Complications of Tubes and Lines: Part II
Complications of Tubes and Lines: Part II
Authors:
Charlotte Derr, MD, RDMS, FACEP, Associate Program Director, Ultrasound Fellowship Director, University of South Florida Emergency Medicine Residency Program, Tampa.
Nathaniel Ronning, MD, University of South Florida Emergency Medicine Residency Program, Tampa.
Peer Reviewer:
Eric Gross, MD, Hennepin County Medical Center, Associate Professor of Emergency Medicine, University of Minnesota Medical School, St. Paul.
This is the second part of our two-part series on complications of tubes and lines. This issue deals with nephrostomy and enterostomy tubes and urinary catheters. It offers straightforward advice about these frequent problems.
Sandra M. Schneider, MD, FACEP, Editor
Nephrostomy Tubes
Case. A 57-year-old man presents with complaints of decreased urine output from his right-sided nephrostomy tube. He reports normal urine output through micturition and is well-appearing and afebrile with normal vital signs. How will you proceed in evaluating the possible causes of this man's chief complaint and provide appropriate treatment?
Nephrostomy tubes are typically placed transcutaneously by an interventional radiologist under ultrasonographic, fluoroscopic, or CT guidance, or by a surgeon during open surgery. Indications for placement include decompression of an obstructive uropathy, a hole in the ureter or urinary bladder, access to the renal pelvis for a procedure (e.g., antegrade stent insertion), or in preparation for surgery (e.g., removal of a large renal stone). Complications due to the insertion of these devices include retroperitoneal hemorrhage (typically a procedural complication and less likely to be encountered by the emergency physician), obstruction, dislodgement, hematuria, infection/sepsis, and pain. Decreased urine output, hematuria, and tube dislodgement represent the triad of complications most commonly encountered in the emergency department.
Decreased Nephrostomy Output. Decreased urine output in patients with adequate renal perfusion and adequate intravascular volume may be the result of tube malfunction, occlusion, human error (stopcock in the off position), or dislodgement. The patient presenting with decreased nephrostomy output should initially be evaluated for any potential causes of reduced renal perfusion or intrinsic renal diseases, such as acute tubular necrosis, interstitial nephritis, etc. Further, the physician should attempt to ascertain the patient's nephrostomy output, keeping in mind that normal output is typically regarded as greater than 30 mL/hr (total urine output) in an average-sized adult. If urine output through the nephrostomy is determined to be inadequate, further evaluation of the nephrostomy tube and its components is indicated.
Initially, inspect the tubing and stopcock to ensure that the stopcock is in the proper position to allow flow and that there are no obvious kinks, clogs, or encrustations in the tubing itself. If no obvious cause is identified, the physician should then attempt flushing the tube. Swab the injection port on the stopcock with alcohol, then turn the stopcock off to the collection bag and slowly instill 5 cc of preservative-free normal saline. Next, turn the stopcock off to the kidney and flush the distal tubing (into the collection bag), and, finally, return the stopcock to the "on" position and observe for return of flow. If unsuccessful, the tube may be dislodged or irreversibly occluded and likely needs to be replaced by interventional radiology or urology.
Abdominal or Flank Pain. Patients presenting with abdominal or flank pain associated with a nephrostomy catheter, especially in the perioperative period, require thorough evaluation by the emergency physician to rule out any underlying life-threatening pathology. Causes of flank/abdominal pain related to nephrostomy catheters include hematoma (e.g., retroperitoneal or perinephric), pyelonephritis, perioperative complications (injury to spleen, liver, bowel, etc.), ureteral stone migration, and hydronephrosis (secondary to tube occlusion or dislodgement). The workup of the majority of these patients requires CT imaging, urinalysis, and basic laboratory tests for signs of infection and assessment of renal function. For cases in which the patient's symptoms are deemed likely to be nephrostomy-related, appropriate disposition and/or follow-up is best decided in consultation with the patient's urologist.
