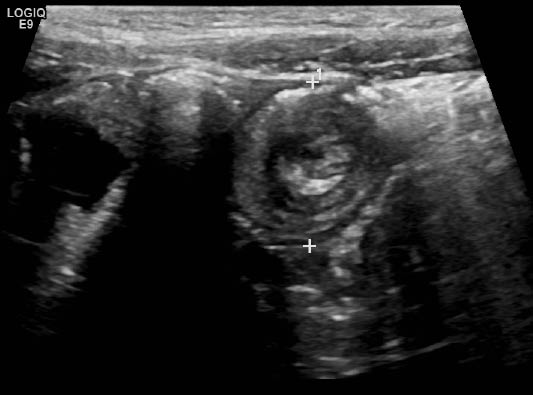
Abdominal radiographs can reveal signs of intussusception. These include a paucity of bowel gas beyond the ileocecal junction, the target sign, two concentric circles overlying the right kidney, which are representative of mesenteric fat of the intussusception, or the crescent sign, indicating a crescent-shaped mass in the transverse colon.14,15 Abdominal radiographs, however, have a sensitivity of only 45%, and may appear completely normal in infants with intussusception.18 Abdominal ultrasound can make the definitive diagnosis of intussusception.15 (See Figure 1.) It is 98-100% sensitive and 88-100% specific for diagnosing intussusception if performed by an experienced sonographer.15,19 Management is reduction with air or barium contrast enema; refractory cases need surgery.
Malrotation/Volvulus. Infants with abnormal or incomplete intestinal fixation or rotation are considered to have malrotation of the small intestine. This disorder occurs in up to 1 in 500 infants in the United States.20 These infants are at high risk to develop acute mid-gut volvulus, a twisting of the small intestine causing obstruction and strangulation of the small bowel. Half of all infants presenting with acute volvulus are younger than 1 month of age; three-fourths present in the first year of life.21,22
Initial symptoms of volvulus include bilious vomiting, bloody stools, anorexia, abdominal distention, signs of abdominal pain (crying with legs up to pulled to torso), and irritability. Alternatively, infants may present with weeks of feeding intolerance that has recently progressed to bilious vomiting.23 Infants with delayed recognition of malrotation with volvulus will present with lethargy and shock. Metabolic acidosis secondary to necrotic or strangulated bowel should be anticipated. An abdominal X-ray is poorly sensitive for acute volvulus, but may indicate malrotation by the presence of bowel loops overlying the liver, with absent bowel gas distal to the point of obstruction, or only scant air-fluid levels in the GI tract.21 An abdominal ultrasound positive for malrotation with volvulus will show inversion or coiling of the superior mesenteric vein in relationship to the superior mesenteric artery. The sensitivity of ultrasound ranges between 67-100% for diagnosing acute volvulus.20
An upper GI is the diagnostic test of choice. In the patient with acute volvulus, the duodenal-jejunal junction is malpositioned to the right side of the patient, with a "cork-screw" like appearance of the small bowel.20,23 Sensitivity and specificity of upper GI approaches 100%. Mid-gut volvulus is a surgical emergency, and an immediate laparotomy is needed to minimize the amount of bowel necrosis and resultant morbidity.
Necrotizing Enterocolitis (NEC). NEC should be considered in the newborn infant presenting with lethargy, mottled skin, and abdominal distention. Although most often a condition that afflicts premature infants, term infants with underlying medical problems, such as congenital heart disease, maternal cocaine exposure, or perinatal birth asphyxia, are at increased risk for NEC.23,24 Term infants account for 5-25% of NEC cases and usually present within the first 7-10 days of life.24 Preterm infants account for the majority of NEC cases and present later, between 1-3 weeks of age.24
Infants with NEC will often have bloody stools, as well as signs of poor circulation and shock. Neonates under stress also exhibit non-specific signs, such as temperature instability, apnea, and respiratory distress. Ominous signs indicating bowel necrosis or perforation associated with NEC include gray discoloration or bruising of the abdomen and a rigid abdomen.
Abdominal X-rays can be very helpful in diagnosing NEC. The pathognomonic finding is "railroad tracks" outlining intestinal walls. This is a double line indicating pneumatosis intestinalis, or air within the bowel wall. Early in the course, dilated bowel loops may be the only radiographical clue. A pneumoperitoneum indicates perforated bowel, a late sign. Blood chemistries and complete blood count panel results are non-specific but can indicate metabolic acidosis, leukocytosis, neutropenia, and thrombocytopenia.24 Coagulation studies may show DIC, a late-stage complication.22
NEC is a surgical emergency. Infants should be kept NPO with IV fluids and broad-spectrum antibiotics started. An NG or OG tube should be placed to decompress the stomach.
