Complications of Tubes and Lines: Part I
Complications of Tubes and Lines: Part I
Authors:
Charlotte Derr, MD, RDMS, FACEP, Associate Program Director, Ultrasound Fellowship Director, University of South Florida Emergency Medicine Residency Program, Tampa.
Nathaniel Ronning, MD, University of South Florida Emergency Medicine Residency Program, Tampa.
Peer Reviewer:
Eric Gross, MD, Hennepin County Medical Center, Associate Professor of Emergency Medicine, University of Minnesota Medical School, St. Paul.
Emergency physicians are often required to adeptly manage patients with unique and challenging clinical problems. This is a two-part paper to provide the emergency physician with a clear, concise review of troubleshooting tubes and lines. Part I will deal with central venous catheters and tracheostomies. A later edition will deal with feeding and nephrostomy tubes.
The Editor
Central Venous Catheters
Since first described in 1952, central venous catheter (CVC) placement has proven to be of great clinical utility with numerous uses and indications. General categories of central venous catheters include tunneled and non-tunneled catheters. Examples of tunneled catheters include Hickman, Broviac, Groshong, and totally implantable venous access devices (TIVADs or "portacaths"). Examples of non-tunneled CVCs include double or triple lumen catheters, Shiley catheters, Cordis catheters, and peripherally inserted central catheters (PICC).
Tunneled catheters are typically utilized to provide medium- or long-term access to patients with chronic conditions or those requiring regular intravenous access. Such patients may include those with total parenteral nutrition (TPN)-dependence, end-stage renal failure requiring dialysis, or malignancy requiring chemotherapy. These catheters typically access the central venous system through an internal jugular or subclavian vein, are tunneled through the subcutaneous tissues, and may or may not exit the skin over the chest wall. The TIVADs, for example, utilize a reservoir buried in the subcutaneous tissue of the chest wall. The patient's own skin and subcutaneous tissues provide a barrier to ascending infection.
Non-tunneled catheters are placed into the central venous system through percutaneous puncture more directly superficial to the targeted central vein (or peripheral vein, as in PICCs). They may be placed in the neck, chest, or extremities. Indications for placement include extended antimicrobial therapy, need for repeated blood sampling, administration of vasopressors or TPN, hemodynamic monitoring, or emergent access when peripheral venous access is not possible. PICCs are more suitable to longer-term use as compared to other non-tunneled CVCs.1,2,3
Common Complications of CVC Insertion
Examples of complications include: pneumothorax; catheter malpositioning; hemothorax; hemopericardium; pericardial tamponade; lymphatic injury with possible chylothorax or chylopericardium; vascular injuries such as arterial puncture and/or large-bore arterial cannulation; hematoma; pseudoaneurysm; AV fistula; guidewire loss; and dysrhythmias. In a 2007 prospective study of 1794 central venous cannulations by experienced operators using landmarks alone (no ultrasound guidance), the overall incidence of mechanical complication or malpositioning was 12.2%.4 Malpositioning represented more than half of these adverse events, while arterial puncture accounted for nearly one-quarter.4 Importantly, several recent studies suggest that these complications may be reduced by the use of bedside real-time ultrasonographic guidance.5,6
The overall incidence of pneumothorax was rather low (0.5%). Right subclavian CVC placement was the highest independent risk of pneumothorax at 1.2%. Second was the right internal jugular with an incidence of 0.7%, followed by left subclavian (0.6%), the right innominate (0.5%), and, finally, the left internal jugular (0.3%).4 A large meta-analysis analyzed data from 10 studies and 3420 catheter placements. They found the incidence of pneumothorax (or hemothorax) to be 1.3% in internal jugular CVC insertion and 1.5% in subclavian catheterizations.7 Although the incidence of iatrogenic pneumothorax may be low in patients receiving CVCs, the clinician must always maintain a high index of suspicion for this potentially life-threatening complication.