Nephrostomy Tube Dislodgement. Tube dislodgement is among the commonest causes of nephrostomy catheter malfunction.1 Patients typically present with decreased nephrostomy output and/or flank pain (secondary to hydronephrosis). Diagnosis requires radiographic evaluation, typically with either CT or nephrostogram (radiograph of the kidney following administration of a contrast agent through the nephrostomy tube). Treatment includes tube replacement by interventional radiology or urology.
Hematuria. Severe hematuria associated with nephrostomy may be secondary to arterial injury with AV fistula or pseudoaneurysm formation, or blunt or penetrating trauma. Hemorrhage requiring blood transfusion occurs in 1-4% of patients with a nephrostomy.1-3 Gross hematuria more than several days post-nephrostomy insertion that is not associated with trauma raises suspicion of AV fistula or pseudoaneurysm-associated bleeding. This may occur if large branches of the renal artery are lacerated during the procedure. Initial management includes clamping/occlusion of the tube to tamponade bleeding and emergent urology or interventional radiology consultation.3,4 Diagnosis requires angiography, with selective angiography and embolization being the gold standard for diagnosis and treatment.2-4
Urinary Catheters
Case. A 25-year-old man is sent to your emergency department from his primary physician's office for a retained urinary catheter. The catheter was placed last week for urinary retention and was scheduled to be removed today. Multiple attempts at removal were unsuccessful at his physician's office. On evaluation of the catheter, you realize that the balloon is unable to be deflated.
Urinary catheters are frequently used for a variety of indications. Catheterization is a common emergency department procedure, and patients are often discharged home from the ED or hospital with indwelling urinary catheters. Therefore, the emergency physician should be familiar with the potential complications associated with catheter placement and their management. Known complications include urinary tract infection, urethral injury or perforation, bleeding, catheter occlusion, paraphimosis, and retained catheter. This article will focus on the management of a retained urinary catheter.
Most urinary catheters have a distal balloon that is filled with 5-10 mL of water to prevent dislodgement and must be emptied in order to remove the catheter. The balloon mechanism is accessed through a side arm port that contains an internal valve functioning to prevent balloon contents from spontaneously emptying and deflating the balloon. Occasionally, the balloon is unable to be deflated, resulting in a retained catheter. Known causes of catheter retention include blockage of the inflation channel by debris, malfunction of the inflation valve from clamping, crushing, or kinking of the inflation channel, and crystallization of the fluid within the balloon.
Figure 1: Obstructed Foley Catheter

Cutting the side arm (inflation port) of an obstructed urinary catheter to remove the valve
Multiple documented techniques have been utilized to deflate retained urinary catheter balloons, and employing these techniques in a stepwise fashion renders the emergency physician capable of safely and effectively removing a retained urinary catheter. A logical algorithmic approach begins with cutting the side arm (inflation valve) to remove the valve mechanism. (See Figure 1.) If this is unsuccessful, the physician may attempt passing a ureteral stent stylet (or similar small diameter wire) to remove any debris from the balloon port. Next, mineral oil may be instilled into the balloon.5 This method is performed by the instillation of 10 mL of mineral oil into the balloon inflation channel. The oil is left in the channel for 15 minutes, and if no result, an additional 10 mL may be injected. Finally, the physician may proceed directly to needle puncture of the balloon (see below). Instillation of water into the balloon until overinflation achieves balloon rupture is not recommended secondary to risk of bladder injury and retained fragments.6 Several of these deflation techniques have been examined individually by Gulmez et al with regard to rates of successful deflation and production of significant remnant debris. (See Table 1.)7
Table 1: Retained Urinary Catheter Balloon Deflation Techniques
|
Technique |
Successful Deflation (%) |
Retained Debris (%) |
|
Comparison of techniques employed to remove a retained urinary catheter balloon. Data from Gulmez, et al. A comparison of various methods to burst Foley catheter balloons and the risk of free fragment formation. Br J Urol 1996;77:716-718. |
||
|
Balloon rupture |
94 |
83 |
|
Ether instillation |
100 |
100 |
|
Ureteral stent stylet |
14 |
0 |
|
Needle |
100 |
0 |
The best approach for direct needle puncture is gender-based. In females, a transurethral approach is recommended, in which a small gauge angiocatheter is passed along the surface of the urinary catheter until the balloon is encountered. The sheath of the angiocatheter can be used during needle advancement to keep the soft tissues protected from iatrogenic injury.8 The needle can then be advanced beyond the sheath upon contact with the balloon to induce puncture. In males, a transurethral approach is more difficult secondary to the length of the urethra. For this reason, a suprapubic approach is often recommended.6 The bladder should be filled with 50 mL of saline prior to the procedure. After anesthetizing the suprapubic region, a 25-gauge spinal needle is inserted through the abdominal wall and into the region of the bladder neck to deflate or rupture the balloon. Ultrasound guidance should be used to locate the balloon within the bladder as the catheter is held in place with gentle traction.