Child Abuse. When presented with a lethargic infant or toddler, the EM physician should consider child maltreatment. Child Protective Services reported that in 2008, the highest rates of child maltreatment (21.7 per 1000) occurred in infants younger than 1 year old.25 This young age group is most vulnerable, as they cannot adequately verbalize what they are experiencing. Children who are victims of non-accidental traumatic brain injury may have clues pointing to prior injury and a pattern of abuse.26
Clinicians should fully examine body areas normally covered with clothing, and carefully scrutinize areas such as inside the mouth and behind the ears. Pay particular attention to the mechanism, timing, and severity of injury as described by the parents in order to determine if the story "fits."27 Warning signs include a vague or inconsistent history. A history that does not include any traumatic event may be due to evasiveness or, alternatively, another caregiver causing the injury.
Infants and toddlers with non-accidental head trauma, including blunt-force trauma and shaken baby syndrome, can present with non-specific symptoms, such as lethargy, poor feeding, apnea, or irritability.26,28,29 These may not be accompanied by obvious presenting signs such as seizure or facial bruising. If suspicious for head trauma, a head CT is indicated. Intracranial hemorrhages due to non-accidental brain injury are frequently small subdural hemorrhages over the frontal lobes or between the hemispheres.26 A skeletal survey should be obtained in children younger than 2 years of age to identify fracture patterns pathognomonic for child abuse, such as metaphysical chip or spiral fractures of extremity bones. A head CT may miss a small, linear skull fracture in the plane of the CT; therefore, skull X-rays should still be performed.26
An ophthalmology consult is useful to evaluate for retinal hemorrhage, especially in the setting of intracranial hemorrhage. Retinal hemorrhages occur in 50-80% of abusive head trauma cases.28
Awareness of risk factors that increase the likelihood of child maltreatment is helpful. These include poor social supports, poverty, young parents, and having multiple young children in the household.27 However, child abuse occurs in all demographic groups and socioeconomic levels, but subconscious biases may prevent physicians from correctly identifying child abuse. An analysis of abusive head trauma in children reported 31% of cases were missed initially and were more likely to be missed if the child was Caucasian, from an intact family, or when initial symptoms were non-specific.29
Cardiac Arrhythmia. Supraventricular tachycardia (SVT), the most common pediatric arrhythmia, commonly presents with lethargy, irritability, poor feeding, pallor, and/or cough in the clinical presentation.30 Infants with prolonged SVT may have signs of congestive heart failure, including an enlarged liver readily palpable several centimeters below the costal margin.
Heart rates in infants with SVT exceed 220 beats per minute, often as high as 280 bpm. Management depends on the clinical picture of the patient. Most infants and children tolerate the high heart rates of SVT very well for several hours and even one to two days. Lethargy occurs when their capacity to withstand the stress of SVT has been exceeded.
If the infant is hemodynamically stable, the emergency physician may attempt vagal maneuvers by applying an ice bag over the nose and mouth for 5-10 seconds to illicit the diving reflex.31 Older children who can follow instructions should try Valsalva maneuvers. In cases in which vagal maneuvers fail to resolve the SVT, adenosine should be administered. In patients who are hemodynamically unstable or in shock, synchronized cardioversion should be performed.32
Botulism. Rarely, the non-specific findings of lethargy and poor feeding in the infant may be due to botulism. Clostridium botulinim is a gram-positive anaerobe that produces a lethal neurotoxin, causing 75-100 cases of infant botulism in the United States per year.33 Infants with botulism present with hypotonia in addition to the lethargy and poor feeding. This hypotonia is a symmetric, descending flaccidity that progresses to paralysis. Cranial nerves are often affected, causing poor suck and swallow and altered gag reflex. Infants exhibit poor head control and ptosis. Constipation is often reported by parents. Although the peak age of presentation is between 2-6 months of age, botulism can present in infants up to 1 year.33 The classic exposure to honey is, in actuality, infrequent. Physicians should be alert to details in the history pointing to more common sources of C. botulinim, such as soil or dust exposure, especially in rural farming regions.
Treatment consists of supportive care. Infants often need prolonged respiratory support and nutritional support, as full recovery can take months. This course has been shown to be shortened by early administration of BabyBIG, human botulism immune globulin.34.
School-Age Children
Diabetic Ketoacidosis. Diabetic ketoacidosis (DKA) accounts for numerous ED visits each year and 65% of hospital admissions for pediatric diabetic patients. Up to 10% of those patients with known type I diabetes mellitus (DM) suffer from DKA each year.35,36 Many new-onset diabetic patients first present with DKA.
Children in fulminant DKA will present with signs of dehydration, vomiting, along with lethargy and Kussmaul respirations. They will have a fruity odor to their breath. Other typical presenting symptoms of DKA include those of new-onset diabetes: polydipsia, polyuria, and polyphagia, along with increasing fatigue and abdominal pain.
Hyperglycemia (blood glucose > 250 mg/dL) and acidosis (serum pH < 7.3) together form the basis for the diagnosis of DKA.35 An assessment of clinical status, including vital signs, protection of the patient's airway if needed, dehydration assessment, and state of consciousness, should be performed while starting a normal saline (NS) bolus of 20 mL/kg to be run over 1 hour. If the patient is acidotic, a regular insulin drip should be started at 0.1 units/kg/hour. Insulin boluses should be avoided. Venous blood gas, serum electrolyte, and blood glucose levels should be checked initially and then rechecked an hour after starting the insulin infusion.