The intravenous loss of a guidewire is perhaps the most easily preventable potentially serious complication of CVC insertion. We have all heard the mantra "never let go of the guidewire." In addition to physician carelessness, unanticipated circumstances and distractions may result in guidewire loss. However personally distressing or embarrassing it may be, management involves immediate interventional radiology consultation and, if necessary, transfer to a facility with an interventional radiology service.
Arterial puncture, perhaps a less easily prevented complication of CVC insertion, can also produce catastrophic outcomes, especially with large-bore arterial dilation or cannulation. This complication has been shown to be associated with AV fistulae, arterial thrombosis, arterial pseudoaneurysm, arterial dissection, stroke, hemothorax, cardiac tamponade, and aortic injury. The incidence of arterial puncture varies based on anatomic location. The study by Reusch et al found internal jugular CVC placement was associated with a higher risk of arterial puncture than subclavian access, with a relative risk of 4.7.7 Eisen et al found the incidence of arterial puncture was 3.2% of subclavian lines, 7.1% of femoral lines, and 5% of internal jugular lines.8 Schummer et al reported similar data, finding the overall incidence of arterial cannulation to be 0.2%, with a significant difference between right subclavian (3.6%) and left subclavian (0.6%) catheterization.4
Several papers reported that large-bore arterial perforation produces physical symptoms in up to 30% of patients, and in these symptomatic patients, mortality may be as high as 20% to 40%.3,9-14 CVP monitoring and radiographic confirmations are effective adjuncts in identifying successful catheterization of the venous system. In addition, the use of real-time ultrasound (US) guidance during needle placement decreases the incidence of arterial puncture.5,6,15,16 Another technique proposed in two recent (2010) papers suggests that ultrasound can be used to confirm appropriate guidewire placement in the venous system prior to cannulation.15,16
Despite the use of real-time US guidance, there remains a certain risk of serious arterial injuries secondary to inadequate needle visualization or improper use of dilators (e.g., deeper than just skin and subcutaneous tissues). In a recent study, a higher than expected percentage of emergency medicine residents penetrated the posterior wall of the internal jugular vein in a "lifelike mannequin" when using cross-sectional windows for US guidance.17 Therefore, it is our recommendation that the longitudinal view of the vessel is used whenever possible, with care to visualize the needle tip at all times from entrance into the subcutaneous tissue until the appropriate vessel is successfully penetrated.
Although a detailed discussion of the techniques used in the management of large-bore arterial cannulation is beyond the scope of this paper, consultation with interventional radiology or vascular surgery is indicated. In cases of large-bore arterial catheterization, the catheter should be left in place and removal should be performed by interventional radiology or vascular surgery.18,19 A study by Chemelli et al successfully managed all cases of inadvertent subclavian artery cannulation with a strategy of balloon catheterization or percutaneous closure device deployment first, and stent-graft insertion for failures or certain complications.20
Common Complications of Established CVCs
Common complications of appropriately placed CVCs include: infection (local or systemic); occlusion (thrombus, fibrin sheath); deep venous thrombosis; and catheter tip migration. Less common complications include: vascular erosion and perforation; catheter fracture with or without fragment embolization; air embolization; extravasation injury.