If the balloon is successfully deflated but the urinary catheter remains fixed within the bladder, the physician should suspect a phenomenon known as balloon cuffing. This occurs when there is "circumferential elevation of the cuff at the equator of a deflated cuff."8 If this condition is suspected, Patterson et al recommend attempting instillation of 0.5 to 1.0 cc water into the balloon port. This should flatten the obstructing ridge and allow for successful catheter removal.
Enterostomy Tubes
Case. An 80-year-old woman with a history of stroke 2 weeks ago resulting in severe dysphagia is sent to your emergency department from her rehabilitation facility after her percutaneous gastrostomy feeding tube fell out one hour ago while performing physical therapy. The tube was placed by interventional radiology 7 days ago. On exam, you find a well-nourished elderly woman complaining of moderate generalized abdominal pain. She is afebrile. Abdominal exam reveals moderate diffuse tenderness with equivocal rebound and voluntary guarding. The patient's nurse asks you if you would like to replace the tube and confirm placement with a contrasted radiograph. Is this appropriate? If not, what is the appropriate acute management and disposition of this patient?
Enteral access for nutrition has long been considered superior to parenteral nutrition in patients with a functional gut, making percutaneous gut access desirable in multiple patient populations. Indications for enteral feeding tube placement include prolonged neurogenic or mechanical dysphagia, prolonged mechanical ventilation, and poor intake (e.g., during chemotherapy). Enterostomy tubes may be placed by interventional radiology under fluoroscopic guidance, endoscopically by a gastroenterologist, or surgically (open or laparoscopic). Relatively common complications related to enterostomy tubes encountered in emergency medicine practice include: tube dislodgement; tube occlusion; leakage around the tube; and skin changes such as hypergranulation, ulceration, erythema, and infection. Some less common problems include: buried bumper syndrome; peritonitis; gastric outlet obstruction; procedural complications such as solid organ injury or bowel injury; gastro-colo-cutaneous fistula; and stomal herniation. In this review, we will focus on tube dislodgement and occlusion, and will briefly cover peritonitis, ostomy leakage, and buried bumper syndrome. See Table 2 for a management overview regarding the most common presenting chief complaints related to enterostomy tube problems.