Potassium should be added to the fluids unless levels are measured greater than 5.5 mEq/L. To avoid hypoglycemia, dextrose should be added to IV fluids once the glucose level drops to 250-300 mg/dL.35 If the patient is no longer acidotic, insulin should be continued in subcutaneous form.
Lethargy or other mental status changes during fluid resuscitation of patients with DKA is an ominous sign warning of cerebral edema. Other warning signs include headache, hypoxia, bradycardia, and hypertension.37 Cerebral edema occurs in approximately 0.5-0.9% of cases of DKA and carries a high mortality rate (40-90%).37,38 Clinicians should be particularly vigilant during hours 3-12 after initiation of DKA treatment. Younger children and those with new-onset DM are more at risk.35,37
Begin immediate treatment if cerebral edema is suspected. Raise the head of the bed to 30 degrees. Administer mannitol at 0.25-1 g/kg IV over 20 minutes and reduce fluids.35
Adrenal Insufficiency. Adrenal crisis can present with lethargy in a child who has congenital or pharmacologically induced adrenal insufficiency. Congenital causes include Addison's disease, panhypopituitarism, or congenital adrenal hyperplasia. Drug-induced causes of adrenal insufficiency include any chronic condition being treated with steroids, such as lupus, cancer, or inflammatory bowel disease. Adrenal crisis can present in all age groups, and occurs in the setting of a body stress, such as fever or trauma. Other symptoms include severe hypotension or shock, dehydration, acute abdominal pain, vomiting, or seizures.39 Parents of children with undiagnosed adrenal insufficiency may report long-term fatigue, anorexia, or failure to thrive.39,40 Those with primary adrenal failure will have hyper-pigmentation, with darkened skin creases and an overall tanned appearance.
Immediately treat patients with known or suspected adrenal insufficiency who present with illness, injury, or crisis, with stress-dose steroids. Hydrocortisone IV/IM should be administered at an initial 50 mg/m2 dose (roughly estimated at 1-2 mg/kg).10,41 For severe stress, such as overwhelming sepsis or trauma, increase stress dosing to 100 mg/m2/day.40 Isotonic fluids should concurrently be bolused at 20 mL/kg and repeated as necessary. Treat hypoglycemia with a dextrose infusion as necessary. An initial cortisol level should be drawn if the patient does not have a known cause of adrenal insufficiency. A cortisol level of < 3 micrograms/dL is pathologic in a stressed individual; however, results should not delay management.41
Reye Syndrome. Reye syndrome is a rare post-infectious encephalitis linked to aspirin use for treatment of upper respiratory illness in children. It has two stages of presentation. The first is lethargy and vomiting, which progresses to a second stage consisting of delirium and combative behavior. In severe cases, this progresses to seizure, coma, decerebrate and decorticate posturing, or death. There is moderate liver enlargement, and liver transaminases (ALT and AST) and ammonia are highly elevated.42 Hypoglycemia is common. The CSF cell counts, glucose, and protein are normal, but opening pressure can be elevated.42 Treatment for Reye syndrome is largely supportive.
Hemolytic Uremic Syndrome (HUS). Hemolytic uremic syndrome (HUS) may occur after toxin-producing E. coli infection or, more rarely, after infection with invasive S. pneumoniae.43 In typical cases, patients exhibit fever and hemorrhagic diarrhea. Laboratory studies indicate thrombocytopenia, anemia, and acute kidney injury/failure. A blood smear will show schistocytes. Stool guaiac is usually positive. In severe cases of HUS, endothelial cell injury involves the CNS, with affected patients exhibiting seizures, combativeness, lethargy, and other mental status changes in the setting of encephalopathy.44
The majority of HUS-related deaths occur in those patients with CNS and extrarenal involvement.44 Dialysis in patients with kidney failure will likely be necessary.
Table 1: Causes of Lethargy by Pediatric Age Group, Subdivided into Medical vs. Structural Etiologies
|
Age Group |
Causes of Lethargy |
|
|
Medical |
Structural |
|
|
Abbreviations: AVM: Arteriovenous malformation, CAH: congenital adrenal hyperplasia, DKA: Diabetic ketoacidosis, ICH: Intracranial hemorrhage, HUS: Hemolytic uremic syndrome, NEC: Necrotizing enterocolitis, SVT: Supraventricular tachycardia, TBI: Traumatic brain injury |
||
|
Infants |
|
Non-accidental Trauma Accidental Trauma
|
|
Toddlers |
|
Non-accidental Trauma Accidental Trauma
|
|
School-age Children |
|
|
|
Adolescents |
|
|
Increased Intracranial Pressure. Elevated intracranial pressure (ICP) can present with lethargy in all pediatric age groups. In addition to mental status change, typical signs of increased ICP include headache and vomiting. Seizures and focal neurological weakness may occur. Elevated ICP can be due to several reasons, including intracranial hemorrhage, tumor, infection, and hydrocephalus. Regardless of etiology, resuscitate the child first, protecting the airway if clinically necessary.