Systemic and local infections are well known yet relatively uncommon complications of CVCs. O'Grady et al cited the incidence of indwelling catheter-related bloodstream infection (CRBSI) to be 5.3 per 1000 catheter days (CDs).3,21 According to recent French articles, the incidence of infection (local and CRBSI) in TIVADs is 0.37 to 0.43 per 1000 catheter days, more than 10-fold lower than the incidence of infection in CVCs with external ports.22,23 A systematic review of 200 published studies found catheter-related bloodstream infection rates of 2.7 per 1000 CDs for short-term CVCs, 1.6 per 1000 CDs for tunneled CVCs, 0.1 per 1000 CDs for central venous ports, 1.7 per 1000 CDs for arterial catheters, and 2.1 per 1000 CDs for peripherally inserted CVCs.24 Another study of 656 emergency department-placed CVCs found the infection rate to be 1.93 per 1000 CDs.25
Indwelling catheters may become infected through a variety of mechanisms, including hematogenous seeding, primary infection via the skin or insertion site, or through contamination of the catheter hub or infusate. The causative organisms most commonly include coagulase-negative Staphylococcus (CONS), Staphylococcus aureus, Candida species, gram-negatives, and Enterococcus. Clinical manifestations include superficial infection at the insertion site, tunnel infection (only in tunneled catheters), pocket space abscess (only in TIVADs), catheter colonization, thrombus (i.e., septic thrombophlebitis), and bloodstream infection/bacteremia.26,27
Local infections, including exit site, tunnel, and pocket infections, characteristically present with local erythema, drainage, induration, and/or overlying tenderness. They are most commonly caused by contamination with skin flora and, therefore, the incidence (of both local and systemic infection) can be reduced by meticulous catheter care, use of topical antimicrobials such as chlorhexidine washes, and antimicrobial barriers.28-30 Insertion/exit site infections typically present with 2 cm or less of surrounding erythema, whereas tunnel infections may present with erythema, induration, purulent drainage, or tenderness that extends more than 2 cm from the exit site.26-28 This distinction is important because infections localized to the exit site may be managed expectantly with appropriate antibiotics, often sparing the catheter, whereas tunnel infections (and pocket abscesses) usually require catheter removal. If present, any drainage should ideally be sent for culture and gram stain.
Catheter-related bloodstream infection is associated with numerous complications with relatively high morbidity and mortality, such as sepsis, septic shock, infective endocarditis, septic thrombophlebitis, septic emboli, etc. The most common causative agents in the United States in 2006-2007 included (in descending order of prevalence) CONS, Enterococcus, Candida species, Staph. aureus, K. pneumonia, Enterobacter spp., P. aeruginosa, E. coli, and A. baumannii.31 The pathogenesis of CRBSI is most frequently extraluminal catheter colonization from the exit site. Less commonly, intraluminal colonization may occur secondary to hub contamination or administration of contaminated infusate, or via hematogenous seeding from a different source of bacteremia.32,33
Emergency department management essentially consists of drawing appropriate cultures (either from the catheter or simultaneously from the catheter and a peripheral source)28,34-36 and initiating appropriate antimicrobial therapy. In most cases, it is not necessary to remove a potentially infected CVC in the emergency department, although this may be appropriate under certain circumstances. If possible, the decision should be made in conjunction with the physician or service utilizing the indwelling catheter. The indications for catheter removal vary based on organism isolated, length of catheter insertion (long term vs. short term), and ability to obtain additional IV access. In short-term CVCs, removal is indicated for CRBSI caused by organisms other than coagulase-negative Staphylococcus (CONS), including gram-negative Bacilli, S. aureus, Enterococcus spp., fungi, and Mycobacteria.28 For CONS CRBSIs, antimicrobial lock therapy (with appropriate systemic antibiotic therapy) may be attempted for catheter salvage. In regard to long-term CVCs, infectious indications for catheter removal include severe sepsis, suppurative thrombophlebitis, endocarditis, CRBSI that persists after 72 hours of appropriate antimicrobial therapy, and any CRBSI involving S. aureus, P. aeruginosa, fungi, or Mycobacteria.28 If none of the above is present, catheter salvage may be attempted.28
In particular, for suspected CRBSI, the Infectious Diseases Society of America 2009 recommendations for empiric therapy include: (1) vancomycin in regions of elevated MRSA prevalence, or daptomycin if local vancomycin mean inhibitory concentration (MIC) is frequently > 2 mcg/mL; (2) gram-negative bacilli coverage based on local susceptibilities (e.g., cefepime, piperacillin/tazobactam, or a carbapenem, with or without an aminoglycoside); (3) if neutropenic or severely ill or previously colonized with Pseudomonas, add locally appropriate coverage for multidrug-resistant Pseudomonas; (4) if the patient has a femoral catheter or is septic with hematologic malignancy, history of solid organ transplant, colonization with Candida spp., or on TPN, add an appropriate antifungal (e.g., micafungin can use fluconazole if no azole exposure within 3 months).34
Catheter Occlusion
Catheter occlusion is a common complication of chronic indwelling venous catheters, occurring in up to 50% of the pediatric population and 66% of adults with long-term CVCs.37 CVC occlusions can be described as complete or partial. In partial occlusions, it is possible to deliver an infusion through the catheter, but one cannot withdraw from the catheter. A complete occlusion is present when one can neither withdraw nor infuse through the catheter.