Table 2: Troubleshooting Enterostomy Tubes
|
Problem |
Possible Causes |
Plan of Action |
|
Vomiting |
Balloon obstruction Malposition |
Check tube length, pull back tube Check placement with contrast study |
|
Diarrhea |
Fistula |
Check for fistula with contrast study |
|
Tube fell out |
Less than 2 weeks old |
Call IR, do not replace tube |
|
2 to 8 weeks old |
Place urinary catheter in stoma and confirm placement with contrast study |
|
|
Greater than 8 weeks old |
Place urinary catheter in stoma and confirm placement with contrast study if any history of trauma or uncertain |
|
|
Tube clogged |
Review medication list for possible causes: ciprofloxacin suspension, lactulose, kayexalate, tube feeds, etc. |
Flush with warm water Flush with carbonated beverage Consider pancrelipase if available |
|
Tube leaking |
Check connection joints and look for holes in tube |
Seal with tape Replace tube |
|
Erythema at site |
Increased tube mobility Skin sensitivity to tape Leakage of gastric acid Granulation tissue infection |
Secure tube Change tape Add acid-blocking agent Cauterize with silver nitrate Start antibiotics |
Accidental tube dislodgement is one of the most common complications of enteral feeding tubes bringing patients to the emergency department. Similar to the management of a dislodged tracheostomy tube, the management of a dislodged enterostomy tube is dependent upon tract maturation, which is temporally mediated. Whereas the important time frames to keep in mind for tracheostomy management are one week and one month, the dates pertinent to enterostomy management are two weeks and two months.
In their 2011 study, Rosenberger et al found the incidence of early dislodgement (defined as within 7 days in their study) to be 4.1%, consistent with the commonly reported range of 0.6 to 4.0%.9 If dislodgement occurs within two weeks of initial insertion, blind reinsertion should be avoided.10 Further, the emergency physician must be aware that, in patients with ascites, malnutrition, or on chronic corticosteroids, tract maturation may require more time, and, therefore, an interval of more than two weeks may be necessary before the tract is mature enough to tolerate any blind reinsertion.11,12 Unless early tube dislodgement is immediately recognized and replaced endoscopically or by interventional radiology, some authors recommend nasogastric suctioning and broad spectrum intravenous antibiotics in order to prevent peritonitis and sepsis, with replacement postponed 7-10 days.11-14 However, on the basis of their 2007 literature review, Minchff et al concluded that, in the absence of clinical peritonitis, these patients do not appear to be at increased risk for septic complications, and may be safely observed, with reinsertion performed in 5-7 days.10
As time progresses and the tract matures, clinical concern for the development of peritonitis and sepsis with accidental tube dislodgement diminishes. In the two-week to two-month time interval, most enterostomy tubes may be safely replaced at bedside, but proper placement should be confirmed with a water-soluble contrast study. Of note, a contrast study should also be utilized for confirmation any time the physician is uncertain about proper placement. As time since initial placement progresses beyond one month to two months, the necessity for radiologic confirmation of placement becomes less obviously indicated. Conversely, as time since actual tube dislodgement progresses beyond 6 to 8 hours, replacement may become increasingly difficult secondary to ostomy stenosis, and the physician should consider endoscopic replacement.14
Figure 2: Dislodged or Misplaced Gastrostomy Tube
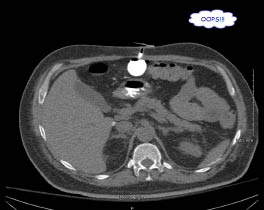
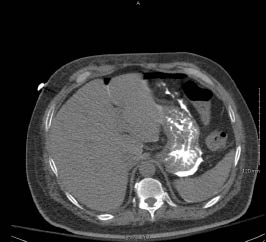
A. Balloon outside of the stomach; B. Intraperitoneal contrast extravasation. Photographs courtesy of Dr. Kamal Massis, Radiology Associates of Tampa.
Case. An 80-year-old woman with a history of multiple strokes and dysphagia is sent to your emergency department from her assisted living facility after the staff were unable to administer her feeding or flush her PEG tube. On exam, you find a well-nourished elderly woman in no distress. You are unable to aspirate or flush her PEG tube.