Table 2: Suggested Initial Management for a Lethargic Pediatric Patient
Management Checklist
- Resuscitate/protect airway if necessary
- Monitor vital signs, cardiac and pulse oximetry monitor
- Oxygen if needed
- IV access (with NS bolus if no concern for CHF or increased ICP)
- Initial diagnostic testing
- FSBG
- 12-lead EKG
- Chemistry panel
- CBC
- Blood culture
- Serum osmolarity
- ABG/VBG
- UA
- Urine/blood toxicology screen
- Acetaminophen/aspirin level
- Pregnancy test
- Further testing if clinically/historically indicated:
- Imaging
- Lumbar puncture
- Ammonia level
- Amino acid levels
- Specific medication levels
- Alcohol screen
- Consult appropriate specialists
Abbreviations: ABG: arterial blood gas, CBC: complete blood count, CHF: congestive heart failure, EKG: electrocardiogram, FAST: Focused Assessment with Sonography for Trauma, FSBG: finger stick blood glucose, ICP: intracranial pressure, VBG: venous blood gas, UA: urinalysis
Intracranial hemorrhage (ICH) can occur with trauma in all pediatric age groups. Children with hemophilia A and sickle cell disease are at increased risk and can have ICH without precipitating trauma.45 Cocaine use is also a known etiology of subarachnoid hemorrhage (SAH) in adolescents; arteriovenous malformation (AVM) causes ICH most commonly in school-age children.
Intracranial tumors can cause elevated ICP resulting in lethargy via mass effect or bleeding. In addition to headache and vomiting, affected children may exhibit signs of malignancy, including fever, weight loss, anorexia, and fatigue. Focal neurological findings may be present.
Figure 1: Intussusception on Ultrasound with Target Sign
Mass effect may also result from infection. Subdural empyema has been reported to cause lethargy in affected patients and can be a complication of sinusitis or otitis media.46,47
Coagulopathy can produce spontaneous bleeding in the central nervous system and result in lethargy. Immune (idiopathic) thrombocytopenic purpura (ITP) is a self-limited autoimmune disorder that may cause ICH if platelet counts are reduced to less than 20 x 109/L.45,48 Diffuse petechiae and/or purpura may be present. Parents may report a history of a generally well-appearing child and recent respiratory illness.
Ruptured arteriovenous malformation (AVM) is the most common cause of intracranial hemorrhage in the school-age child.45 This can present with seizures, lethargy, and weakness, and can progress to coma. Patients can also have nuchal rigidity, meningitic signs, and/or hemiparesis, depending on the hemorrhage distribution. A previous history of frequent severe headaches is a diagnostic clue.
Figure 2: Shunt Failure with Accumulation of CSF Resulting in Hydrocephalus and Lethargy in the Child
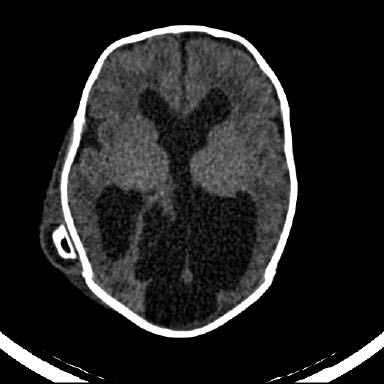
Children with hydrocephalus often have a shunt in place to drain cerebrospinal fluid. Approximately 18,000 CSF shunts are placed each year, with an average 5% annual failure rate.49 Shunt failure occurs most commonly in the first year of placement. Children with shunt malfunctions can present with signs of increased intracranial pressure, including lethargy, that, along with drowsiness and swelling around the shunt site, are predictive of shunt malfunction.49,50 Other signs include headache, nausea, and vomiting. Infants may have a bulging fontanel. Fever may be present if the shunt is infected. Appropriate imaging studies include a head CT (see Figure 2) and radiographs of the shunt (a.k.a., "shunt series"). Shunt malfunction is an important condition to detect, as shunt failure can cause mortality in up to 2% of cases.49,51
Adolescence
Traumatic Brain Injury. Lethargy and other mental-status changes are common in children of all ages who have suffered traumatic brain injury (TBI). The GCS score should be assessed, and for those patients with a GCS of 8 or less, intubation is recommended. A neuroprotective rapid sequence intubation procedure should be followed, with avoidance of ketamine and succinylcholine, as both have been reported to cause elevated intracranial pressure.52 Etomidate (0.3 mg/kg) and rocuronium (1 mg/kg) are good alternatives. These may be used in conjunction with lidocaine for further cerebroprotective effect. Elevate the head of bed to 30 degrees. In those patients with intracranial hemorrhage and/or signs of intracranial hypertension (posturing, dilated or non-reactive pupils, or Cushing's triad), mannitol (0.5-1 g/kg) should be given to reduce chance of herniation. Treat hyperthermia aggressively, as this has been shown to lead to poorer outcomes.52 Cooling therapy or therapeutic hypothermia for TBI in children is controversial. Hyperventilation also has varying effect on elevated intracranial pressure.