Causes of CVC occlusion include mechanical obstruction, intraluminal infusate precipitation, and thrombosis. Mechanical obstructions may be secondary to pinch-off syndrome, port abutment of the vessel wall, catheter kink, tight suture, closed clamp, catheter fracture or disconnection, malpositioned subcutaneous access needle (in TIVADs), or thrombotic obstruction.37 Thrombotic obstruction may be secondary to intraluminal thrombosis, fibrin sheath formation, or deep venous thrombus occluding the vessel lumen or catheter ports.3,37,38
A systematic approach may help to guide diagnosis and management. First, evaluate the patient for signs or symptoms of deep venous thrombosis (DVT) and evaluate the catheter for any obvious external mechanical causes, such as kinks in the catheter tubing, excessively tight sutures, or subcutaneous needle malposition. If signs or symptoms of DVT are present, consider duplex ultrasonography. Catheter-related thrombosis is managed similarly to non-catheter-related DVT, typically with anticoagulation and hospital admission. Recent literature suggests that select patients with newly diagnosed deep venous thrombosis may be managed safely on an outpatient basis. The safety and efficacy of outpatient management of CVC-associated DVTs is not clear. A 2005 study found the 2-month mortality (all causes) was increased by 14% in patients with CVC or pacemaker associated DVT vs. patients without these indwelling devices.39 In certain circumstances (i.e., clots with a low risk of embolization in patients who are not dependent on the catheter), the catheter may be removed by the emergency physician. In most circumstances, however, it is prudent for the emergency physician to leave the catheter in place throughout the patient's ED course.
Figure 1: Pinch-off Syndrome
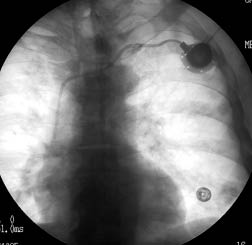
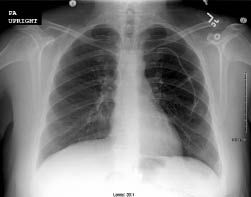
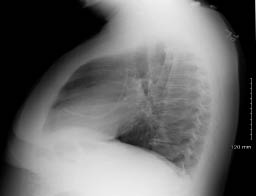
A. Pinch-off Syndrome. Note the small blush of contrast extravasation at the inferior margin of the clavicle. B and C. PA and lateral chest radiograph illustrating catheter fracture and tip embolization into the right ventricle. Photographs courtesy of Dr. Kamal Massis, Radiology Associates of Tampa.
If there are no signs or symptoms of DVT and no obvious external causes, repeat attempts at catheter access after repositioning the patient by elevating the ipsilateral arm. If positional, a chest radiograph with or without a contrast-enhanced study of the catheter, referred to as a "linogram," may be helpful. (See Figure 1.) If compression is present, remove the catheter immediately to avoid fracture and embolization. If not positional and there is a partial occlusion (i.e., can flush, cannot aspirate), likely causes include fibrin sheath, catheter tip thrombus, or vessel wall abutment (can be positional). If completely occluded, there is likely an intraluminal catheter thrombus. Attempts may be made with thrombolytic instillation, followed by repeated attempts at withdrawal and flushing. If unsuccessful, consider ultrasonography to rule out occult catheter-related venous thrombosis. Ultimately, interventional radiology may need to be consulted. See Table 1 for an overview of the etiology, workup, and management of catheter occlusion.
