The Red Eye
The Red Eye
Author:
Pamela Arsove, MD, FACEP, Associate Residency Director, Department of Emergency Medicine, Long Island Jewish Medical Center, New Hyde Park, NY; Assistant Professor of Emergency Medicine, Hofstra North Shore – Long Island Jewish School of Medicine.
Peer Reviewer:
Jason Knight, MD, Vice Chair and Medical Director, Department of Emergency Medicine, Maricopa Medical Center, Phoenix, AZ.
Introduction
The number of adult ED visits for eye-related complaints is largely limited to data on eye injuries.1,2 According to the latest National Health Statistics Report, 1.2 % of all injury-related visits to emergency departments (EDs) in the United States in 2007 were for eye injuries.1 A recent analysis showed that infectious and non-infectious diseases of the eye accounted for 1.6% of ED pediatric visits in 2002.3 Given the diversity of non-traumatic ocular pathology, this likely represents a gross underestimation of the number of patients presenting with complaints related to the eye.
Most patients presenting to the ED with eye complaints have a red eye. The differential diagnosis includes a broad range of local infectious, inflammatory, and traumatic processes, as well as systemic diseases that affect the globe. Patients must be evaluated expeditiously if a potential ocular emergency exists, such as a chemical injury. Clues to potentially serious ocular pathology are eye pain, visual disturbance, eye trauma, or a patient in distress; these patients must be given a high triage priority. This article will discuss the more important inflammatory causes of red eye. Determining the exact etiology or causative agent may not be possible. In fact, many of the conditions discussed have wide overlap in symptoms, etiologic agents, and associated systemic diseases. In addition, certain disease states can present with a spectrum of ocular conditions. For example, systemic lupus erythematosus may present with conjunctivitis, scleritis, keratitis, or uveitis. Eye trauma, although an important consideration in the evaluation of red eye, will not be discussed in this article.
Ocular Anatomy and Pathophysiology
The eyeball is divided into three layers or "tunics": the outer fibrous tunic, the middle vascular tunic, and the inner neural tunic. The outer layer of the eyeball, the fibrous tunic, is a collagenous membrane that is composed of the sclera and the cornea.
The middle layer of the eye is the vascular tunic, which is composed of the choroid posteriorly and the iris and ciliary body anteriorly. The choroid is rich in vasculature and pigment. The ciliary body is composed of the ciliary processes that secrete aqueous humor and the ciliary muscle that attaches to the lens.
The retinal or nervous tunic forms the innermost layer of the eyeball and contains the photoreceptors.
Aqueous and Vitreous Humor. The vitreous humor is located in the posterior chamber and the aqueous humor is located in the anterior chamber. Small changes in the production or drainage of aqueous humor have a big effect on intraocular pressure. A significant increase in intraocular pressure can compress the optic nerve in the posterior portion of the eye and cause visual loss.
Initial Approach to the Patient with Red Eye
History. Important data include past ocular diseases or procedures, a complete medical history, and a history of prescription and non-prescription medication use. The ED physician should determine the duration of symptoms and the presence or absence of certain indicators of eye pathology such as pain, discharge, change in visual acuity, contact lens use, photosensitivity, trauma, or foreign body exposure.
Examination of the Eye. Visual acuity testing is done bilaterally and is generally done first. The patient should wear his or her corrective lenses if available. Pinhole visual acuity testing is performed if the patient's corrective lenses are unavailable or when visual acuity is abnormal to correct for possible refractive errors. Check the pupils for irregularity in size or reactivity and photophobia. The eye should then be examined from the outside in, starting with the lids and lashes. Evert the lid to look for a conjunctival foreign body. Perilimbic conjunctival injection ("ciliary flush") is indicative of more serious pathology such as acute iritis or acute glaucoma. The cornea should be examined for abrasions and ulcerations using fluorescein eye stain and a blue light source. A slit-lamp examination should be performed to magnify the external ocular structures and anterior chamber. The anterior chamber should be assessed for depth and the presence of inflammatory cells or blood. Unless a ruptured globe is present or suspected, ocular pressure should be measured on the anesthetized eye.
Simple causes of red eye are easily treated in the ED. Consult an ophthalmologist when serious complaints exist or if the diagnosis is uncertain and there are symptoms of more serious ocular pathology, as listed above or for any condition that warrants topical steroids.
Inflammatory and Infectious Causes of Red Eye
Conjunctivitis. Conjunctivitis, or inflammation of the conjunctiva, is the most common cause of red eye. Vascular dilatation, cellular infiltration, and exudation are the hallmark of the disease. The conjunctiva is divided into the bulbar conjunctiva, which covers the globe, and the tarsal conjunctiva, which lines the eyelids. Conjunctivitis may involve one or both of these structures.
The etiology of conjunctivitis is usually attributed to bacteria and viruses, but may also be due to allergens and other irritants, and less commonly to fungus, parasites, and systemic disease. For simplicity, conjunctivitis is often classified as infectious or non-infectious. Infectious causes are further subdivided into bacterial or viral, and non-infectious causes are subdivided into allergic and non-allergic.
The presenting complaint in conjunctivitis is acute, diffuse conjunctival injection that rapidly spreads from one eye to the other, and eye discharge. Eyelid edema may or may not be present. The diagnosis of simple conjunctivitis should be questioned if the patient presents with eye pain, photophobia, decreased visual acuity, an abnormally reactive pupil, increased intraocular pressure, corneal opacification, or localized conjunctival injection. These complaints should alert the physician to look for other more serious eye pathology. Overall, conjunctivitis is a benign, self-limited disease that rarely causes severe sequelae. It is a clinical diagnosis in most settings, and cultures are rarely taken. Distinguishing between the different types of conjunctivitis based on clinical presentation is not reliable.4
Infectious Conjunctivitis. Bacterial Conjunctivitis. Nearly 4 million cases of bacterial conjunctivitis were seen in the United States in 2005, with direct and indirect costs estimated at nearly $600 million.5 Because conjunctival cultures are rarely obtained, the true incidence of this disease may be misrepresented by available data. Disruption in the epithelial surface allows pathogens to infect the conjunctiva and cause an inflammatory reaction. Infection in surrounding structures, such as obstructed nasolacrimal ducts, pharyngitis, bacterial sinusitis, or otitis media, may spread to the conjunctiva. In addition, disorders of the lids or tear film production may inhibit the normal protective barrier and make the patient susceptible to infection. The clinical presentation usually begins with unilateral conjunctival injection, mucopurulent discharge, and eyelid edema that rapidly progresses to bilateral eye involvement in 1 to 2 days. Patients may experience a "gritty" sensation in their eyes, but true pain is unusual. Common pathogens are Staphylococcus aureus, Staphylococcus epidermidis, Haemophilus influenzae, Streptococcus pneumoniae, and Moraxella species.6 Different bacterial pathogens predominate in different age groups and with different acuity.