Seizure. Subclinical seizures in pediatric patients can present with only lethargy or altered mental status. This diagnosis is more likely in children who have a seizure history or family history of seizures. Eye deviation or loss of bladder/bowel function may be the only diagnostic clues evident. Treat suspected seizures with IV or IM lorazepam. Patients with seizures refractory to lorazepam should be administered a loading dose of phenytoin. Remember to protect the airway and administer oxygen if clinically necessary.
If the child is on regular seizure medications, levels of these drugs should be drawn for later analysis. Some antiepileptic drug levels, especially the second-generation agents, are often send-out laboratory values that take up to a week for results to be returned and, therefore, will not be useful in emergent management.
Encephalitis. Infectious encephalitis can cause lethargy or altered mental status, along with headache, seizure, or focal neurological signs. CSF will usually show an elevated lymphocyte count in the setting of a mildly elevated WBC count, normal glucose, and normal/mildly elevated protein level.53 As there are numerous causes of encephalitis, specific diagnostic testing should be guided by patient history, time of year, geographic region, and clinical suspicion. However, for most cases of encephalitis, a specific etiology is never discovered.
HSV, EBV, Mycoplasma pneumoniae, and enterovirus are all causes of encephalitis that can be diagnosed via CSF PCR.53 Cerebellar ataxia or lymphadenopathy should raise suspicion for EBV, and preceding respiratory infection is suspicious for Mycoplasma. Enterovirus and Mycoplasma can both cause a diffuse rash in addition to encephalitis.54 Start all patients with suspected infectious encephalitis on acyclovir at 10 mg/kg IV every 8 hours and broad-spectrum antibiotics at meningitic dosing.53,54 Brain MRI is more sensitive than CT in detecting edema and focal change associated with encephalitis, but both studies may be normal.
Acute disseminated encephalomyelitis (ADEM) is a post-infectious cause of encephalitis thought to be immunologically mediated. Children and adolescents affected often have had an upper respiratory infection or immunization a few days to weeks prior. Several vaccinations have been implicated, including tetanus toxoid, diphtheria, rabies, influenza, pertussis, yellow fever, and measles.54,55 There is a seasonal peak to ADEM that mimics the incidence of respiratory infections; the majority of cases occur in the winter and early spring.56 CNS symptoms include altered mental status ranging from lethargy to coma, seizures, and/or sensory, ocular, or motor neurological deficit. Patients often complain of fever, myalgias, headache, and vomiting. The CT scan will often miss ADEM lesions. T2-weighted and FLAIR MRI images will show high-intensity lesions in the gray and white matter.54,56 In addition to supportive care, treatment consists of high dose IV methylprednisolone at 20 mg/kg.55
Toxicology
Regardless of the age of the child, some common drugs can cause lethargy when ingested in toxic amounts. (See Table 3.) Consult the Poison Control Center or a toxicologist for help with acute management specific to the case.
Table 3: Ingestions that May Cause Lethargy
- Alcohols
- Ethanol
- Ethylene glycol
- Isopropyl alcohol
- Methanol
- Cardiac Drugs
- Calcium channel blockers
- Clonidine
- Digoxin
- Anticholinergics
- Organophosphates
- Psychiatric medications
- Atypical antipsychotics
- Selective serotonin- reuptake inhibitors (SSRIs)
- Tricyclic antidepressants
- Illicit Drugs
- Cocaine
- Opiates/narcotics
- Barbiturates
- Benzodiazepines
- Anti-seizure medications
- Carbamazepine
- Phenytoin
Common signs and symptoms with toxic ingestion with tricyclic antidepressants include lethargy, tachycardia, coma, hyponatremia, and hypotension.57 There are often cardiac abnormalities on ECG, with QRS and QT prolongation.57 Symptoms can start between 2 to 6 hours after ingestion.
Clinical symptoms from toxic ingestion of atypical antipsychotics, such as clozapine and olanzapine, include a spectrum from lethargy and somnolence to agitation, irritability, and combativeness.58 Tachycardia, seizures, ataxia, hypersalivation, and hypotension are other common signs.58 Symptoms can last up to several days. Supportive care is the mainstay of treatment.
Sedating medications, such as benzodiazepines and opioids, also may cause lethargy and respiratory depression. In addition to lethargy and respiratory depression, opioids may produce miosis, nausea/vomiting, and constipation.
Overdose of cholinergic medication or organophosphate ingestion presents differently depending on age group. Older adolescents will display those symptoms indicating overdrive of the parasympathetic system: hypersalivation, miosis, diaphoresis, urination, defecation, and bradycardia.59 This is not the case in younger children in whom lethargy, seizures, and other CNS signs are more common.60 Atropine should be administered for organophosphate toxicity.