Table 1: Trouble-shooting a Clogged Central Venous Catheter
|
Evaluation |
Causes |
Treatment |
|
*Alteplase dosing: 2 mg per dose, typically using 2 mL of a 1 mg/mL solution, placed in the catheter lumen and given 30 minutes intraluminal time. Attempt to aspirate, then flush. If occlusion remains, repeat x 1. Alternatively (for smaller catheters), administer 110% of the catheter volume (of the 1 mg/mL solution) up to a maximum of 2 mg per dose.37 If occlusion remains, the physician may attempt passing a guidewire through the catheter to dislodge the clot. If catheter remains occluded, consult IR. If successful, lock with heparin solution. |
||
|
Examine external catheter and anatomy |
Break or Kink? Wear at the clamp site? Closed clamp? Tight suture? Signs or symptoms of DVT? |
Correct any external causes. May require replacement of CVC. If suspect DVT, proceed with venous US. |
|
Attempt to Flush and Aspirate Catheter |
Unable to aspirate, able to flush:
Unable to aspirate or flush:
|
Reposition and retry Radiograph (consider linogram with contrast): Kinks? Loops? Twists? Detached reservoir? Pinch-off syndrome? Consider ultrasound Alteplase* |
|
Patient on TPN or Drug Infusions |
Review drug and possible precipitations |
TPN: Instill 70% EtOH37 Acidic drug: NaCO3 |
Tracheostomy Tubes
This section will narrow the primary focus to three tracheostomy-related problems potentially requiring immediate intervention by the emergency physician, including tracheoinnominate artery fistula (TIF), the dislodged tracheostomy, and the occluded tracheostomy.
A bleeding and/or pulsatile tracheostomy can herald one of the most feared and life-threatening complications of tracheostomy, the tracheoinnominate artery fistula (TIF). The incidence of TIF has been reported to be 0.6% to 0.7%, with the majority (78%) occurring within the first 4 weeks of placement.40,41 Without urgent operative intervention, the mortality of this complication is believed to be near 100%.42 The pathophysiology involves pressure necrosis and erosion of the soft tissues between the ostomy (or the anterior trachea just distal to the ostomy) and the innominate artery. This soft-tissue injury may be secondary to persistent compression by the elbow of the tracheostomy tube, an overinflated cuff, or by the tip of the tube. At least 48 hours are required for the necessary soft-tissue changes and necrosis to develop before any possible TIF-associated bleeding may occur.42 Risk factors include a mobile tracheostomy tube, a relatively caudally placed ostomy, and an overinflated cuff.
Patients may present with mild to severe bleeding through the ostomy, mild to massive hemoptysis, or a pulsatile tube. Nearly 50% of patients will experience a sentinel bleed preceding any massive bleeding or hemoptysis; therefore, any bleeding 3 days to 6 weeks after tracheostomy should be considered TIF until proven otherwise.42 However, if bleeding on presentation is mild or absent, it is prudent for the emergency physician to rapidly exclude other more common causes of bleeding initially, including tracheobronchitis, pneumonia, or ostomy wound bleeding.43,44 Granulation tissue formation is another common cause of tracheostomy–associated bleeding, but is rarely life-threatening and requires fiberoptic evaluation for diagnosis. Once the aforementioned causes have been ruled out, or if the patient presents initially with brisk bleeding, TIF must be considered and the patient managed accordingly with appropriate medical resuscitative interventions (intravenous fluids, blood transfusion) and immediate surgical consultation. The diagnosis of TIF is clinical, and angiography is often non-diagnostic. Therefore, any decision to pursue imaging in diagnosis should likely be made in cooperation with the consulting surgeon.
