Abstract & Commentary: Cephalosporin-resistant Gonorrhea: It's Just a Matter of Time
Abstract & Commentary
Cephalosporin-resistant Gonorrhea: It's Just a Matter of Time
By Stan Deresinski, MD, FACP, FIDSA, Clinical Professor of Medicine, Stanford University, Associate Chief of Infectious Diseases, Santa Clara Valley Medical Center, is Editor for Infectious Disease Alert.
Synopsis: Cephalosporin "MIC creep" in Neisseria gonorrhoeae is occurring in the United States, with full-fledged resistance to follow.
Sources: Centers for Disease Control and Prevention (CDC). Cephalosporin susceptibility among Neisseria gonorrhoeae isolates United States, 2000 2010. MMWR Morb Mortal Wkly Rep 2011;60:873-877.
By 2007, the prevalence of fluoroquinolone resistance among N. gonorrhoeae had become so widespread in the United States that the CDC recommended against use of drugs of that class in the treatment of gonorrhea. When the CDC updated its guidelines for the treatment of sexually transmitted diseases in 2010, they noted that, at that time, the only class of antibiotics acceptable for empiric treatment of gonorrhea were cephalosporins. However, they also noted that approximately 50 cases of failure of oral cephalosporins (cefixime is recommended for oral therapy in the United States) had been reported worldwide, with most having occurred in Asia. One possible case of failure of cefixime therapy in Hawaii had, however, been reported. While cephalosporins still generally remain effective for treatment of gonorrhea in the United States, CDC has now documented "MIC creep" of N. gonorrhoeae, a harbinger of worse to come.
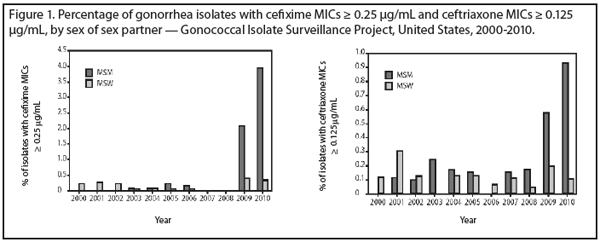
An average of almost 6,000 isolates from male urethras recovered at U.S. sentinel sites were tested against each year from 2000-2010 and the proportion of isolates with cefixime MIC ≥ 0.25 µg/mL and ceftriaxone with MIC ≥ 0.125 µg/mL was examined. Reduced susceptibility to these antibiotics is defined by CLSI as an MIC > 0.5 µg/mL; there are no defined breakpoints for resistance defined for N. gonorrhoeae.
The percentage of isolates with cefixime MIC ≥ 0.25 µg/mL increased from 0.2% to 1.4% during 2000-2010 (P < 0.001), while those with ceftriaxone MIC ≥ 0.125 µg/mL increased from 0.1% to 0.3% during 2000-2010 (P = 0.047). In the Western region of the United States, the proportion of isolates with cefixime MIC ≥ 0.25 µg/mL increased from 0% to 3.3% (P < 0.001), and the percentage of isolates with ceftriaxone MICs ≥ 0.125 µg/mL increased from 0% to 0.5% (P < 0.001). This reduced susceptibility was most marked in Honolulu, with cefixime MICs ≥ 0.25 µg/mL, increasing from 0% in 2000 to 7.7% of isolates in 2010 (P < 0.001). In California, the comparable results were 0% in 2000 and 4.5% in 2010 (P < 0.001). An increase in ceftriaxone MICs also was observed in California (0% in 2000 and 0.6% in 2010; P = 0.001). Overall in the United States, reduced susceptibility was more marked among men who have sex with men (MSM) than in others (see Figure 1). Although azithromycin resistance has been previously reported, isolates in this study remained susceptible to this azalide.
Commentary
The pattern of reduced susceptibility to cephalosporins noted by CDC is eerily reminiscent of the pattern observed with fluoroquinolones before full resistance emerged in N. gonorrhoeae. Thus, these data are almost certainly a harbinger of continuing evolutionary change in this organism in the face of β-lactam exposure that will eventually lead to a high prevalence of full-fledged resistance to cephalosporins. The emergence of reduced susceptibility to cefixime surely predicts that ceftriaxone will follow. This is problematic, since agents from this class are currently critical to effective therapy of gonorrhea and no other effective antibiotic treatment options that have been well-studied are currently available.
CDC now recommends treating uncomplicated gonorrhea with ceftriaxone 250 mg intra-muscularly and azithromycin 1 g by mouth. Patients should undergo test-of-cure and cultures, rather than just nucleic acid amplification tests, obtained from those with possible treatment failure so that antibiotic susceptibility can be determined. Patients with cefixime treatment failure should be re-treated with 250 mg ceftriaxone intramuscularly and 2 g azithromycin orally.
By 2007, the prevalence of fluoroquinolone resistance among N. gonorrhoeae had become so widespread in the United States that the CDC recommended against use of drugs of that class in the treatment of gonorrhea.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