Neonatal conjunctivitis (ophthalmia neonatorum) occurs within the first 28 days of life. Many cases are acquired by passage through the birth canal. In the United States, it occurs in 1-2% of births and is most often caused by Chlamydia trachomatis, Staphylococcus aureus, Staphylococcus epidermidis, Escherichia coli, Neisseria gonorrhea, other gram-negative bacteria, and herpes simplex virus.7 Chlamydia trachomatis is the most common cause, but Neisseria gonorrhea is the most destructive and must be excluded in all cases. Neisseria conjunctivitis typically presents earlier, in the first 24-48 hours of life, while Chlamydia conjunctivitis manifests later, usually at 1-2 weeks. Neonates also get conjunctivitis as the result of a blocked tear duct. Because topical silver nitrate instillation at birth is no longer a common procedure, chemical conjunctivitis is less common.
Childhood conjunctivitis is predominantly caused by Streptococcus pneumoniae and Haemophilus influenzae. Large outbreaks may occur in daycare or school settings.
Adult conjunctivitis is usually caused by Staphylococcus species.
Acute conjunctivitis is the most common type of bacterial conjunctivitis and usually lasts less than 4 weeks. Chronic bacterial conjunctivitis is present if the infection lasts longer than 4 weeks. In this circumstance, symptoms are often mild but recurrent, and infection is usually caused by coagulase-positive and -negative staphylococci.8
Hyperacute bacterial conjunctivitis deserves special mention because it is an aggressive form of conjunctivitis that may progress rapidly to involve the cornea and intraocular cavities. It is primarily caused by Neisseria gonorrhea and is most often seen in neonates and young sexually active adults. In adults, a concurrent genital infection is usually present, but may be asymptomatic. Symptoms occur within 24 hours and present as a painful hyperemic conjunctiva, profuse mucopurulent discharge, marked conjunctival chemosis, and preauricular adenopathy.
Routine culture is not indicated for simple bacterial conjunctivitis. Cultures should be performed in neonates, immunocompromised patients, and in those with severe acute conjunctivitis or hyperacute conjunctivitis when gonorrhea is suspected. It should also be considered in chronic or recurrent conjunctivitis, or those cases not responding to treatment. Bacterial conjunctivitis has an extremely high rate of spontaneous remission and rarely causes serious ocular sequelae. Because bacterial conjunctivitis is highly contagious, local hygienic practices, such as frequent hand-washing, must be encouraged. It is established medical practice to treat acute bacterial conjunctivitis with broad-spectrum topical antibiotics, although there is significant literature to suggest that no treatment or delayed treatment yields similar outcomes.9-14 The choice of which agent to use does not usually affect outcome.15 Topical fluoroquinolones, trimethoprim/polymyxin B, sulfacetamide, erythromycin, and gentamicins are among the available preparations used. Topical azithromycin ophthalmic solution was approved in 2007 for use in bacterial conjunctivitis, with the advantage of once- to twice-daily dosing. Reports show similar efficacy to other currently available preparations.16,17
Adult gonococcal conjunctivitis is treated with single-dose intramuscular ceftriaxone, plus or minus topical antibacterials. In neonatal conjunctivitis, gonococcal infection is treated with single-dose ceftriaxone, 100 mg/kg IV or IM. Pediatric chlamydial infection is treated with erythromycin 50 mg/kg PO 4 times daily for 14 days.18 Topical medication alone is not effective in either gonococcal or chlamydia conjunctivitis of the newborn, and not necessary when systemic treatment is administered.
Viral Conjunctivitis. Viral conjunctivitis is classically described as having a watery discharge, an acute follicular conjunctival reaction, preauricular lymphadenopathy, and a longer clinical course than bacterial conjunctivitis, often lasting several weeks. Viral conjunctivitis presents as an isolated conjunctivitis or as part of a generalized viral syndrome, especially in children. While a number of viruses cause viral conjunctivitis, adenovirus is the most common. Other causes include enterovirus, varicella zoster virus, herpes simplex virus (HSV), Epstein-Barr virus, poxviruses, and coxsackievirus. Adenovirus and HSV are the two viruses discussed in detail here; the former because of its prevalence, and the latter because of its potential for serious complications if misdiagnosed.
Adenovirus has many different serotypes, 19 of which cause conjunctivitis.19 It is extremely contagious, usually through upper respiratory droplets or finger to eye contact during the first few days of symptoms. Adenovirus presents as one of several well-described syndromes: as a follicular conjunctivitis, as pharyngoconjunctival fever, or as epidemic keratoconjunctivitis. They are often indistinguishable early in the disease course. The mildest form is follicular conjunctivitis, which usually presents with mild, unilateral conjunctival injection, watery discharge, follicular and papillary conjunctival reactions, and preauricular lymphadenopathy on the involved side. As the name implies, pharyngoconjunctival fever presents as a febrile illness with pharyngitis and conjunctivitis, and is often confused with influenza. It is most commonly seen in school-age children. A follicular conjunctival reaction is seen with watery discharge, conjunctival injection, chemosis, and preauricular lymphadenopathy in the majority of cases.
Epidemic keratoconjunctivitis (EKC) is a more severe, contagious form of conjunctivitis that can involve the cornea as well. It is associated with sporadic outbreaks and persistent infection. EKC is usually a bilateral follicular conjunctivitis accompanied by pain and photophobia, characteristics less commonly seen in other viral conjunctivitis. Preauricular adenopathy and serous discharge are common, but the hallmark of the disease is the presence of conjunctival membranes (often termed "pseudomembranes"), epithelial and subepithelial corneal infiltrates, and preauricular lymphadenopathy. The membranes are most common on the tarsal plates and may be missed on examination. The corneal infiltrates seen in EKC are present in only about one-third of the patients.19 Central corneal infiltrates, or ones that coalesce, may reduce visual acuity.