Anticholinergic overdose results in mydriasis, lethargy or delerium, decreased secretions, tachycardia, urinary retention, and hypertension. There may be QRS prolongation on ECG.61
Alcohols. Ethanol, isopropyl alcohol, ethylene glycol, and methanol are all alcohols that can cause lethargy when ingested in toxic amounts.
Ethanol is commonly imbibed by adolescents during high school or college parties. A teenager who has had limited previous exposure to alcohol will be more susceptible to its CNS and respiratory depressant effects. Ethanol ingestion often causes hypoglycemia in children.
Isopropyl alcohol is found in many household cleaning and automotive products. Toxicity from isopropyl alcohol occurs via rapid absorption through gastrointestinal mucosa, usually within a half-hour after ingestion.62 Isopropyl alcohol is up to 2.5 times more potent as a CNS depressant than ethanol, and can cause symptoms of toxicity after ingestion of only 0.5-2 mL/kg.62,63 Isopropyl alcohol is metabolized to acetone in the liver, with peak concentration of metabolites at 10 hours after ingestion. During this time period, children can present initially with mild CNS effects, including "drunken" behavior, confusion, and headache, as well as gastrointestinal symptoms including vomiting and abdominal pain. Levels of isopropyl alcohol greater than 50 mg/dL are thought to be significant, although children can have symptoms at any level.62 Laboratory studies indicate an osmolar gap without metabolic acidosis. Treatment is largely supportive, unless the patient has life-threatening serum isopropyl alcohol levels higher than 400 mg/dL or is hemodynamically unstable, in which case hemodialysis should be performed.63
Methanol is used in household fuels, cleaners, and antifreeze. Its toxicity is due to its metabolism via alcohol dehydrogenase to formaldehyde and formic acid, both highly poisonous to cells. Peak symptoms do not occur until 8-24 hours after ingestion.63 Ingestion can initially cause symptoms similar to ethanol intoxication, including symptoms of dizziness, slowed cognitive thought processes, nausea, and cerebellar dysfunction. Visual disturbance is a common complaint, ranging from blurry or cloudy vision, to altered color perception, to blindness.64 CNS symptoms can progress to lethargy, coma, and death. Formic acid accumulation results in a severe metabolic acidosis and high osmolar gap. A methanol level of 20 mg/dL or higher is associated with long-term sequelae, including permanent visual loss and death, if untreated.63 Fomepizole is used to inhibit alcohol dehydrogenase and prevent the production of the toxic metabolites formaldehyde and formic acid in patients with methanol poisoning.
Ethylene glycol ingestion is severely toxic, causing metabolic acidosis and calcium oxalate deposits in organ tissue. Effects of ethylene glycol occur within 3 hours of ingestion.63 Children may exhibit nausea, vomiting, and hemodynamic compromise, in addition to lethargy and other ethanol-intoxication-like CNS effects.
Hypocalcemia can be profound and should be treated with IV calcium gluconate. Sodium bicarbonate is indicated for severe acidosis. Thiamine (0.5 mg/kg) and pyridoxine (1-2 mg/kg) are important adjunct therapeutic agents to reduce the formation of toxic metabolites.63 Ethylene glycol can result in renal failure, in which case hemodialysis may be necessary.
Cardiac Drugs. Digoxin is well known to have a narrow therapeutic index. When young children overdose on digoxin, they are less likely to develop cardiac toxicity than adults. Ingestion leading to digoxin levels greater than 4 ng/mL can produce vomiting, visual disturbance, electrolyte disturbance, and lethargy.63 If serum digoxin levels exceed 10 ng/mL, children are at risk for life-threatening AV dissociation, heart block, and severe hyperkalemia. Hemodynamic status should be monitored closely. Treat cardiac arrhythmias and hyperkalemia as indicated.
Severe digoxin toxicity can be treated with an infusion of digoxin-specific antibody fragments. The antidote dose needed is dependent on the digoxin amount ingested.63 If the amount ingested is unknown, the number of necessary antidote vials can be obtained using the child's weight (kg) and the digoxin level.63
Calcium channel blockers taken in an overdose can cause lethargy and other neurological symptoms such as dizziness and seizure activity. These can progress to coma and death. Children who ingest calcium channel blockers often exhibit bradycardia and hypotension, along with a range of cardiac arrhythmias. Symptoms also include nausea and vomiting, bowel ileus, hyperglycemia, and metabolic acidosis. Children usually exhibit signs of toxicity between 30 minutes and 2 hours after ingestion, although this time frame is longer if the medication is sustained-release.65
Clonidine, an alpha adrenergic anti-hypertensive agent, is also widely prescribed for ADHD, mania, and psychosis. Like digoxin, it has a narrow therapeutic index, and can cause serious harmful effects when ingested in toxic quantities. Ingestions can cause hemodynamic instability with bradycardia and hypotension, respiratory depression, miosis, and dizziness or ataxia. Lethargy has been reported in 70-80% of patients with clonidine ingestion.66,67
Conclusion
The ED clinician who encounters the lethargic child has a broad differential to contend with. After providing the appropriate resuscitative and supportive care, further ED management of the pediatric patient should be guided by an age-based approach with targeted laboratory and imaging studies.