Feeding tube occlusion is another relatively common complication of enteral feeding tube placement. Infusion of medications and/or feeds, inadequate water irrigation, use of the tube for assessing residual volumes (i.e., aspiration of gastric contents), and mechanical tube factors such as long length, small caliber, and kinking are associated with feeding tube occlusion. See Table 2 for a list of medications commonly associated with feeding tube occlusion. Options for restoration of tube patency include flushing with warm water, cola, cranberry juice, and/or pancreatic enzymes.15 Marcuard and Stegall found that when tube occlusion is believed to be secondary to precipitation of feeds, pancreatic enzyme injection (e.g., pancrelipase crushed and dissolved in 5 cc sodium bicarbonate to activate the enzymes) into the feeding tube followed by clamping for 30 minutes resolved the obstruction in 96% of cases.16
Case. A malnourished 82-year-old man with history of PEG tube placement one month ago is brought to the emergency department with diffuse abdominal pain and subjective fevers progressing over the past 12 hours since his tube feeds were resumed. Twenty-four hours ago he had been managed by one of your colleagues for gastrostomy tube dislodgement and the tube was replaced without documented incident. Abdominal exam reveals diffuse moderate tenderness with rebound and voluntary guarding.
Peritonitis is potentially the most serious and life-threatening complication associated with enterostomy tube placement. Though classically associated with tube dislodgement prior to tract maturation (typically within 7-10 days of insertion) with peritoneal extravasation of gastric or jejunal contents, several cases have been reported in patients with greater than one month of tract maturation.14 Consider abdominal radiographs with injection of water soluble contrast to evaluate for free air and peritoneal contrast extravasation. Management of these patients is straightforward and includes medical resuscitation, broad spectrum antibiotics, immediate cessation of feeds, emergent surgical consultation, and nasogastric suctioning.
Another relatively common problem encountered by patients with enterostomy tubes is leakage of enteral contents around the ostomy. Leakage of large amounts of fluid from the tube site may be secondary to dislodgment of the tube into the subcutaneous tissue tract or tube migration into the gastric outlet, producing gastric outlet obstruction. Subcutaneous dislodgement can produce peritonitis (see above). Diagnosis is based upon water soluble contrast study of tube position. Subcutaneous dislodgement requires immediate tube removal and replacement, whereas gastric outlet obstruction may be managed simply by decreasing balloon volume from 5 mL to 3 mL. If unsuccessful in mitigating leakage, these patients may require conversion to gastrojejunostomy. Small to moderate amounts of leakage may commonly be associated with poor wound healing, skin infection, hypergranulation tissue, buried bumper syndrome, gastric hypersecretion, and excessive cleansing with hydrogen peroxide.11,12 Management of these patients initially includes use of zinc oxide-containing barrier creams and institution of antisecretory therapy.11
Buried bumper syndrome is a rare, late potential cause of peristomal leakage, with few cases reported within 2 months of initial tube placement.17 These patients may present with an immobile enterostomy tube, increased leakage around the tube, pain with feeds, or resistance to feeds. It is believed to be secondary to increased tension between the internal and external tube bolsters, resulting in erosion of the internal bolster superficially into the tract. Over time, this epithelializes and becomes fixed in place.12,18 Treatment usually involves tube replacement.
Some minor cutaneous complications of enterostomy tubes also include hypergranulation tissue, pressure ulcer formation, and infection. Hypergranulation tissue formation is a potential early cause of peristomal leakage and characteristically produces clear/yellow or serosanguinous drainage.19,20 Treatment is with silver nitrate cautery or low-dose topical steroid cream, such as 0.5% or 0.1% triamcinolone TID.18-20 Cutaneous ulceration is a complication of long-standing indwelling enterostomy tubes. Associated risk factors include excessive tube mobility causing side torsion with ulceration in the tract, and excessive skin pressure from tightly anchored bumpers.12,21 Treatment includes loosening the external bolster if too tight and utilization of a commercial securing device or improvised device if the tube is too mobile. Such patients are also at risk of ulceration of the affected gastric mucosa and may present with signs and symptoms typical of peptic ulcer disease.21
Conclusion
With the relatively common utilization of various indwelling devices, including central venous catheters, tracheostomy tubes, nephrostomy tubes, urinary catheters, and enterostomy tubes, it is important for the emergency physician to be familiar with the diagnosis and management of the most common and life-threatening complications associated with these devices. It is our hope that this review has succinctly provided sufficient information relevant to the emergency management of these patients to have covered the vast majority of potential emergency department presentations.