HSV conjunctivitis (HSVC) occurs more often in children and adults and is most often caused by herpes simplex virus type 1 (HSV-1). When it occurs in neonates, it is usually caused by herpes simplex virus type 2 contracted during passage through the birth canal. It causes a papillary conjunctivitis that is often accompanied by eyelid infection, and is similar to adenovirus conjunctivitis.20 Signs that distinguish this infection from adenovirus are its tendency to be unilateral, the presence of a vesicular or ulcerating rash on the eyelids, and dendritic epithelial disruption of the cornea. It is the most common cause of blindness in third-world countries. There are about 500,000 cases diagnosed in the United States each year.21
Herpes zoster ophthalmicus (HZO, or "ocular shingles") is caused by the varicella zoster virus (VZV). Immunocompromised patients are at higher risk for developing this disease. Vesicles on the tip of the nose are a clue to this disorder and are the result of nasocilary (V1) nerve involvement (Hutchinson's sign). Since this nerve branch also innervates the cornea, patients with Hutchinson's sign and conjunctival injection must be suspected of having ocular involvement. A small number of patients may have eye involvement without the characteristic rash, and ocular involvement can occur with a rash anywhere on the forehead. While conjunctivitis itself is usually self-limiting, secondary bacterial superinfection may occur. Any patient who presents with herpetic vesicles in the distribution of the trigeminal nerve should undergo examination of the conjunctiva, cornea, and anterior chamber. In a recent study in Academic Emergency Medicine, eye redness in patients with HZO was found to be 100% predictive of moderate to severe eye disease. They recommend ophthalmology consultation in these patients, but not necessarily in patients without eye redness, even in the presence of Hutchinson's sign.22
Potential complications of adenovirus and herpes conjunctivitis include conjunctival scarring and involvement of the cornea or deeper ocular structures, with the possibility of vision loss. For adenovirus conjunctivitis, there is no effective antiviral medication and treatment is aimed at relief of symptoms. Cool compresses, vasoconstrictive eye drops, and artificial tears may ease symptoms. If bacterial superinfection exists, a broad-spectrum topical antibiotic should be prescribed. Careful hygiene is warranted to prevent the spread of infection to others. In severe cases, topical steroids may be prescribed to decrease symptoms and minimize scarring, but this should be done by the consulting ophthalmologist. In EKC with pseudomembrane formation, debridement of the membrane may provide increased comfort to the patient.
HSV conjunctivitis may be treated with antiviral medication to prevent infection from spreading to the cornea. Oral (acyclovir, valacyclovir, famciclovir) or topical (ganciclovir gel, trifluridine solution, vidarabine ointment) antivirals can be used. Topical antivirals may produce epithelial toxicity. They should be tapered rapidly as the patient improves, and be limited to a two-week course of treatment. Topical antibiotics are given to prevent bacterial infection. Neonatal ocular herpes infection requires prompt infectious disease consultation due to the high incidence of systemic and neurologic disease in this age group. In VZV infections, the oral antivirals listed previously are helpful to prevent ocular involvement if given to patients with cutaneous involvement only in the first three days from onset of the rash. Topical antibiotics may be given to prevent infection. Topical antivirals have not been shown to be effective once conjunctivitis is diagnosed. Topical steroids are indicated in the treatment of herpetic stromal keratitis, which involves the middle, transparent layer of the cornea most susceptible to scarring and subsequent visual loss. When this diagnosis is suspected, ophthalmology consultation is essential.
Non-infectious Conjunctivitis. Allergic Conjunctivitis. Four types of allergic reaction are described: seasonal and perennial allergic conjunctivitis (SAC and PAC), vernal keratoconjunctivitis (VKC), atopic keratoconjunctivitis (AKC), and giant papillary conjunctivitis (GPC). Each of these may present a similar initial picture, with bilateral findings of eyelid edema, conjunctival injection and edema, watery discharge, and itching or burning in the affected eyes. Purulent discharge is uncommon. The patient with allergic conjunctivitis will often have a personal or family history of atopy. The presence of nasal congestion, sneezing, and rhinorrhea with conjunctivitis (rhinoconjunctivitis), by history or on physical examination, is a clue to the allergic nature of the patient's symptoms.
Seasonal and Perennial Conjunctivitis. Ocular allergies are usually due to SAC and PAC. They are acute, mild, and uncomplicated. SAC predominates during the spring to fall months when airborne allergens are abundant. Patients with PAC have symptoms year-round. Environmental, animal, and household allergens are thought to trigger their symptoms.
Vernal and Atopic Keratoconjunc-tivitis. These two types of ocular allergy represent approximately 2% of allergic conjunctivitis.23 Chronic inflammation is believed to involve different cytokines and effector cells such as eosinophils and basophils.24,25 Patients frequently have a personal or family history of atopy, such as asthma, eczematous dermatitis, or allergic rhinitis. VKC and AKC are more chronic, recurrent, and potentially serious processes. The complications seen with both of these diseases are due to corneal involvement and the potential for visual loss secondary to corneal scarring. VKG predominates in childhood and has several unique physical exam findings to distinguish it from other forms of allergic conjunctivitis. It may involve the tarsal plate, where the papillae enlarge up to 7-8 mm, and are described as having a "cobblestone" appearance, one of the hallmarks of the disease. (See Figure 1.) The limbus may reveal yellow-appearing infiltrates and neovascularization. Itching is severe, and photophobia is common. It is not unusual for patients to have repeated episodes of conjunctivitis throughout the year. AKC occurs more commonly in men between the ages of 30 and 50. As opposed to VKC, the lower lid is more commonly involved.23 These more severe types of ocular allergy are more complicated to treat than simple allergic conjunctivitis (SAC and PAC) and usually involve the use of topical corticosteroids under the supervision of an ophthalmologist.
Figure 1: Large Papillae with a Classic "Cobblestone" Appearance Seen in Vernal Keratoconjunctivitis

Image used with permission from: Dr. Sunil Kumar.
Giant Cell Papillary Conjunctivitis. GPC is commonly classified with other allergic entities because it involves mast cell degranulation and elicits a type 1 hypersensitivity reaction. However, it differs in its immunologic features that are not truly typical of an allergic reaction. GPC is a condition caused by chronic micro-trauma, most commonly seen in contact lens wearers.26 Patients with GPC present with typical conjunctivitis symptoms. The examination reveals large papillae on the upper tarsal palpebrae and a stringy mucus discharge. There is no mortality with this disease, and treatment, although variable, generally yields favorable results. Temporarily stopping contact lens use is essential until GPC resolves. Topical combination antihistamine/mast cell stabilizers, nonsteroidal anti-inflammatory drugs (NSAIDs), and occasionally corticosteroids are used. A subset of patients will never tolerate contact lenses due to this entity.