References
1. Karthikeyan G, Premkumar K. Neonatal sepsis: Staphylococcus aureus as the predominant pathogen. Indian J Pediatrics 2001;68:715-717.
2. Ganatra HA, Stoll BJ, Zaidi AJ. International perspective on early-onset neonatal sepsis. Clinics in Perinatology 2010;37:501-523.
3. Mann K, Jackson MA. Meningitis. Pediatrics in Review 2008;29:417-430.
4. Caserta MT. (2009) Neonatal Bacterial Meningitis. Merck Manuals Online Medical Library for Health Professionals.
5. Curtis S, Stabart K, Vandermeer B, et al. Clinical features suggestive of meningitis in children: A systematic review of prospective data. Pediatrics 2010;126:952-960.
6. Chayavichitsilp P, et al. Herpes simplex. Pediatrics in Review 2009;30:119-130.
7. Donn SM, Banagale RC. Neonatal hyperammonemia. Pediatrics in Review 1984;5: 203-208.
8. Burton BK. Inborn errors of metabolism. In: Infancy: A Guide to Diagnosis. Pediatrics. 1998:102.
9. Ward JC. Inborn errors of metabolism of acute onset in infancy. Pediatrics in Review 1990;11:205-216.
10. Agus M. Endocrine Emergencies. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine, 6th Edition. Philadelphia: Lippincott, Williams, and Wilkins: 2010.
11. Rehman H. Methemoglobinemia. West J Med 2001;175:193-196.
12. Stevens MW, Henretig FM. Vomiting. In: Fleiserh GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine. Philadelphia: Lippincott Williams and Wilkins; 2010.
13. Godbole A, Concannon P, Glasson M. Instructive case: Intussusception presenting as profound lethargy. J Pediatr Child Health 2000;36:392-394.
14. Waseem M, Rosenburg HK. Intussusception. Pediatric Emerg Care 2008;24:793-800.
15. Applegate KE. Intussusception in children: Evidence-based diagnosis and treatment. Pediatric Radiology 2009;39(Suppl 2):S140-S143.
16. Yamamoto LG, et al. Stool appearance in intussusception: Assessing the value of the term "currant jelly." Am J Emerg Med 1997;15:293-298.
17. McCann T, Lin J. Gastrointestinal Emergencies. In: Crain EF, Gershel JC, eds. Clinical Manual of Emergency Pediatrics, 5th Edition. New York: Cambridge University Press: 2010.
18. Weihmiller SN, Buonomo C, Bachur R. Risk stratification of children being evaluated for intussusception. Pediatrics 2011;127:e296-e303.
19. Clayton KM. Focus on diagnosis: Pediatric abdominal imaging. Pediatrics in Review 2010;31:506-510.
20. Applegate K. Evidence-based diagnosis of malrotation and volvulus. Pediatric Radiology 2009;39:S161-S163.
21. Bachur RG. Abdominal Emergencies. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine. Philadelphia: Lippincott, Williams and Wilkins; 2006.
22. Selbst SM. The Septic-Appearing Infant. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine. Philadelphia: Lippincott, Williams, and Wilkins; 2006.
23. McCollough M, Sharieff GQ. Abdominal pain in children. Pediatric Clin North Am 2006;53:107-137.
24. Okada PJ, Hicks BA. Nontraumatic surgical emergencies. In: Fuchs S, et al, eds. APLS: The Pediatric Emergency Medicine Resource. Boston: Jones and Bartlett; 2006.
25. Centers for Disease Control and Prevention. Child Maltreatment: Facts at a Glance, N.C.f.I.P.a. Control, Editor. 2010: Washington, DC.
26. Bechtel K, Berger R. Inflicted traumatic brain injury: Making the diagnosis in the emergency department. Clin Pediatric Emerg Med 2006;7:138-142.
27. Asnes AG, Leventhal JM. Managing child abuse: General principles. Pediatrics in Review 2010;31:47-55.
28. Sirotnak AP. Grigsby T, Krugman RD. Physical abuse of children. Pediatrics in Review 2004;25:264-267.
29. Jenny C, et al. Analysis of missed cases of abusive head trauma. JAMA 1999;281:621-626.
30. Rindal K, Davis CO. Acute cardiovascular events in infants. Emerg Med Rep 2009;30:221-231.
31. Craig JE, et al. Fat necrosis after ice application for supraventricular tachycardia termination. J Pediatrics 1998;133:727.
32. Gewitz MH, Woolf PK. Cardiac Emergencies. In: Crain EF, Gershel JC, eds. Clinical Manual of Emergency Pediatrics, 5th edition. New York: Cambridge University Press; 2010.
33. Clemmons MR, Bell L. Infant botulism presenting with poor feeding and lethargy. Pediatric Emergency Care 2007;23:492-494.