Correction
In the July 4, 2011, issue of Emergency Medicine Reports, the text on page 189 should say, "Moving into a dark environment, classically described as the patient going into a darkened movie theater, acutely dilates the pupil and may precipitate an attack."
References
1. Hausegger KA, Portugaller HR. Percutaneous nephrostomy and antegrade ureteral stenting: Technique indications complications. Eur Radiol 2006;16:2016–2030.
2. Guzmán Martínez-Valls PL, et al. Lower massive hematuria deferred by arteriovenous fistula following percutaneous nephrostomy. Arch Esp Urol 2003;56:1158-1160.
3. Saad Wael EA, et al. Percutaneous nephrostomy: Native and transplanted kidneys. Tech Vasc Interventional Rad 12:172-192.
4. Mahler S, Vaidyanathan L. Complications of urologic devices. In: Cline D, Stead L, eds. Abdominal Emergencies. New York: McGraw-Hill. 2008. P172-180.
5. Murphy GF, Wood DP Jr. The use of mineral oil to manage the nondeflating Foley catheter. J Urol 1993;149:89-90.
6. Daneshmand S, Youssefzadeh D, Skinner EC. Review of techniques to remove a Foley catheter when the balloon does not deflate. Urology 2002;59:127-129.
7. Gulmez I, et al. A comparison of various methods to burst Foley catheter balloons and the risk of free fragment formation. Br J Urol 1996;77:716-718.
8. Patterson R, Little B, Tolam J, et al. How to manage a urinary catheter balloon that will not deflate. International Urology and Nephrology 2006;38:57-61.
9. Rosenberger LH. Late accidental dislodgement of a percutaneous endoscopic gastrostomy tube: An underestimated burden on patients and the health care system. Surg Endosc 2011 May 2.
10. Minchff TV. Early dislodgement of percutaneous and endoscopic gastrostomy tube. J S C Med Assoc 2007;103:13-15.
11. Schrag SP, et al. Complications related to percutaneous endoscopic gastrostomy (PEG) tubes. A comprehensive clinical review. J Gastrointen Liver Dis 2007;16:407-418.
12. McClave SA, Chang WK. Complications of enteral access. Gastrointestinal Endoscopy 2003;55:739-751.
13. Marshall JB. Early accidental dislodgement of PEG tubes. J Clin Gastroenterol 1994;18:210-212.
14. Taheri MR, et al. Peritonitis after gastrostomy tube replacement: A case series and review of literature. J Parenter Enteral Nutr 2011;35:56-61.
15. Napolitano L. Endoscopic placement of feeding tubes. In Rippe JM, Irwin RS, eds. Irwin and Rippe's Intensive Care Medicine, 6th ed. Lippincott Williams & Wilkins; Philadelphia; 2008.
16. Marcuard SP, Stegall KS. Unclogging feeding tubes with pancreatic enzyme. JPEN J Parenter Enteral Nutr 1990; 14:198-200.
17. Tzong-Hsi L, Jaw-Town L. Clinical manifestations and management of buried bumper syndrome in patients with percutaneous endoscopic gastrostomy. Gastrointestinal Endoscopy 2008;68.
18. Friedman JN. Enterostomy tube feedings: The ins and outs. Paediatr Child Health 2004;9:695-699.
19. Crawley-Coha T. A practical guide for the management of pediatric gastrostomy tubes based on 14 years of experience. J WOCN 2004;193-200.
20. Tracey DL, Patterson GE. Care of the gastrostomy tube in the home. Home Healthcare Nurse 2006;24:381-386.
21. Frolich T, et al. Review article: Percutaneous endoscopic gastrostomy in infants and children. Aliment Pharmacol Ther 2010;31:788–801.
This is the second part of our two-part series on complications of tubes and lines. This issue deals with nephrostomy and enterostomy tubes and urinary catheters. It offers straightforward advice about these frequent problems.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