Diagnosis of allergic conjunctivitis in the ED is based on the history and clinical presentation. Treatment of allergic conjunctivitis depends on the cause, but usually involves topical agents that have a rapid onset of action. Studies have shown that topical ocular treatment may be equal to nasal or oral agents for the ocular component of simple allergic conjunctivitis.27,28 Oral antihistamines are helpful, however, in cases of allergic rhinoconjunctivitis to relieve the nasal symptoms that accompany the ocular allergy. Popular treatments are the antihistamines and combination drops that have two or more antihistamine/mast cell stabilizer/anti-inflammatory components. Vasoconstrictive agents, mast cell inhibitors, NSAIDs, and, in complicated cases, corticosteroids, may be used alone or in combination. Topical vasoconstrictors may cause a rebound effect or a hypersensitivity reaction that should be taken into account when prescribing this medication, and should not be used as first-line agents. Table 1 provides a list of some common topical medications used for the various types of allergic conjunctivitis. Avoidance of known allergens is important when counseling these patients.
Table 1: Common Topical Medications for Allergic Conjunctivitis
|
Antihistamines |
Mast Cell Stabilizers |
NSAIDs |
Antihistamine/Mast Cell Stabilizer Combination |
Corticosteroids |
|
Selective histamine (H1) receptor antagonists |
Stops the release of inflammatory mediators from the mast cell |
Inhibit the production of prostaglandins and thromboxane |
See the individual properties of each. In addition, some have anti-inflammatory properties. |
Work intracellularly to inhibit production of pro-inflammatory mediators |
|
Antazoline 0.05% |
Alamast 0.1% |
Ketorolac 0.5% |
Olopatadine 0.01% |
Loteprednol etabonate 0.5% |
|
Azelastine 0.05% |
Opticrom 4% |
Diclofenac 0.1% |
Ketotifen 0.025% |
Prednisolone acetate 1.0% |
Chemical Conjunctivitis. The normal pH of the eye is approximately 7.0-7.4. Acids and alkali can seriously damage the conjunctiva and deeper structures of the eye. Ocular burns occur in up to 18% of patients who present with eye trauma29; the majority of these are from chemical burns. Chemical burns can denature and coagulate eye proteins and cause ischemia to vascular structures. Patients with chemical exposure to the eye should undergo immediate copious irrigation prior to a complete history and physical examination. The determining factor in morbidity from acid or alkali burns to the eye is the duration of exposure and the pH of the offending agent. Alkali burns are lipophilic and penetrate the eye quickly, causing deep necrosis, a condition termed "liquefactive" necrosis. If the pH is higher than 12, irreversible damage will likely occur.
Acids produce a coagulation necrosis. They denature proteins, causing eschar formation that functions as a barrier to further acid penetration, and a resultant superficial burn. The exception to the superficial nature of acid burns is hydrofluoric acid, which is a weak acid with rapid penetration into the cornea, causing a liquefactive necrosis injury. Treatment of acid or alkali burns in the ED involves prompt and copious irrigation to return the eye to a pH of about 7.4. A topical anesthetic applied prior to irrigation helps in pain management. A Morgan lens can be used to deliver low-pressure irrigation using 1 to 2 liters of irrigation solution (tap water is acceptable) over 30 to 60 minutes, at which time the pH is checked using litmus paper. The pH should be rechecked 30 to 60 minutes after an optimal pH is reached, and irrigation restarted if necessary. Ophthalmology should be consulted if there is significant injury to the cornea or sclera.
Corneal Abrasion and Corneal Ulcer
Keratitis refers to inflammation of the cornea and includes both corneal abrasion and corneal ulcer. Corneal abrasions are scratches to the superficial layers of the cornea and usually have a good prognosis with no visual loss. When injury or inflammation involve the deeper layers of the cornea, it is termed corneal ulcer, a more serious process that may cause scar formation and resultant visual loss if disruption occurs in the visual field. In severe cases, corneal perforation may occur. In areas of the developing world, corneal ulcers are a major cause of blindness.30 Predisposition for both of these processes occurs in individuals with conditions that cause dry eyes or decreased corneal sensation. Examples of this include patients with Bell's palsy and incomplete eyelid closure, or patients with corneal viral infections or chemical burns causing decreased corneal sensation.
Infection is the leading cause of corneal ulceration, and often complicates non-infectious etiologies as well. Bacteria, viruses, fungi, and amoeba are common causes and, with the exception of viruses, usually invade a traumatized cornea. In some cases, the trauma is not apparent, as seen with contact lens use. Bacteria are the most common infectious cause of corneal ulcers. In the United States, the most common bacteria are Pseudomonas aeruginosa and Staphylococcus aureus.31 In addition, immune-related conditions such as Lyme disease may localize to the eye as a non-infectious corneal inflammation, or present with ocular complaints as part of a systemic process. Risk factors for corneal ulcer include contact lens use, corticosteroid eye drop use, immunocompromised states, eye trauma, and warm climates.
Unlike conjunctivitis, patients typically complain of eye pain and a foreign body sensation. They usually present with blurry vision, photophobia, and excessive tearing. The physical exam reveals these findings, along with conjunctival injection or ciliary flush. Miosis, chemosis, and lid edema may be present. The physical exam must include upper lid eversion to detect and remove any foreign bodies that may be present. A defect in the surface of the cornea can be seen with the slit lamp and confirmed with fluorescence testing. Corneal abrasion and ulcer can be differentiated by the depth and color of the lesion. Corneal abrasions appear clear on slit lamp examination. In corneal ulcer, opacification of the cornea occurs and appears as a white opacity often seen on simple visual inspection. (See Figure 2.) Slit lamp confirms the finding. If herpetic keratitis is present, classic dendrite formation is seen with florescence application.
Figure 2: Corneal Ulcer Appearing as a White Opacification on the Surface of the Eye
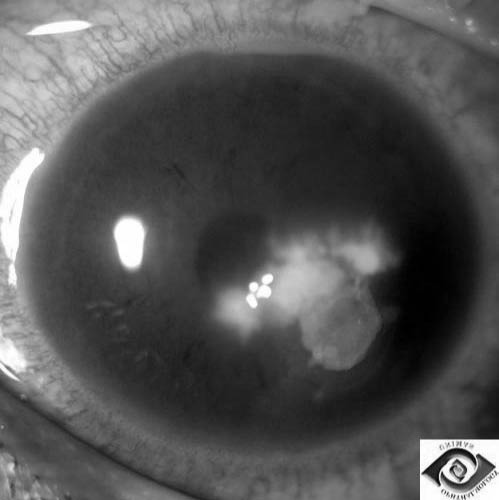
Reproduced with permission from: Dr. Chua Chung Nen, Department of Ophthalmology, UNIMAS.