Several maneuvers can be performed by the emergency physician that may serve as a temporizing measure to bridge the temporal gap to definitive treatment in the operating room. These interventions have been shown to be successful in controlling hemorrhage in more than 80% to 90% of cases.42,43 The emergency physician should be familiar with the following three interventions: 1) overinflation of the tracheostomy tube cuff; 2) placement of a translaryngeal (oral or nasal) endotracheal tube; and 3) digital compression through the ostomy. A logical sequence includes initially attempting tracheostomy cuff overinflation. If this fails, one may attempt slight withdrawal of the tracheostomy tube and repeated cuff overinflation with the cuff at the level of the ostomy. If this fails to achieve hemostasis, next place an endotracheal tube orally with the cuff positioned just distal to the tracheostomy tube (with the tracheostomy tube left in place), and the cuff overinflated. If bleeding persists despite the above interventions, the physician should remove the tracheostomy tube and perform the Utley maneuver, which consists of inserting a digit into the ostomy and compressing the tracheoinnominate artery against the posterior sternum.42-44
Decannulation
Accidental decannulation represents a relatively common complication of tracheostomy placement, especially in the pediatric population.45,46 Emergency department management of these patients is largely based on two important factors: oxygenation and airway protection; and time since tracheostomy was placed. At less than one week since placement, the tract is immature and, therefore, recannulation carries significant risk of misplacement or mediastinal intubation.45 If more than one month has passed since initial tracheostomy placement, the tract is likely well-formed, and risk of inadvertent misplacement is low. Between one week and one month, the risk of misplacement is proportional to the amount of time passed since initial cannulation. Clinically, this is relevant because tract maturity determines the appropriate method of recannulation. In a stable patient with a mature tract, it is reasonable to simply replace the tracheostomy at bedside. This can be done blindly, with a "fingertip" technique, or with a Seldinger technique (discussed below). Patients who are stable with an immature tract (less than seven days) should have their ostomy recannulated only by an experienced provider with fiberoptic visual confirmation. Fiberoptic visual confirmation is also indicated in the seven-day to one-month group.45 In any patient who is unstable or is unable to protect his or her airway, orotracheal intubation is indicated, especially in the setting of an immature ostomy tract.
Several techniques for bedside recannulation have been proposed, including the "fingertip" technique and the Seldinger technique, which may improve success rates. The fingertip technique involves insertion of a gloved fingertip through the ostomy to identify the tract and elevate the thyroid isthmus or any other soft tissues potentially occluding the tract. The tracheostomy tube is then advanced as the gloved finger is withdrawn. Another technique, which seems intuitively safe and easy to utilize, is the Seldinger technique, in which a nasogastric tube (or fiberoptic bronchoscope, gum elastic bougie, etc.) is inserted into the trachea through the ostomy tract as a "guidewire," and the tracheostomy tube inserted over the "guidewire."46 This technique may also be useful in cases of tracheostomy tube obstruction to either remove the obstruction or to replace the tracheostomy tube.
Obstruction of the tracheostomy tube may result from mucous plug formation, thick secretions, granulation tissue, blood clot formation, kinking or tube dislodgement, or foreign bodies. Depending on the relative severity of obstruction, these patients may present with mild shortness of breath to complete respiratory arrest. Following an ordered algorithmic approach to intervention will allow the emergency physician to relieve the obstruction in a majority of cases. Begin your assessment with a rapid evaluation of the tracheostomy to ensure proper positioning and the absence of any obvious external causes of obstruction. If the patient has a speaking valve attached, remove the speaking valve or at least deflate the cuff.47 Next, the physician or other care provider should attempt to pass a suction catheter. If the suction catheter passes (at least 10 cm or past the distal tip of the tube), reassess and observe for adequacy of oxygenation and ventilation. If inadequate, consider replacing the tracheostomy tube (using the aforementioned Seldinger technique) or oral intubation. If the initial attempt at passing a suction catheter is unsuccessful, remove the inner cannula (if present) and remove any obstructing material. If respiratory distress persists, replace the airway or attempt oral intubation.47
References
1. Paauw JD, et al. The incidence of PICC line-associated thrombosis with and without the use of prophylactic anticoagulants. JPEN J Parenter Enteral Nutr 2008;32(4):443-447.
2. Amerasekera SSH, Jones CM, Patel R, et al. Imaging of the complications of peripherally inserted central venous catheters. Clin Radiology 2009;64:832-840.