34. Arnon SS, et al. Human botulism immune globulin for the treatment of infant botulism. N Engl J Med 2006;354:462-471.
35. Fogel N, Zimmerman D. Diabetic ketoacidosis in the ED. Clin Pediatric Emerg Med 2009;10:246-251.
36. Agus MS, Wolfsdorf JI. Diabetic ketoacidosis in children. Pediatric Clin North Am 2005;52:1147-1163.
37. Dunger DB, et al. ESPE/LWPES consensus statement on DKA in children and adolescents. Arch Dis Child 2004;89: 188-194.
38. Glaser N, et al. Risk factors for cerebral edema in children with diabetic ketoacidosis. The Pediatric Emergency Medicine Collaborative Research Committee of the AAP. N Engl J Med 2001;344:264-269.
39. Arit W, Allolio B. Adrenal insufficiency. Lancet 2003;361:1881-1893.
40. Antal Z, Zhou P. Congenital adrenal hyperplasia: Diagnosis, evaluation, and management. Pediatrics in Review 2009;30:e49-e57.
41. Shulman D, Palmert M, Kemp S. Adrenal insufficiency: Still a cause of morbidity and death in childhood. Pediatrics 2007;119:e484-e494.
42. Gupta A. Index of suspicion. Pediatrics in Review 2005;26:23-33.
43. Bechtel K, Iragorri S. Hepatosplenomegaly and reticulocytopenia as prominent features of atypical hemolytic uremic syndrome. Pediatric Emergency Care 2010;26:510-511.
44. Scheiring J, Andreoli SP, Zimmerhackl LB. Treatment and outcome of Shiga-toxin-associated hemolytic uremic syndrome (HUS). Pediatric Nephrology 2008;23:1749-1760.
45. Rivkin M, Volpe J. Strokes in children. Pediatrics in Review 1996;17:265-278.
46. Swischuk LE. Headache, increasing lethargy, and left-side weakness. Pediatric Emergency Care 2002;18:289-391.
47. Bockova J, Rigamoniti D. Intracranial empyema. Pediatric Infect Dis J 2000;19:735-737.
48. Blanchette V, Bolton-Maggs P. Childhood immune thrombocytopenic purpura: Diagnosis and management. Pediatric Clin North Am 2008;55(2).
49. Kim TY, et al. Signs and symptoms of cerebrospinal fluid shunt malfunction in the pediatric emergency department. Pediatric Emergency Care 2006;22:28-34.
50. Garton HJ, Kestle JR, Drake JM. Predicting shunt failure on the basis of clinical symptoms and signs in children. J Neurosurgery 2001;94:202-210.
51. Sainte-Rose C, Piatt JH, Pierre-Kahn A. Mechanical complications in shunts. Pediatric Neurosurgery 1992;17:2-9.
52. Mansfield RT. Severe traumatic brain injuries in children. Clin Pediatric Emerg Med 2007;8:156-164.
53. Lewis P, Glaser CA. Encephalitis. Pediatrics in Review 2005;26:353-363.
54. Tunkel AR, et al. The management of encephalitis: Clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis 2008;47:303-327.
55. Carstairs SD, Carstairs K. Emergency department presentation of acute disseminated encephalitis. Pediatric Emergency Care 2007;23:109-111.
56. Murthy SNK, et al. Acute disseminated encephalomyelitis in children. Pediatrics 2002;110:e21.
57. Olgun H, et al. Clinical, electrocardiographic, and laboratory findings in children with amitriptyline intoxication. Pediatric Emergency Care 2009;23:170-173.
58. Antia SX, Sholevar EH, Baron DA. Overdoses and ingestions of second-generation antipsychotics in children and adolescents. J Child Adolesc Psychopharmacology 2005;15:970-985.
59. Barry JD. Diagnosis and management of the poisoned child. Pediatric Annals 2005;34:937-946.
60. Fox,J, et al. Index of suspicion. Pediatrics in Review 2005;26:263-270.
61. Bruns JJ, Tarabar A. Anticholinergic toxicity. EMedicine.com. 2009.
62. Goldberg K, et al. Lethargy from an unsuspected culprit. Curr Opin Pediatrics 1999;11:28-30.
63. Osterhoudt K, et al. Toxicologic emergencies. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine, 6th edition. Lippincott, Williams, and Wilkins: Philadelphia; 2010.
64. Sutton T, Foster R, Liner S. Acute methanol ingestion. Pediatric Emergency Care 2002;18:360-363.
65. Anderson AC. Calcium-channel blocker overdose. Clin Pediatr Emerg Med 2005;6:109-115.
66. Klein-Schwartz W. Trends and toxic effects of pediatric clonidine exposures. Arch Pediatric Adolesc Med 2002;156:392-396.
67. Spiller HA, et al. Toxic clonidine ingestion in children. J Pediatrics 2005;146:263-266.
Stedman's medical dictionary defines lethargy as a "relatively mild impairment of consciousness resulting in reduced alertness and awareness; this condition has many causes but is ultimately due to generalized brain dysfunction."Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