The physician may encounter several distinct types of keratitis:
UV keratitis occurs from exposure to natural or artificial ultraviolet light. It is seen in welders, people with exposure to bright sunlight or reflected light off snow or sand, and in patients using tanning salons without protective eyewear. Symptoms are usually delayed for hours after exposure. It causes a painful punctuate keratitis, often on the inferior portion of the cornea, which appears as small pinpoint defects on fluorescein examination.
Herpes keratitis may occur with HSV (usually type 1) or varicella-zoster virus. It is recognized by a classic dendritic pattern on fluorescein staining of the eye.
Contact lens acute red eye (CLARE) is caused by protein or bacteria deposits on the contact lens that inflame the cornea. It is more common in extended-use lenses. Gram-negative bacteria (Pseudomonas, Serratia, and Haemophilus influenzae) are isolated.
Diffuse lamellar keratitis is a complication of Lasik surgery seen most commonly the day after surgery, but can occur within the first week. Sterile infiltrates elicit an inflammatory reaction in the cornea around the surgical incision site. Because it may be difficult for the ED physician to distinguish this entity from infectious keratitis, an ophthalmologist should be involved in the care of these patients.
Because the etiology is often not initially known, and to prevent bacterial super infection, topical antibiotics are usually given. Pain may be treated with oral medication. In corneal ulcers, topical NSAIDs have been used with good results.32 Cycloplegics may alleviate ciliary spasm. For viral keratitis, topical or oral antivirals such as acyclovir, and topical or oral steroids should be considered. Fungal keratitis is a contraindication to topical corticosteroid use. Ophthalmology should be consulted when a corneal ulcer is present and topical steroids are prescribed.
Scleritis/Episcleritis
Inflammation of the sclera may occur in the more superficial episclera, which lies just beneath the conjunctiva, or in the adjacent inner layer of the sclera. These two processes are termed episcleritis and scleritis, respectively, and differ significantly in severity and potential for visual loss. They are relatively uncommon disorders.
Episcleritis is a benign disease affecting the external portion of the eye and the superficial vascular plexus of the sclera. Its exact pathogenesis is unknown. Like other forms of inflammatory eye disease, it has been linked to certain diseases, such as systemic lupus, rheumatoid arthritis, and inflammatory bowel disease,33-35 which occur in a minority of patients. The majority of cases occur in young to middle-aged females.36 Episcleritis has two clinical presentations: simple and nodular. Usually episcleral hyperemia is localized, often with inflammation of the overlying bulbar conjunctiva. Nodular episcleritis presents with a focal nodule surrounded by hyperemia. Unlike simple episcleritis, which usually resolves spontaneously in several weeks, nodular episcleritis is slower to clear and is more likely to have recurrence. Both can present with acute, focal, or generalized hyperemia and mild eye discomfort. Symptoms may be bilateral, and visual acuity is unaffected. It is differentiated from conjunctivitis by its focality and lack of discharge, although tearing is common. On slit lamp examination, there is episcleral vascular congestion and edema of the surrounding episcleral tissue. A localized whitish-appearing nodule may be seen within the area of hyperemia. Uveitis and keratitis may complicate a minority of cases. Treatment is usually supportive. Topical corticosteroids, on a limited basis, may be prescribed to decrease inflammation in the extremely symptomatic patient. They are effective in treating episcleritis, but the risk of their use should be considered in treating this relatively benign disease. Topical NSAIDs have been suggested, but are of questionable efficacy.37 Oral NSAIDs may also be used. This condition may be recurrent, but does not progress to scleritis or cause visual loss.
Scleritis. Scleritis a more serious disease and should be approached by the ED physician as an ocular emergency. An immune-complex vasculitis-type reaction may account for the pathogenesis of this disease.38 In up to 50% of patients, it occurs in conjunction with an autoimmune systemic inflammatory disease.39 Rheumatoid arthritis and Wegener's granulomatosis are the most frequently associated autoimmune diseases.40 In a minority of patients, it is caused by infection, as a complication from ocular surgery, a medication reaction, or secondary to malignancy. Scleritis involves the anterior or posterior portion of the eye. When anterior, the inflamed scleral structures are visible on exam, but when the disease is posterior, the ocular changes are hidden from view.
There are five clinically recognized subtypes in this disease: four anterior and one posterior. Approximately 90% of cases are of the anterior type.41 Anterior scleritis is either diffuse, nodular, necrotizing with inflammation, or necrotizing without inflammation (scleromalacia perforans). The most common subtypes are anterior diffuse and anterior nodular. Anterior necrotizing scleritis with inflammation is not common, but is the most severe form of the disease and the most likely to result in loss of vision. Posterior scleritis occurs in 2-12% of cases.42 The patient usually presents with a normally appearing painful eye and, therefore, the diagnosis is often missed. Eye pain and a decrease in visual acuity should prompt the clinician to consider this diagnosis. It is associated with anterior scleritis in over 50% of cases, at which point sclera inflammation is visible. Complications include uveitis, retinal detachment, retinal hemorrhage, macular edema, cataracts, and ophthalmoplegia.
Aside from sclera vascular injection, the prominent symptom in scleritis is pain, which may be orbital or periorbital. Patients typically present with gradual onset of symptoms. Reduced visual acuity may be present. Examination is variable depending on the subtype but classically reveals tenderness to palpation, and focal or diffuse hyperemia, which is described as having a violaceous hue due to the depth of the involved blood vessels. Proptosis and lid edema may be present. There may be an associated underlying systemic disorder, which should increase the clinician's suspicion for this diagnosis.
It can be difficult to differentiate episcleritis from scleritis. On exam, episcleral vessels will move when prodded with a cotton-tipped swab. Also, if you instill a 10% solution of pilocarpine, episcleral vessels will blanch, while deeper sclera vessels will not. Imaging modalities useful in diagnosing scleritis include b-mode ultrasonography and CT scanning,40 which are helpful in elucidating disease at the back of the eye. Treatment of non-infectious scleritis is always systemic. Non-necrotizing scleritis is treated with oral NSAIDs, oral steroids, or a combination of both. Peri-oribital or subconjunctival steroid injections have been used if necrotizing disease is present but are beyond the scope of ED practice. In necrotizing scleritis, oral steroids, immunosuppressants such as cyclophosphamide or cyclosporine, or a combination of both is used. In posterior scleritis, the underlying cause must be treated along with oral NSAIDs, steroids, or both. In infectious scleritis, antimicrobial treatment is given systemically with or without topical antimicrobials. Because of the high incidence of associated systemic disease, patients with scleritis should be referred to a primary practitioner for a thorough medical evaluation.