3. Kusminsky RE. Complications of central venous catheterization. J Am Coll Surg 2007;204:
681-696.
4. Schummer W, et al. Mechanical complications and malpositions of central venous cannulations by experienced operators. A prospective study of 1794 catheterizations in critically ill patients. Intensive Care Med 2007;33:1055-1059.
5. Cavanna, Luigi, et al. Ultrasound-guided central venous catheterization in cancer patients improves the success rate of cannulation and reduces mechanical complications: A prospective observational study of 1,978 consecutive catheterizations. World J Surgical Oncology 2010;8:91.
6. Serafimidis K, et al. Ultrasound-guided catheterization of the internal jugular vein in oncologic patients; comparison with the classical anatomic landmark technique: A prospective study. Int J Surg 2009;7:526-528. Epub 2009 Sep 12.
7. Ruesch S, et al. Complications of central venous catheters: Internal jugular versus subclavian access-a systematic review. Crit Care Med 2002;30:454-460.
8. Eisen LA, et al. Mechanical complications of central venous catheters. J Intensive Care Med 2006;21:40.
9. Jain U, et al. Subclavian artery laceration and acute hemothorax on attempted internal jugular vein cannulation. J Cardiothor Vasc Anesth 1991;5:608-610.
10. Kron IL, et al. Arch vessel injury during pulmonary artery catheter placement. Ann Thorac Surg 1985;39:223-224.
11. Robinson JF, et al. Perforation of the great vessels during central line placement. Arch Int Med 1995;155:1225-1228.
12. Schwartz AJ, et al. Carotid artery puncture with internal jugular cannulation using the Seldinger technique: Incidence, recognition, treatment and prevention. Anesthesiology 1979;51:S160.
13. Shah PM, et al. Arterial misplacement of large caliber cannulas during jugular vein catheterization: Case for surgical management. J Am Coll Surg 2004;198:939-944.
14. Wicky S, et al. Life threatening vascular complications after central venous catheter placement. Eur Radiol 2002;12:901-907.
15. Gillman LM, et al. Ultrasound confirmation of guidewire position may eliminate accidental arterial dilatation during central venous cannulation. Scand J Trauma Resusc Emerg Med 2010;18:39.
16. Stone MB, et al. Ultrasound detection of guidewire position during central venous catheterization. Am J Emerg Med 2010;28:82–84.
17. Blaivas M, Adhikari S. An unseen danger: Frequency of posterior vessel wall penetration by needles during attempts to place internal jugular vein central catheters using ultrasound guidance. Crit Care Med 2009;37:2345-2349.
18. Nicholson T, Ettles D, Robinson G. Managing inadvertent arterial catheterization during central venous access procedures. Cardiovasc Intervent Radiol 2004;27:21-25.
19. Pikwer A, et al. Management of inadvertent arterial catheterisation associated with central venous access procedures. Eur J Vasc Endovasc Surg 2009;38:707-714.
20. Chemelli AP, et al. Endovascular management of inadvertent subclavian artery catheterization during subclavian vein cannulation. J Vasc Interv Radiol 2010;21:470-476. Epub 2010 Feb 20.
21. O'Grady NP, et al. Guidelines for the prevention of intravascular catheter-related infections. CDC. MMWR Recomm Rep 2002;51(RR 10):1-29.
22. Barbut F, et al. Totally implantable venous access ports: Frequency of complications and analysis of bacterial contamination after ablation. Pathol Biol (Paris) 2004;52:566-574.
23. Crisinel M, et al. Incidence, prevalence and risk factors for a first infectious complication on a totally implantable venous-access port. Med Mal Infect 2009;39:252-258.
24. Maki DG, Kluger DM, Crnich CJ. The risk of bloodstream infection in adults with different intravascular devices: A systematic review of 200 published prospective studies. Mayo Clin Proc 2006;81:1159-1171.
25. LeMaster, Christopher H, et al. Infection and natural history of emergency department–placed central venous catheters. Ann Emerg Med 2010;56:492-499.