Uveitis/Iritis
The uvea is the portion of the eye that includes the iris and the ciliary body anteriorly and the choroid posteriorly. Inflammation of the anterior uveal tract may involve the iris, a condition known as uveitis or iritis. If it involves the iris and ciliary body, it is called iridocyclitis. When posterior uveal structures are involved in inflammatory processes, the condition has multiple terms, including pars planitis, intermediate uveitis, retinitis, or chorioretinitis. Panuveitis is inflammation of all uveal structures. The pathogenesis of uveitis is unknown, but it is strongly associated with a variety of systemic disorders. Causes include infectious, idiopathic, and localized or systemic autoimmune disorders. In addition, the term "masquerade syndrome" is used to refer to disorders, often malignant, that cause similar symptoms to uveitis, although they do not represent the true disease. Occasionally, trauma and drugs may also cause this disease. Factors in the history that should heighten the clinician's suspicion for uveitis include a past or current history of autoimmune disorders, HIV disease, recent ocular surgery, or diseases such as syphilis, tuberculosis, toxoplasmosis, sarcoidosis, and Lyme disease.
Uveitis may be classified as anterior or posterior, or by clinical course as acute, recurrent, or chronic.44 In the Western world, the most common form of uveitis is acute, anterior, nongranulomatous disease associated with HLA-B27 positive patients.45,46 In patients who have symptomatic systemic disease, most are male with a spondyloarthropathy such as ankylosing spondylitis or reactive arthritis.
Figure 3: Keratic Precipitates Seen on the Cornea in Anterior Uveitis
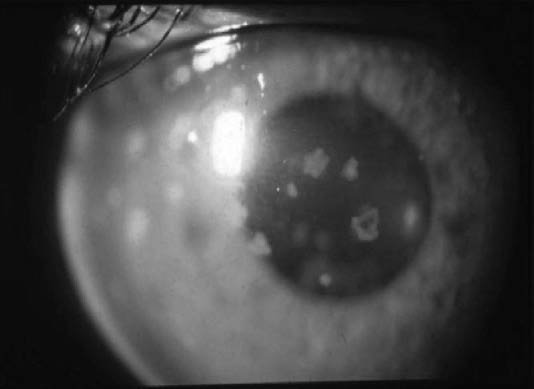
Copyright © Indian Journal of Ophthalmology, reproduced with permission from Dr. Rupish V. Agrawal.
Anterior uveitis commonly presents with pain that occurs over hours to days, ciliary flush, photophobia, miosis, and variable visual loss. Posterior uveitis patients do not typically have a red eye or pain but variable degrees of visual loss or the perception of "floaters" in the visual field. Slit lamp exam may show white cell deposits on the cornea, termed "keratic precipitates." (See Figure 3.) The hallmark of uveitis is flare and cells in the anterior chamber on slit lamp exam. Inflammatory white blood cells are directly visualized, while the "flare," a foggy haze, results from proteinaceous material leaking out of inflamed vasculature into the anterior chamber. If white cells settle in a meniscus-like fashion in the anterior chamber, it is termed a hypopyon. (See Figure 4.) Posterior uveitis requires specialized equipment to visualize the back of the eye, which is out of the scope of the ED physician's practice.
Figure 4: Classic Appearance of a Hypopyon in the Anterior Chamber
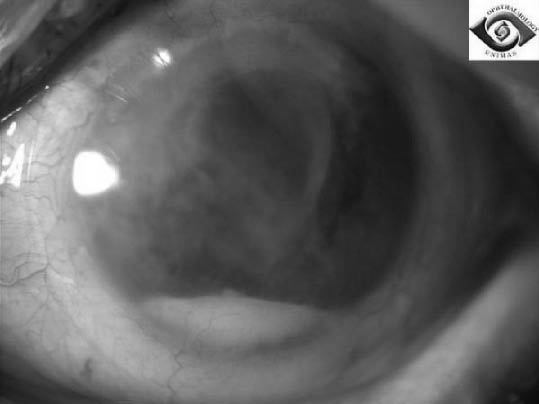
Reproduced with permission from: Dr. Chua Chung Nen, Department of Ophthalmology, UNIMAS.
The goal in managing anterior uveitis is to relieve pain and inflammation and to prevent possible complications such as glaucoma, synechiae formation, and visual loss. The mainstay in treatment of non-infectious uveitis is topical corticosteroids, often with a topical mydriatic agent, such as atropine, that helps relieve spasm and prevent posterior synechiae from forming. If topical steroid treatment fails, systemic or peri-ocular steroids can be given. When posterior uveitis is diagnosed, systemic corticosteroids are used. Recent developments in treatment of posterior uveitis have focused on local, intraocular corticosteroids and surgically implanted devices that deliver steroids in a sustained-release manner.47 In infectious uveitis, the underlying cause is treated. The main causes of visual loss are from macular edema, glaucoma, and cataract formation, which are usually seen in patients with chronic disease.
Acute Angle Closure Glaucoma
Acute angle closure glaucoma (AACG) is a true ocular emergency that requires emergent ophthalmology consultation in the ED. It occurs when there is a sudden, rapid increase in intraocular pressure due to closure of the anterior chamber angle. Under normal circumstances, aqueous humor drains out of the anterior chamber of the eye via the trabecular meshwork located at the angle of the anterior chamber in Schlemm's canal. Anything that blocks this angle will prevent drainage of aqueous humor and increase the normal pressure inside the eye. There are two main types of AACG: Primary angle closure and secondary angle closure. In primary angle closure, patients have anatomic abnormalities that account for angle narrowing or closure. In secondary angle closure, a secondary process such as hemorrhage or tumor is responsible for mechanically deforming the structures at the angle of the anterior chamber to cause its closure. Overall, the most common cause of obstruction occurs when the peripheral iris is pushed up against the angle and becomes a mechanical barrier to normal drainage. In this situation, physical contact between the iris and lens occurs, which is termed "pupillary block."
The highest rates are seen among patients of Eskimo or Asian decent.48 Risk factors include farsighted individuals who have inherently shallow anterior chambers and narrow anterior chamber angles. Because hyperopia (farsightedness) occurs with advancing age, it is more common in the elderly. In addition, with advanced age comes enlargement of the lens, which pushes the iris forward and predisposes individuals to this disease. Other risk factors include a family history of AACG. Medications, used topically or systemically, can also contribute to AAGC. Adrenergic agents, anticholinergic and cholinergic medication, and sulfa-based drugs have been implicated.49 The mechanisms in drug-induced AACG are pupillary dilatation, anterior displacement of the lens-iris diaphragm, and ciliary body edema.50 Moving from a dark environment to a light one, which is classically described as coming out of a dark movie theatre into bright light, can acutely dilate the pupil and precipitate an attack. Although unusual, bilateral AACG can occur and is often attributed to underlying systemic disease.51-53
Table 2: Common Medications in Acute Angle Closure Glaucoma
|
Class of Medication |
Mode of Action |
Representative Drug |
Initial Dose |
|
Note: Adult doses are listed; AH = aqueous humor |
|||
|
Alpha-adrenergic agonists |
↓ AH production ↓ Resistance to aqueous outflow |
Apraclonidine |
1 drop of 0.5 or 1% |
|
Beta blockers |
↓ AH production |
Timolol maleate |
1 drop of 0.25 or 0.5% |
|
Parasympathomimetics |
Contracts ciliary muscle: miosis ↑ Outflow of AH |
Pilocarpine |
1-2 drops |
|
Prostaglandin analogs |
↑ Outflow of AH |
Latanoprost |
|
|
Carbonic anhydrase inhibitors |
↓ AH production |
Acetazolamide |
500 mg IV, then 500 mg PO |
|
Steroids |
↓ Intraocular inflammation |
Prednisolone |
1-2 drops |
|
Hyperosmotic medication |
Osmotic diuresis ↓ Vitreous volume |
Mannitol Isosorbide |
1.5-2 g/kg IV (20% solution) over 30 minutes 1.5 g/kg PO |
|
Antiemetic |
↓ Nausea |
Ondansetron |
4 mg IV or PO |
|
Analgesic |
↓ Pain |
NSAID, narcotic |
|
Symptoms of AACG include eye pain, headache, nausea, blurred vision, tearing, and the appearance of halos around lights (a result of corneal edema). Abdominal pain, nausea, and vomiting have also been reported. The exam may reveal ciliary flush, generalized conjunctival hyperemia, a fixed or sluggish mid-dilated pupil, corneal edema, and a shallow anterior chamber. Visual acuity is diminished and intraocular pressure is elevated. In normal eyes, intraocular pressure ranges 8-21 mmHg. When an eye suffers from acute angle closure, the pressure often rises above 30 mmHg. Prophylactic treatment is often initiated in the unaffected eye if a narrow angle is seen on slit lamp examination. Rarely, the process resolves spontaneously, but recurrences are common. If untreated, optic neuropathy rapidly ensues and permanent vision loss may occur in hours to days. Emergent treatment, therefore, is aimed at initially lowering the intraocular pressure to below 40 mmHg as quickly as possible. Examples of common medications to reduce intraocular pressure, reverse angle closure, and treat the systemic symptoms of AACG are listed in Table 2. A common approach is to give topical beta-blockers, alpha-adrenergic blockers, or prostaglandin inhibitors and oral or intravenous acetazolamide to initially decrease intraocular pressure. Pressure should be rechecked one hour later. Once intraocular pressure is lowered, pilocarpine is given to vasoconstrict the pupil in hopes of widening the anterior chamber angle. If pilocarpine is given before intraocular pressure is reduced, it is generally ineffective because elevated intraocular pressures cause paralysis of the iris. Medication to dilate the eye must be avoided, as this will exacerbate AACG. If these modalities are unsuccessful in decreasing intraocular pressure, laser peripheral iridotomy is performed by an experienced ophthalmologist.
References
1. Niska R, Bhuiya F, Xu J. National Hospital Ambulatory Medical Care Survey: 2007 Emergency Department Summary. National Health Statistics Reports 2010;26:1-32.
2. McGwin G, Owsley C. Incidence of emergency department–treated eye injury in the United States. Arch Ophthalmol 2005;123:662-666.
3. Alessandrini E, Alpern E, Chamberlain J, et al. A new diagnosis grouping system for child emergency department visits. Acad Emerg Med 2010;17:204-213.
4. Rietveld R, Van Weert H, Ter Riet G, et al. Diagnostic impact of signs and symptoms in acute infectious conjunctivitis: Systematic literature search. BMJ 2003;327:789.
5. Smith A, Waycaster C. Estimate of the direct and indirect annual cost of bacterial conjunctivitis in the United States. BMC Ophthalmology 2009:9:13.
6. Jeng B. Bacterial conjunctivitis: A review for internists. Cleve Clin J Med 2008;75:507-512.
7. Palafox SKV, Jasper S, Tauber AD, Foster SC. Ophthalmia Neonatorum. J Clinic Experiment Ophthalmol 2010;1:119. doi:10.4172/2155-9570.1000119.
8. Hovding G. Acute bacterial conjunctivitis. Acta Ophthalmologica 2008;86:5-17.
9. Hutnik C, Thomas M. Evidence-based treatment of acute infective conjunctivitis Breaking the Cycle of Antibiotic Prescribing. Can Fam Physician 2009;55:1071-1075.
10. Everitt HA, Little PS, Smith PW. A randomised controlled trial of management strategies for acute infective conjunctivitis in general practice. BMJ 2006;333:321–324.
11. Rose PW, Harnden A, Brueggemann AB, et al. Chloramphenicol treatment for acute infective conjunctivitis in children in primary care: A randomised double-blind placebo-controlled study. Lancet 2005;366:37–43.
12. Rietveld RP, Ter Riet G, Bindels PJE, et al. The treatment of acute infectious conjunctivitis with fusidic acid: A randomised controlled trial. Br J General Pract 2005:55:924–930.
13. Rose P. Management strategies for acute infective conjunctivitis in primary care: A systematic review. Expert Opin Pharmacother 2007;8:1903–21.
14. Davis H, et al. Relative impact of clinical evidence and over-the-counter prescribing on topical antibiotic use for acute conjunctivitis. Br J Gen Pract 2009;59:897.
15. Hutnik C, Mohammad-Shahi M. Bacterial conjunctivitis. Clin Ophthalmol 2010;4:1451–1457.
16. Abelson M, Protzko E, Shapiro A, et al. A randomized trial assessing the clinical efficacy and microbial eradication of 1% azithromycin ophthalmic solution vs tobramycin in adult and pediatric subjects with bacterial conjunctivitis. Clin Ophthalmol 2007;1:177-182.
17. Protzko E, Bowman L, Abelson M, et al. Phase 3 safety comparisons for 1.0% azithromycin in polymeric mucoadhesive eye drops versus 0.3% tobramycin eye drops for bacterial conjunctivitis. Invest Ophthalmol Vis Sci 2007;48:3425-3429.