26. Campbell PM. Troubleshooting central venous catheters in the emergency department. J Emerg Nursing 1996;22:416-421.
27. Greene JN. Catheter-related complications of cancer therapy. Infect Dis Clin North Am 1996;10:255-295.
28. Weber DJ, Rutala WA. Central line–associated bloodstream infections: Prevention and management. Infect Dis Clin N Am 2011;25:77–102.
29. Munoz-Price LS, Hota B, Stemer A, et al. Prevention of bloodstream infections by use of daily chlorhexidine baths for patients at a long-term acute care hospital. Infect Control Hosp Epidemiol 2009;30:1031–1035.
30. Climo MW, Sepkowitz KA, Zuccotti G, et al. The effect of daily bathing with chlorhexidine on the acquisition of methicillin-resistant Staphylococcus aureus, vancomycin-resistant Enterococcus and healthcare-associated bloodstream infections: Results of a quasi-experimental multicenter trial. Crit Care Med 2009;37:1858–1865.
31. Hidron AI, et al. NHSN annual update: Antimicrobial-resistant pathogens associated with healthcare-associated infections: Annual summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2006-2007. Infect Control Hosp Epidemiol 2008;29:
996-1011.
32. Guidet B, Nicola I, Barakett V, et al. Skin versus hub cultures to predict colonization and infections of central venous catheter in intensive care patients. Infection 1994;22:43–48.
33. Salzman MB, Isenberg HD, Shapiro JF, et al. A prospective study of the catheter hub as the portal of entry for microorganisms causing catheter-related sepsis in neonates. J Infect Dis 1993;167:487–490.
34. Mermel LA, Allon M, Bouza E, et al. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 update by the Infectious Disease Society of America. Clin Infect Dis 2009;49:1–45.
35. Raad I, Hanna H, Maki D. Intravascular catheter-related infections: Advances in diagnosis, prevention, and management. Lancet Infect Dis 2007;7:645–657.
36. Safdar N, Fine JP, Maki DG. Meta-analysis: Methods for diagnosing intravascular device-related bloodstream infection. Ann Intern Med 2005;142:451–466.
37. Baskin JL, Pui CH, Reiss U, et al. Management of occlusion and thrombosis associated with long-term indwelling central venous catheters. Lancet 2009;374:159-169.
38. Kurul S, Saip P, Aydin T. Totally implantable venous-access ports: Local problems and extravasation injury. Lancet 2002;3:684-692.
39. Hingorani A, et al. Risk factors for mortality in patients with upper extremity and internal jugular deep venous hrombosis. J Vas Surg 2005;41:476-478
40. Epstein SK. Late complications of tracheostomy. Respir Care 2005;50:542–549.
41. Sue RD, Susanto I. Long-term complications of artificial airways. Clin Chest Med 2003;24:
457–471.
42. Grant CA, et al. Tracheo-innominate artery fistula after percutaneous tracheostomy: Three case reports and a clinical review. British J Anaesthesia 2006;96:127–131.
43. Cohen JE, et al. Exsanguinating tracheoinnominate artery fistula repaired with endovascular stent-graft. Surgical Neurology 2008;69:306–309.
44. Thorp A. Tracheoinnominate artery fistula: A rare and often fatal complication of indwelling tracheostomy tubes. Pediatr Emerg Care 2005;21:763-766 .
45. O'Connor H, White A. Tracheostomy decannulation. Respiraory Care 2010;55:1076-1081.
46. Pattanong P. Dislodged tracheostomy. J Prapokklao Hosp Clin Med Educat Center 2007;24:304-308.
47. Regan K, Hunt K. Tracheostomy management. Contin Educ Anaesth Crit Care Pain 2008;8:31-35.
Emergency physicians are often required to adeptly manage patients with unique and challenging clinical problems. This is a two-part paper to provide the emergency physician with a clear, concise review of troubleshooting tubes and lines. Part I will deal with central venous catheters and tracheostomies. A later edition will deal with feeding and nephrostomy tubes.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