18. Workowski KA, Berman S. Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep 2010;59(RR-12):1.
19. Aoki K, Kaneko H, Kitaichi N, et al. Clinical features of adenoviral conjunctivitis at the early stage of infection. Japanese Journal of Ophthalmology 2011;55:11-15.
20. Uchio E, Takeuchi S, Itoh N, et al. Clinical and epidemiological features of acute follicular conjunctivitis with special reference to that caused by herpes simplex virus type 1. Br J Ophthalmol 2000;84:968–972.
21. Liesegang TJ. Herpes simplex virus epidemiology and ocular importance. Cornea 2001;20:1-13.
22. Adam RS, Vale N, Bona MD, et al. Triaging herpes zoster ophthalmicus patients in the emergency department: Do all patients require referral? Acad Emerg Med 2010;17:1183-1188.
23. Guglielmetti S, Dart J, Calder V. Atopic keratoconjunctivitis and atopic dermatitis. Curr Opin Allergy Clin Immunol 2010;10:478–485.
24. Kumar S. Vernal keratoconjunctivitis: A major review. Acta Ophthalmologica 2009;87:133–147.
25. Pokharel S, Shah DN, Joshi SN, et al. Tearfilm immunoglobulin E (IgE) level in vernal keratoconjunctivitis by ELISA. KUMJ 2009;7:104-108.
26. Forister JF, Forister EF, Yeung KK, et al. Prevalence of contact lens-related complications: UCLA contact lens study. Eye Contact Lens 2009;3:176-180.
27. Spangler D, Abelson M, Ober A, et al. Randomized, double-masked comparison of olopatadine ophthalmic solution, mometasone furoate monohydrate nasal spray, and fexofenadine hydrochloride tablets using the conjunctival and nasal allergen challenge models. Clin Ther 2003;25:2245-2267.
28. Crampton HJ. Comparison of ketotifen fumarate ophthalmic solution alone, desloratadine alone, and their combination for inhibition of the signs and symptoms of seasonal allergic rhinoconjunctivitis in the conjunctival allergen challenge model: A double-masked, placebo- and active-controlled trial. Clin Ther 2003;25:1975-1987.
29. Merle H, Gerard M, Schrage N. Ocular burns. J Fr Ophtalmol 2008;3:723-734.
30. Singh SK. Corneal ulcer. Nepal J Ophthalmol 2011;3:1-2.
31. Marquart M. Animal models of bacterial keratitis. J Biomed Biotechnol 2011;2011:680642.
32. Schechter B, Trattler W. Efficacy and safety of bromfenac for the treatment of corneal ulcer pain. Adv Ther 2010;27:,756-761.
33. Taxiarchis F, Konstantinos K, Kitsanou M, et al. Spectrum and frequency of ophthalmologic manifestations in patients with inflammatory bowel disease: A prospective single-center study. Inflamm Bowel Dis 2009;15:29-34
34. Hochberg M, Johnston S, John A. The incidence and prevalence of extra-articular and systemic manifestations in a cohort of newly-diagnosed patients with rheumatoid arthritis between 1999 and 2006. Curr Med Res Opin 2008;24:469-480.
35. Pavesio C, Meier F. Systemic disorders associated with episcleritis and scleritis. Curr Opin Ophthalmol 2001;12:471-478.
36. Quinn C, Skorin L. Episcleritis. In: Pine J, Dernoski N, Gunning F, eds. Ocular Therapeutics Handbook, 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005: 272-274.
37. Williams CP, Browning AC, Sleep TJ, et al. A randomised, double-blind trial of topical ketorolac vs. artificial tears for the treatment of episcleritis. Eye 2005;19:739-742.
38. Fong LP, Sainz de la Maza M, Rice BA, et al. Immunopathology of scleritis. Ophthalmology 1991;98:472.
39. Smith JR, Mackensen F, Rosenbaum J. Therapy insight: Scleritis and its relationship to systemic autoimmune disease. Nat Clin Pract Rheumatol 2007;3:219-226.
40. Akpek E, Thomas J, Qazi F, et al. Evaluation of patients with scleritis for systemic disease. Ophthalmology 2004;111:501-506.
41. Watkins A, Kempen J, Dongsoek C, et al. Ocular disease in patients with ANCA-positive vasculitis. J Ocul Biol Dis Infor 2010;3:12–19.
42. Galor A, Thorne J. Scleritis and peripheral ulcerative keratitis. Rheum Dis Clin North Am 2007;33:835-854.
43. Platnik J, Crum A, Soohoo S, et al. The globe: Infection, inflammation, and systemic disease. Semin Ultrasound CT MR 2011;32:38-50.
44. Agrawal R, Murthy S, Sangwan V, et al. Current approach in diagnosis and management of anterior uveitis. Indian J Ophthalmol 2010;58:11–19.
45. Monner D, Breban M, Hundry C, et al. Ophthalmic findings and frequency of extraocular manifestations in patients with HLA-B27 uveitis: A study of 175 cases. Ophthamology 2004;111:802-809.
46. Wakefield D, Chang J, Amjadi S, et al. What is new HLA-B27 acute anterior uveitis? Ocul Immunol Inflamm 2011;19:139-144.
47. Taylor S, Hazlita I, Lavnish J, et al. New developments in corticosteroid therapy for uveitis. Ophthalmologica 2010;224:46-53.
48. Vajaranant TS, Nayak S, Wilensky, J, et al. Gender and glaucoma: What we know and what we need to know. Curr Opin Ophthalmol 2010;21:91-99.
49. Razeghinejad MR, Myers J, Katz L. Iatrogenic glaucoma secondary to medications. Am J Med 2011;124:20-25.
50. Tripathi RC, Tripathi BJ, Haggerty C. Drug-induced glaucomas: Mechanism and management. Drug Saf 2003;26:749–767.
51. Cristol S, Baumblatt J, Icasiano E, et al. Bilateral acute angle-closure associated with systemic lymphoma: A report of 2 cases. J Glaucoma 2011;20:115-117.
52. Baxter J, Alexander P, Maharajan VS. Bilateral, acute angle-closure glaucoma associated with Guillain-Barré syndrome variant. BMJ Case Reports 2010;doi:10.1136/bcr.11.2009.2487.
53. Hunter T, Chong G, Asrani S, et al. Simultaneous bilateral angle closure glaucoma in a patient with giant cell arteritis. J Glaucoma 2010;19:149-150.
The number of adult ED visits for eye-related complaints is largely limited to data on eye injuries.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
